Abstract
Materials and Methods
Materials
Chemicals and Reagents
Standards of myricitrin (Mytr) and myricetin-3-
Preparation of Isolated Fractions
Based on the references,
26,27
a macroporous adsorption resin column was used to separate the water extract and prepare the active fractions of
Determination of Phenolic Contents
The phenolic contents of Fr1-Fr7 were determined following the published method, with some changes.
13
At first, Fr1-Fr7 at 1 mg/mL and the various concentrations of gallic acid (1.3-9.2 μg/mL) were separately prepared in 50% ethanol solution. Then, 1 mL of each sample solution was added to a mixture of 1 mL of Folin-Ciocalteu reagent (0.1 N) and 10 mL of 50% ethanol solution and incubated in the dark at RT for 5 minutes. Afterward, 10 mL of 7% sodium carbonate solution was added to the mixture, which was diluted to 25 mL with a 50% ethanol solution. Subsequently, the mixture was incubated in the dark at RT for 30 minutes. Finally, the absorbance of the mixture was tested at 760 nm using an ultraviolet (UV) spectrophotometer (CARY 50, Varian, USA). The phenolic content in the sample was expressed in terms of gallic acid and calculated from the standard curve equation (
DPPH-Scavenging Assay
DPPH-scavenging activities of Fr1-Fr7 were determined following the reported procedure. 28 Briefly, the various concentration solutions (1-350 μg/mL) of Fr1-Fr7 were prepared in 50% ethanol, and the DPPH solution of 0.1 mM was made in ethanol. Then, 2 mL of each sample solution was mixed with 2 mL of DPPH solution, and the mixture was placed in the dark at RT for 30 minutes. Afterward, the absorbance of the mixture was measured at 517 nm using a UV spectrophotometer (CARY 50, Varian, USA). Vc was used as positive control. The DPPH scavenging rate by the sample was calculated using the following equation:

where
A dose-effect relationship curve was established based on the sample concentration and the scavenging rate, and the concentration of the sample at the scavenging rate of 50% (IC50) was determined from the curve.
The same method was used to determine the IC50 of Mytr, Myt, Myt-Glc, Myt-Gal, and Fr5 on the DPPH-free radical.
Nitrite-Scavenging Assay
The nitrite-scavenging activities of Fr1-Fr7 were determined according to the reported method.
14
Briefly, the various concentration solutions (0.1-4 mg/mL) of Fr1-Fr7 were prepared in 50% ethanol, and sodium nitrite (NaNO2) solution of 5 mg/L was prepared in water. Each sample solution of 1 mL was mixed with 1 mL of NaNO2 solution and 1 mL of citrate buffer (pH = 3, 0.5 M), and then the mixture was incubated for 30 minutes in a water bath (37 °C). Next, 1 mL of 4 g/L amino benzene sulfonic acid sodium (in 20% hydrochloric acid) was added to the mixture; 3 minutes later, 0.5 mL of 2 g/L hydrochloric acid naphthalene ethylenediamine (in water) was also added. After another 15 minutes, the absorbance (

A dose-effect relationship curve was established based on the sample concentration and the scavenging rate, and the concentration of the sample at the scavenging rate of 50% (IC50) was determined from the curve.
The same method was used to determine the IC50 of Mytr, Myt, Myt-Glc, Myt-Gal, and Fr5 on nitrite.
Inhibition of NAs Formation Assay
Formation of NAs under simulated gastric juice conditions
The inhibition effects of Fr1-Fr7 on NDMA and NDEA formation were studied in vitro simulated gastric juice according to the published method. 29 The various concentration solutions (1.5-30 mg/mL) of Fr1-Fr7 were prepared in dimethyl sulfoxide (DMSO). Briefly, 0.5 mL of dimethylamine hydrochloride (0.4 M) and 0.5 mL of NaNO2 (0.1 M) were added to 7 mL of sodium citrate-hydrochloric acid buffer (pH = 3, 0.1 M), and the resulting mixture was called the original synthetic solution of NDMA. Then, 2 mL of each sample solution was added to the original synthetic solution of 8 mL to obtain a reaction solution of the sample (Fr1-Fr7); 2 mL of each sample solution was added to 7 mL of sodium citrate-hydrochloric acid buffer (pH = 3,0.1 M) and 1 mL water to obtain the blank solution of the sample (Fr1-Fr7); 2 mL of DMSO was added to 8 mL of the original synthetic solution to obtain the control reaction solution. Afterward, the reaction solution, blank solution of sample, and the control reaction solution were incubated in a water bath (37 °C) for 4 hours, after which the reaction was quenched by adding ammonium sulfamate (500 mg). Vc was used as the positive control. For the synthesis of NDEA, 0.5 mL of diethylamine hydrochloride (0.8 M) and 0.5 mL of NaNO2 (0.2 M) were added to 7 mL of sodium citrate-hydrochloric acid buffer (pH = 3,0.1 M) as the original synthetic solution. The other steps were the same as in the NDMA formation experiment.
Determination of NAs
The resulting NDMA and NDEA in the above final solutions were extracted with dichloromethane following the published method. 13 NaCl (5 g) was added to the final solution, and then the mixture was extracted twice with 20 mL of dichloromethane each time. The dichloromethane solution was dried with sodium sulfate and filtered. The sodium sulfate was washed twice with dichloromethane. Next, the dichloromethane solutions were combined and concentrated to 5 mL with a KD concentrator and filtered through a 0.45 µm syringe filter. The peak area of NDMA and NDEA in the dichloromethane-concentrated solution and standard solutions (0.5-10 μg/mL in dichloromethane) were tested on a high-performance liquid chromatography (HPLC) system (Agilent 1260, Agilent, USA) equipped with a Zorbax XDB C18 reversed-phase column (250 mm × 4.6 mm, 5 µm), according to the reported method. 30 HPLC parameters were set as follows: injection volume 10 µL, flow rate 1 mL/min, column temperature 32 °C and detection wavelength of 230 nm. The mobile phase was methanol/water (10:90 V/V).
The peak area was plotted against the concentration of NDMA and NDEA to generate a standard curve and obtain the standard curve equation:

where
A dose-effect relationship curve was established based on the sample concentration and the inhibition rate, and the concentration of the sample at the inhibition rate of 50% (IC50) was determined from the curve.
The same method was used to measure the inhibition rate of Mytr, Myt, Myt-Glc, Myt-Gal, and Fr5 at concentrations of 0.5, 1, and 2 mg/mL on NDMA formation.
Analysis and Identification of the Active Ingredients in Fr5 by LC-MS and HPLC
The reaction solution and blank solution of Fr5 were prepared according to the method described in the “Inhibition of NAs Formation Assay” section. The stock solution of Fr5 was prepared in chromatography grade methanol. The concentrations of the above 3 solutions of Fr5 were kept the same.
LC-MS analysis
The 3 solutions of Fr5 were filtered through a 0.22 µm syringe filter and analyzed using an LC system (Dionex UltiMate 3000, Dionex, USA) equipped with a Hypesil GOLD column (100 mm × 2.1 mm, 1.9 µm) and a diode array detector (DAD). The analysis was carried out by gradient elution with a mobile phase of 0.1% formic acid and methanol mixture at a flow rate of 0.2 mL/min. The percentage of methanol in the mobile phase was programmed as follows: 11% (0 minutes)-11% (2 minutes)-15% (6 minutes)-20% (10 minutes)-26% (13 minutes)-60% (15 minutes). The other experimental conditions were: injection volume 2 µL, column temperature 25 °C, and detection wavelength 210‐400 nm.
Then, the chromatograms of the 3 solutions were compared to discover peaks of the active ingredients that inhibited the formation of NAs. Finally, the possible active ingredient peaks were analyzed by a Thermo Scientific Q Exactive (Thermo, USA) equipped with an electrospray ionization (ESI) source. ESI-MS was applied for qualitative analysis under negative ion (MS) mode. The parameters were set as: mass scan range
HPLC analysis
Standards of Mytr, Myt, Myt-Gal, and Myt-Glc were used to verify the qualitative results of liquid chromatography-mass spectrometry (LC-MS) and to measure their contents in Fr5 and
Mixed standard solutions (1-2000 μg/mL) of Mytr, Myt, Myt-Gal, and Myt-Glc were prepared in chromatography grade methanol. Fr5 was dissolved in methanol of HPLC grade. The
The above standard and sample solutions were filtered through a 0.45 µm syringe filter and analyzed by HPLC (Agilent 1260, Agilent, USA) equipped with a Tnature C18 column (250 mm × 4.6 mm i.d., 5 µm), according to the method established in our laboratory, with slight modification. 31 In brief, the analysis was carried out by gradient elution with a mobile phase of 0.05% phosphoric acid and acetonitrile mixture at a flow rate of 1 mL/min. The percentage of acetonitrile in the mobile phase was programmed as follows: 13% (0 minutes)-18% (20 minutes)-18% (33 minutes)-20% (40 minutes)-20% (45 minutes)-22% (47 minutes)-22% (60 minutes). The other experimental conditions were: injection volume 10 µL, column temperature 25 °C, and detection wavelength 360 nm.
The contents of the 4 active ingredients in Fr5 and
Statistical Analysis
Statistical analysis was conducted with SPSS 22.0 software (USA), and the experimental results were expressed as means ± SD of triplicate measurements. Values of
Results and Discussion
The Phenolic Contents of Isolated Fractions
The Folin-Ciocalteu method was used to determine the phenolic contents of Fr1-Fr7 (Table 1), which were significantly different (
The Phenolic Contents, DPPH-Scavenging, Nitrite-Scavenging, and Inhibition of NAs Formation Activities of Fr1-Fr7.

Abbreviations: Abbreviations: DPPH, 1,1-diphenyl-2-trinitrophenyl hydrazine; IC50, half-maximal inhibitory concentration; NDEA,
Different letters of a, b, c, d, e, f, g, h in the same column indicate the significant difference (
Antioxidant, Nitrite Scavenging, and NAs Formation Inhibitory Activity of Isolated Fractions
The antioxidant activities of Fr1-Fr7 were measured using the DPPH-scavenging assay.
33
As shown in Table 1, the 7 isolated fractions presented DPPH-free radical scavenging capacity to some extent, with IC50 values from 2.8 ± 0.1 to 89.9 ± 1.1 µg/mL. Fr5 showed the strongest scavenging ability, followed by Fr4, Fr2, Fr6, Fr7, Fr3, and Fr1. This order is consistent with the sequence of the phenolic content of the isolated fractions. Thus, the extracts from
The nitrite-scavenging activities of Fr1-Fr7 are shown in Table 1. Fr1 had no significant effect on nitrite within the measured concentration range. Fr2-Fr7 had a certain scavenging effect; their effects were proportional to their phenolic contents and scavenging activities on the DPPH-free radical. These are consistent with the reported results.
14,15
These studies also found that the nitrite-scavenging activities of the phenolics were more closely related to their antioxidant activities. In this study, the scavenging activity of Fr5 on nitrite, with an IC50 value of 47.8 ± 1.1 µg/mL, was significantly greater than those of other fractions (
The stomach is the main organ for the formation of NAs in the human body.
11
NDMA and NDEA are the most common types of NAs and are classed as 2A group carcinogens by IARC.
34
In the study, samples were added to the original synthetic solution of NDMA and NDEA to evaluate their inhibitory effect on the formation of NDMA and NDEA in simulated gastric juice conditions. The contents of NDMA and NDEA in the control reaction and sample reaction solutions were determined by HPLC. The chromatographic peaks of NDMA and NDEA had good symmetry and perfect separation under the detection conditions. As shown in Table 1, the effects of Fr1-Fr7 on the formation of NDMA and NDEA were quite different (
All of the above results indicated that the capacities of antioxidant, nitrite scavenging, and inhibition of NAs formation of Fr1-Fr7 were significantly related to their phenolic contents. Fr5 was the main active fraction and the phenolics were the main chemicals for these activities.
Active Ingredients in Fr5
The chromatograms of the reaction, blank, and stock solutions of Fr5 were compared to discover the active ingredients in Fr5 that inhibited the synthesis of NAs. The chromatograms of Fr5 blank and stock solutions showed no difference, which indicated that the composition of Fr5 had not significantly changed in the system for the synthesis of NAs. However, the chromatographic peaks of the 5 ingredients (A, B, C, D, and E) in the Fr5 reaction solution were significantly decreased when compared with the corresponding peaks in the other 2 solutions. This suggested that the 5 ingredients played a major role in the inhibition of NAs synthesis by Fr5.
LC-MS was used for qualitative analysis of the 5 ingredients. By comparison with reference compounds and literature data, A was identified as Mytr,
35
B as Myt,
36
C as Myt-Gal,
36
D as Myt-Glc,
37
and E as myricetin-3-
The contents of Mytr, Myt, Myt-Gal, and Myt-Glc in Fr5 and
Active Ingredients and Their Contents in Fr5 and

Abbreviations: Abbreviations: DB, dry basis; MSn, tandem mass spectrum; MW, molecular weight; Myt, myricetin; Myt-Ara, myricetin-3-
λmax is the maximum absorption wavelength.
Contents (mg/100 g DB) indicates the contents of the compound in Fr5 or
The Activities of Mytr, Myt, Myt-Gal, and Myt-Glc
The effects of Mytr, Myt, Myt-Gal, and Myt-Glc on DPPH-free radical, nitrite, and NDMA formation were tested. As shown in Table 3, the IC50 of Myt and Mytr on the DPPH-free radical and nitrite was significantly lower than those of Myt-Glc, Myt-Gal, and Fr5 (
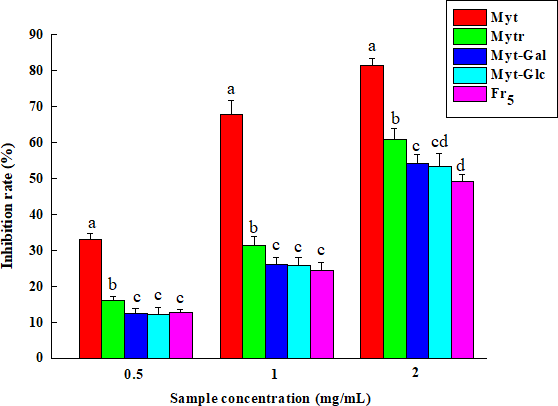
The inhibition rate of Fr5, myricitrin (Mytr), myricetin (Myt), myricetin-3-
The DPPH-Scavenging and Nitrite-Scavenging Activities of Myt, Mytr, Myt-Gal, and Myt-Glc.
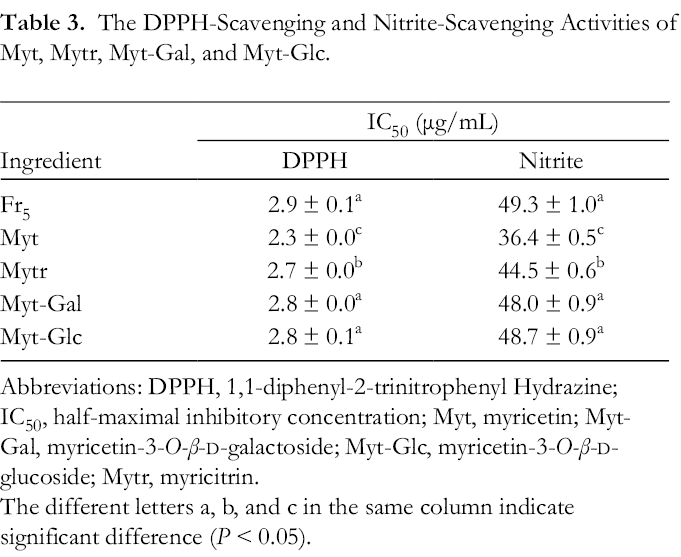
Abbreviations: Abbreviations: DPPH, 1,1-diphenyl-2-trinitrophenyl Hydrazine; IC50, half-maximal inhibitory concentration; Myt, myricetin; Myt-Gal, myricetin-3-
The different letters a, b, and c in the same column indicate significant difference (
Conclusion
In this study, the effects of the phenolic extracts from
Supplemental Material
Supplementary Material 1 - Supplemental material for Nitrite Scavenging and Inhibition of N -Nitrosamines Formation by Phenolic Extracts From Diospyros lotus L. Leaves and Active Ingredients
Supplemental material, Supplementary Material 1, for Nitrite Scavenging and Inhibition of
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Department of Science and Technology (the Key Projects of Shanxi Province Key R&D Program, China) (Nos. 201603D14010 and 201603D3114015).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
