Abstract
Objective
Heliciopsis terminalis (Kurz.) Sleum (family Proteaceae) is distributed in India, Mianma, Thailand, Cambodia, Vietnam, and China. In Vietnamese traditional medicine, it is used as an antidote for detoxifying the liver and detoxifying alcohol, a contraceptive, and to treat infections. The purpose of this study is to determine the chemical constituents of the plant, as well as their in vitro hepatoprotective and antioxidant activities.
Methods
The dried powdered sample was ultrasonically extracted with MeOH, and the extract was fractionated by various chromatographic methods, including high-performance liquid chromatography. The structures of the isolated compounds were identified by NMR, high-resolution electrospray ionization mass spectrometry, and CD spectral analysis, and then screened for their in vitro hepatoprotective and antioxidant activities.
Results
One new phenolic glycoside, named helitermioside (1), and eight known compounds (2-9) were isolated from the trunk and branches of H terminalis. All of the isolates were reported from H terminalis for the first time. Compound 6 showed antioxidant activity in thiobarbituric acid reactive substances (TBARS) lipid peroxidation inhibition assays, with an IC50 value of 61.16 ± 1.17 µg/mL, compared to that of Trolox (positive control), IC50 = 7.06 ± 0.11 µg/mL. At a concentration of 100 µg/mL, none of the isolates exhibited hepatocellular protective activity.
Conclusion
Nine phenolic compounds (1–9) were recorded from H terminalis. Of these, compound 1 was new. Compound 6 exhibited moderate antioxidant activity in TBARS lipid peroxidation inhibition assays, but none of the compounds exhibited hepatocellular protective activity against CCl4-induced toxicity.
Introduction
Heliciopsis terminalis (Kurz.) Sleum (Proteaceae) grows wild in Vietnam. The wood of this plant has the effect of detoxifying the liver and detoxifying alcohol, and is used as a contraceptive and to treat infections in traditional medicine in Vietnam. 1 In previous papers, several macrocyclics and phenolic glycosides have been reported for H terminalis and H lobata (Merr.) Sleum, along with their in vitro antioxidant and hepatoprotective activities.2-5 The wood of H lobata and H terminalis are very similar, so according to folk experience, these materials are used interchangeably in the treatment of liver diseases without distinguishing them. Therefore, the wood of H terminalis was chosen for further study of its in vitro antioxidant and hepatoprotective activity. In this paper, we report one new (1) and eight known phenolics (2–9) from H terminalis, and their hepatoprotective and antioxidant effects.
Results and Discussion
Helitermioside (1) was isolated as a colorless powder. In the IR spectrum of
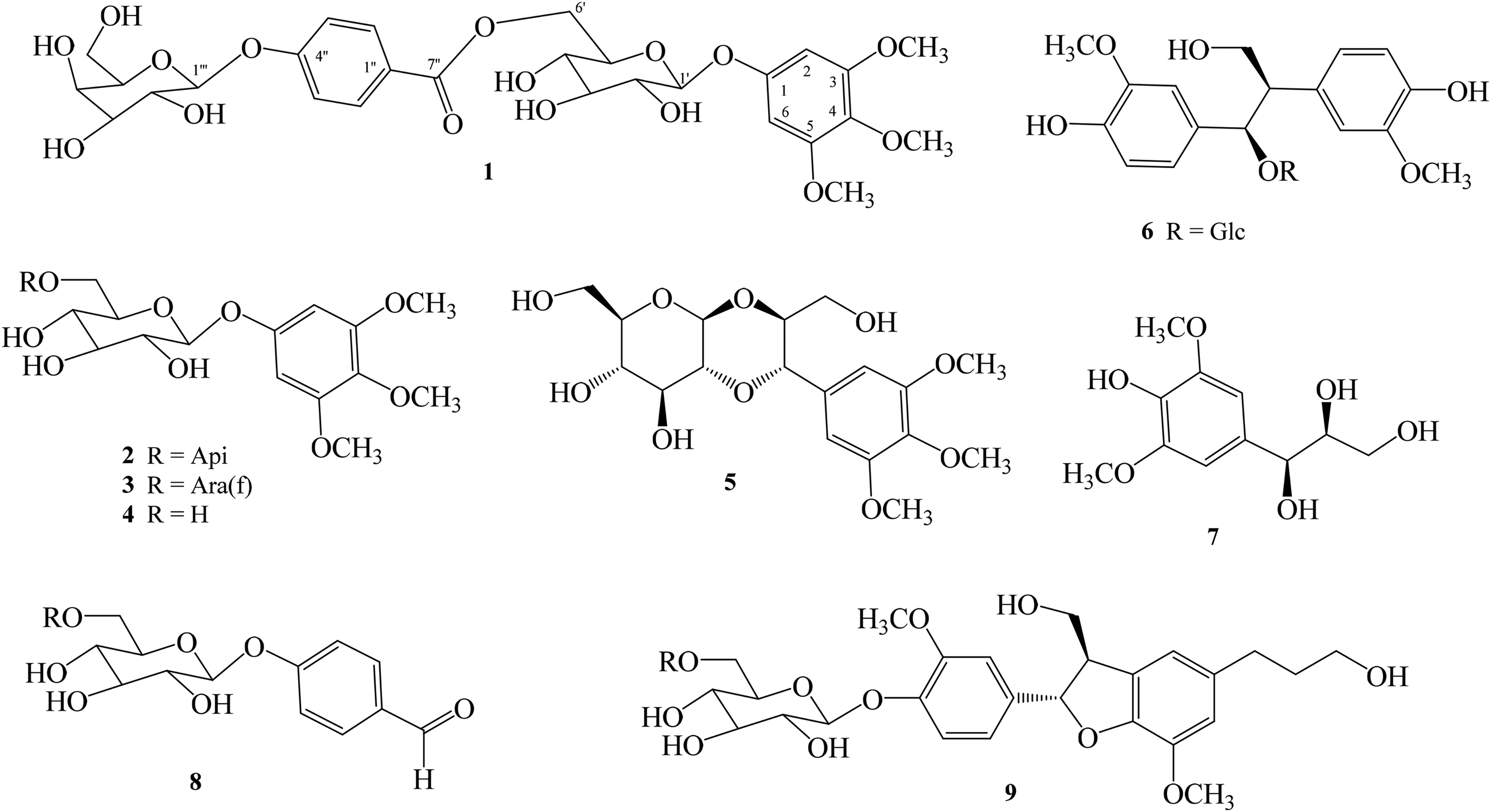
Chemical structures of 1–9.
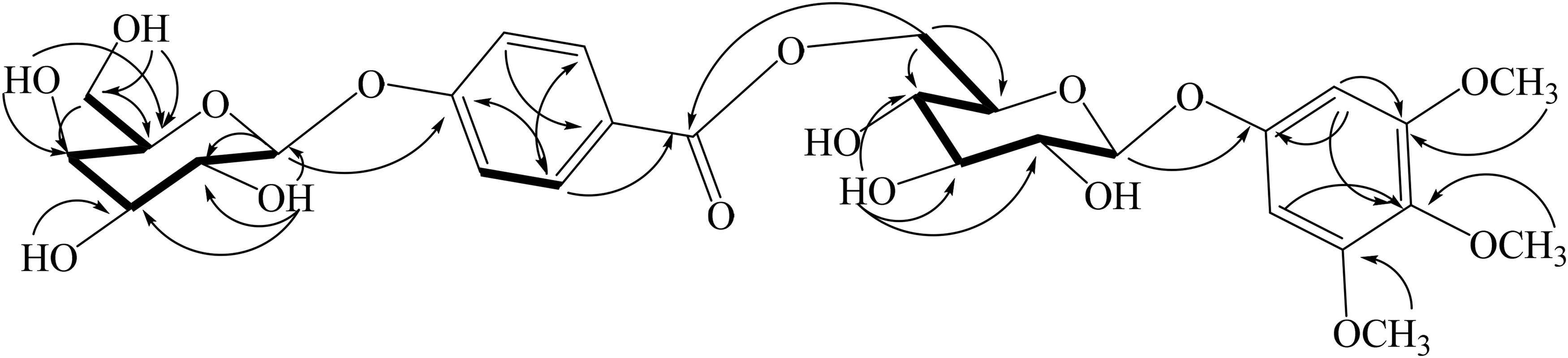
Key HMBC and COSY correlations of 1. Abbreviations: COSY, correlation spectroscopy; HMBC: heteronuclear multiple bond correlation.
1H NMR and 13C NMR Spectroscopic Data for 1 in DMSO-d6.

a125 MHz, b500 MHz.
The other compounds were identified as 34,5-trimethoxyphenyl 1-O-β-D-apiofuranosyl-(1ʹʹ→6ʹ)-β-D-glucopyranoside (2), 10 rhyncoside C (3), 11 34,5-trimethoxyphenyl 1-O-β-D-glucopyranoside (4), 12 ficuscarpanoside B (5), 13 hovetrichoside A (6), 14 threo-syringylglycerol (7), 15 helicide (8), 16 and (7R,8S)-dihydrodehydrodiconeferyl alcohol 4-O-β-D-glucopyranoside (9) 17 by comparisons of their physical and NMR data (including CD spectral data for 9) with those of reported data. This is the first isolation of 2–9 from H terminalis.
Compounds 1–9 were tested for their hepatoprotective and antioxidant activities. Compound 6 showed moderate antioxidant activity in thiobarbituric acid reactive substances (TBARS) lipid peroxidation inhibition assays with an IC50 value of 61.16 ± 1.17 µg/mL compared to the IC50 value of 7.06 ± 0.11 µg/mL of Trolox (Supplemental Tables S1 and S2). None of the isolates showed hepatocellular activity against the toxic induction caused by CCl4, as the viability of cells was lower than that of the blank compound, DMSO.
Materials and Methods
General
Refer to Supplemental Material. 5
Plant Materials
The H terminalis (Kurz.) Sleum sample (coded: HTG0821) was harvested in December 2021 at BacKan, Vietnam and identified by Dr ND Trong (Hanoi University of Pharmacy).
Extraction and Isolation
H terminalis trunk and branches (10 kg, dried powder) were extracted ultrasonically by MeOH (3 times, each 30 L, 60 min) to obtain the extract (120 g). This was suspended in H2O and partitioned successively with EtOAc to obtain the EtOAc extract (32.0 g) and water layer (HTW). The HTW was fractionated on a diaion column, eluted by MeOH/H2O (1/4, 1/1, 4/1, and 1/0 each 3.0 L) to get four fractions, HTN1–HTN4. HTN2 (8.2 g) was separated on a silica gel column, eluting with CH2Cl2/MeOH (gradient 10/1–3/1) to obtain 3 fractions, HTN2A-HTN2C. HTN2A (430 mg) was fractionated by high-performance liquid chromatography (HPLC), eluting with MeOH in H2O (1/6) to give 8 (10.6 mg, tR 6.5 min) and 7 (10.6 mg, tR 7.8 min). HTN2B (1.2 g) was purified by HPLC eluting with MeOH in H2O (1/6) to give 5 (83.1 mg, tR 13.5 min). HTN2C (220 mg) was purified by HPLC eluting with MeOH in H2O (1/5) to yield 4 (7.5 mg, tR 8.5 min). HTN3 (9.6 g) was fractionated on a silica gel column, and eluted with CH2Cl2/MeOH (gradient 20/1–4/1) to obtain 3 fractions, HTN3A-HTN3C. HTN3A (310 mg) was purified by HPLC, eluting with MeOH in H2O (1/6) to give 3 (4.2 mg, tR 16.2 min). HTN3B (250 mg) was treated by HPLC eluting with MeOH in H2O (1/6) to yield 3 (7.5 mg, tR 18.0 min). HTN3C (240 mg) was purified by HPLC eluting with MeOH in H2O (1/5) to give 2 (7.5 mg, tR 22.3 min). HTN4 (3.5 g) was isolated using a silica gel column by eluting with CH2Cl2/MeOH (10/1) to give 2 fractions, HTN4A and HTN4B. HTN4B (510 mg) was purified by HPLC, eluting with MeOH in H2O (1/4) to give 1 (35.3 mg, tR 24.6 min) and 9 (21.3 mg, tR 28.4 min).
Helitermioside (1)
Colorless powder,
Antioxidant and Hepatoprotective Assay
See supporting information.
Acid Hydrolysis
See supporting information.
Conclusions
In this study, one new compound, named as helitermioside (1), and eight phenolics (2–9) were recorded from H terminalis. Compound 6 exhibited moderate antioxidant activity in TBARS lipid peroxidation inhibition assays, but compounds 4, 6, 8, and 9 exhibited moderate hepatocellular protective activity against CCl4 toxic induction.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231174993 - Supplemental material for Hepatoprotective and Antioxidant Activities of Phenolic Compounds from Heliciopsis terminalis
Supplemental material, sj-docx-1-npx-10.1177_1934578X231174993 for Hepatoprotective and Antioxidant Activities of Phenolic Compounds from Heliciopsis terminalis by Bui V. Trung, Duong H. Anh, Pham H. Viet and Phan Van Kiem in Natural Product Communications
Footnotes
Author Contribution
Research idea was conceived by BVT and PHV; isolation, bioactivity assay were performed by BVT and DHA; structure elucidation and writing were handeled by BV Trung and PV Kiem.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
