Abstract
Twenty-one novel luotonin derivatives were synthesized from 2-aminobenzamide in excellent yields. The prepared compounds, when tested against 8 strains of bacteria and many plant pathogen fungi showed diverse and promising antimicrobial activities. Notably, compounds
In nature, tricyclic heteroaromatic skeletons are very important moieties that are widespread in a large family of natural products with a broad range of attractive bioactivities. 1 Luotonin A (Figure 1) and derivatives, which possess a novel kind of tricyclic heteroaromatic scaffold, have displayed important biological activities such as anticancer, cytotoxic, and antiproliferative activity. 2,3 Because of this wide scope of biological activities, a great deal of study toward the synthesis and antifungal activities of luotonin A has been reported. 4 -17 Therefore, we focused on structural modification of luotonin A in the hope that a lead compound could be found for the discovery of novel antimicrobial agents. Herein, we report the synthesis and bioactivity of a series of novel luotonin analogs using 2-aminobenzamide as the starting material.

Structure of luotonin A.
To the best of our knowledge, the antimicrobial activities of the synthetic compounds are reported for the first time.
Materials and Methods
Instruments and Chemicals
All chemicals were purchased from commercial sources and purified based on standard procedures before use. Analytical thin-layer chromatography was performed with silica gel plates using silica gel 60 GF254 (Qingdao Haiyang Chemical Co., Ltd., China). The 1H nuclear magnetic resonance (NMR; 500 MHz) and 13C NMR (125 MHz) spectra were obtained on an AM-500 FT-NMR spectrometer (Bruker Corporation, Switzerland) with deuterated chloroform (CDCl3) as the solvent and trimethylsilane as the internal standard. Melting points were taken on an electrothermal digital apparatus (Beijing, China) and were uncorrected. Mass spectra (MS) were recorded under electrospray ionization (ESI) conditions using an LCQ Fleet instrument (Thermo Fisher, Waltham, MA, USA). The title compounds were synthesized under a nitrogen atmosphere. Yields were not optimized.
Synthesis
The general synthetic methods for luotonin derivatives are depicted in Scheme 1.
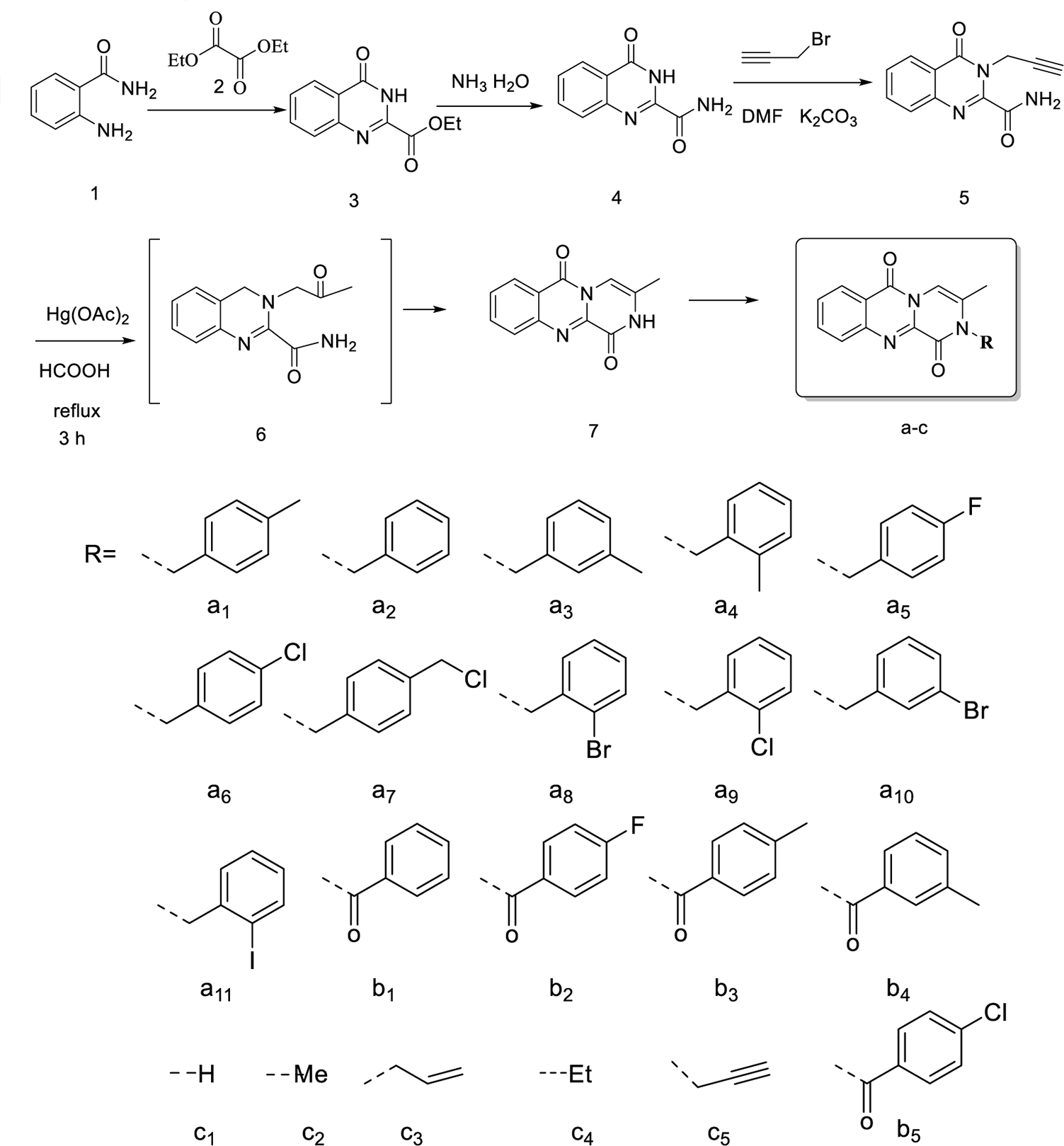
Synthetic route for the title compounds.
Synthesis of compound 3
A mixture of 2-aminobenzamide (2.72 g, 20 mmol) and diethyl oxalate (20 mL) was refluxed at 185.5 °C for 5 hours. The reaction mixture was then allowed to cool to room temperature (r.t). The precipitate was collected by filtration using a Hirsch funnel to give compound
Synthesis of compound 4
Compound
Synthesis of compound 5
To a stirred solution of compound
Synthesis of compound 7
A mixture of crude residue
Synthesis of compounds a1-a11
Compound
Synthesis of compounds b1-b5
A solution of compound
Synthesis of compounds c1-c5
A solution of compound 7 (0.20 g, 0.9 mmol) and potassium carbonate (0.25 g, 1.8 mmol) in N, N-dimethylformamide (10 mL) was stirred at 0 °C for 0.5 hours. The solution was allowed to stir for 2-3 hours at r.t. The corresponding desired reagent was added to the flask and then extracted with dichloromethane (3 × 15 mL). The combined organic extracts were dried over anhydrous sodium sulfate and evaporated under reduced pressure yielding a white solid. All the crude residues were purified by silica-gel column chromatography and evaporated under reduced pressure to provide the desired compounds
3-Methyl-2-(4-methylbenzyl)-2H-pyrazino[2,1-b]quinazoline-1,6-dione (a1)
Yellow solid, m.p., 231-233 °C, Formula: C20H17N3O2 (81% yield), 1H NMR (500 MHz, CDCl3), δ (ppm): 8.38 (dd, J = 1.0 Hz, 8.0 Hz, 1 H, 7 H), 8.07 (d, J = 9.0 Hz, 1 H, 10 H), 7.86 (m, 1 H, 9 H), 7.60 (m, 1 H, 8 H), 7.52 (m, 1 H, 4 H), 7.18 (m, 2 H, 15-CH × 2), 7.12 (m, 2 H, 14-CH × 2), 5.27 (s, 2 H,12-CH2), 2.31 (s, 3 H, 17-CH3), 2,28 (s, 3 H, 3-CH3).13C NMR (125 MHz, CDCl3), δ (ppm): 157.98 (C = O), 157.06 (C = O), 146.91 (10a-C), 139.83 (11a-C), 137.70 (13 C), 134.94 (9-CH), 132.75 (16 C), 129.62 (15-CH × 2), 129.51 (8-CH, 128.28 (10-CH), 127.02 (3 C), 126.99 (14-CH × 2), 125.53 (7-CH), 119.77 (6a-C), 101.60 (4-CH), 47.45 (12-CH2), 21.07 (17-CH3), 17.45 (3-CH3). ESI-MS: [M + H]+ 332.1399 ; found 332.1387.
The data of the other compounds and their NMR spectral details can be found in the Supplemental Material.
Biological Activity
The antimicrobial activity of the N-protected luotonin derivatives was measured according to the previously reported method. 18,19
Minimal inhibitory concentration
Antibacterial activities were evaluated by the micro broth dilution method in 96-well culture plates using Mueller-Hinton broth, according to the National Committee for Clinical Laboratory Standards. 20,21 All compounds were tested in triplicate at each concentration.
Inhibition of spore germination method
All experiments were conducted in triplicate. The half-maximal effective concentration values were determined by performing the bioassay as described above with concentrations of 100, 75, 50, 37.5, and 25 µg/mL, respectively.
Results and Discussion
Design and Synthesis of Luotonin Analogs
The synthetic route is outlined in Scheme 1. Twenty-one luotonin analogs, including the previously prepared
Antimicrobial Activity
All the target compounds were screened for antibacterial activities against 5 Gram-negative (Pseudomonas solanacearum, Pseudomonas aeruginosa, Escherichia coli, Ralstonia solanacearum, and Pseudomonas syringae pv. actinidiae) and 3 Gram-positive bacteria (Staphylococcus aureus, Bacillus subtilis, and B. cereus). The minimum inhibitory concentrations (MICs) were determined by the double-dilution method using penicillin sodium and fosfomycin sodium as the positive controls. The MIC values are summarized in Table 1. The target compounds showed moderate antibacterial activities against S. aureus, E. coli, P. aeruginosa, B. cereus, P. syringae pv. actinidiae, P. solanacearum, and R. solanacearum. Compounds
Antibacterial Activity of Luotonin Derivatives Against B. c., B. s., S. a., E.c., P. a., P. s., P. s. a., and R. s.

Abbreviations: Abbreviations: B. c, Bacillus cereus; B. s., Bacillus subtilis; E. c., Escherichia coli; F, fosfomycin sodium; MIC, minimum inhibitory concentration; P, penicillin sodium; P. a., Pseudomonas aeruginosa; P. s., Pseudomonas solanacearum; P. s. a., Pseudomonas syringae pv. actinidiae; R. s., Ralstonia solanacearum.
Note. P and F were used as positive controls.
The activity of the synthesized compounds toward plant pathogen fungi was also screened, and the results are listed in Table 2. A mycelial growth inhibition assay was utilized, with carbendazim as the positive control, to evaluate the activity of the 21 synthesized N-protected luotonin derivatives against Alternaria alternata, Fusarium oxysporum f. sp. niveum, Colletotrichum gloesporioides, Sclerotinia sclerotiorum, Gibberella zeae, Botrytis cinerea, and Curvularia lunata (Walk) Boedijn at a concentration of 100 µg/mL. Among the synthesized compounds,
Inhibitory Effects of Luotonin Derivatives Against A.a., F.O.N., C.g., S.s., G., B.c., and C.l.b.

Abbreviations: Abbreviations: A.a., Alternaria alternata; B. c., Botrytis cinerea; C. g, Colletotrichum gloesporioides; C. l. B., Curvularia lunata (Walk) Boedijn; EC50, half maximal effective concentration; FON, Fusarium oxysporum f. sp. niveum; G, Gibberella zeae; S. s., Sclerotinia sclerotiorum.
Note. The carbendazim was used as the positive control.
Although it is difficult to extract apparent structure-activity relationships from the bioassay results, some conclusions can still be obtained. First, N-substituents (
Conclusions
Twenty-one luotonin derivatives were synthesized using 2-aminobenzamide as the starting material via either alkylation or acylation at the N-position, and their antimicrobial activities have been evaluated against 8 strains of bacteria and many plant pathogen fungi. The majority of the synthesized compounds showed potent activities. Notably, compounds
Supplemental Material
online supplementary file 1 - Supplemental material for Synthesis and Antimicrobial Characterization of Luotonin Derivatives
Supplemental material, online supplementary file 1., for Synthesis and Antimicrobial Characterization of Luotonin Derivatives by Rui Zhu, Rui Guo, Ke Han, Wenying Xi, Shaojun Zheng and Jiwen Zhang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the National Key Research and Development Program of China (No. 2017YFD0201402), the National Natural Science Foundation of China (21502073), the Natural Science Foundation of Jiangsu Province (Grants No BK20150465 and BK20180978), and the Key Research and Development Program (Modern Agriculture) of Zhenjiang City (NY2018002), and Zhoushan Public Welfare Science and Technology Project (2019C31066).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
