Abstract
Colorectal cancer (CRC) is a malignant tumor with high incidence and bad prognosis. Therapies, which are more safe and effective, are urgently needed. Trypsin is proved to be crucial to cancer proliferation and migration, therefore, it is possible to control cancers by modulating its activity. Fisetin is a flavone with trypsin inhibition properties that was screened from more than 45 compounds derived from traditional Chinese medicine (TCM). However, the effects and mechanisms of fisetin on CRC have not been well investigated. In this study, we evaluated the effects of fisetin on 2 different CRC cell lines. Fisetin remarkably inhibited CRC cell proliferation and migration, as well as induced cell apoptosis and Go/G1 phase arrest in a dose-dependent manner. Mechanistic studies revealed that these effects were mediated partially through signaling pathways involving cell cycle regulators p21, p27, cyclinD1, and NF kappa B (NF-κB) p65. Administration of fisetin also significantly suppressed the tumor growth in tumor-bearing NOD/Shi-scid-IL2R gamma (null) (NOG) mice that had been inoculated with human HCT116 cells. Fisetin at the given dosage did not induce significant acute or chronic toxicity in rats. These data provide a potential therapeutic strategy for CRC.
Introduction
Colorectal cancer (CRC) is the fourth deadliest cancer, accounting for almost 900,000 deaths annually in the world. 1 The incidence and mortality of CRC do not reduce significantly, even with effective cancer screening measurements and great advancements in cancer treatment.2,3 Surgical resection with adjuvant chemotherapy is effective for early stage CRC treatment, nevertheless, there is no effective therapy for advanced stage patients, especially for those with metastatic cancer.4,5 Therefore, it is urgent to find more effective and safe therapies for the prevention and treatment of CRC.
Trypsin is a digestive enzyme that relates to the development of neoplasia, invasion, and metastasis of several cancers.1,6 Trypsin positivity significantly contributes to the depth of lymphatic and venous invasion, lymph node metastasis, distant metastasis, and recurrence of CRC.7,8 Previous studies have shown that the overall survival and disease-free survival periods of patients with trypsin-positive CRC was significantly shorter than those with trypsin-negative CRC. Meanwhile, cetuximab resistance was found to be related to the level of trypsin (PRSS1) expression secreted by CRC cells. 9 Trypsin (PRSS) inhibited the blocking of cetuximab on epidermal growth factor receptor and diminished the therapeutic effects. However, the combination of cetuximab with SPINK1 (a trypsin inhibitor [TI]) suppressed tumor growth more efficiently than cetuximab alone in a xenograft model. Thus, TIs may reduce the drug resistance and improve the prognosis in patients with CRC.
Most of the reported TIs are naturally derived proteins or polypeptides. 10 However, some traditional Chinese medicines (TCMs) have demonstrated trypsin inhibitory activity.11,12 These facts indicated that TCMs could be an important potential source of TIs. Current methods for TI screening are based on enzyme activity assays with high enzyme consumption, and which are very time consuming, and more importantly, sometimes inefficient. 13 Thus, an effective TI screening method is of great significance to new drug discovery and design. Recently, with advancement in proteins/enzymes and other target immobilization methods, and development of state-of-the-art analytical approaches,14,15 the combination of liquid chromatography with a bioassay unit has been used for natural products screening and drug discovery.16,17 We have established a new immobilized TI screen based on a poly(GMA-co-PEGDA) monolithic column, and 45 flavonoid compounds derived from TCM have been screened (Supplementary Table S1).15,18 Fisetin (3, 7, 3′, 4′-tetrahydroxyflavone, Figure 1) has been shown to display the best trypsin inhibitory activity (62%) among these Chinese herb-derived flavonoids. Fisetin is a dietary flavonoid found in Chinese traditional herbs, such as Cotinus coggygria Scop.19,20 With a polyphenolic structure, fisetin is a potent antioxidant agent and free radical scavenger, both in vitro and in vivo.21–23 Fisetin also showed promising therapeutic effects against several cancers, such as breast, leukemia, liver, and prostate.24–26

The structure of fisetin.
In this study, we investigated the antitumor effects of fisetin via the trypsin inhibition pathway on 2 different CRC cells (HCT116 and HT-29), and further evaluated the therapeutic effects in a CRC mouse model. The underlying mechanisms of interaction between fisetin, trypsin, and tumor cells were also explored. Finally, we also evaluated the safety of fisetin on normal rats.
Materials and Methods
Cell Culture
Two CRC cell lines (HCT116 and HT-29), purchased from the American Tissue Type Culture Collection (Manassas, VA, USA), were maintained in Dulbecco’s modified Eagle medium (DMEM, Gibco BRL) containing 10% fetal bovine serum (Gibco BRL) and 100 U/mL penicillin/streptomycin at 37 °C under 5% CO2.
Cell Proliferation Assay Induced by Trypsin
For cell proliferation, the cells were seeded into 96-well plates at a density of 2 × 104 cells per well. After culturing for 24 h, trypsin (100 nM) and fisetin at different concentrations (30, 60, and 120 μM, final concentration) were added to the medium and incubated. At the indicated time points (0, 24, 48, and 72 h after incubation), 20 μL of 5 mg/mL 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma) was added, and the plates were incubated at 37 °C for an additional 4 h. Then, the supernatant was removed and washed 3 times with phosphate buffer saline (PBS) solution. The residue was dissolved in 100 μL of dimethyl sulfoxide. The optical density (OD) at 490 nm was determined with a microplate reader. Cell viability was represented by the absorbance. Three independent experiments were performed and the average data were obtained for analysis.
Transwell Migration Assays Induced by Trypsin
The transwell assays were performed according to the manufacturer's instructions. The transwell (Costar, USA) and polycarbonate membrane insert (Corning, NY, USA) with an 8 μm pore were used. Cells (1.5 × 104) were seeded in the upper compartment and incubated with trypsin (100 nM) and the indicated concentration of fisetin (Energy Chemical). After incubation for 24 h at 37 °C in a humidified incubator with 5% CO2, the insert was removed, along with the culture medium. The cells that remained on the upper side of the filter membrane were gently removed. The cells on the lower side of the insert filter were quickly fixed with 5% glutaraldehyde for 10 min and stained with 1% crystal violet. The migration was evaluated by counting the cells that had migrated on the lower side of the filter. Each assay was performed in triplicate and repeated 3 times.
Cell Cycle Analysis
Cells (2.5 mL/well) were plated at a density of 1 × 106 cells/mL into 6 well plates followed by treatment with different concentrations of fisetin for 48 h. After removal of the culture mixture, the cells were fixed with 70% ethanol at 4 °C overnight, and then incubated with 50 μg/mL propidium iodide (PI) and 100 μg/mL RNase (Sigma) at room temperature in the dark for 30 min. The cell cycle phase was analyzed by flow cytometry (Beckman Coulter). The experiment was repeated 3 times and the average data were used for analysis.
Cell Apoptosis Analysis
Cells were seeded into 96-well plates at a density of 1 × 104 cells per well and cultured for 24 h. Fisetin at different concentrations was added to the medium. Following culture for another 48 h, the medium was removed and the cells were washed with PBS for 3 times. The cells were harvested and resuspended in the binding buffer, and incubated with PI and Annexin V/fluorescein isothiocyanate (FITC) (Sigma) for 15 min in the dark. Apoptosis was then detected by a flow cytometer (Beckman Coulter). The experiment was independently repeated 3 times and the average data were used for analysis.
Western Blot Assay
HCT116 and HT-29 cells were seeded in 6 well plants and cultured for 24 h. Trypsin (100 nM), fisetin (60 and 120 μM, final concentration), and NF kappa B (NF-κB) p65 expression vector (10μM, final concentration) were added to the medium and cultured for another 48 h. Protein extraction from the cell lines was performed using a standard protocol. Equal amounts of protein were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis followed by transfer of the proteins to nitrocellulose membranes (Bio-Rad). The membranes were first incubated with primary antibodies against trypsinogen-2 (PRSS2), p21, p27, cyclinD1, NF-κB p65, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH, Abcam), followed by incubation with the horse radish peroxidase-conjugated secondary antibodies. The results were visualized using an enhanced chemiluminescence detection system (Amersham Biosciences). Each experiment was repeated thrice.
Human CRC Xenograft Tumor Models in NOG Mice
The following animal experiments were performed to study the therapeutic effect and safety of fisetin on CRC in vivo. NOD/Shi-scid-IL2R gamma (null) (NOG) and Kunming mice, and Wistar rats were purchased from the Guangzhou Jennio Biotech Co., Ltd (Guangzhou, China). All animal experiments were approved by the medical ethics committee of the First Affiliated Hospital of Jinan University, and carried out in accordance with the guidelines for Care and Use of Laboratory Animals of the First Affiliated Hospital of Jinan University.
A total of 16 male NOG mice (weight: 26-32 g, 4-6 weeks old) were injected subcutaneously in the right axillary with 5 × 106 HCT116 cells. Once the tumor volume had grown to 100 mm3, the mice were randomly divided into 4 groups (n = 4/group): control group (treated with saline) and experimental groups (treated with 30, 60, and 120 mg/kg of fisetin). NOG mice in the experimental groups were injected intraperitoneally with the indicated dose of fisetin, while mice in the control group were administrated with isovolumetric saline. The tumor size was monitored every other day. The largest (a) and smallest (b) superficial diameters of tumors were measured with vernier calipers, and the tumor volume was calculated as follows: tumor volume (mm3) = a × b2 × 0.5. After the last injection, NOG mice were sacrificed and the tumors were collected.
Acute and Chronic Toxicity Assay
Forty Kunming mice (weight: 18-22 g, 4-6 weeks old) were randomly divided into 2 groups (n = 20, male: female = 50:50). Mice in the fisetin group were injected intraperitoneally with 223 mg/kg of fisetin dissolved in 0.1 mL of PEG200:DMSO (7:3; v:v), as described previously, while the control group was injected with 0.1 mL saline. 27 The general condition of the mice (changes in skin, mucous membrane, fur color, eyes, conscious behavior, respiration, circulation, and central nervous system) was observed daily. Body weight was also recorded weekly.
A total of 160 Wistar rats (weight: 75-100 g, 4-6 weeks old) were randomly divided into 4 groups (n = 40, half male and female), which included a control group, low-dose group (L, fisetin 15 mg/kg), medium-dose group (M, fisetin 30 mg/kg), and high-dose group (H, fisetin 60 mg/kg). Rats in the experimental groups were injected with the indicated dosage of fisetin once a day for 6 weeks, followed by a recovery period of 2 weeks. Those in the control group were administered normal saline in the same way. Rats were observed daily for abnormal behavior and other adverse signs of toxicity. Body weight and food consumption were measured and recorded once a week. All animals were euthanized by CO2 inhalation at the end of the experiment, and blood samples and organs (brain, lung, heart, liver, kidney, spleen, ovaries, and testes) were collected for biochemical assays (hematological assessments, liver and renal function, and serum electrolyte tests).
Statistical Analysis
The statistical software package SPSS 16.0 (SPSS, Inc.) was used for statistical analysis. Data were expressed as the mean ± SEM. Quantitative data were calculated by independent sample t-test followed by Mann–Whitney U test. Categorical data were determined by either the χ2 or Fisher’s exact tests, according to the absolute numbers included in the analysis. The level of significance was set to P < 0.05.
Results and Discussion
Expression of PRSS2 was Increased in CRC Cells
Tumor-associated trypsin has been recognized as a significant factor in cancer progression and metastatic processes. 28 As the precursor of trypsin, PRSS2 seems to be strongly correlated with cancers in 4 trypsin isoforms. 29 Several studies revealed high trypsinogen expression in different CRC cell lines.30,31 Thus, the expression of PRSS2 was first evaluated in 6 different tumor cell lines, including non-small-cell lung cancer cell line A549, CRC cell lines HCT116 and HT-29, pancreatic carcinoma cell line Panc-1, gastric carcinoma cell line SGC-7901, and hepatocellular carcinoma cell line HepG2. As shown in Figure 2, the expression of PRSS2 was higher in HepG2, HCT116, and HT-29 compared with A549, Panc-1, and SGC-7901. Recent research also demonstrated the possibility of PRSS as a predictive marker of the mCRC response to cetuximab treatment. 9 These findings suggest that trypsin may play an important role in the development of CRC, and the TI fisetin may be beneficial for CRC treatment.
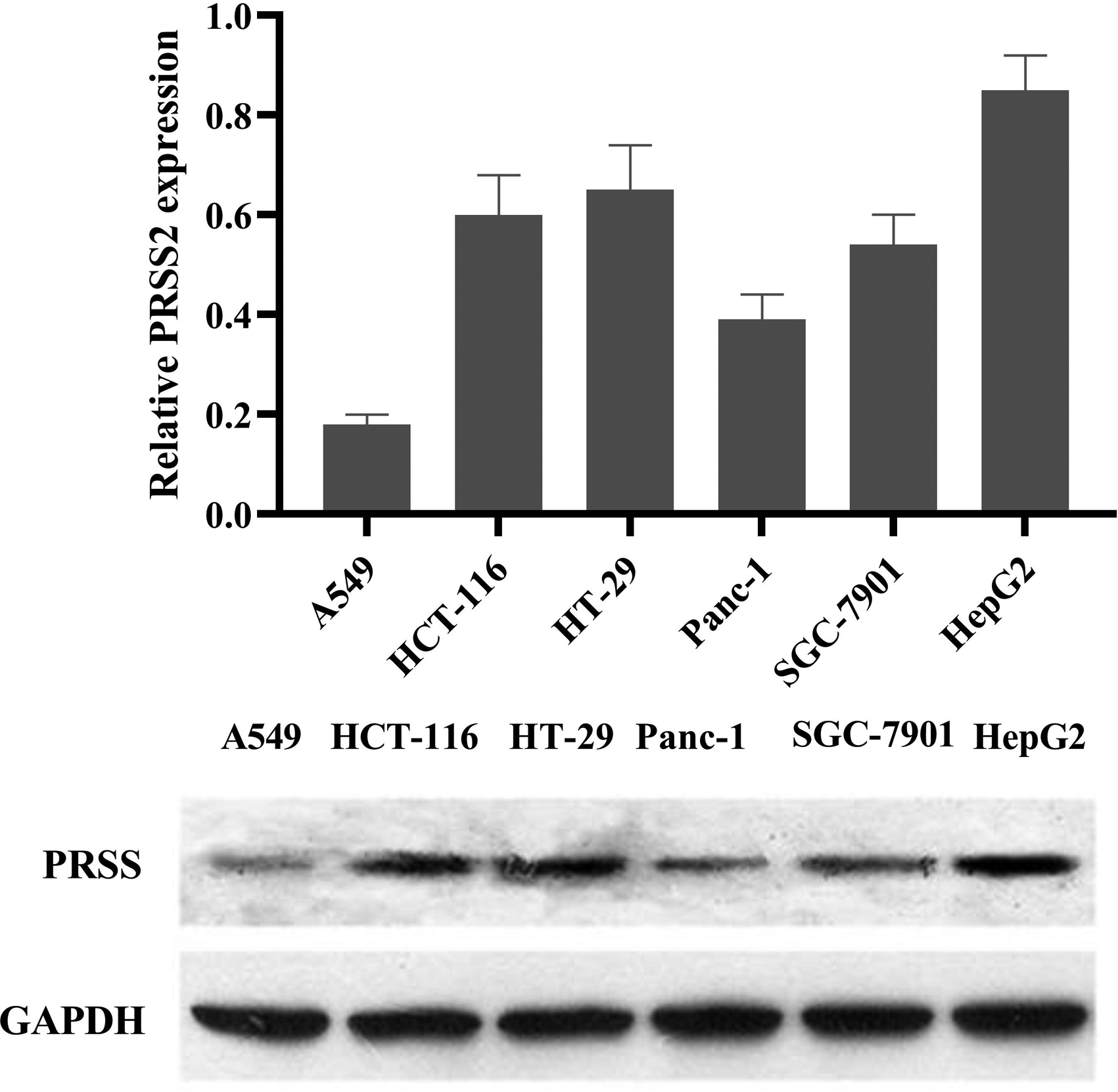
Expression of trypsin PRSS2 in different cancer cell lines.
Fisetin Inhibited the Proliferation of CRC Cells Induced by Trypsin
To evaluate the effect of fisetin on the proliferation of CRC cells induced by trypsin, we determined the cell viability of HCT116 and HT-29 cells with 3 concentrations of fisetin (30, 60, and 120 μM) at different times (0 h, 24 h, 48 h, and 72 h). As shown in Figure 3, after incubation with trypsin, the cell viability was significantly increased, both in HCT116 (Figure 3A) and HT-29 (Figure 3B) which means that trypsin promoted CRC propagation. Fisetin treatment (from 24 h to 72 h) reversed the cell viability in a concentration-dependent manner. At a concentration of 30 μM, fisetin significantly reduced the cell viability compared to the control group. These data demonstrated that fisetin was cytotoxic and effective in inhibiting CRC tumor growth induced by trypsin. Similar to our findings, previous studies exploring the effects of fisetin on CRC indicated that fisetin dramatically suppressed the proliferation of HT-29 cells, and induced HCT116 cell apoptosis.32–34

Effects of fisetin on the proliferation induced by trypsin of HCT116 and HT-29 cells.
Fisetin Promoted the Apoptosis of CRC Cells
Then, the effect of fisetin on CRC cell apoptosis was investigated. As shown in Figure 4, after an incubation of 48 h, fisetin treatment significantly increased the cells in early apoptosis and later apoptosis both in HCT116 and HT-29 cells (Figure 4C). The statistical results (Figure 4A and B) demonstrated that fisetin remarkably promoted CRC cell apoptosis in a concentration-dependent manner.

Effects of fisetin on the apoptosis of HCT116 and HT-29 cells.
Fisetin Suppresses the Cell Cycle Progression of CRC Cells
Cell cycle arrest is an important mechanism of cancer treatment drugs, so the effects of fisetin on the cell cycle were also analyzed. Flow cytometry results revealed that fisetin (60 μM and 120 μM) inhibited cell cycle progression by increasing the proportion of G1/G0 phase and decreasing the number of S phase cells in HCT116 (Figure 5A and 5C) and HT-29 (Figure 5B and 5C) cell lines compared to the control group (Figure 5). These results indicate that fisetin obstructs cell transition from the G1 to S phase and inhibits cell cycle progression.

Effects of fisetin on the cell cycle of HCT116 and HT-29 cells.
Fisetin Inhibited the Migration of CRC Cells Induced by Trypsin
Cell migration is crucial for tumor evasion and metastasis. Previous study had revealed that fisetin remarkably inhibited adhesion, migration, and invasion of lung cancer A549 cells, 35 but the effects on CRC were vague. To explore the effects of fisetin on the migration of CRC cells induced by trypsin, transwell assays were performed. Trypsin treatment (100 nM) significantly promoted HCT116 and HT-29 migration (Figure 6C). Fisetin (30 μM, 60 μM, and 120 μM) obviously inhibited the migration of both types of cells compared with the trypsin-treated group (Figure 6A and 6B). The migration of the cells of the 30 μM fisetin incubated group was even less than that of the control group. These findings indicated that fisetin can effectively inhibit the migration of CRC.

Effects of fisetin on the migration induced by trypsin of HCT116 and HT-29 cells.
Fisetin and Trypsin Affect CRC Cells Proliferation and Apoptosis Through the Modulation of NF-κB Pathways
To explore the molecular mechanisms by which fisetin affects pro-apoptosis CRC cells, the cell proliferation and apoptosis-related proteins were examined. As demonstrated in Figure 7, the expression of p21 and p27 was markedly elevated, while that of cyclin D1 and NF-κB p65 were significantly decreased in both HCT116 and HT-29 cells treated with fisetin (60 μM and 120 μM); the opposite results were observed in the trypsin-treated group, although there was no statistical difference compared with the control group (Figure 7A and 7B). We then explored the role of NF-κB in CRC cell proliferation and apoptosis mediated by fisetin and trypsin. After loading NF-κB expression vector into HCT116 cells, the increased expression of p21, p27, cyclin D1, and NF-κB p65 induced by fisetin were significantly repressed (Figure 8). These findings suggested that trypsin may promote the growth of CRC cells through the NF-κB signaling pathway, and the inhibitory effect of fisetin on cell growth is also through this signaling pathway.
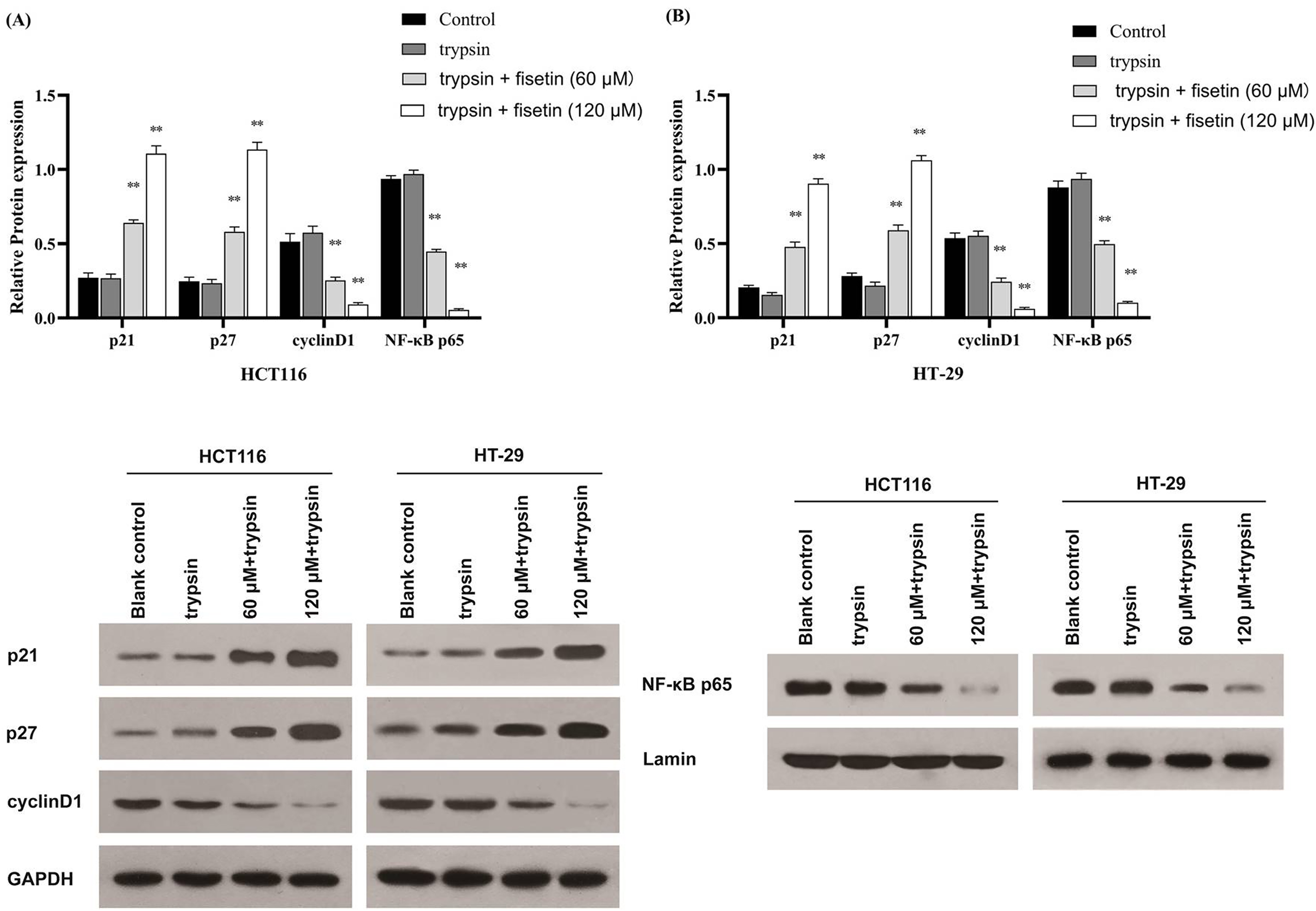
Fisetin affects colorectal cancer (CRC) cells proliferation and apoptosis through the modulation of NF kappa B (NF-κB) pathways.

NF kappa B (NF-κB) expression vector reversed the effect of fisetin on colorectal cancer (CRC) cells.
The effects of fisetin on cancer have been studied, but the mechanism of action remained unclear. The NF-κB pathway seems to play a vital role in fisetin against cancer.36–38 Overexpression of NF-κB consequently induced a deterioration of cancers. For example, several studies have demonstrated that activation of the NF-κB signaling pathway promotes inflammation-associated cancer, and inhibition of it in hepatocytes attenuates the onset of hepatocellular carcinoma related to inflammation.37,38 In this study, treatment with fisetin increased p21 and p27 expression levels and decreased cyclin D1 and NF-κB p65 levels, while treatment with trypsin reversed the changes in the CRC cell lines, suggested that activation of NF-κB p65 and cyclin D1 played an important role in trypsin-induced CRC cell growth. Moreover, we also found that supplementation with NF-κB remarkably neutralized the effects of fisetin on anti-apoptotic and pro-apoptotic protein levels. These data demonstrated that fisetin induced cell apoptosis in CRC by inhibiting the activation and transcriptional activity of NF-κB. However, the role of NF-κB in cancer remains controversial. Some research indicated that the NF-κB signaling pathway suppressed, but not promoted, the progress of oncology, implying a tumor suppressor role for NF-κB in cancers.39–41 Further investigation using cell and animal models is, therefore, necessary to elucidate the causative mechanism on how fisetin exerts its anti-carcinogenic effects, in addition to the NF-κB pathway.
Fisetin Suppresses the Growth of Cancer in Tumor-Bearing NOG Mice
To assess the in vivo antitumor activity of fisetin, the subcutaneous tumor-bearing NOG mice model was used with HCT116 cells. Fisetin at different dosage (30, 60, and 120 mg/kg) and saline were injected intraperitoneally for 6 weeks. As shown in Figure 9, administration of fisetin significantly decreased the tumor growth compared with the control group, especially at high dosage. The morphology of the tumor shown in Figure 9 revealed that the tumor in mice treated with fisetin was obviously smaller compared with that in the saline treated mice. These findings indicated that fisetin induces a forceful antineoplastic activity in CRC in vivo.

Fisetin suppressed the growth of cancer in tumor-bearing NOG mice.
Fisetin Causes no Acute or Chronic Toxicity in Animals
To investigate the safety of the clinical use of fisetin, acute and chronic toxicity tests were conducted in Kunming mice and Wistar rats, respectively. At the dosage of 223 mg/kg, fisetin showed no significant toxicity in mice, without mortality or other abnormal changes, including in skin, mucous membrane, fur color, eyes, circulation, central nervous system, respiration, and conscious behavior. In addition, the growth of mice in the experimental group was not significantly different from that of the control group (Table 1). After a long-term administration of 6 successive weeks, fisetin also did not cause significant differences in body weight (Table 2) and food consumption (Table 3), neither were there noteworthy abnormalities in the hematology (Table 4) and biochemistry (Table 5), or obvious changes in the major organs of the rats. The results indicate that fisetin, at the studied dosage, does not cause acute and chronic toxicity in animals.
Body Weight Changes of Mice in the Acute Toxicity Test.

Values are expressed as mean ± SEM. Fisetin-treated group showed nonsignificant changes when compared with the control group.
Body Weight Changes of Rats in Chronic Toxicity Test.
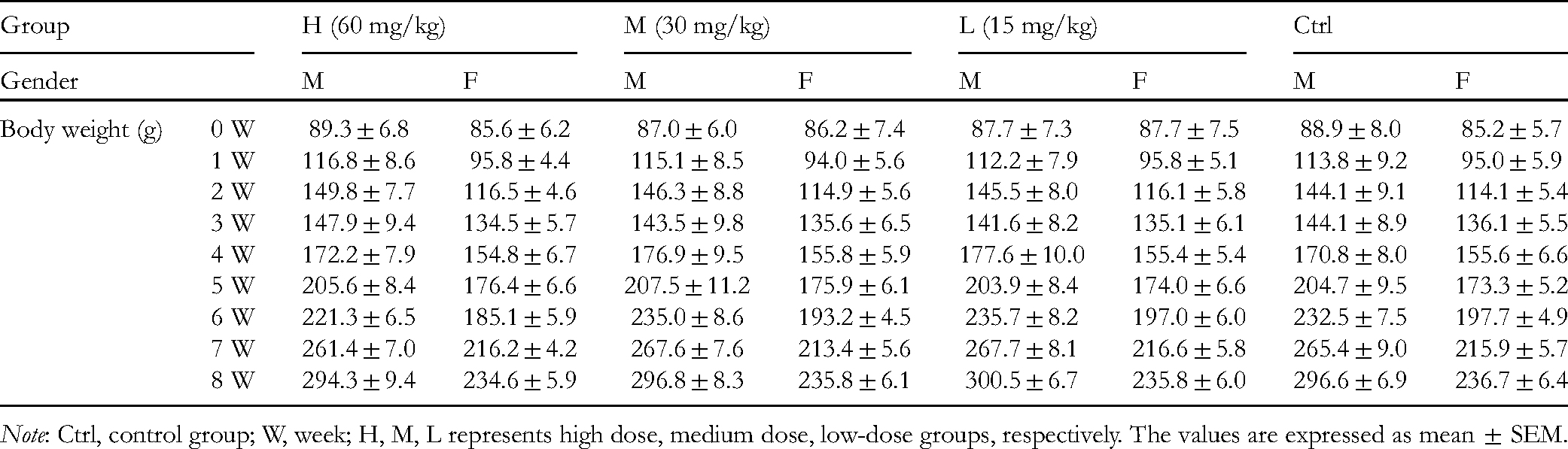
Note: Ctrl, control group; W, week; H, M, L represents high dose, medium dose, low-dose groups, respectively. The values are expressed as mean ± SEM.
Food Consumption of Rats in Chronic Toxicity Test.
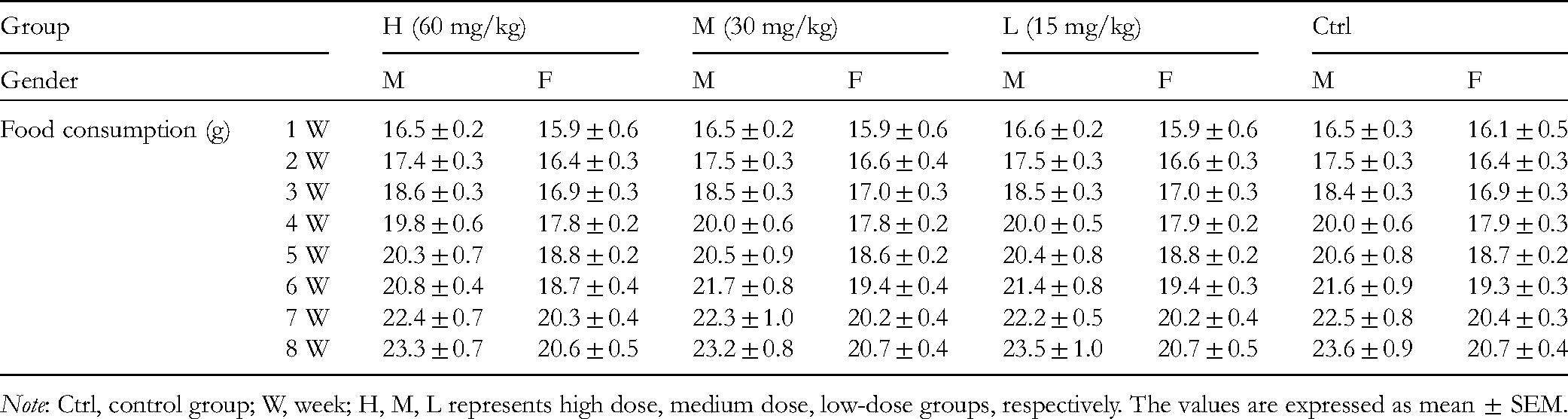
Note: Ctrl, control group; W, week; H, M, L represents high dose, medium dose, low-dose groups, respectively. The values are expressed as mean ± SEM.
Hematological Parameters of Rats in Chronic Toxicity Test.

Note: Ctrl, control group; H, M, L represents high dose, medium dose, low-dose groups, respectively. The values are expressed as mean ± SEM.
Abbreviations: HGB, hemoglobin; LYMPH, lymphocyte; MONO, mononucleosis; NEUT, neutrophil; PLT, blood platelets; RBC, red blood cell; WBC, total white blood cell.
Biochemical Parameters of Rats in Chronic Toxicity Test.

Note: Ctrl, control group; H, M, L represents high dose, medium dose, low-dose groups, respectively. The values are expressed as mean ± SE.
Abbreviations: ALT, alanine aminotransferase; AST, aminotransferase; ALB, albumin; BUN, blood urea nitrogen; CRE, creatinine; GLU, glucose; TC, total cholesterol.
Conclusions
In conclusion, fisetin significantly promoted CRC apoptosis, arrested the cell cycle, and suppressed CRC proliferation and migration induced by trypsin, partially through modulating NF-κB pathways and inhibiting trypsin activity. Fisetin also showed cancer growth inhibition in a tumor-bearing NOG mice model. It is safe at an effective dosage in animals. Fisetin is a promising drug for CRC treatment. Our findings provide useful information for understanding the therapeutic effects of fisetin on CRC, but the possible mechanism involved in the inhibition of CRC growth and migration induced by fisetin needs further study.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221115511 - Supplemental material for Fisetin Inhibits Trypsin Activity and Suppresses the Growth of Colorectal Cancer in Vitro and in Vivo
Supplemental material, sj-docx-1-npx-10.1177_1934578X221115511 for Fisetin Inhibits Trypsin Activity and Suppresses the Growth of Colorectal Cancer in Vitro and in Vivo by Lin Li, Min Wang, Hongyan Yang, Yuting Li, Xiaoling Huang, Jialiang Guo and Zheng Liu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Field Projects (Intelligent Manufacturing) of General Universities in Guangdong Province (2020ZDZX2057), International Science & Technology Cooperation Program of Guangzhou, china (201807010022), National Natural Science Foundation of China (81872832, 82073806, and 82173781), Guangdong Basic and Applied Basic Research Foundation (2019A1515010806), and the Scientific Research Projects (Characteristic Innovation) of General Universities in Guangdong Province (2019KTSCX195).
Ethical Approval
All animal experiments were approved by the Ethics Committee of the Institute of Laboratory Animal Science of Jinan University (N2021054), Guangzhou, China, and the animal protocols fulfilled the institutional guidelines on the National Institutes of Health guidelines (NIH, USA).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
