Abstract
Objective
This study aims to evaluate the phytochemical composition, antioxidant, and antihyperglycemic (in vivo, in vitro, and in silico) activities and acute toxicity of Crataegus monogyna Jacq (C monogyna) aqueous extracts.
Methods
The study analyzed the aqueous extract of C monogyna through various methods such as phytochemical screening, and the high-performance liquid chromatography-ultraviolet (HPLC-UV)-visible analysis. The extract was also tested for antioxidant potential, acute toxicity, antihyperglycemic effect, and inhibitory effect on the pancreatic α-amylase enzyme. Additionally, the study used the molecular docking approach to identify the most potent ligands in the extract.
Results
The phytochemical screening of the aqueous extract of C monogyna showed the presence of flavonoids, tannins, coumarins, sterol, and triterpene. The extract was rich in total polyphenols (1.65 ± 0.04 mg gallic acid equivalent per gram of extract [GAE/g] DM), total flavonoids (0.33 ± 0.03 EQ/g DM), and condensed tannins (0.28 ± 0.01 EC/mg DM). HPLC-UV-visible analysis identified 9 phenolic compounds, with high levels of gallic acid and caffeic acid. The C monogyna extract has a high antioxidant activity with an IC50 of 9.23 ± 0.01 mg/mL by DPPH and 8.32 ± 0.02 mg/mL by FRAP. The aqueous extract of C monogyna was not toxic to albino mice. The glucose tolerance test showed a significant antihyperglycemic effect, with an IC50 of 0.070 ± 0.008 mg/mL for the inhibition of pancreatic α-amylase activity by the aqueous extract of C monogyna. The in vivo inhibitory effect of the extract on the pancreatic α-amylase enzyme was confirmed. Two flavonoids, catechin, and rutin, were identified as potent inhibitors of the activity of α-amylase in the in silico part of the study, compared to the native ligand, Acarbose.
Conclusion
The study found that C monogyna has significant antioxidant and antihyperglycemic properties. The presence of catechin and rutin may contribute to these effects. The results suggest that C monogyna could be used as a dietary supplement to prevent and treat diabetes.
Introduction
Medicinal plants are considered finished products, that are ready for consumption and raw materials that can be used to obtain active substances. They represent a significant resource for many people, and they have numerous therapeutic virtues, which have been demonstrated by experiments. 1 Throughout history, natural products have been employed for medical purposes, particularly in traditional medicine, where plants and herbal preparations have played a vital role in preventing and alleviating a variety of human health conditions.2,3 Recently, herbal medicines have received significant attention as a potential solution for treating various human illnesses. The therapeutic properties of medicinal plants are believed to be attributed to the presence of various bioactive compounds. Of these compounds, edible plants are particularly promising as they have minimal adverse effects.4,5 The Mediterranean basin, comprising only 1.6% of the land area, is home to over 22,500 plant species, accounting for 10% of known plants. 6 Remarkably, nearly 60% of these species are exclusive to the region. The “Conservation International” organization, has identified the Mediterranean basin as 1 of the 25 hotspots due to its rich biodiversity, including 11,700 endemic species, representing 52% of endemism. The islands, peninsulas, rocky cliffs, and mountain peaks within the region contain over 47% of the total endemic species, making it a significant hub of unique flora. 7
Belonging to the Mediterranean basin, Morocco is floristically rich. The capacity of medicinal plants is immense and encompasses a vast array of species with diverse characteristics. This presents a crucial avenue for scientific exploration, particularly in the area of natural substances. 8 The presence of secondary metabolites with beneficial biological properties plays a crucial role in the therapeutic effectiveness of these plants. It is worth noting that over 60% of the pharmaceutical drugs approved have their origins in natural sources, with plants being the primary source. 9 In addition to the bioactive role of secondary metabolites, there are certain primary molecules, including iminosugars, which have attracted the attention of researchers in recent years due to their pharmacological capacities. Iminosugars are a diverse group of plant and microbial compounds that are of interest due to their ability to interact with human glycosidase, other proteins, and the sugar receptor10-12 Iminosugars are of particular interest because they are known to act as glycosidase and glycosyl transferase inhibitors, influencing a wide range of biological processes. 13 These compounds can play a crucial role in controlling diabetic patients. 1-deoxynojirimycin is a powerful α-glucosidase inhibitor, and its derivative N-hydroxyethyl-1-deoxynojirimycin (miglitol) is used to treat type II diabetes.12,13
Crataegus monogyna Jacq. (common hawthorn), part of the Moroccan flora is a species of the Rosaceae family that also grows in Europe, and Asia, where it is usually found as a shrub or small tree 5 to 10 m high. 14 Hawthorn contains aromatic amines, essential oils, phenolic acids, flavonoids (hyperin, quercetin, spirein, rutin, and apigenin), and proanthocyanidins as bioactive compounds. 15 It has an important place in traditional medicine for its sedative, vasculoprotective, and antioxidant properties. 16 Recently, attention has focused on antioxidant activity because of its role in the prevention of chronic diseases. Some parts (such as flowers, flower buds, and leaves) are used for the treatment of certain diseases, including irritability, insomnia migraines, confusion, and memory loss.17,18 The unripe fruit juices of hawthorn have been used in cosmetic applications and for some skin applications, arthritis, and muscle pain. Dried fruits also have diuretic properties.18,19 The leaves, flowers, and fruits of hawthorn are used as coronary vasodilators, cardiotonic, and hypotensive. An ethnobotanical study in Algeria demonstrated the use of hawthorn as an antidiabetic plant in traditional medicine. 20 Diabetes mellitus is a severe condition that is prevalent worldwide, and it is characterized by chronic hyperglycemia due to metabolic disorders. 21 Presently, there are several artificial medications available to treat diabetic patients. In our study, we focused on evaluating both the antioxidant activity and antihyperglycemic properties of a specific plant extract. The rationale for studying both activities stems from their interconnection in the context of diabetes. Oxidative stress, characterized by an imbalance between reactive oxygen species production and antioxidant defense, is closely linked to the onset and progression of diabetes. By investigating the antioxidant activity of the extract, we aimed to assess its potential in mitigating oxidative stress, which is a crucial factor in diabetes management. Simultaneously, we examined the antihyperglycemic activity of the extract, specifically targeting enzyme inhibition involved in glucose metabolism. This approach aligns with the objective of reducing postprandial hyperglycemia, a key concern in diabetes treatment. By exploring the extract's ability to inhibit α-glucosidase and α-amylase enzymes, we aimed to identify natural compounds that could effectively regulate glucose absorption and control blood glucose levels. natural plant extracts have gained attention as promising α-glucosidase and α-amylase inhibitors in recent years. 22 Furthermore, there has been a recent emphasis on promoting safer and more tolerable inhibitors for both enzymes that can be naturally extracted from medicinal plants, fruits, and vegetables at a lower cost.
The objectives of our study revolve around the aqueous extract of C monogyna leaves and flowers. We aimed to extract secondary metabolites from these plant parts using the decoction method. Subsequently, we conducted qualitative and quantitative phytochemical analyses to identify and characterize the bioactive components present in the extract. In addition, we focused on the evaluation of its antioxidant activity, toxicity, antihyperglycemic, and pancreatic α-amylase inhibitory effects.
Results and Discussion
Yield of Extraction
The solid–liquid extraction of phenolic compounds performed by decoction resulted in a crude extract with a yield of 9.05 ± 0.3%. This yield is close to those of the leaves and flowers of C. oxyacantha species obtained by decoction, which are respectively in the order of 6.7% and 9.75%. 23 However, the comparison of our yield results with those of the bibliography appears to be difficult because the yield is only relative and depends on the method, the polarity of the solvent and the conditions, under which the extraction was performed, as well as the species and its geographical origin.
Qualitative Phytochemical Screening
The process of phytochemical testing involved qualitative analysis to detect various categories of compounds in the leaves and flowers of the C monogyna extract. This analysis was performed using precipitation and turbidity reactions, examination under ultraviolet (UV) light, or a particular change in color. The findings of this characterization of the aqueous C monogyna extract were recorded and can be seen in Table 1.
Phytochemical Characterization of the Main Secondary Metabolites Contained in C monogyna Leaves and Flowers Aqueous Extract.

(−)negative result, (+) weak positive result, (++) positive result, (+++) strong positive result.
Indeed, the phytochemical screening tests revealed the absence of saponosides and alkaloids and the presence of flavones, bile tannins, and mucilage, leucoanthocyanins, oses, and holosides in very large quantities in the aqueous extract of the leaves and flowers of C monogyna. In addition, the presence of coumarin was also detected in our extract but with a small amount compared to other compounds. These results are consistent with the work of Boughrara and Belgacem, 24 who confirmed the presence of anthocyanins, coumarin, flavonoids, quinones, saponins, tannins, and sterols in the genus Crataegus. 24 Similarly, our results are consistent with the findings of Bouzid 1 who affirmed the presence of flavonoids, tannins, and coumarin in C monogyna fruits extract. Likewise, a separate investigation conducted by Khokhlova et al 25 demonstrated that among 15 species of the Crataegus genus, C monogyna exhibited a high abundance of flavonoids.
It is crucial to emphasize the role of these phenolic compounds in hawthorn. Flavonoid content in the fruit is 1% to 2% which is responsible for the red or blue color of the hawthorn berry. 15 Polyphenols are involved in the food quality of fruits, and anthocyanins and some flavonoids participate in the coloring of ripe fruits. 26 Phenolic compounds determine the flavor of fruits: Tannins are responsible for the astringent sensation of unripe fruits; flavanones are responsible for the bitterness of Citrus fruits and can give rise, by chemical transformation, to sweet dihydrochalcones. 23 Quinic acid and its derivatives such as caffeoylquinic acids are also responsible for the astringent sensation and have also been isolated from C monogyna.27,28
Total Polyphenols, Flavonoids, and Tannins contents
By utilizing the Folin–Ciocalteu method to determine the total phenols, it was discovered that the average amount of polyphenols, measured in terms of caffeic acid equivalents, per gram of extract was 1.65 ± 0.04 mg AGE/g DM. Quantitative estimation of flavonoids showed that the aqueous extract of C monogyna contains 0.33 ± 0.03 EQ/g DM. Condensed tannins determination gave a content in the order of 0.28 ± 0.01 EC/mg DM. According to 1 study, the level of polyphenols in 9 samples of C monogyna aerial parts ranged from 117.729 to 204.286 mg gallic acid equivalent per gram of extract [GAE/g]. 29 Whereas the levels of flavonoids in the previous extracts ranged from 2.12 to 32.62 mg/g. By comparing the results obtained with those of the scientific literature, where other species of the same genus were studied, we find that the polyphenol content of the methanolic extract of C. pinnatifida leaves found by Yoo et al 30 was about 8.17 mg/g extract. The hydroalcoholic extract had a flavonoid content of 4.07 mg Eq/g, which was lower compared to the results of Barros et al 18 for methanolic extracts of C monogyna flowers and fruits (330.32 and 247.03 mg/g extract, respectively). Furthermore, our findings differed from those reported by Belabdelli et al, 31 who found high levels of polyphenols (473.4 mg GAE/g extract) and flavonoids (80.9 mg CE/g extract) in the ethanolic extract of C monogyna leaves. However, it is difficult to compare these results with other studies due to the varying extraction methods, solvent polarities, and species used, which reduces the reliability of a comparison. 32 The content of phenolic compounds is influenced by several factors, including extrinsic factors such as geographical and climatic conditions, genetic factors, the degree of ripening, and storage duration of the plant. Recent studies have shown that these factors have a significant impact on the polyphenol content.33,34
High-Performance Liquid Chromatography Coupled With Diode Array Detector Analysis
The chemical compounds of the aqueous extract of C monogyna were characterized using high-performance liquid chromatography coupled to diode array detector (HPLC-DAD) method by comparing their retention times and UV spectra with those of standards. A total of 9 compounds were identified in the aqueous extract of C monogyna with a total identified percentage of 33.9% (Figure 1). Flavonoids such as rutin and catechin, tannins such as tannic acid, phenolic acids such as gallic acid, caffeic acid and rosmarinic acid, ascorbic acid, coumaric acid, and catechol. Our results are in agreement with those presented in previous studies. Keser et al 35 reported that the aqueous extracts of the leaves and flowers of C monogyna are rich in rutin-type flavonoids, and other types of flavonoids. In addition, Belabdelli et al 31 identified 9 procyanidine telic phenolic compounds, epicatechina, catechin, chlorogenic acid, caffeic acid, vitexin-2-O-rhamnoside, vitexin, rutin, and hyperoside in an ethanolic extract of C monogyna leaves. The same authors identified syringic acid, myricetine, quercetin, naringenin, kaempferol, using the chloroform extract of C monogyna leaves. Another phytochemical study conducted on the extract of flowers of C monogyna showed the richness of this extract in phenolic compounds such as rutin, and quercetin. 32 Abuashwashi and Palomino 29 identified rutin, apigenin, and quercetin among the compounds in methanolic extracts of aerial parts of 9 samples of C monogyna growing spontaneously in Spain. A study conducted on the fruits of 2 hawthorn species (C monogyna, C pentagyna) for the identification of organic acids showed that both of them contain ascorbic acid in variable amounts depending on the species. 36 The ascorbic acid discovered in C monogyna may contribute to the nutritional value and medicinal properties of the plant. For example, flowers contain high levels of tocopherols, and ascorbic acid, and have a good ratio of n-6/n-3 fatty acids, compared to ripe fruits, while unripe fruits generally contain the highest level of polyphenols. 18 Furthermore, the low percentage of ascorbic acid detected in our extracts may be due to the flowering period of the plant which was justified by Glew et al, 37 who noted that while the malic acid level increases continuously, the ascorbic acid level decreases throughout the development of the fruit, with both acids reaching the maximum and minimum limits at 161 days after complete flowering, 428 and 8.4 mg /100 g DM, respectively. Overall, the chemical composition of C monogyna extracts remains relatively different from part to part and thus in even part. This variation could be attributed to the samples’ geographical origin, climatic and ecological conditions, harvest season and drying process, extraction procedures, and solvent type used.9,38
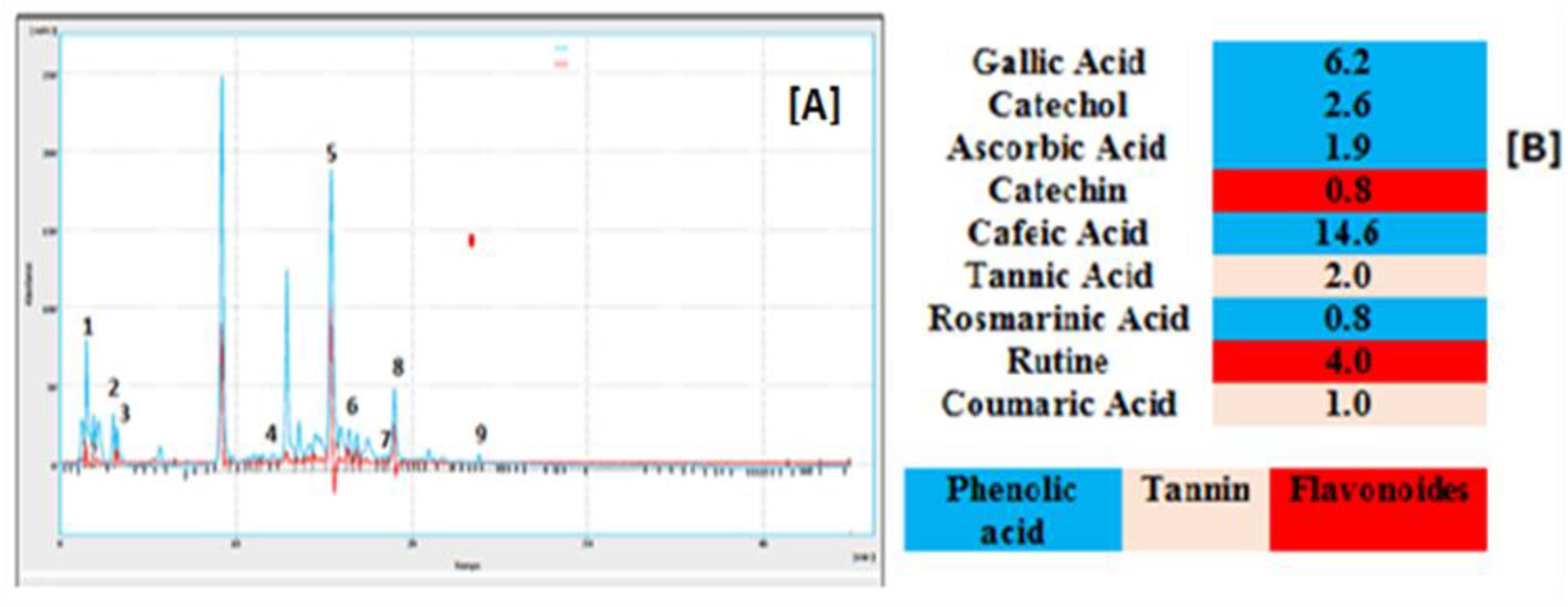
High-performance liquid chromatography (HPLC) chromatogram of the aqueous extract of C monogyna: (A) Chromatogram, (B) relative quantification (%) of compounds identified in the aqueous extract of C monogyna. (1) gallic acid (6.2%), (2) catechol (2.6%), (3) ascorbic acid (1.9%), (4) catechin (0.8%), (5) caffeic acid (14.6%), (6) tannic acid (2.0%), (7) rosmarinic acid (1.9%), (8) rutin (4.0%), (9) coumaric acid.
Biological activities
Antioxidant Activity of C monogyna Extract
The DPPH radical is commonly utilized to assess the capacity of substances to serve as radical scavengers or hydrogen donors and to evaluate their antioxidant properties. This radical is stable under normal conditions and can receive an electron or hydrogen to become a stable paramagnetic molecule. The antioxidant potential of the aqueous extract of C monogyna toward the DPPH free radical was determined by measuring the reduction of this radical spectrophotometrically at 517 nm, which is accompanied by a color change from purple to yellow.
The results of the percentage of antioxidant activity are shown in the figures belowe (Figure 2A). The results of the antioxidant activity of the aqueous extract of C monogyna flowers and leaves showed a high value of antioxidant effect, up to 87.78%, this value was close to that of the ascorbic acid, for which the maximum value was around 90.49%. Similarly, the IC50 values are in the order of 1.86 ± 0.01 mg/mL and 9.23 ± 0.01 mg/mL for ascorbic acid, and C monogyna, respectively. Our results demonstrated that the aqueous extract of C monogyna had less radical trapping activity than the ethanolic extract of IC50 leaves (22.5 µg/mL) found by Fouzia et al. 31 Furthermore, Amor and Belhattab 39 reported an IC50 of C monogyna aqueous leaf extract ranging from 45 to 725 µg/mL at different vegetation stages. Coimbra et al 40 reported that the extracts obtained from C monogyna demonstrated an IC50 value of 36.11 g/mL. We assume that the aqueous extract of C monogyna with significant antioxidant activity was richer in flavonoid compounds with free-radical scavenging activity, which is strictly linked to the structure of the flavonoid compound itself, which has been proven in numerous studies to be related to the structure and anti-free-radical activity of flavonoids.9,38 This structure requires 3 criteria, the ortho-dihydroxy structure on the B ring, the C2-C3 double bond in conjunction with the 4-oxo function, and the presence of OH group in positions 3 and 5 in combination with the C2-C3 double bond which gives maximum antiradical activity. The HPLC analyses confirm this hypothesis, given the presence of 2 powerful flavonoids as antioxidants, namely catechin and rutin, the latter is also known to have anti-inflammatory effects, and anticancer properties and to reduce the deleterious effect of oxidized bad cholesterol. 41 In addition, tannins, coumarin, and caffeic acid present in both plants also possess antioxidant activity and can protect cells from free radical damage. 42 Reducing power is the ability of a so-called “antioxidant” compound to donate an electron and/or a hydrogen atom, thus preventing biomolecules such as lipoproteins and polyunsaturated fatty acids from the action of free radical species. 43 The obtained results (Figure 2B) show that the aqueous extract of C monogyna has an iron-reducing power as well as the reference molecule (ascorbic acid). Moreover, this extract has a lower reducing activity (0.690) than ascorbic acid (1.859). Likewise, these results were confirmed by the EC50 values, 2.71 ± 0.01 and 8.32 ± 0.02 mg/mL for ascorbic acid and C monogyna, which would be due to the richness of the said extract in phenolic compounds in general and in flavonoids in particular indicated by the chemical analysis which demonstrated a high content of total phenols, flavonoids, and condensed tannins which can provide good sources of natural antioxidants. For this reason, the phenolic compounds present in the extract have powerful effects against iron reduction activity. However, Singleton and Rossi (1965), found that the radical scavenging activity is not diverse. They react differently depending on the number of phenolic groups they contain. 44 Specifically, the phenolic compounds do not necessarily incorporate all the antioxidants present in the extracts. Therefore phenolic compounds are sometimes correlated with several plant species. 45 The structure-activity relationship hypothesis is consistent with the findings of other authors. Yuting et al, 46 Jovanovic et al 47 showed that flavonoids containing hydroxyl groups at the 3′, 4′ positions on the B-ring and at the 5, 7 positions on the A-ring are endowed with antioxidant properties. Moreover, Ariga et al 48 evaluated the antioxidant activities of flavin derivatives (catechins, proanthocyanidins B1 and B3) extracted from Vigna angularis seeds. They particularly insisted on the number of phenolic groups in these compounds that could explain the antioxidant potentialities. In conclusion, we can emphasize the multiple interests of the potential use of Crataegus or its fractionated extracts as antioxidants. Let's start by recalling that Crataegus extracts are used in the composition of “anti-aging” creams or cosmetology and that our results, even if they are not obtained with the same extracts, partially support this type of use, with a tendency to prefer natural additives to which a prejudice of iniquity is attached that should be verified.

Antioxidant activity of the aqueous extract of C monogyna and standard ascorbic acid measured by DPPH (A) and FRAP (B) methods. Data are expressed as mean ± SD.
The attribution to certain polyphenols as an important part of the antioxidant activity of Crataegus tissue extracts in vivo and in vitro can contribute to improving the knowledge of potentially usable antioxidants. Finally, in the field of therapy and despite the high activities observed, we cannot currently affirm that Crataegus extracts and their most active phenolic molecules can be used against certain pathologies linked to the overproduction of free radicals. We know that the way from in vitro action in pharmacological tests to application in medicine is long and so complicated.
Acute Oral Toxicity of C monogyna Sample
The results of the acute toxicity test indicated that the aqueous extract of C monogyna was not toxic, even at a dosage of 2 g/kg. No signs of toxicity such as diarrhea, vomiting, abnormal mobility, or mortality were observed during the monitoring period. Given its use in cooking, it can be assumed that hawthorn has minimal adverse effects. At therapeutic doses, hawthorn is associated with very few adverse effects, including sweating, headache, mild skin rash, palpitations, drowsiness, restlessness, and gastrointestinal issues. 49 A systematic review of 5577 patients who were administered standardized hawthorn extracts revealed that most adverse effects were mild to moderate. Clinical studies have shown that hawthorn consumption does not have any significant adverse effects to date. 50
Antihyperglycemic effect of C monogyna Sample
Figure 4 displays the findings of the oral glucose tolerance test. The aqueous extract of C monogyna at a dose of 400 mg/kg, when administered orally to normal rats 30 min before glucose overload, significantly reduced postprandial hyperglycemia at 60 min (P < .01; 1.07 ± 0.14 g/L) and at 90 min (P < 0.001; .86 ± 0.01g/L). Similarly, glibenclamide had a significant inhibitory effect on postprandial hyperglycemia for up to 2 hours after glucose overload at 60 min (P < .01; 1.08 ± 0.09 g/L) and at 90 min (P < .001; 1.09 ± 0.10 g/L). At 150 min, no significant difference in blood glucose was observed between all groups compared to the pretreated group with distilled water (at 60 min (1.37 ± 0.16 g/L), at 90 min (1.22 ± 0.11 g/L), and 150 min (0.76 ± 0.07 g/L) (Figure 3A). Furthermore, the area under the glucose curve (AUC glucose) was significantly lower (P < .001) in rats treated with C monogyna (53.13 g/L/h) compared to rats treated with distilled water (62.91 ± 4.32 g/L/h). Additionally, the area under the curve of glibenclamide was significantly (P < .001) lower (55.95 ± 1.69 g/L/h) compared to the area under the curve of distilled water-treated rats (62.91 ± 4.32 g/L/h) (Figure 3B). In conclusion, the results of the oral glucose tolerance test suggest that the aqueous extract of C monogyna can enhance glucose tolerance.

Change in postprandial blood glucose (A) and area under the postprandial blood glucose curve (B) in normal rats after administration of the test products (aqueous extract of C monogyna and glibenclamide). Values are means ± standard mean error (SEM) (n = 6). *P < 0.05; **P < 0.01; ***P < 0.001.
This plant effect could be due to its property to significantly induce peripheral tissues to use glucose in a very active way, which explains the improvement of glucose tolerance in rats. Also, its capacity to block intestinal glucose absorption, and/or to potentiate insulin production by the pancreas.51,52 The chemical analyses of this extract showed that it is composed of gallic acid, catechol, ascorbic acid, catechin, caffeic acid, tannic acid, rosmarinic acid, rutin, and coumaric acid. In the literature, caffeic acid and its derivatives have been shown to have impressive antidiabetic potential by stimulating glucose uptake in cultured C2C12 cells through the AMPK pathway.53,54 Similarly, A study confirmed that the consumption of catechin-rich green tea (intake of 500 mg per day of catechin) in type 2 diabetic patients under insulinotropic treatment would increase insulin levels and improve the decrease of glycated hemoglobin levels. 55 Another study showed that supplementation with a mixture of polyphenols (333 mg per day for 6 weeks) from strawberries and cranberries containing anthocyanins, proanthocyanins, ellagitannins, phenolic acids, and flavonols would improve insulin sensitivity in overweight nondiabetic individuals. 56
In Vitro α-Amylase Activity of C monogyna Extract
One of the methods used to manage hyperglycemia in diabetics is inhibiting enzymes involved in the digestion of complex carbohydrates, such as α-amylase. This approach aims to reduce the digestion and absorption of ingested carbohydrates in the gastrointestinal tract . 51 The study found that C monogyna's aqueous extract has a dose-dependent inhibitory effect on pancreatic α-amylase, as shown in Figure 4. The presence of the extract significantly inhibited the activity of pancreatic α-amylase, with an IC50 of 0.07 ± 0.008 mg/mL. Additionally, the inhibitory effect of the aqueous extract on pancreatic α-amylase activity was greater than that of acarbose, with an IC50 of 0.37 ± 0.03 mg/mL.

Inhibitory effect of aqueous extract of C monogyna and acarbose on pancreatic α-amylase activity, in vitro. Values are means ± standard mean error (SEM), (n = 3).
In Vivo Pancreatic α-amylase Activity of C monogyna Extract
To assess the impact of the C monogyna extract on the pancreatic α-amylase activity in the gut, a study was conducted on normal rats in vivo (Figure 5). The rats were orally administered the aqueous extract of C monogyna flowers and leaves at a dose of 400 mg/kg, 30 min before being overloaded with starch. The results showed a significant decrease in postprandial hyperglycemia at 60 min (P ≤ .001, 0.99 ± 0.06 g/L), at 90 min (P ≤ .001, 0.73 ± 0.04 g/L), and at 150 min (P < .05, 0.79 ± 0.07), compared to rats treated with distilled water. Acarbose also significantly inhibited postprandial hyperglycemia for 2 hours after starch overload (Figure 5A). The area under the curve was significantly (P ≤ .001) lower in rats treated with the extract (48.39 ± 2.06 g/L/h) than in those treated with distilled water (61.82 ± 1.53 g/L/h). Similarly, the area under the acarbose curve was significantly (P < .001) lower (52.05 ± 4.27 g/L/h) (Figure 5B). Inhibitors of α-amylase activity such as acarbose, voglibose, and miglitol are commonly used to treat type II diabetes, either alone or in combination with other drugs, but they can cause several side effects, including liver problems, flatulence, and abdominal cramps. 57 In addition, some of them may increase the incidence of kidney tumors, liver damage, and acute hepatitis. 58 To avoid the adverse effects of these synthetic molecules, studies are focusing on the isolation and identification of new α-amylase inhibitors from natural resources with few side effects. Plants have always been an exemplary source of drugs and many currently available active ingredients are derived directly or indirectly from plants. 59 On the other hand, several α-amylase inhibitory phytoconstituents, such as flavonoids, alkaloids, terpenoids, anthocyanids, glycosides, and phenolic compounds have been identified. 60 Natural flavonoids as α-amylase inhibitors have been the subject of several investigations. 61 These natural α-amylase inhibitors may represent an alternative and attractive strategy for the control of hyperglycemia, 62 hence the rationale for the important results of our work, thanks to the richness of the decoctate in phenolic acids, flavonoids, and tannins. Some experiments suggest that gallic acid could be involved in the inhibition of pancreatic α-amylase63-66 Similarly, rutin has been demonstrated to inhibit α-amylase in a competitive mode with quercetin. 60

Effect of aqueous extract of C monogyna and acarbose on the variation of postprandial blood glucose in normal rats (A), with the area under the curves (B). Values are mean ± standard mean error (SEM) (n = 6). ***P < 0.001: In comparison with control.
In silico α-amylase Activity of C monogyna Extract
Molecular docking is an advanced computing method that is commonly used to provide valuable insight into the molecular mechanisms of pharmacologically active medicines.67,68 Molecular docking was employed in this investigation to reveal the possible mechanism of action connected with the activity of α-amylase characteristics of C monogyna components. The reported data, in the form of binding affinity values, may suggest that the researched molecule has a higher or lower affinity for the specified target when compared to a native ligand (acarbose), assuming that binding energy decreases as compound affinity rises. The α-amylase active sites are surrounded mainly by the amino acid residues
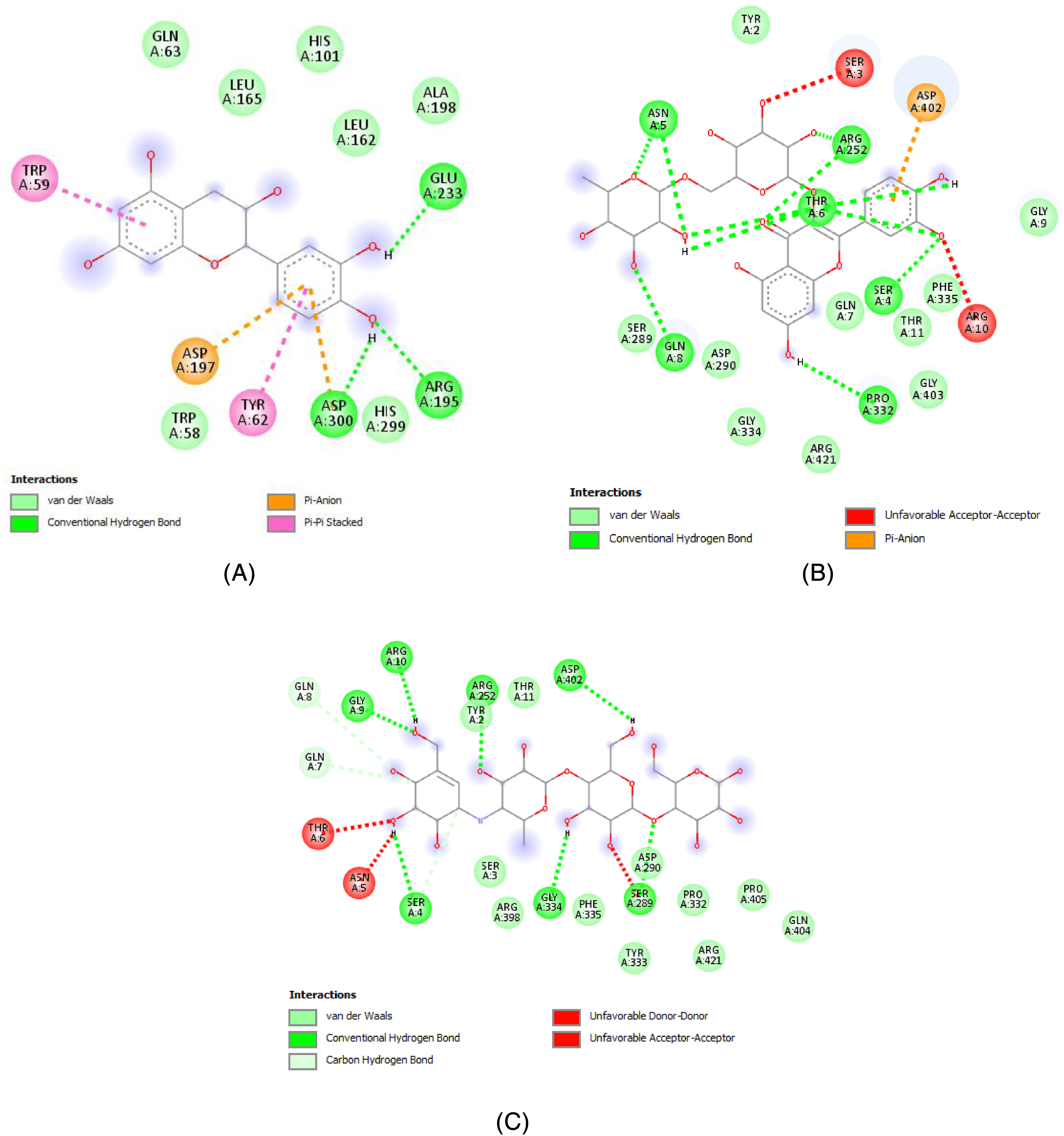
Two-dimensional schemes of the interactions with the amino acid residues of the 2 potent flavonoids found in C monogyna, (A) catechin, (B) rutin, and the native ligand, acarbose (C).
Binding Energy, H-bonds, and Interacting Amino Acids of Phytocompounds Found in C monogyna Target Protein.

Acarbose, a native ligand of α-amylase.
*The potent ligands in comparison to the native ligand.
Conclusion
The management of diabetes mellitus requires precise control of postprandial glycemia, which can be achieved by consuming plant extracts with an antihyperglycemic effect. C monogyna leaf and flower extract was found to have an antihyperglycemic effect thanks to its rich secondary metabolites in addition to significant antioxidant activity. The plant was also nontoxic. Overall, our results confirm the traditional use of C monogyna leaves and flowers to treat diabetes mellitus. Consequently, this plant may hold promise for alleviating hyperglycemia in diabetics, encouraging further studies to complement the results found in diabetic rats and to further test subacute toxicity.
Materials and Methods
Plant Material
The collection of C monogyna leaves and flowers took place in Ifrane, which is located in the Middle Atlas region of Morocco. The Laboratory of Botany and Plant Ecology at the Scientific Institute in Rabat, Morocco, conducted the plant species identification. The plant material was air-dried in the shade at room temperature for 15 days, processed, and subsequently kept in a damp and dark storage area.
Extract Preparation
A modified version of the traditional Moroccan population method, 72 was used to produce the aqueous extract of C monogyna leaves and flowers. To prepare the extract, 30 g of the plant material in powdered form was placed in a 1-liter Erlenmeyer flask. Then, 300 mL of distilled water was added, and the mixture was heated on a hot plate for 1 hour until it boiled. After cooling and filtering, the water was removed by vacuum evaporation and dried in an oven at 40 °C. A portion of the extract was preserved for future reference by storing it in a dark environment at a temperature of 4 °C.
Qualitative Screening
The chemical composition of the plant extract was analyzed by conducting several tests, such as staining reactions and precipitation, to identify the primary categories of chemical constituents present in the plant species under investigation. The protocols outlined in references73-75 were followed to carry out these tests.
Quantitative Screening
Quantification of Total Phenols
The Folin–Ciocalteu method,
76
with modified procedures was used to determine the total phenol content of the C monogyna aqueous extract. To obtain a blue coloration, a certain amount of the extract was mixed with 1.5 mL of Folin–Ciocalteu reagent (10%) and 1.5 mL of 7.5% (m/v) sodium carbonate (Na2CO3) in a 100 mL volumetric flask, which was then filled with distilled water. After 2 hours at room temperature, the absorbance was measured against a blank value at 765 nm using a spectrophotometer (UV mini-1240). A calibration curve was made using gallic acid as a positive control under the same conditions. The results were expressed in mg GAE/g and calculated using the equation Y = ax + b obtained from the calibration curve.

Determination of Total Flavonoids
The method used to measure the amount of flavonoids in the C monogyna extract was adapted from Djeridane et al. 77 First, 100 µl of the extract was mixed with 20 mL of distilled water and allowed to sit for 5 min. Then, 100 µl of aluminum trichloride (AlCl3) at a concentration of 10% (w/v) was added, and the solutions were adjusted to 50 mL with pure methanol. After shaking and keeping the solution in the dark for 30 min at room temperature, the absorbance was measured at 333 nm using a spectrophotometer (UV mini-1240) against a blank value. The flavonoid content was quantified using a calibration curve made with a standard flavonoid (quercetin) that underwent the same analytical conditions as the samples, with concentrations ranging from 5 to 30 μg/mL. The total flavonoid content was expressed as milligrams of quercetin equivalent per gram of extract (mg EQ/g).
Determination of Condensed Tannins
To estimate the condensed tannins, the vanillin acid method, 78 was used. First, different concentrations of catechin solution (2 mg/mL) were prepared and mixed with 3 mL of 4% (m/v) vanillin/methanol solution, followed by the addition of 1.5 mL of concentrated hydrochloric acid. After a 20-min reaction at room temperature, the absorbance was measured at 499 nm using a spectrophotometer against a blank. The same procedure was used to determine the condensed tannin content in the aqueous extract of C monogyna, replacing the catechin with the sample. The tannin content was expressed in milligram equivalents of catechin per gram of dry matter weight, using the calibration curve.
HPLC-DAD Analysis
The C monogyna aqueous extract underwent HPLC analysis using a Waters e2695 HPLC analysis system with a C18 column of dimensions (5 µm, 250 mm × 4.6 mm). Elution was achieved by using water/acetic acid (2% v/v) and acetonitrile (pH = 2.6). The injection volume was 10 µL, and separation was carried out using a gradient mode with a constant flow rate of 0.3 mL/min. 79 Detection was done using a diode array detector with a wavelength range of 280-360 nm. To identify the compounds, their retention time and λmax were compared with authentic standards such as gallic acid, catechol, catechin, caffeic acid, ascorbic acid, tannic acid, rosmarinic acid, rutin, coumaric acid, quercetin, vanillin, and apigenin.
Biological Activities
Antioxidant Activity
DPPH Free Radical Scavenging Test
The DPPH radical scavenging assay is based on the ability of the extract to capture the dark violet-colored free radical DPPH and reduce it to a yellowish color. The procedure, described in the work of Braca et al. 80 was slightly modified. A solution of 2.4 mg of DPPH in 100 mL of ethanol was prepared, and the extract was dissolved in ethanol at a rate of 1 mg/mL to create a stock solution, which was then diluted to obtain a range of concentrations from 0.05 to 0.1 mg/mL. Two microliters of the compound to be tested and 2.8 mL of DPPH solution were mixed to perform the test, and the same concentrations were prepared with ascorbic acid as a positive control. A blank was also prepared with absolute ethanol. The samples were left in the dark for 30 min, and the reduction in discoloration was measured at 517 nm compared to the negative control (DPPH solution only).
The results were expressed as the percentage reduction of DPPH (AA %).
The IC50 values, which represent the concentration of the sample required to scavenge 50% of DPPH-free radicals, were determined through linear regression analysis of the concentration–absorbance graph of the extract. Since there is no standard measure of antioxidant activity, results are often compared to a reference antioxidant like ascorbic acid.
Ferric Reducing Antioxidant Power
The method described by Koncic et al 81 was used to determine the ability of the phenolic extract to reduce ferric iron (Fe3+) to ferrous iron (Fe2+). In this method, 0.5 mL of extract at various concentrations (ranging from 0.1 to 1.0 mg/mL) was mixed with 2.5 mL of a 0.2 M phosphate buffer solution (pH = 6.6) and 2.5 mL of a 1% solution of potassium ferricyanide K3Fe(CN)6 in test tubes. The mixture was then incubated in a water bath at 50 °C for 20 min. The reaction was stopped by adding 2.5 mL of 10% trichloroacetic acid and centrifuging the mixture at 3000 revolutions for 10 min. The supernatant of each concentration was mixed with 2.5 mL of distilled water and 0.5 mL of the aqueous solution of FeCl3 at 0.1%. The absorbance of the reaction mixture was measured at 700 nm and compared to a blank prepared in the same way, with distilled water used to calibrate the spectrophotometer. The reducing power of the extracts tested was determined by measuring the absorbance, with an increase in absorbance indicating an increase in reducing power. The EC50 was determined by linear regression from the concentration-absorbance graph of the extract. Standard solutions (ascorbic acid) were used as positive controls, and their absorbance was measured under the same conditions as the samples.
Animals
Animal studies were conducted on male and female Wistar rats and albino mice. The animals were raised in the animal house of the Biology Department at the Faculty of Sciences of Oujda, where they were exposed to a photoperiod of 12 h of light and 12 h of darkness and a temperature of 22 ± 2 °C. The animals were kept under optimal rearing conditions and provided with unlimited access to food and water.
Acute Oral Toxicity of C monogyna Sample
In accordance with the OECD recommendations (425), 82 acute oral toxicity was evaluated in albino mice that had been fasting for 14 h, with 2 batches of mice (n = 6; ♂/♀ = 1; 20-35 g) randomly divided into 4 groups (n = 6; ♂/♀ = 1). The Control group was given a dose of 10 mL/kg of distilled water, while the treated groups were given different doses (0.5, 1, or 2 g/kg) of the aqueous extract of C monogyna leaves and flowers. At the beginning of the study, the mice were weighed, then orally administered a single dose of the extract and monitored for 10 h for any signs of apparent toxicity. Over the next 14 days, the mice were closely observed every day for any additional clinical or behavioral indications of toxicity.
Study of the Aqueous Extract's Antihyperglycemic Effect
In order to evaluate the antihyperglycemic properties of the extracts in vivo, the researchers utilized the oral glucose tolerance test, as outlined in a previous study. 51 Normal rats were allocated into 3 groups (n = 6; ♂/♀ = 1). The Control group was given distilled water at a dose of 10 mL/Kg, while the test group was administered an aqueous extract of C monogyna at a dose of 400 mg/Kg. The positive control group received glibenclamide (2 mg/mL). The oral glucose tolerance test was carried out as follows: Blood glucose measurements were taken at t0, immediately following the administration of the test product (distilled water, aqueous extract of C monogyna, or glibenclamide), and again after 30 min. Following this, the rats were overloaded with D-glucose (2 mg/Kg), and the change in blood glucose levels was monitored at 60, 90, and 150 min.
Study the Inhibitory Effect of the Aqueous Extract of C monogyna on the Activity of Pancreatic α-Amylase
In Vitro Test
To determine the aqueous extract's impact on α-amylase activity, the method outlined by Laaraj et al
83
was followed with some modifications. Firstly, 200 µL of C monogyna extract (0.89, 0.45, 0.22, 0.11, 0.06 mg/mL) or acarbose solution (1, 0.8, 0.6, 0.4, 0.2 mg/mL) was added to 200 µL of phosphate buffer solution (0.1 M, pH 6.9). Then, 200 µL of enzyme solution (3 UI/mL; E.C.3.2.1.1) was added to all tubes, except for the blank, which received phosphate buffer instead of enzyme solution. Following a 10-min preincubation at 37 °C, 200 µL of starch solution (1%) was added to each tube. After a 15-min incubation at 37 °C, 600 µL of 3,5-dinitrosalicylic DNSA reagent (prepared by dissolving 1 g of DNSA in 20 mL of 2N NaOH, followed by combining it with 30 g of KNaC4H4O6, and the final volume was adjusted to 100 mL with distilled water), was added to stop the enzymatic reaction. The tubes were then boiled for 8 min and placed in an ice water bath before 1 mL of distilled water was added to each tube. The absorbance was measured at 540 nm using a spectrophotometer against a blank containing only buffer solution. The percentage inhibition was calculated using the following formula:
In Vivo Test
Fasting rats (14 h) with a normal weight range of 180 to 250 g were randomly assigned into 3 groups (n = 6; ♂/♀ = 1). The control group was administered with 10 mL/Kg of distilled water, the test group was given 400 mg/Kg of the decoctate, and the positive group was treated with 10 mg/mL of acarbose. The oral starch tolerance test was conducted by measuring the blood glucose levels at t0, just before administering the test material (either distilled water, aqueous extract of C monogyna, or acarbose). After 30 min, another blood glucose measurement was performed, and then the rats were overloaded with 3 g/Kg of starch. The change in blood glucose was measured at 60, 90, and 150 min after the starch overload.
In silico Testing
Molecular docking analysis was carried out exactly as reported in Kandsi et al. 84 The crystal structure of α-amylase (PDB ID: 1SMD), was retrieved from the RCSB protein data bank (http://www.rcsb.org/pdb) (accessed on 19 November 2022) set up at Brookhaven National Laboratory in 1971. Water molecules were removed using AutoDock Tools, and polar hydrogens and Kollman charges were added. The co-crystallized ligands were also removed, and the protein was stored in the “pdbqt” format. Ligands were downloaded in “sdf 3D” format from (https://pubchem.ncbi.nlm.nih.gov/) (accessed on November 19, 2022), then converted to a “pdb” file using PyMol. Using AutoDock Tools (version 1.5.6), the ligand final pdbqt file was obtained. To best fit, the active binding site, the grid box used to depict the docking search space was enlarged. The grid box coordinates are as follows, the centers (x, y, and z), 8.349; 58.705; 19.096, and a grid box size of 40. The results for the docked ligand complexes were expressed as kcal/mol values for the ΔG binding energies. Acarbose was the native ligand used in this computational part of the study. The creation of two-dimensional molecular interaction diagrams as well as the analysis of protein-ligand binding interactions was done using Discovery Studio 4.1 (Dassault Systems Biovia, San Diego, CA, USA).
Statistical Analysis
The data was presented as the mean value along with the standard mean error. The statistical analysis of the data was conducted using one-way analysis of variance followed by the Turkey post-test, with the help of Graph Pad Prism 5, San Diego, CA, USA. A P-value of less than .05 was considered statistically significant.
Footnotes
Acknowledgments
The authors extend their appreciation to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R141), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. Thankful also to Researchers Supporting Project number (RSP2023R379), King Saud University, Riyadh, Saudi Arabia.
Data Availability Statement
All the data supporting the findings of this study are included in this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R141), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. Thankful also to Researchers Supporting Project number (RSP2023R379), King Saud University, Riyadh, Saudi Arabia.
Ethical Approval
This research was carried out in accordance with the principles and guidelines outlined in the Declaration of Helsinki. The study protocol was reviewed and approved by the Institutional Review Board of the Faculty of Sciences, Oujda, Morocco, under the reference numbers 01/20-LBBEH-04 and 09/01/2020.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
