Abstract
Research Objective
This study was aimed to determine the main ingredients of Huajie Xiaoliu Formula (HJF) and investigate the anti-colon cancer effect of its combination with Tegafur, Gimeracil, and Oteracil Potassium Capsules (S-1) in vivo as well as to preliminarily explore its possible mechanism.
Materials and Methods
The chemical composition of HJF was determined by ultra-high performance liquid chromatography with quadrupole time-of-flight mass spectrometry (UPLC-QTOF-MS). The murine colon carcinoma cell (CT 26.WT) subcutaneous xenograft model of colon cancer mice was established to evaluate the in vivo anti-colon cancer effect of HJF. Evaluation of pathological changes in tumor tissue was evaluated by hematoxylin–eosin staining. Enzyme-linked immunosorbent assay (ELISA) was used to measure the expressions of interleukin-1β (IL-1β), inflammatory cytokines interleukin (IL-6), and tumor necrosis factor-α (TNF-α) in mice serum. Immunohistochemistry was used to evaluate B cell lymphoma-2 (Bcl-2), Bax, and Ki67 protein expression in tumor tissue.
Results
Twenty chemicals in HJF were identified by UPLC-QTOF-MS. In vivo experiments showed that HJF has obvious inhibitory effects on the subcutaneous xenograft model of colon cancer mice. Results of ELISA showed HJF is able to increase mice body weight, inhibit tumor growth, and reduce the serum levels of IL-1β and IL-6 along with TNF-α. Immunohistochemistry results demonstrated that HJF can significantly inhibit the levels of Bax, Bcl-2, and Ki67 in mice with colon cancer transplant tumors. Furthermore, the combination of HJF and S-1 demonstrated a more significant antitumor effect than any other group in vivo.
Conclusions
Huajie Xiaoliu Formula is effective in CT 26.WT subcutaneous xenograft model of colon cancer mice by inhibiting tumorigenesis, metastasis, and angiogenesis through the Bcl-2/Bax signaling pathway, resulting in anti-colorectal tumor effects. Huajie Xiaoliu Formula provides a strong theoretical basis for its clinical use and also provides a basis for further research.
Keywords
Introduction
Colorectal cancer, the fifth most common cancer in the world, is one of the leading causes of cancer death worldwide.1,2 The etiology and pathophysiology of colorectal cancer are closely related to unhealthy dietary habits and lifestyle changes. 2 Now, with rapid economic development and as living standards continue to improve, more and more people have the characteristics of lack of exercise, hyperglycemia, high-fat feeding, and lack of dietary fiber intake, which leads to the increasing incidence of colorectal cancer.3,4 The etiology and pathogenesis of colorectal cancer are complex and unclear, involving multiple links and pathways, as well as gene regulation and protein expression. In addition, the environment is also an equally significant factor in the pathogenesis of colorectal cancer. There are many methods to treat colon cancer, among which surgery, chemotherapy and radiotherapy are very traditional, but play very important roles. 5 However, the therapeutic effectiveness of these treatments is unsatisfactory, and after resection of colon cancer, the 5-year overall survival rate is only about 60%, and the recurrence rate is high. 6 Obviously, it is necessary to find new medicines to reduce the adverse side effects of undergoing chemotherapy in patients and maintain their clinical efficacy. Traditional Chinese medicine (TCM) has been proven to be able to effectively improve the curative effect, enhance the antitumor efficacy of various chemotherapeutic drugs, and reduce adverse reactions.7,8 Recently, it has been reported that Teng-Long-Bu-Zhong-Tang had a significant synergistic effect with 5-Fluorouracil in a subcutaneous xenograft model of colon cancer mice. 9 Another Chinese herbal formula, called Weichang'an, exhibited synergism in combination with Bevacizumab in an HCT-116 colorectal cancer xenograft model by inhibiting VEGFR-1. 10 Therefore, it is urgently required to establish an effective herbal formula for colorectal cancer treatment.
Huajie Xiaoliu Formula (HJF) is a TCM herbal formula that has been used clinically for tumor therapy for many years. In the early stage, the laboratory conducted basic research on the preparation process and quality standards of HJF and formed a stable in-hospital preparation for clinical use. Huajie Xiaoliu Formula contains: Astragali Radix, Glycyrrhiza glabra Linne, Poria cocos, Atractylodis macrocephalae, the roots of Actinidia chinensis, Coix seed, Scutellaria barbata, Angelica sinensis, Pericarpium Citri Reticulatae, Pinellia ternata (Thunb.) Breit, Lobelia chinensis, Hedyotis diffusa Willd., Scolopendra subspinipes Curcuma phaeocaulis Valeton, and Solanum lyratum. As a Chinese medicine compound based on clinical experience, several components of HJF have been verified to have antitumor effects. Of these herbs, both the roots of Actinidia chinensi and Astragali Radix are frequently used in antitumor formulas.11,12 Radix Astragali 13 and Hedyotis diffusa Willd 14 inhibit cancer cell proliferation. Accumulating evidence suggests that PI3K/Akt/mTOR signaling plays multiple roles in regulating the cell cycle, participating in tumor angiogenesis, affecting tumor invasion and metastasis, regulating apoptosis and autophagy, as well as participating in tumor resistance. 15 The antiapoptotic effect of the PI3K-AKT signaling pathway may be via the Bad pathway, a related death promoter for B cell lymphoma-2 (Bcl-2), and AKT can phosphorylate its 5136 site to inactivate Bad and prevent it from binding to the antiapoptotic protein Bcl-x. 16 We speculated that the mechanism of action of HJF is closely related to the Bcl-2/Bax pathway. However, there is little relevant theoretical research, and, therefore, this study established a murine colon carcinoma cell subcutaneous xenograft model of colon cancer mice and studied the anticancer effect of HJF on CRC cells in vivo. Furthermore, this study explored whether HJF inhibits tumors through the Bcl-2/Bax pathway by immunohistochemistry (IHC), hematoxylin–eosin (HE), and other modern molecular biological techniques.
Methods and Materials
Instruments and Materials
In this study, we used a Waters Xevo G2-XS QTOF Time-of-Flight Mass Spectrometer (Waters Corporation), an ACQUITY UPLC M-Class system (Waters Corporation), BT25S 100,000th analytical balance (Saidoris), XW-80A vortex mixer, KQ-500VD ultrasonic cleaner, TGL-16 desktop high-speed refrigerated centrifuge (Xiangyi), and UPT-II-10 T ultrapure water machine (UPT).
Dulbecco's modified Eagle medium, 10% heat-inactivated fetal bovine serum (FBS), and phosphate-buffered saline (PBS) were purchased from Gibco Company. Interleukin (IL)-6, tumor necrosis factor-α (TNF-α), and IL-1β enzyme-linked immunosorbent assay (ELISA) kits were purchased from Wuhan Elite Biotechnology Co. Ltd.
Astragali Radix, Glycyrrhiza glabra Linne, Poria cocos, Atractylodis macrocephalae, the roots of Actinidia chinensis, Coix seed, Scutellaria barbata, Angelica sinensis, Pericarpium Citri Reticulatae, Pinellia ternata (Thunb.) Breit, Lobelia chinensis, Hedyotis diffusa Willd., Scolopendra subspinipes Curcuma phaeocaulis Valeton, and Solanum lyratum were all characterized by Professor Duan Xueyun from Hubei University of TCM. All conformed to the provisions of the 2020 edition of Chinese Pharmacopoeia. Voucher samples of each are conserved in the herbarium of the Hubei Institute of TCM with samples numbers: 20211012, 20211013, and 20211014; the standard reference compounds, including protocatechuic acid (110809-201906), deacetylasperulosidic acid methyl ester (111786-201602), chlorogenic acid (110753-202119), asperulosidic acid (B24028), paclitaxel (100382-201904), glycyrrhizic acid (B20417), ferulic acid (110773-201915), calycosin-7-O-β-D-glucoside (111920-201907), scutellarin (110842-202010), rutoside (100080-202012), hesperidin (110721-202019), isoliquiritin (B21524), ononin (B20214), 10-deacetylbaccatin III (B21747), calycosin (B20846), formononetin (111703-201504), heterophyllin B (B21236), 6-gingerol (111833-202007), pachymic acid (B27710), and ursolic acid (110742-201823) were purchased from China Food and Drug Identification Institute. Tegafur, gimeracil, and Oteracil Potassium Capsules (batch number: 210620NF) were purchased from Jiangsu Hengrui Pharmaceuticals Co., Ltd.
Preparation of Huajie Xiaoliu Decoction
Huajie Xiaoliu Formula is composed of 17 medicinal herbs, which were obtained from the Hubei Provincial Hospital of TCM. First, all herbs were soaked in water for 30 min, boiled 3 times, for 1.5 h each time, filtered, and the filtrate collected and then concentrated to 2.84 g/mL.
Analysis of Components in HJF
Chromatographic and mass spectrometry conditions
Separation was performed by ultra-high performance liquid chromatography (UPLC) using a BEH C18 column (100 × 2.1 mm, 1.7 μm). Gradient elution was carried out with water (A) and methanol (B) (0.1% formic acid) as the mobile phase, 0 to 20 min (B: 5%-60%), 20 to 30 min (B: 60%-80%), 30 to 38 min (B: 80%-98%), 38 to 41 min (B: 98%), 41 min to 42 min (B: 98%-5%), 42 min to 46 min (5%); column temperature, 40 °C; flow rate: 300 μL/min; and injection volume, 1 μL.
The ESI source was scanned in positive and negative ion modes. Data acquisition was performed in the MSE mode. Scanning range: m/z 50–1200 Da; desolvation temperature: 500 °C; ion source temperature: 100 °C; cone gas flow: 50 L·Hr-1; desolvation gas flow: 500 L·Hr-1/600 L·Hr-1; cone voltage: 40 V/20 V; capillary voltage: 2.5 kV/3 kV.
Cell Culture and Xenograft Model Procedure
The CT 26.WT cell line was purchased from the Chinese Academy of Sciences cell bank. Cell culture was carried out using 10% FBS, 1% penicillin/streptomycin in Eagle's medium, 5% carbon dioxide (CO2) humidifier, and the incubator temperature was 37 °C. When the cells were in the exponential growth phase, the cell concentration was adjusted to 1.5 × 107/mL with PBS. Forty BALB/c mice (male, 200 g ± 20 g) were provided by the Hubei Laboratory Animal Research Center. The certificate number of these mice was SCXK (E) 2017–0012. Feeding was under standard conditions, with 12 h light/12 h dark, and a temperature of 25 ± 1 °C. Adaptive feeding was for 1 week. All experiments were ratified by the Animal Ethics Committee of Hubei Provincial Hospital of TCM (No. HBZYY-2018-043). The mice were subcutaneously injected in the armpit with about 1 × 106 CT 26.WT cells. When the tumor increased to about 100 to 200 mm3, it was stripped. The fish-like tissues with strong growth ability were cut into small squares of 1 mm3 and placed in physiological saline. The tumor block was inoculated subcutaneously in the anterior armpit of 30 BALB/c mice. The other 5 mice were the control group. When the tumors had increased to about 100 to 200 mm3, the mice were randomly divided into 6 groups (n = 5), and given corresponding doses of drugs twice a day by gavage: the low-dose HJF group (HJF-L, 25.84 g/kg/0.4 mL), high-dose HJF group (HJF-H, 51.69 g/kg/0.4 mL) or the same volume of distilled water, or given S-1 (30 mg/kg/0.4 mL, twice a day), or HJF-L + S-1 and HJF-H + S-1 treatment. During the medication, the mice were weighed DD, and the tumor’s width (W) and length (L) were measured with a caliper. After 2 weeks of continuous medication, the mice were sacrificed, and the tumors were stripped and weighed. The serum of mice was also collected for further experiments.
Hematoxylin–Eosin Staining and IHC
All mice tumor tissues were fixed with paraformaldehyde, wax embedded, and then sliced into 5 μm thick sections. All tumor sections were removed and hydrated, and finally stained with HE.
The tumors of all the mice were fixed with 10% paraformaldehyde for about 12 h, embedded in paraffin, dewaxed with xylene, and rehydrated with ethanol. The antigen was extracted with a high-pressure cooker in 0.01 M citric acid buffer (pH 6.0) and incubated for 3 min. It was then washed 3 times with PBS and incubated overnight with anti-Ki-67 antibody (1:600), anti-Bcl-2 antibody (1:400), and anti-Bax antibody (1:400), and then incubated with the corresponding HRP-labeled secondary antibody at 37 °C for 20 to 30 min. Then the sections were washed with PBS, stained with DAB, counterstained with hematoxylin, and dehydrated. Then the expression of Ki-67, Bcl-2, and Bax proteins in tissue sections was evaluated by optical inverted microscope (×200).
Enzyme-Linked Immunosorbent Assay
Serum in mice was diluted with the working solution, and then, according to the manufacturer's instructions, IL-1β (EMC001a.96), IL-6 (EMC004a.96), and TNF-α (EMC102a.96) ELISA Kits (neo biological) were used to determine cytokines in the supernatant.
Statistical Analysis
GraphPad Prism version 8.0 software was used to process the data. Results were recorded in means ± standard deviation. Student t test or variance analysis was used for statistical analysis. P < .05 indicated that the difference was statistically significant.
Results
Ultra-High Performance Liquid Chromatography With Quadrupole Time-of-Flight Mass Spectrometry Analysis of Components in HJF
The identification of the chemical composition in HJF was determined by UPLC-QTOF-MS. The compounds in HJF were ionized in negative and positive ion polarity modes, and the identification of 20 components of HJF was made by Extraction Ion Chromatography (EIC) (Figure 1).

(A and B) Representative extraction ion chromatograms of Huajie Xiaoliu Formula (HJF) detected in positive ion polarity mode. (C and D) Representative extraction ion chromatograms of HJF detected in the negative ion polarity mode.
The retention time (min), chemical formula, and component names of the constituents of HJF are shown in Table 1. All components were validated by accurate measurement of fragments.
Identification of 20 Metabolites in Plasma by UPLC-QTOF-MS Analysis.
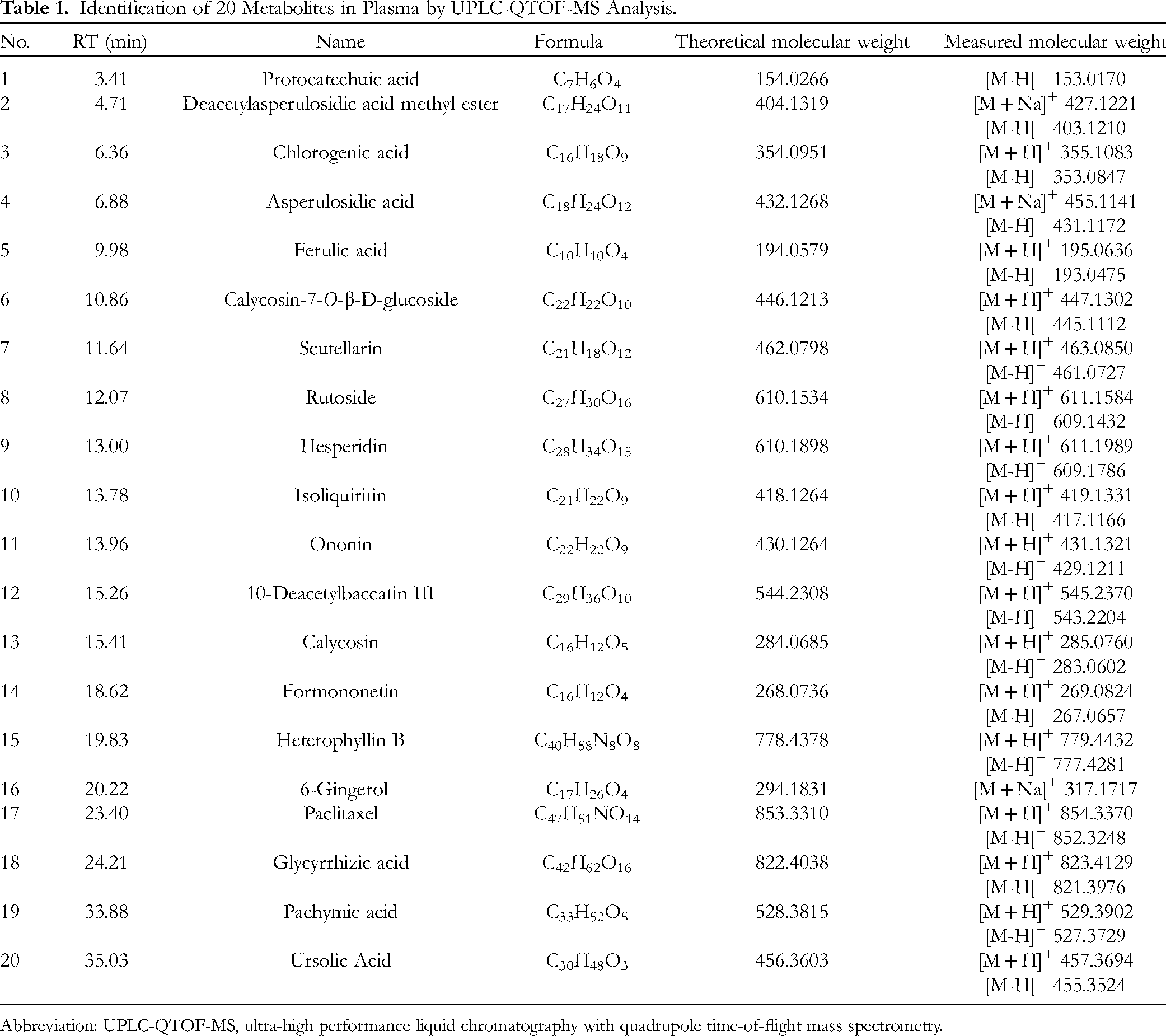
Abbreviation: UPLC-QTOF-MS, ultra-high performance liquid chromatography with quadrupole time-of-flight mass spectrometry.
In Vivo Inhibitory Effect of HJF on Colon Cancer in Mice
Effects of HJF on growth of CT 26 subcutaneous xenograft model of colon cancer mice
The tumor volume of mice was measured by a vernier caliper every day to observe the tumor growth status. The growth curve of tumor-bearing mice showed that the tumor volume of the 6 groups increased with time, and the order of tumor volume was as follows: Molder > HJF-L > HJF-H > S-1 >HJF-L + S-1 >HJF-H + S-1(Figure 2A).

(A) Tumor growth curves based on different treatments. ****P < .01 versus Molder group.
The formula for calculating tumor volume and tumor weight inhibition rate is as follows:
Tumor Weight and Inhibition Rate Based on Drug Treatment (Mean ± SD, n = 5).

Abbreviation: HJF, Huajie Xiaoliu Formula.
Huajie Xiaoliu Formula improves the pathological changes of colon cancer xenograft in mice by reversing the levels of inflammatory cytokines
As shown in Figure 3A, the IL-6, TNF-α, and IL-1β serum levels in the model group were significantly higher than those in the other groups, whereas those in the S-1 group, different doses of HJF group, and the combination group were significantly decreased. The serum IL-6, TNF-α, and IL-1β levels in the HJFL + S-1 group and HJFH + S-1 group were significantly lower than those in the S-1 group. The results were statistically significant (P < .01) The results revealed that HJF at different doses significantly reduced the levels of TNF-α and IL-6, as well as stimulating the production of anti-inflammatory cytokine IL-1β (P < .01). The samples of tumor tissue were sliced and stained. Changes in histopathology were observed in the different groups (Figure 3B). In the model group, tumor tissue cells had dense but irregular arrangement, were of different sizes, and had diverse morphology, accompanied by enlargement of the tumor cell nucleus, less cytoplasm, dense growth of tumor cells, and the formation of the tumor cell mass. After drug treatment, the distribution of tumor cells in all treated groups was more sparse than that in the model group, showing a verified form and varying degrees of lamellar necrosis, among which the HJFL + S-1 and HJFH + S-1 groups had more necrotic areas. Taken together, the results suggested that different doses of HJF and the combination group of HJF + S-1 can regulate the necrosis of the tumor and improve the quality of life of mice.

(A) Expression levels of interleukin (IL)-1β, IL-6, and tumor necrosis factor-α (TNF-α) (n = 5).*P < .05 versus Molder group, **P < .01 versus Molder group, ****P < .0001 versus Molder group, ####P < .0001 versus Control group. (B) Histopathological examination of tumor tissue (×200, n = 3).
Huajie Xiaoliu Formula inhibits tumorigenesis via the Bcl-2/Bax signaling pathway and apoptosis
In order to further explore the mechanism of inhibiting colorectal cancer in mice, we detected proteins that may be involved in tumor growth in tissues. The results of IHC revealed that the protein expression of Bcl-2 and ki67 in the tumor of the 5 treatment groups was significantly lower than that in the model group (Figure 4). Moreover, the expression of Bax protein in the tumors of the 5 treatment groups was significantly increased (Figure 4). These results showed that HJF and S-1 significantly inhibit Bcl-2/Bax signaling in colorectal cancer mice. The combination produced more powerful inhibition of tumor growth than either treatment alone.
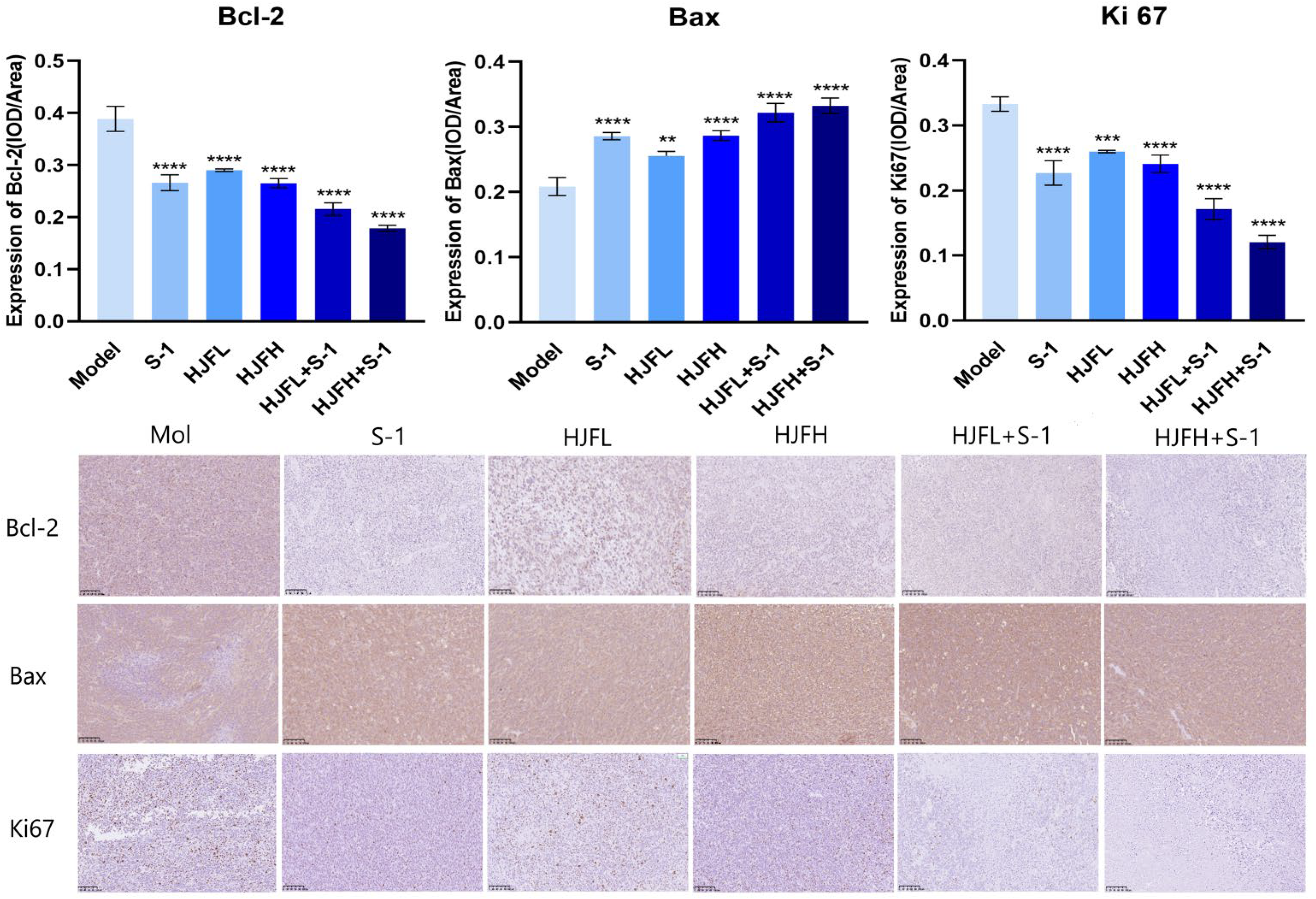
(A) Quantitative analysis of protein levels of B cell lymphoma-2 (Bcl-2), Bax, and Ki67. (B) The protein levels of Bcl-2, Bax, and Ki67 in the tumor tissues were detected by immunohistochemistry. Representative micrographs from 5 mice per group are shown, Scale bar = 200 μm. (×200, n = 3).
Discussion and Conclusions
Colorectal cancer, one of the most common gastrointestinal tumors at present, has high morbidity and mortality. The majority of patients are discovered in the middle and advanced stages of cancer. 17 At present, many anticancer drugs have therapeutic benefits, but it is worrying that the many chemotherapy drugs used in clinic have various side effects, including hepatotoxicity, renal injury, nerve sensory irreparability, thromboembolic events, and granulocytic lesions. 18 These problems are caused by the single treatment method and drugs, and thus it is extremely important to find new drugs derived from medicinal plant sources. 19 TCM has played a positive role in clinical treatment and has been commonly used as a supplementary treatment in the whole course instead of only at the terminal stage of colorectal cancer.20,21 The HJF studied in this experiment is a pure Chinese medicine formula developed under the guidance of the theory of clearing heat and expelling toxin, replenishing qi and blood, strengthening body health, and resisting cancer. Huajie Xiaoliu Formula is a commonly used formula in the clinical treatment of malignant tumors of the digestive tract and is composed of 17 kinds of TCMs such as vine pear root, southern yew, half-sided lotus, half-branched lotus, white flower snake tongue grass, astragalus, coix kernel, and licorice. Actinidia chinensis Planch. has been shown to be able to inhibit the proliferation of liver cancer cells (HepG2) and regulate apoptotic signaling pathways. 22 Lin et al confirmed that Shenling Baizhu San was able to inhibit TGF-β1-induced epithelial–mesenchymal transition (EMT) and myeloid infiltration in the CRC model. 23 Peng et al showed that Jianpi Jiedu decoction effectively inhibited the proliferation of HCT116 cells and induced their apoptosis by inhibition of migration, invasion, and angiogenesis of colon cancer cells by regulating the mTOR/HIF-1α/VEGF pathway. 24 In our study, the results indicated that HJF significantly inhibited tumorigenesis of CT-26.WT mice with colorectal cancer. HJF + S-1 had an additional inhibitory activity on the growth of colorectal cancer CT-26.WT mice subcutaneous xenografts. In this study, we found that HJF can inhibit the growth of CT-26.WT xenotransplantation in colorectal cancer and achieve the corresponding effect by downregulating expression of the Bcl-2/Bax signaling pathway-related proteins. At the same time, the combination of HJF and S-1 groups demonstrated a more significant antitumor effect than any other group. There are proapoptotic proteins and antiapoptotic proteins in the Bcl-2 protein family. The proapoptotic protein Bcl-2 is located in the outer membrane of mitochondria, which is closely related to promoting cell survival and inhibiting the occurrence of proapoptotic proteins. 25 Studies have proved that Bcl-2 is positively correlated with the genesis and development of various cancers.26,27 In our study, we found that HJF and its combination groups inhibit the expression of Bcl-2 in colon cancer in vivo and promoted the expression of the protein Bax from the same family. Among them, combinations are more effective. What is more, we found that the Ki67 protein level in mice colorectal cancer tissues and colorectal cancer cell lines can be significantly inhibited after HJF intervention. Furthermore, we also found that HJF was more effective in combination with S-1 in addition to HJF treatment alone. In our study, HJF and HJF + S-1 treatments inhibited the expression levels of IL-6, IL-1β, and TNF-α and the protein expressions of Bcl-2, Bax, and Ki67 in neoplasm tissues. HJF + S-1 displayed an additive antineoplastic activity. The mechanism may also be related to the role of HJF in regulating the Bcl-2/Bax signal. Although TCM has played a critical role in the treatment of tumors, the active ingredients which play a therapeutic effect in the formula have not been clearly verified. Therefore, it is essential to explore the effective components in TCM or Chinese herbal compounds. In this study, UPLC-QTOF-MS was used to detect 20 chemical constituents in HJF, and several of them have been proven to have significant inhibitory effects on tumors. Xie et al discovered that protocatechuic acid can inhibit the growth of epithelial ovarian cancer cells by apoptotic induction and autophagosome. 28 Chlorogenic acid has been confirmed to restrain lung cancer proliferation by targeting annexin. 29 Paclitaxel can prolong the survival time of rat liver graft, inhibit the growth of liver cancer cells, and has a good therapeutic effect on liver cancer patients. 30
B cell lymphoma-2/Bax signaling has a critical effect on tumor proliferation and degeneration. The main mechanism to achieve the effect of HJF antiangiogenesis and promoting tumor cell apoptosis may be closely related to the Bcl-2/Bax signaling pathway. Furthermore, HJF promotes the antitumor effect of S-1 by inhibiting Bcl-2 and Bax to regulate tumor immunity. The combination of HJF and S-1 groups demonstrated a more significant antitumor effect than any other group. All of these provide a theoretical foundation for our group to further study the mechanism of HJF against colon cancer.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231185201 - Supplemental material for A Chinese Medicine, Huajie Xiaoliu Formula, Enhances the Anti-Colon Cancer Effect of Tegafur, Gimeracil, and Oteracil Potassium Capsules Through Bcl-2/Bax Signaling Pathway
Supplemental material, sj-docx-1-npx-10.1177_1934578X231185201 for A Chinese Medicine, Huajie Xiaoliu Formula, Enhances the Anti-Colon Cancer Effect of Tegafur, Gimeracil, and Oteracil Potassium Capsules Through Bcl-2/Bax Signaling Pathway by Peili Tang, Wanjin Sun, Sifang Xie, Sicheng Yang, Ju Huang, Kai Deng, Xueyun Duan and Fan Heng in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X231185201 - Supplemental material for A Chinese Medicine, Huajie Xiaoliu Formula, Enhances the Anti-Colon Cancer Effect of Tegafur, Gimeracil, and Oteracil Potassium Capsules Through Bcl-2/Bax Signaling Pathway
Supplemental material, sj-docx-2-npx-10.1177_1934578X231185201 for A Chinese Medicine, Huajie Xiaoliu Formula, Enhances the Anti-Colon Cancer Effect of Tegafur, Gimeracil, and Oteracil Potassium Capsules Through Bcl-2/Bax Signaling Pathway by Peili Tang, Wanjin Sun, Sifang Xie, Sicheng Yang, Ju Huang, Kai Deng, Xueyun Duan and Fan Heng in Natural Product Communications
Footnotes
Acknowledgments
This study was financially supported by the Affiliated Hospital, Hubei University of Chinese Medicine, Wuhan 430061, China. Peili Tang, Wanjin Sun and Sen Li contributed equally to the work.
Authors’ Note
The study was approved by the Animal Ethics Committee of Hubei Provincial Hospital of Traditional Chinese Medicine Review Board (No. HBZYY-2018-043). The datasets generated for this study are available on request to the corresponding authors.
Authors Contribution
Peili Tang and Wanjin Sun are equally responsible for conducting the experiments and writing the manuscript. Sifang Xie, Sicheng Yang, Ju Huang and Kai Deng assisted with the experiments and data analysis. Xueyun Duan and Heng Fan are responsible for designing and guiding the experiments. Peili Tang and Wanjin Sun contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Seventh Batch of Traditional Chinese Medicine Experts in China (No. [2022] 76), National Training Project for Traditional Chinese Medicine Technology Inheritance Talent (No.〔2023〕96) and Provincial Key Laboratory Research Project -9
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
