Abstract
Fungal bioactive secondary metabolites are considered promising sources for the production of unique compounds with exceptional chemical structures. Of these bioactive secondary metabolites, meleagrins are prenylated indole alkaloids characterized by a triazaspirocyclic skeleton that is generally obtained from Penicillium species. Meleagrins are alkaloids with promising biological activities. Here is a survey of previously published research on meleagrins: their sources, biosynthetic pathways, synthesis, and bioactivities.
Introduction
Fungi isolated from marine environments are recently considered as potential sources for the production of unique bioactive secondary metabolites leading to the isolation of lead compounds for drug discovery.1,2 Fungal secondary metabolites have exhibited significant applications in agriculture and the pharmaceutical industry, as well as the food industry, and have played a crucial economical role.1,3-5 Penicillium spp are the most known producers of biologically active byproducts.6,7 Meleagrins are considered as one of the most reported active compounds obtained from Penicillium spp8,9 (Figure 1).
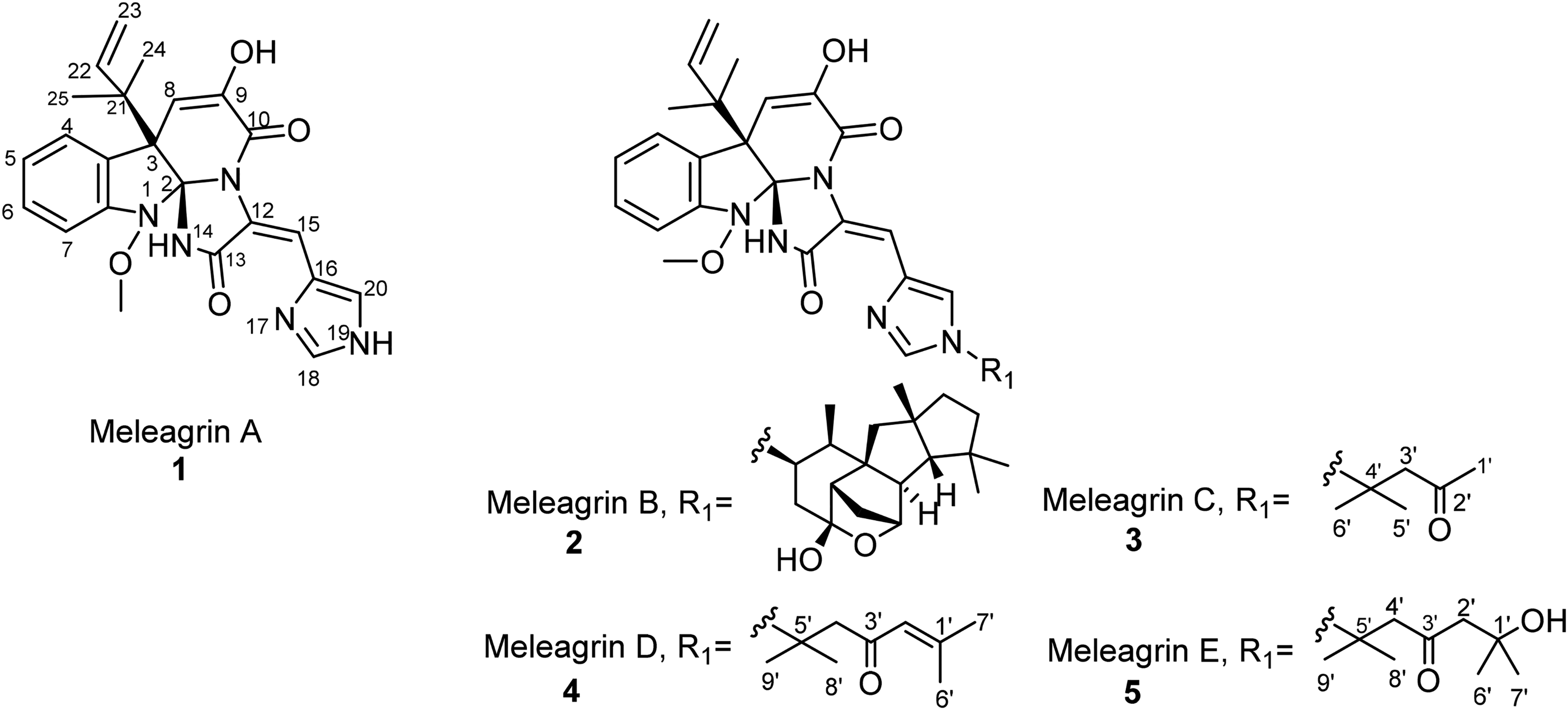
Chemical structure of meleagrins. doi: 10.1002/chem.201501150. 10
Meleagrin A (
Meleagrins have been detected in over 30 Penicillium spp (Table 1). Further, meleagrin A (
Penicillium spp. as Sources of Meleagrins.

Meleagrins possess various biological characteristics including inhibition of the biosynthesis of bacterial type II fatty acid,22,31 antiproliferative activity,9,13,29,30 inhibiting the polymerization of tubulin in Jurkat cells, and a protection effect against bleomycin-induced pulmonary fibrosis in mice. 11
In the present review, we describe the different sources of meleagrins, their biosynthetic and synthetic pathways, as well as their biological activities for the first time.
Biosynthetic Pathway of Meleagrins
Enzymes catalyze the biosynthesis of secondary metabolites in fungi. Gene clusters encode these enzymes, which are frequently coregulated at the transcriptional level. 32 Gene clusters can be created by de novo assembly of specific genes from other cellular functions. Still, new gene clusters are also formed through the reorganization of progenitor clusters and distributed by horizontal gene transfer. 32
Roquefortine/Meleagrin Gene Cluster
The roquefortin/meleagrin gene cluster in the Penicillium chrysogenum genome underwent a series of RNA silencing experiments, which enabled a putative biochemical mapping of each step of the biosynthetic pathway.33,34
García-Estrada et al investigated the biosynthetic pathway of the mycotoxins roquefortine C/meleagrin originated from a single path in P. chrysogenum through a single gene cluster. 20 This cluster includes Pc21g15480, Pc21g15430, Pc21g15460, Pc21g15470, Pc21g15450, and Pc21g15440 genes (Scheme 1).

Roquefortine C/meleagrin pathway in Penicillium chrysogenum. The arrow with the symbol (×) refers to the silenced steps.
Pc21g15430 (rpt) encodes the prenyltransferase enzyme required for the biosynthesis of roquefortine C (
Driessen and his coworkers described the biosynthetic process used in the filamentous fungus P. chrysogenum to produce meleagrin A and other correlated chemical compounds. 21 Histidyl-tryptophanyl-diketopiperazine (HTD), dehydro-histidyl-tryptophanyl-diketopiperazine (DHTD), roquefortine D, roquefortine C, glandicoline A, and glandicoline B were detected as abundant metabolites of this route.
Each stage of an enzymatic reaction was assigned by a specific set of genes. L-tryptophan (

Roquefortin/meleagrin biochemical pathway for meleagrin biosynthesis.
In another publication, Driessen and his coworkers endorsed the roquefortin C/meleagrin biochemical pathway for meleagrin A (
Oxaline Biosynthetic Gene Cluster
Sherman and his coworkers reported the Oxa gene cluster from the fungus Penicillium oxalicum F30 alongside the representation of OxaD, a flavin-dependent oxidase that creates roquefortine L (15). Oxa D is considered as a nitrone-bearing intermediate in the biogenesis of meleagrin A (1) and then the biogenesis of oxaline (17) (Scheme 3).19,36

The Oxa gene cluster from the fungus Penicillium oxalicum F30.
Acetate–mevalonate Pathway
Du et al postulated a biosynthetic pathway of meleagrin D (

Biosynthetic pathway of meleagrins D and E.
Synthetic Pathway of Meleagrin A (1 )
Sunazuka and his coworkers synthesized meleagrin A (
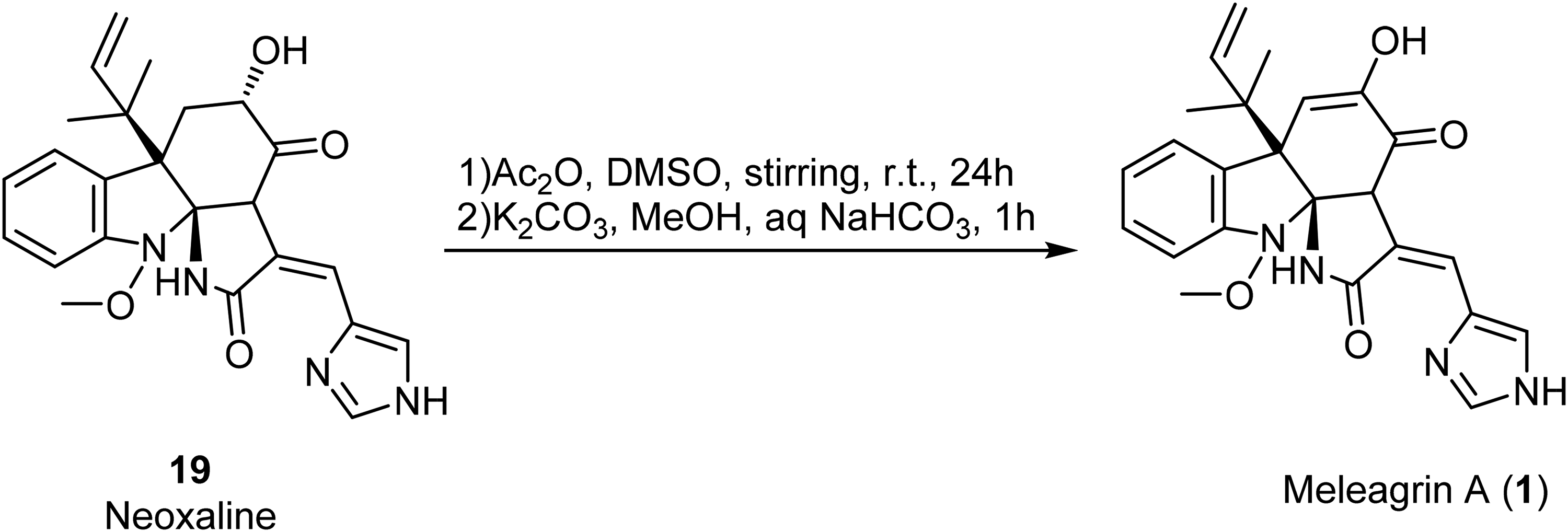
Synthesis of meleagrin A (
Meleagrin A (1 ) Derivatives
Some meleagrin derivatives were obtained via chemical modification of some functional groups such as the hydroxyl and amine groups of meleagrin A (Scheme 6).
22
As a result of the demethoxylation of compound

Some derivatives of meleagrin A (
Biological Activities
Meleagrin A (
On the other hand, meleagrin A (
In another publication, the efficacy of meleagrin A (
Additionally, meleagrin A (
Meleagrin A (
Subsequent studies were conducted to investigate the influence of meleagrin on the life cycle of A. amphiplanus as a causative agent of marine fouling. 40 Fifty proteins have been characterized by applying isobaric tags for relative and absolute quantitation coupled with 2D LC−MS/MS proteomic analysis to investigate the effect of meleagrin on the proteomic expression profile of cyprid development and aging in the barnacle Balanus amphitrite. It was found that 50 proteins of B. amphitrite were differentially expressed in response to meleagrin treatment: 26 proteins were associated with development/aging in the cypris larvae, and, of those, 24 were associated specifically with meleagrin treatment. 40 Analysis of data from the Kyoto Encyclopedia of Genes and Genomes indicated that meleagrin affected several pathways, including metabolic pathways, extracellular matrix–receptor interactions, and the regulation of the actin cytoskeleton. Among the 24 proteins affected by meleagrin treatment, a vitellogenin-like protein was upregulated, indicating endocrine disruption and prevention of the larval molting cycle. With the exception of mediation of the molting process involving chitin-binding proteins and adenosine triphosphatase (ATPase)-mediated energy, most of the major proteins in this study were not found to be significant in butenolide and polyether B-treated larvae, suggesting that meleagrin exhibits a distinct and specific mode of action. 40
Meleagrin A (
Meleagrin A (
Moreover, meleagrin A (
In 2021, Hamed et al reported that meleagrin A (1), obtained from E. dentata Nq45, exhibited effective cytotoxic activity toward KB-3-1, a human cervix carcinoma (IC50 = 3.07 μmol/L), besides its activity against multidrug-resistant subclone (IC50 = 6.07 μmol/L). 13
In two successive studies by Du et al in 2009 and 2010, concerning the activity of melaegrins A (
Recently, in a study on the effect of meleagrin A (1) against pulmonary fibrosis caused by bleomycin in mice, it was found that meleagrin A (1) has a protective effect. 11 Its effectiveness could be attributable to restoring the oxidant–antioxidant balance through reducing MDA contents and elevating the activities and levels of antioxidant molecules. 11
Conclusion
Meleagrins are bioactive fungal secondary metabolites mainly isolated from Penicillium species. The active components of these species were examined in order to get a deeper understanding of the biosynthetic processes and structure–activity connections in this family of metabolites, which resulted in the isolation of meleagrins. The gene cluster encoding for meleagrin production is a unique pathway in the biosynthesis of these compounds. The gene cluster encoding the production of meleagrins was identified using whole genome sequencing, analysis, and substrate modeling with P. chrysogenum. Through genetic studies within the roquefortin/meleagrin gene cluster in the P. chrysogenum genome, besides RNA silencing experiments, it has been confirmed that there is a putative biochemical mapping for each step of the biosynthetic pathway to reach the production of meleagrins. It can be confirmed by previous biological studies that meleagrin A is a unique alkaloid with promising biological properties, especially its ability to inhibit microbes, including Gram-positive and Gram-negative bacteria. On the other hand, it has the potential to be an anti-cancer agent. Additionally, in a distinct study, its ability to be an anti-fouling agent was demonstrated by a life cycle study of A. amphiplanus as a marine fouling agent without affecting its life cycle. This suggests that meleagrin A has no toxic effect.
Footnotes
Author Contributions
Eslam R. El-Sawy and Gilbert Kirsch: contributed to the conception of the study, collected literature, and revised it critically; Eslam R. El-Sawy: evaluated the literature and wrote the initial draft; Mohamed S. Abdel-Aziz: helped perform the analysis with constructive discussions; and Eslam R. El-Sawy, Mohamed S. Abdel-Aziz, and Gilbert Kirsch: contributed to writing the final draft and corrected the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
