Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) mainly affects respiratory tracts including the trachea and lungs. As inflammation and cytokine storm are major pathological features in a Syrian hamster for SARS-CoV-2 infection, reducing inflammatory responses or antiviral therapy is an effective strategy for treating SARS-CoV-2 infections. Herbal medicines and natural substances are applied worldwide due to their health-beneficial effects. Although chemical and pharmacological verifications have not been made for all substances, the pharmacological effects of many substances are being studied. In this study, Boswellia serrata, Commiphora myrrha, and propolis known to have anti-inflammatory and antimicrobial properties were selected as candidates for therapeutic herbal medicine targeting SARS-CoV-2. When this herbal mixture was treated to SARS-CoV-2 infected Vero E6 and Calu-3 cells, effective antiviral effects were demonstrated. Next, this herbal mixture was administered to SARS-CoV-2-infected Syrian hamsters, and histopathological lesions were significantly reduced. Viral spike protein and viral enzyme gene were also significantly less detected in immunohistochemistry and quantitative real-time PCR results, respectively. Thus, it can be concluded that a mixture of Boswellia serrata, Commiphora myrrha, and propolis as natural substances has the potential to reduce lesions of respiratory tracts upon SARS-CoV-2 infection.
Introduction
Herbal medicine is the use of herbs, herbal materials, herbal preparation, and herbal products to treat diseases and improve general health. 1 Herbal products as medication have been used for thousands of years, beginning with ancient civilizations. In present times, its role is being studied in detail through scientific analysis. The use of herbal products is increasing and expanding due to their anti-inflammatory to anti-cancer effects. 2 However, various problems associated with the usage of herbal products are increasingly reported. Intoxication by heavy metals such as arsenic, mercury, and lead has been continuously reported worldwide.3,4 Hepatotoxicity is known as the most fatal and representative side effect. 5 To prevent these disadvantages, many purification techniques are being developed and a method of extracting active components rather than ingesting whole-plant is being used. 6
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, which has become a global pandemic since early 2020, is still ongoing after more than 2 years. SARS-CoV-2 has shown a capability to constantly mutate, resulting in several notable variants. And as of 2022, the lineage B.1.1.529, the so-called Omicron variant, has outcompeted other variants of concern. 7 The severity of patients admitted to hospitals with SARS-CoV-2 infections has been shown to be lower with an Omicron variant infection than with a Delta variant infection. 8 Clinical symptoms of SARS-CoV-2 infection range from no symptoms to severe respiratory and death. The most common symptoms reported are fever, cough, myalgia, fatigue, sputum, and headache. 9 To relieve these clinical symptoms and to find antiviral activities, a range of herbal medications have been examined. Especially in China, many studies have been published regarding the efficacy, safety, pros, and cons of using herbal medicine in clinical experience with meta-analysis.10–12 Although not all herbs are effective against SARS-CoV-2, quite a few have been shown to be able to relieve clinical symptoms and exhibit antiviral effects.
In the present study, we examined the effects of a mixture of three different herbal materials on respiratory lesions from SARS-CoV-2 infection. Syrian hamster as an experimental animal model has been widely used in SARS-CoV-2 studies. 13 Unlike humans, Syrian hamster does not show clinical symptoms such as fever after SARS-CoV-2 infection. In addition, they can recover quickly with low mortality. 14 However, microscopic lesions of respiratory tracts caused by a viral infection can be observed in Syrian hamsters, with viral clearance and reduced lesions occurring 7 days after infection. 15 Thus, we studied the effects of a mixture of herbs on respiratory tract lesions. Since representative respiratory lesions are inflammatory cell infiltration and interstitial pneumonia in both SARS-CoV-2 infected Syrian hamsters and humans,15,16 we believe that our results could be applied to human medicine.
Results
HPLC Results of YSK-A
The concentration of YSK-A provided by YESKIN, KOREA is 20 g/L in DW and we administered 6 mL/kg of the solution orally to Syrian hamsters. This means that the concentration of YSK-A that was administered to the Syrian hamsters is 120 mg/kg. By taking 5 different compounds of YSK-A shown in Figure 1 and Table 1, we determined the administered concentration of the solution to be 0.066 mg/kg (a), 0.7872 mg/kg (b), 1.2312 mg/kg (c), 0.3516 mg/kg (d), 0.186 mg/kg (e), respectively (a, Cinnamic acid; b, Galangin; c, Furanoeudesma 1,3-diene; d, KBA and e, AKBA).
Quantitative Analysis Results of YSK-A.

Abbreviations: KBA, 11-keto-β-boswellic acid; AKBA, acetyl-11-keto-β-boswellic acid; STD, standard; RSD %, percent relative standard deviation (n = 5).

HPLC chromatograms of chemical standards (1: Cinnamic acid, 2: Galangin, 3: Furanoeudesma 1,3-diene, 4: 11-keto-β-boswellic acid (KBA), and 5: acetyl-11-keto-β-boswellic acid (AKBA). Standards and YSK-A extracts.
YSK-A Inhibits SARS-CoV-2 Propagation
To determine whether YSK-A displays an anti-SARS-CoV-2 activity, Vero E6 cells were either mock-infected or infected with SARS-CoV-2 (MOI = 0.01) in the absence or presence of different concentrations of YSK-A. At 2 dpi, the cytopathic effect (CPE) was monitored by light microscopy. As shown in Figure 2a, CPE was prominently observed by the morphological change in SARS-CoV-2-infected Vero E6 cells. We showed that SARS-CoV-2-induced CPE was markedly inhibited by 50 μg/mL YSK-A. Of note, SARS-CoV-2-induced CPE was almost blocked in the presence of 100 μg/mL YSK-A. To verify whether YSK-A exerts an anti-SARS-CoV-2 activity, Vero E6 cells were either mock-infected or infected with SARS-CoV-2 (MOI = 0.01) in the absence or presence of various concentrations of YSK-A. At 24 hours postinfection, SARS-CoV-2 NP levels were determined by immunoblot analysis. As expected, SARS-CoV-2 NP expression levels gradually decreased with YSK-A in a dose-dependent manner (Figure 2b). We also demonstrated that viral protein expression is nearly blocked with 100 μg/mL YSK-A. YSK-A consists of Boswellia serrata, Commiphora myrrha, and propolis. To further analyze which component of these herbal extracts induces antiviral activity, Vero E6 cells were either mock-infected or infected with SARS-CoV-2 (MOI = 0.01) in the absence or presence of various concentrations of each herbal extract. Figure 2c showed that SARS-CoV-2 NP levels were not altered by Boswellia serrata regardless of its concentration. Meanwhile, Commiphora myrrha inhibits SARS-CoV-2 NP level in a dose-dependent manner (Figure 2d). Consistently, SARS-CoV-2 NP levels were decreased by propolis in a dose-dependent manner (Figure 2e). Interestingly, anti-SARS-CoV-2 activity of propolis is stronger than that of Commiphora myrrha. Collectively, these data suggest that YSK-A may be a potent anti-SARS-CoV-2 agent. Next, we evaluated the antiviral efficacy of YSK-A against emerging SARS-CoV-2 variants. As shown in Figure 2f, YSK-A decreased SARS-CoV-2 NP of both Delta (left) and Omicron (right) variants in a dose-dependent manner. It is noteworthy that the antiviral efficacy of YSK-A is more efficient in variants than wild-type SARS-CoV-2 (Figure 2b vs 2f). Calu-3 has been used to verify in vitro results for drug development. To further verify the antiviral effect of YSK-A on SARS-CoV-2 propagation in lung cells, Calu-3 cells were infected with SARS-CoV-2 at an MOI of 0.1 in the presence of various concentrations of YSK-A. As shown in Figure 2g, NP levels of wild-type SARS-CoV-2 decreased on treatment with 100 ug/mL YSK-A (left panel). Consistently, SARS-CoV-2 NP levels of Delta decreased by YSK-A (Figure 2g, right panel), verifying that YSK-A suppresses SARS-CoV-2 propagation in human lung cells. Although antiviral activities minimally differ by variants, these data indicate the potentiality of YSK-A as an effective antiviral against not only wild-type but also SARS-CoV-2 variants.

In vitro study of YSK-A with various severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variants on Vero E6 and Calu-3 cells. (A) Vero E6 cells were either mock-infected or infected with SARS-CoV-2 WT (MOI = 0.01) for 1 hour in the absence or presence of indicated amounts of YSK-A. At 48 hours postinfection, the cytopathic effect (CPE) in Vero E6 cell was visualized under a light microscope. (B-E). Vero E6 cells were either mock-infected or infected with SARS-CoV-2 for 1 hour in the absence or presence of indicated amounts of YSK-A (b), Boswellia serrata (c), Commiphora myrrha (d), and propolis (e), and further cultured in media containing the indicated concentrations of herbal extracts. At 24 hours postinfection, SARS-CoV-2 NP levels were determined by an immunoblot assay using the indicated antibodies. (F) Vero E6 cells were either mock-infected or infected with SARS-CoV-2 Delta (MOI = 0.1) (left panel) or Omicron (MOI = 1) (right panel) for 1 hour in the absence or presence of indicated amounts of YSK-A. (G) Calu-3 cells were either mock-infected or infected with SARS-CoV-2 WT (MOI = 0.1) (left panel) or Delta (MOI = 1) (right panel) for 1 hour in the absence or presence of indicated amounts of YSK-A. (f, g) At 24 hours postinfection, SARS-CoV-2 NP levels were determined by an immunoblot assay using the indicated antibodies.
Bodyweight of SARS-CoV-2 Infected Syrian Hamster Is Not Affected by YSK-A Treatment
Since the antiviral activity of YSK-A was confirmed by in vitro experiment, the SARS-CoV-2 infection Syrian hamster model was used for the therapeutic effects of YSK-A. SARS-CoV-2 was inoculated via intranasal route to Syrian hamster and YSK-A was administered via oral route once a day for 4 and 7 days. As a result of measuring the body weight once a day during the experiment period, the NC group showed continuously increased weight. Meanwhile, PC and YSK-A groups showed a continuously decreasing trend of body weight, although a tendency of increasing body weight was observed from 5 dpi. There was no significant difference in body weight change between PC and YSK-A groups (Figure 3).
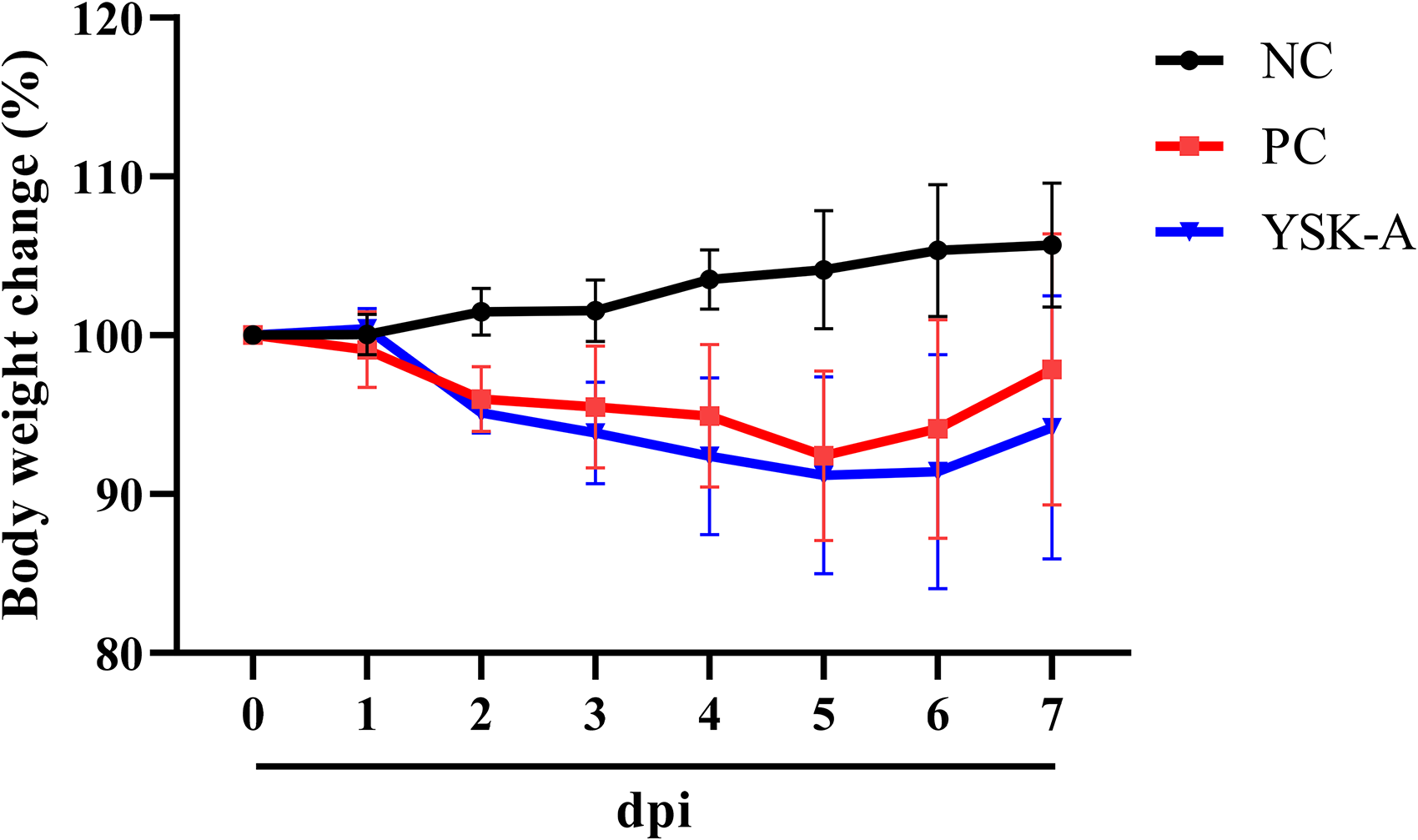
Weight change rates in experimental groups during the experiment period.
YSK-A Treated Group Shows Less Microscopic Lesions in Respiratory Tracts Than PC Group
Histopathological examination of the respiratory tract revealed various degrees of inflammatory lesions and tissue damage in PC and YSK-A groups compared to the NC group at 4 dpi. In the case of the trachea epithelium, morphological damages such as vacuolation in the cytoplasm and stratified epithelium were observed. Infiltration of inflammatory cells in the lamina propria was observed in PC and YSK-A groups. In the PC group, the transformation was observed from pseudostratified columnar to stratified cuboidal epithelial structure in the tracheal epithelium while tracheal cilia were not observed. Meanwhile, the tracheal epithelium of the YSK-A group was more intact. It maintained a pseudostratified columnar shape compared to that of the PC group. Tracheal cilia were not observed either. Infiltrated inflammatory cells in the lamina propria were less in the YSK-A groups than in the PC group. Inflammatory cell infiltration in the peribronchiolar region, bronchiolar lumen, and the perivascular region was observed to be relieved in the YSK-A group compared to that in the PC group. Interstitial pneumonia, in which the alveolar wall thickened due to the proliferation of alveolar cells and infiltration of inflammatory cells, was observed with reduced inflammatory reactions and/or mild tissue damage levels in the YSK-A group compared to that in the PC group (Figure 4a).

Histopathological examination and its representative images. Microscopic changes of the respiratory tract at (A) 4 dpi and (B) 7 dpi (H&E). (C) Evaluation of histopathological lesions scoring following specific criteria. All data are presented as mean ± SD (n = 6 per group).
At 7 dpi, the trachea showed mild morphological changes in tracheal epithelium such as loss of cilia in PC and YSK-A groups. Mild infiltration of inflammatory cells into the lamina propria was observed. Inflammatory cell infiltration in the peribronchiolar region, bronchiolar lumen, and the perivascular region was observed in both PC and YSK-A groups. However, the recruitment of these inflammatory cells was less in the YSK-A group than in the PC group. Severe interstitial pneumonia was observed in both PC and YSK-A groups (Figure 4b).
According to the evaluation criteria, as a result of histopathological examination of the respiratory tract at 4 dpi, for both trachea and lung, the YSK-A group showed a significantly lower histopathological score than the PC group (P-values were .0022 and .0002, respectively). At 7 dpi, microscopic differences were significant only in the lung. The YSK-A group showed a lower histopathological score than the PC group (P-value = .0269) (Figure 4c).
YSK-A Treated Group Shows Less Viral Positive Areas in Respiratory Tracts Than PC Group
Immunohistochemistry (IHC) analysis for SARS-CoV-2 NP in the respiratory tract revealed a clear positive area in the trachea, lung parenchyma, and bronchiolar epithelium of PC and YSK-A groups at 4 and 7 dpi. At 4 dpi, a positive area was observed in the trachea epithelium, bronchiolar epithelium, and recruited inflammatory cells. The pulmonary parenchyma did not show positive staining for SARS-CoV-2 NP (Figure 5a). At 7 dpi, a positive area was not detected in the tracheal epithelium. However, strong positive reactions were detected in pulmonary parenchyma, bronchiolar epithelium, and recruited inflammatory cells (Figure 5b). IHC quantification results revealed that the YSK-A group had a lower percentage of positive area in both trachea and lung than the PC group at 4 dpi (P-value < .0001 for both). At 7 dpi, the trachea did not show significant differences among the study groups. However, the YSK-A group showed a significantly lower percentage of the positive area in the lung than the PC group (P-value < .0001) (Figure 5c).

Immunohistochemistry (IHC) results of the lung at (A) 4 dpi and (B) 7 dpi. DAB chromogen and counterstain with methyl green. (C) Quantification of IHC positive areas in respiratory tracts for SARS-CoV-2 nucleocapsid protein (NP). All data are presented as mean ± SD (n = 10 per group).
SARS-CoV-2 RdRp Enzyme Is Less Expressed in Respiratory Tracts of the YSK-A Treated Group Compared to That in the PC Group
Quantitative real-time PCR (qRT-PCR) was performed to compare the expression level of SARS-CoV-2 RdRp in the respiratory tract. In the trachea at 4 and 7 dpi, SARS-CoV-2 RdRp expression showed no significant differences between the PC and YSK-A groups. Meanwhile, SARS-CoV-2 RdRp was significantly less expressed in the lung of the YSK-A group than that in the PC group at both 4 and 7 dpi (P-value < .0001 for both) (Figure 6).

Relative expression of SARS-CoV-2 RdRp by qRT-PCR. All data are presented as mean ± SD (n = 6 per group).
Infectious SARS-CoV-2 Is Only Detected in the Lung at 4 dpi Without Significant Differences Between PC and YSK-A Groups
50% Tissue culture infectious dose (TCID50) was determined to identify pathogenic viruses in the respiratory tract. CPE was confirmed in the lung of both PC and YSK-A groups at 4 dpi. Virus titer was slightly lower in the lung of the YSK-A group than that in the PC group. However, it showed no statistically significant difference between PC and YSK-A groups (Figure 7).

Viral titration with TCID50 in the lung at 4 dpi. All data are presented as mean ± SD (n = 6 per group).
Discussion
Phytochemicals (from the Greek word Phyto—meaning plant) are chemical compounds from plants that are used for disease treatments and health benefits. 17 Because plant-origin compounds are safe and easily available, thousands of candidates have been tested for medical usage. In the past, the efficacy of various unrefined plants in traditional medicine has been passed down within the region. However, recently, various extraction techniques have been studied to increase safety by extracting desired specific components. 18 In the present study, we used three natural plant/compounds, Boswellia serrata, Commiphora myrrha, and propolis. The beneficial effects of these three components on health are summarized in Table 2.
List of beneficial effects of Boswellia serrata, Commiphora myrrha, and propolis.

Boswellia serrata has been applied to many inflammatory diseases, such as ulcerative colitis, 20 rheumatoid arthritis, 21 and chronic colitis. 22 However, the beneficial effects of Boswellia serrata are not well studied yet compared to the other two plants/compounds. Commiphora myrrha is mainly used for its antimicrobial activities and cytotoxic properties.41–43 It also has analgesic 26 and anti-inflammatory effects. 27 A previous study has shown that a mixture of Commiphora myrrha and Boswellia carterii has significant anti-inflammatory and analgesic effects. 39 Propolis is a resinous mixture that honeybees collect from various plant sources and mix them with wax and exudates from their salivary glands. 32 As propolis is derived from plant resins, its contents can vary because of geological and environmental habitats. 33 Propolis has been applied as a natural remedy since ancient times. 34 The most well-known beneficial effect of propolis is its anti-microbial activities against bacteria, fungi, and viruses.34,36 Especially, flavonoids in propolis are known for having antibacterial, antifungal, and antiviral action. 44 In the present study, our propolis extract powder was from Australia and it contained 8.97% of flavonoids. Propolis has also been used for wound healing and anti-inflammation.34,35,38,40,44,45 Moreover, many previous studies have indicated that propolis has an anti-tumor effect on immunomodulatory function.34,44,46,47 As propolis has a long history of application to humans, its beneficial effects are actively studied with numerous medical benefits demonstrated. These three ingredients show a common scientifically evident anti-inflammation effect. However, they might show other unidentified potential effects. Indeed, in our preliminary study, we confirmed that the isolated murine splenic natural killer (NK) cells were activated following YSK-A treatment when NK cells were co-cultured with NK-sensitive cell target (YAC-1). The flowcytometric result indicated that as the concentration of YSA-A increased (50, 100, and 200 μg/mL) the percentage of NK-specific lysis also significantly increased (data was not shown).
As we discussed above and summarized in Table 2, the antiviral effects of Boswellia serrata, Commiphora myrrha, and propolis were ambiguous, especially against SARS-CoV-2. In this regard, we investigated whether they had an antiviral effect for SARS-CoV-2 with in vitro study. By measuring SARS-CoV-2 NP using immunoblot analysis, we figured out that YSK-A had an antiviral effect for the SARS-CoV-2 WT strain. Especially when each component was examined, not Boswellia serrata but Commiphora myrrha and propolis showed an antiviral effect against SARS-CoV-2 WT on Vero E6 cells (Figure 2b-2f). Also, as a result of examining whether this antiviral effect of YSK-A applied to other SARS-CoV-2 variants, antiviral effect was confirmed on both Delta and Omicron variants (Figure 2f). Moreover, the feasibility of the antiviral effect for YSK-A on Calu-3 cells, which originated from bronchial adenocarcinoma and most representative cell line to drug screening for pulmonary disease, was also confirmed (Figure 2g). These in vitro study results indicated that the YSK-A had a clear antiviral effect for SARS-CoV-2 and its variants.
After confirming that YSK-A has an antiviral effect on in vitro studies, we investigated the therapeutic possibility of YSK-A in vivo. Our in vivo study results indicated that a mixture of Boswellia serrata, Commiphora myrrha, and propolis had a therapeutic effect on SARS-CoV-2 infection in Syrian hamsters when respiratory lesions were examined (Figure 4c). A previous study has indicated that representative microscopic respiratory lesions are inflammatory cell infiltration, interstitial pneumonia, and tracheal epithelium damage in SARS-CoV-2-infected Syrian hamsters. 15 Consistent with the previous study, we could observe those microscopic findings in SARS-CoV-2-infected groups. Both YSK-A and PC groups showed microscopic lesions. However, the YSK-A group had milder lesions than the PC group. When lesions were quantified through objective criteria, they showed statistically significant differences between the two groups. As we summarized in Table 2, previous studies indicated that Boswellia serrata, Commiphora myrrha, and propolis have anti-inflammatory activities, respectively. Therefore, the administration of YSK-A, which is a herbal mixture of the three extracts mentioned above, may have reduced the infiltration of inflammatory cells and subsequent microscopic changes caused by SARS-CoV-2 infection. In practice, in human medicine, not only anti-viral medications but also anti-inflammatory medications are prescribed. For example, nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen and meloxicam are frequently used for the relief of pain and inflammation against SARS-CoV-2 infection. 48 In this regard, we assume that based on in vivo results and references the main mechanism of action of reduced respiratory lesions in the present study is the anti-inflammatory response following YSK-A treatment. In addition, although there is a risk of misuse and abuse, herbs are safer than chemical medications with fewer side effects. 49
In the case of Boswellia serrata, it is known to have strong anti-inflammatory activities through inhibition of tumor necrosis factor-alpha, interleukin-1 beta, and mitogen-activated protein kinases. 50 An in silico study has revealed that some ligands from Boswellia serrata can interact with proteins of SARS-CoV-2. 51 Similarly, Commiphora myrrha containing the product (Tiryaq-E-Wabai) has a good docking power for SARS-CoV-2 protein in an in silico study. 19 Propolis also has powerful anti-viral, anti-inflammatory, and immunoregulating activities. Many research papers have suggested that propolis is likely to be effective against SARS-CoV-2. 31 Although we could not confirm which components played a key role in reducing lesions in the present experiment, we confirmed that their mixture shows a positive therapeutic effect on SARS-CoV-2 infection in Syrian hamsters. To apply this remedy in humans for SARS-CoV-2 infection, clinical trials are required. We hope that YSK-A can be widely used as an alternative treatment for SARS-CoV-2 infection.
Unlike the clear histopathological results, results of IHC, qRT-PCR, and TCID50 were not satisfying for proving the presence of the virus. In the case of the viral enzyme RdRp with qRT-PCR and NP with IHC as methods for measuring the presence of a viral antigen, the YSK-A group showed significantly lower measurements than the PC group (Figures 5c and 6). Infectious virus was only detected in the lung at 4 dpi (Figure 7). Because the former two methods measure RNA and protein of the virus, it is difficult to clearly conclude that YSK-A prevented viral propagation on in vivo environment. According to previous studies, Syrian hamster infected with SARS-CoV-2 showed peak viral load at 2 to 4 dpi. Viral load then decreased, with no infectious virus observed at 7 dpi.15,30 Boswellia serrata, 51 Commiphora myrrha, 19 and propolis 31 are known to have antiviral effects against some viruses. They can bind to SARS-CoV-2 in the in silico experiment. Also, our in vitro study revealed that YSK-A had an antiviral effect for various SARS-CoV-2 variants (Figure 2). Furthermore, it is difficult to conclude that YSK-A had a lack of antiviral effect in in vivo environment, because YSK-A was administrated orally. Thus, it is necessary to consider how substances act after being administered and metabolized. To overcome these limitations, an experimental animal model is needed in which infectious viral loads in the respiratory system can be detected for a long time.
Conclusions
In conclusion, we examined the therapeutic effects of a mixture of Boswellia serrata, Commiphora myrrha, and propolis as herbal medicine. Our in vitro data showed that SARS-CoV-2 protein levels were markedly decreased by YSK-A in both Vero E6 and Calu-3 cells. We further demonstrated that YSK-A displayed antiviral activities against SARS-CoV-2 variants, including highly pathogenic Delta and highly transmissible Omicron. When this mixture was administrated orally to SARS-CoV-2-infected Syrian hamsters, we observed an improvement in respiratory lesions compared to the experimental group not administrated with the mixture. We believe that this mixture has the potential as a healthy functional food that can relieve respiratory lesions caused by SARS-CoV-2 infection.
Materials and Methods
Preparation of Infectious SARS-CoV-2
Vero E6 cells and Calu-3 cells were maintained in complete DMEM containing 10% heat-inactivated FBS. Each variant of SARS-CoV-2; NCCP-43331 (WT), NCCP-43405 (Delta), and NCCP-43412 (Omicron) were provided by the National Culture Collection for Pathogens, Republic of Korea. All experiments were conducted in a biosafety level 3 (BL3) laboratory at the Korea Zoonosis Research Institute, Jeonbuk National University. VeroE6 cells were infected with SARS-CoV-2 diluted in OPTI-MEM for 1 hour. Cells were further cultured in DMEM supplemented with 2% FBS, 1% penicillin-streptomycin, and HEPES (Invitrogen, USA). At 2 days postinfection (dpi), viral titers were determined by the median TCID50 assay.
Sample Preparation
A powder mixture (1:1:1) of Boswellia serrata, Commiphora myrrha, and propolis (named as YSK-A) used in this study was provided by YESKIN, KOREA. Boswellia serrata ethyl alcohol (EtOH) extract powder (UMALAXM ORGANICS Pvt. Ltd, June 2020) and propolis extract powder (AC biotech Pty. Ltd, October 2020) were purchased as commercially available products. Commiphora myrrha extract powder was obtained as follows. Commiphora myrrha (SAMIN pharma. INC., December 2021) was extracted with 50% EtOH at 90° for 8 hours. The extract was concentrated and prepared as a freeze-dried powder.
Standard compounds cinnamic acid, galangin, 11-keto-β-boswellic acid (KBA), acetyl-11-keto-β-boswellic acid (AKBA), and furanoeudesma 1,3-diene were purchased from Coresciences, Korea.
High-Performance Liquid Chromatography Analysis
For all separations, a Kinetex 5 μm EVO (C18, 5 μm, 4.6 ×
The YSK-A provided by YESKIN, KOREA was dissolved in distilled water at a final concentration of 20 g/L. It was then used as the sample for in-vivo experiments.
Western Bolt Analysis
Cells were washed in cold PBS and lysed in a cell lysis reagent (Promega). The samples were centrifuged at 15,000 r/min for 10 minutes at 4 °C. Equal amounts of proteins were subjected to SDS-PAGE and electro-transferred to a nitrocellulose membrane. The membrane was blocked in Tris-buffered saline containing 5% nonfat dry milk for 1 hour and then incubated overnight at 4 °C with the indicated antibodies in TBST buffer containing 1% BSA. The SARS-CoV-2 nucleoprotein was detected using an anti-nucleoprotein antibody (40143-V08B, Sino Biological). An anti-actin antibody was purchased from Sigma-Aldrich. The membrane was then incubated with either horseradish peroxidase (HRP)-conjugated goat anti-rabbit antibody or goat anti-mouse antibody (Jackson ImmunoResearch Laboratories) in TBST buffer for 1 h at room temperature. Proteins were detected using an ECL kit (Amersham Biosciences)
Animal Infection and Sample Collection
All animal experiments including maintaining, infection, sample treatments, physical examination, and necropsy were performed in an animal BL3 (ABL3) facility. All researchers were approved and qualified for ABL3 studies. All experimental procedures were reviewed and approved by the Animal Ethics Committee of Jeonbuk National University (Approval No. JBNU 2021-019).
Thirty-six male Syrian hamsters (Mesocricetus auratus) at 6-week-old and 100 g-weight were purchased from Central Laboratory Animal, Korea. Before the experiment, all animals were kept per two in individually ventilated isolator cages under optimal physical environments (24 ± 2 °C, 50 ± 5% humidity). Experimental groups were divided into 4 dpi and 7 dpi. Each dpi group was subdivided into three groups as follows: no viral infection and no treatment (negative control, NC), SARS-CoV-2 infection and no treatment (positive control, PC), and SARS-CoV-2 infection and YSK-A treatment. One week period was applied to animals for adaption. They were maintained on a 12 h/12 h light/dark cycle. After adaption, 1 × 106 PFU SARS-CoV-2 in a 100 μL volume was inoculated intranasally to each hamster under light isoflurane anesthesia. YSK-A was administered orally at a dose of 6 mL/kg once a day for 4 or 7 days.
Measurement of Virus Infectivity
Either lung or trachea tissue samples were collected from SARS-CoV-2-infected hamsters at the indicated time points and then homogenized in serum-free media. Samples were centrifuged at 3000 r/min for 10 minutes to remove debris. Vero E6 cells cultured in 96-well plates were inoculated with a 10-fold serially diluted virus. At 5 days after virus inoculation, TCID50 of SARS-CoV-2 was measured by detecting the presence or absence of CPE in cells. It was calculated using the Reed–Muench method. 23
RNA Extraction and qRT-PCR
Total RNAs were isolated from the lung and trachea tissues using Trizol (Tri-RNA Reagent, FARRR 001, Favorgen) and Hybrid-R RNA purification kit (305-101, GeneAll Biotechnology) following each manufacturer's instructions. RNAs were incubated with DNase I including an RNase inhibitor and then reverse transcribed to complementary DNAs (cDNAs) using a ReverTra Ace qPCR RT Master Mix (FSQ-301, TOYOBO) in accordance with the manufacturer's protocol. cDNAs were amplified via qRT-PCR on a CFX96™ Real-Time PCR Detection System (Bio-Rad Laboratories) using SYBR Green Master Mix (PB20, PCR Biosystems). Relative messenger RNA (mRNA) expression level was compared with that of hypoxanthine–guanine phosphoribosyltransferase as an internal control. PCR primer sequences are listed in Supplementary Table 1.
Histopathology and IHC
All samples were collected and fixed in 10% neutral buffered formalin. After 48 hours of viral inactivation, formalin-fixed tissues were taken out from the ABL3 facility. Fixed tissues were routinely processed and embedded in paraffin (Surgipath Paraplast, Leica Biosystem Inc.). Formalin-fixed-paraffin-embedded tissue blocks were sectioned at 4 μm in thickness and placed onto silane-coated glass slides. Before staining, deparaffinization in xylene and hydration in serial ethyl alcohol were performed for tissue slides. Tissue slides were stained with hematoxylin and eosin (H&E) with a standard laboratory protocol for microscopic examination. To evaluate and quantify microscopic lesions, previously published criteria for SARS-CoV-2 infected Syrian hamsters 15 were applied.
For IHC, antigens were retrieved with citrate acid buffer (pH 6.0) at 95 °C for 30 minutes and room temperature for 20 minutes. After antigen retrieval, slides were incubated with a SARS-CoV-2 nucleocapsid protein (NP) antibody (40143-V08B, Sino Biological) at a 1:500 dilution in antibody diluent (E09-300, GBI labs) at 4 °C overnight. A horseradish peroxidase-conjugated anti-rabbit IgG antibody (MP-7500, Vector Laboratory) was used to label SARS-CoV-2 NP. To conjugate peroxidase-labeled antibodies, 3,3’-diaminobenzidine (DAB, SK-4105, 1:300, Vector Laboratory) was used. Methyl green was used for counterstaining.
To quantify the positive area of IHC stained tissue, ten images were randomly taken from each group in a defined magnification field and area (400 magnification field, 0.144 mm2). The percentage of dark-brown colored positive cells was calculated using the image analysis software TS Auto 5.1 (Olympus).
All slides were examined using a light microscope (BX53, Olympus). All microscopic images were obtained using a mounted camera (DP80, Olympus). All analyses were conducted in a double-blinded manner with trained veterinary pathologists at Jeonbuk National University Veterinary diagnostic center.
Statistical Analysis
All data are expressed as means ± standard deviations (SD). One-way analysis of variance (ANOVA) was used for confirming the statistical significance among study groups. To measure specific differences among pairs of means, Duncan's multiple range test was used as a post hoc test. A P-value of less than 0.05 was considered statistically significant. P-values less than 0.05 are flagged with one asterisk (*). P-values less than 0.01 are flagged with two asterisks (**). P-values less than 0.001 are flagged with three asterisks (***). P-values less than .0001 are flagged with four asterisks (****). All statistical analyses were performed using SAS statistical software Version 9.4 (SAS Institute Inc.). All graphs were drawn using GraphPad Prism Version 8.0.1 (GraphPad Software) and PowerPoint Version 2204 (Microsoft).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231180709 - Supplemental material for Antiviral and Therapeutic Effects of a Mixture of Boswellia serrata, Commiphora myrrha, and Propolis for SARS-CoV-2
Supplemental material, sj-docx-1-npx-10.1177_1934578X231180709 for Antiviral and Therapeutic Effects of a Mixture of Boswellia serrata, Commiphora myrrha, and Propolis for SARS-CoV-2 by Myeon-Sik Yang, Yun-Sook Lim, Byungkwan Oh, Seok-Chan Park, Daram Yang, Soon B Hwang and Bumseok Kim in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was fully supported by YESKIN, KOREA and partially supported by Basic Science Research Program through the National Research Foundation (NRF) funded by the Ministry of Education (2019R1A6A1A03033084).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
