Abstract
Neuroinflammation is a critical factor in the onset and progression of various brain disorders. Upon exposure to inflammatory stimuli, glial cells become activated, releasing excessive proinflammatory mediators that exacerbate neuropathological conditions. Nitric oxide contributes to neuroinflammation, neurotoxicity, and neuronal death, making its regulation in activated glial cells essential for treating neuroinflammation. Lespedeza bicolor, a traditional medicine used for fever, cough, and nephritis treatment, has a neuroprotective effect attributed to the pterocarpan 1-methoxylespeflorin G11 (MLG), as identified in our previous study. This study examines the protective role of MLG in lipopolysaccharide (LPS)-stimulated glial cells. We found that MLG treatment attenuated LPS-induced nitrite release by downregulating inducible nitric oxide synthase expression in astrocytes. Additionally, MLG impeded LPS-induced nuclear translocation of the p65 subunit of nuclear factor-kappa-light-chain-enhancer of activated B cells (NF-κB) and decreased LPS-induced phosphorylation of extracellular signal-regulated kinase (ERK). The MLG demonstrated similar inhibitory effects on nitrite release and p65 nuclear translocation in microglial cells. Molecular docking analyses unveiled the potential binding mode of MLG to ERK. Collectively, our results indicate that MLG may possess therapeutic potential in treating neuroinflammatory diseases by suppressing nitrite release in glial cells.
Introduction
Lespedeza bicolor (LB) is a plant species widely distributed across Eastern Asia, Australia, and North America, 1 and is traditionally employed to treat various ailments such as fever, nephritis, diabetes, diuresis, azotemia, and headache. 2 The LB extract is known to contain molecules exhibiting antioxidant, anti-inflammatory, and antiglucotoxic properties.3–5 Studies have shown that ethanol extract of LB can ameliorate amyloid beta-induced memory impairment in the brain by enhancing brain-derived neurotrophic factor, cAMP response element binding, Akt, and extracellular signal-regulated kinase (ERK) pathways. 6 Various bioactive compounds, including flavonoids, isoflavonoids, catechin, daidzein, luteolin, and genistein have been identified in LB.3,6
Neuroinflammation plays a significant role in the onset and progression of numerous neurodegenerative diseases. 7 During neuroinflammation, proinflammatory cytokines and mediators display abnormal elevation, which exacerbates pathological conditions and neuronal damage. Nitric oxide (NO), a bioactive free radical, is crucial in brain function, neuronal death, and neuroinflammation. Low levels of NO contribute to maintaining the supply of oxygen and nutrients by regulating brain vascular tone. 8 Additionally, NO is implicated in learning, memory, and presynaptic/postsynaptic outgrowth.9,10 However, excessive NO production is associated with various neurodegenerative diseases, such as Alzheimer's disease and Parkinson's disease.11,12 Excessive NO triggers neuronal death through demyelination and N-methyl-d-aspartate receptor-mediated excitotoxicity,13,14 as well as inducing apoptosis in neurons by increasing oxidative and nitrosative stress in conjunction with mitochondrial dysfunction. 15
NO is produced by 3 NO synthase (NOS) enzymes: endothelial NOS (eNOS), inducible NOS (iNOS), and neuronal NOS (nNOS). Among these, iNOS is expressed in glial cells, astrocytes, and microglia. 16 In contrast to eNOS and nNOS, iNOS is believed to induce toxic effects, as it is responsible for excessive NO production in glial cells during neurotoxic damage or under pathological conditions such as ischemia or trauma.16,17 Moreover, elevated NO production stimulates glial cells, leading to the overproduction of several proinflammatory mediators and aggravating neuroinflammation. 15 Therefore, appropriate regulation of NO release from activated glial cells may offer a strategy to combat neuroinflammation-induced brain damage.
Although LB has been reported to exhibit antioxidant properties, its potential anti-inflammatory activities in the brain remain unexplored. Since LB has demonstrated the ability to partially counteract the effects of amyloid beta exposure in animal models, it may possess protective effects on the brain. The bioactive compound responsible for these positive effects, however, has remained elusive until recently. In our previous study, we identified 1-methoxylespeflorin G11 (MLG) as a bioactive compound in the methanolic extract of LB and demonstrated that MLG confers protective effects against glutamate-induced neurotoxicity by inhibiting the generation of reactive oxygen species (ROS) and preventing mitochondrial dysfunction. 18 In the present study, we investigated the regulatory effect of MLG on NO release in glial cells during neuroinflammation.
Materials and Methods
Isolation of MLG from LB
The isolation and structural elucidation of MLG have been previously described. 19 The EtOAc fraction (51.1 g) obtained from LB was subjected to silica gel CC and eluted using a gradient of CH2Cl2-MeOH (100:1 to 1:100, v/v) to yield 4 fractions (E1-E4). Fraction E1 (16.4 g) was further purified using silica gel CC (n-hexane-EtOAc, 10:1 to 2:1, v/v; CH2Cl2-MeOH, 15:1, v/v) to obtain 5 subfractions (E1-1-E1-6). Subfraction E1-1 (3.5 g) was then subjected to reversed-phase C18 medium-pressure liquid chromatography (RP C18-MPLC) with a gradient of 10% MeOH to 100% MeOH, yielding 7 subfractions (E1-1-1-E1-1-7). MLG (purity >98%, as determined by high-performance liquid chromatography [HPLC] analysis, Figure S1A) was isolated from subfraction E1-1-4 via preparative HPLC using a CH3CN-H2O mixture (13:7).
Materials
Dulbecco's modified eagle medium/F-12 (DMEM/F-12), fetal bovine serum (FBS), and penicillin/streptomycin were acquired from Thermo Fisher Scientific. Lipopolysaccharide (LPS; O55:B5) and ERK inhibitor PD184352 (PZ0181) were obtained from Merck (Sigma-Aldrich). Antibodies targeting ERK (9102S), p-ERK (9101S), c-Jun N-terminal kinase (JNK; 9252S), p-JNK (9251S), p38 (9212S), p-p38 (9211S), and p65 (8242S) were sourced from Cell Signaling Technology. The iNOS antibody (PA3-030A), 4′,6-diamidino-2-phenylindole (DAPI; D1306), and Trizol were procured from Thermo Fisher Scientific, while the β-actin (sc-47778) and glial fibrillary acidic protein (GFAP, sc-33673) antibodies were purchased from Santa Cruz Biotechnology. Horseradish peroxidase-conjugated secondary antibodies, including goat antimouse IgG (1706516) and goat antirabbit IgG (1706515), were acquired from Bio-Rad Laboratories. Alexa Fluor 488 donkey antimouse IgG (ab150105) and Alexa Fluor 594 donkey antirabbit IgG (ab150076) were obtained from Abcam. GoScript™ Reverse Transcriptase was sourced from Promega and Midori Green Advance was purchased from NIPPON Genetics Europe.
Animal Care and Ethics
All experimental procedures adhered to the Principle of Laboratory Animal Care guidelines 20 and received approval from Chung-Ang University's Institutional Animal Care and Use Committee (IACUC) (approval number 2017-00093).
Cell Culture and Treatments
Primary astrocytes were isolated from the cortices of postnatal day 1 Sprague-Dawley rats, following a previously reported method. 21 Rat pups were acquired from Young Bio. Cortices were extracted postdecapitation, dissociated into single cells via mechanical pipetting, and cultured on poly-D-lysine (PDL)-coated T75 flasks in DMEM/F12 containing 10% FBS, 100 μg/mL streptomycin, and 100 U/mL penicillin. Cells were maintained at 37 °C in a humidified incubator with 5% CO2, with the culture medium replaced every 2-3 days. On day in vitro (DIV) 7, astrocytes were detached using 0.25% trypsin, seeded onto PDL-coated plates or coverslips, and allowed to grow for an additional 5 days. On DIV 12-13, astrocytes were pretreated with either MLG or PD184352 (5 μM) for 1 h before LPS treatment.
BV-2 mouse microglia cells (ICLC catalog code: ATL03001) were provided by Dr. Dong-Kug Choi (Konkuk University, Chungju-si, Chungcheongbuk-do, Republic of Korea). These cells were cultured in DMEM supplemented with 5% FBS, 100 μg/mL streptomycin, and 100 U/mL penicillin at 37 °C in a humidified environment containing 5% CO2. Astrocytes and BV-2 cells were treated with 10 and 100 ng/mL LPS, respectively, while vehicle (Veh) treatment entailed the use of 0.1% dimethyl sulfoxide (DMSO).
Nitrite Measurement
NO release was assessed using Griess reagent (1% sulfanilamide in 5% H3PO4 and 0.1% naphthylethylenediamine). The supernatant was incubated with Griess reagent for 5 min at 20 °C-25 °C, and absorbance was recorded at 540 nm with a microplate reader (BioTek Instruments). The amount of nitrite released was determined by comparing the results to a standard curve generated with sodium nitrite.
Western Blot
Cell lysis was performed using RIPA buffer (Biosesang, Inc.), and protein concentrations were quantified via the BCA assay. Proteins (20-30 μg per well) were separated on a 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel and subsequently transferred to a nitrocellulose membrane (Whatman, Cytiva). The membranes were blocked with 5% skim milk and incubated with primary antibodies at 4 °C overnight. After washing, the membranes were incubated with horseradish peroxide-conjugated secondary antibodies for 1 h at 20 °C-25 °C and visualized using a chemiluminescence system (Vilber). Protein band intensities were analyzed using ImageJ software (version 1.38; National Institutes of Health).
RNA Extraction and Reverse Transcription-Polymerase Chain Reaction
Total RNA was isolated using Trizol (Invitrogen), and complementary DNA (cDNA) was synthesized from 1 μg of total RNA using GoScript reverse transcriptase. Polymerase chain reaction (PCR) amplification of cDNA was performed using the following primers: iNOS (forward: 5′-AGG CAC AAG ACT CTG ACA CC-3′, reverse: 5′-GGT AGG GTA GAG GAG GGG AG-3′) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; forward: 5′-GGA CCA GGT TGT CTC CTG TG-3′, reverse: 5′-CAT TGA GAG CAA TGC CAG CC-3′). The PCR program consisted of 25-30 cycles of 95 °C for 2 min, 55 °C for 30 s, and 72 °C for 1 min. The PCR products were separated on an agarose gel containing 1× Midori Green Advance (NIPPON Genetics Europe) and visualized. Band intensities were quantified using ImageJ software (version 1.38; National Institutes of Health).
Cell Viability
Cell viability was evaluated using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Astrocytes or BV-2 cells were pretreated with specified concentrations of MLG and incubated with or without LPS for 24 h. The MTT was added at a final concentration of 100 μg/mL, and the cells were incubated for 2 h at 37 °C. Afterward, the medium was removed, and the insoluble formazan crystals were dissolved in DMSO. The absorbance was measured at 540 nm.
Immunocytochemistry
For immunocytochemical analysis, cells were fixed using 4% paraformaldehyde for 15 min at room temperature (20 °C-25 °C) and permeabilized with 0.1% Triton X-100 for an additional 15 min at the same temperature. Coverslips were then incubated with primary antibodies in a blocking solution composed of 1% bovine serum albumin and 3% FBS in Dulbecco's phosphate-buffered saline at 4 °C overnight. Following wash steps, the coverslips were exposed to secondary fluorescence antibodies for 1 h at room temperature (20 °C-25 °C) before staining nuclei with 0.5 μg/mL DAPI solution for 5 min at room temperature. Images were captured using a confocal microscope (LSM 800, Carl Zeiss).
Binding Affinity Assay
The ChemOffice software was employed to construct the ligand's 3-dimensional structure and conduct energy minimization analysis. The extracellular ERK crystal structure was obtained from the Protein Data Bank (6RQ4). 22 The molecular docking of MLG to ERK's crystal structure was simulated using LeDock software (http://lephar.com), yielding 10 conformations ranked by predicted binding energies (score), with the highest-scoring molecule having a binding energy of −7.23 kcal/mol. The predicted conformations were visualized using PyMOL software (PyMOL Molecular Graphics System, Version 1.2r3pre) and Discovery Studio (BIOVIA, Dassault Systèmes, BIOVIA Discovery Studio).
Statistical Analysis
Results were reported as mean ± SEM derived from a minimum of 3 independent experiments. Statistical analyses were conducted using GraphPad Prism 5.0 software (version 5.01; GraphPad Software, Inc.). Data were assessed using a one-way analysis of variance followed by Tukey's posthoc test, and differences were considered significant when P < .05.
Results
Effects of MLG on NO Release and iNOS Expression in LPS-Stimulated Primary Astrocytes
To elucidate the regulatory impact of MLG on nitrite release in glial cells, we assessed nitrite levels in LPS-stimulated astrocytes. Primary astrocytes were preincubated with MLG for 1 h before LPS stimulation for 24 h. The LPS exposure significantly increased nitrite release, which was attenuated by MLG pretreatment (Figure 1A). Excessive nitrite production in glial cells, mediated by iNOS, contributes to neurotoxicity during inflammation. 16 Therefore, we examined the influence of MLG on iNOS expression. The MLG pretreatment mitigated LPS-induced upregulation of iNOS protein levels (Figure 1B) and messenger RNA expression (Figure 1C). To determine whether MLG affected cytotoxicity, we assessed cell viability using the MTT assay. A concentration of 5 μM MLG did not significantly reduce cell viability, while 10 μM MLG was cytotoxic (Figure 1D). Collectively, these findings demonstrate that MLG suppresses LPS-induced nitrite release by inhibiting iNOS expression in astrocytes.

Effects of MLG on LPS-induced nitrite release in primary astrocytes. Astrocytes were pretreated with MLG at indicated concentrations for 1 h prior to LPS treatment. Nitrite levels, protein and mRNA expression, and cell viability were analyzed after 24 h incubation with LPS. (A) Nitrite levels measured using the Griess reagent. (B) Representative images of western blot analysis; iNOS protein expression levels normalized to β-actin. (C) Relative band intensity of iNOS with GAPDH as a loading control. (D) Viability of astrocytes treated with MLG assessed by the MTT assay. ###, P < .001 compared with Veh; **, P < .01 compared with Veh + LPS; ***, P < .001 compared with Veh + LPS.
Effects of MLG on LPS-Induced Mitogen-Activated Protein Kinase Activation
Mitogen-activated protein kinase (MAPK) is known to control cellular nitrite levels and iNOS expression in glial cells in response to inflammatory stimuli. 23 In line with our previous research, LPS exposure increased iNOS expression and MAPK activation in the cortex and cultured astrocytes. 24 To investigate the involvement of MAPK activation in MLG-mediated nitrite release suppression, astrocytes were pretreated with MLG for 1 h before LPS exposure, and MAPK phosphorylation was analyzed by western blot after 1 h of LPS treatment. The LPS enhanced the phosphorylation of ERK, JNK, and p38. However, MLG treatment significantly reduced p-ERK levels and marginally decreased p-JNK and p-p38 levels (Figure 2). These findings suggest that MLG selectively diminishes ERK phosphorylation in LPS-stimulated astrocytes.

Effects of MLG on MAPK activity in LPS-stimulated astrocytes.
Effects of MLG on Nuclear Translocation of p65 in LPS-Stimulated Astrocytes
Several transcription factors are implicated in iNOS expression, among which the nuclear factor κ-light-chain-enhancer of activated B cells (NF-κB) p50/p65 complex plays a critical role in regulating iNOS gene expression.15,17 Inhibitor of NF-κB (IκB) sequesters the NF-κB complex in the cytosol and mediates its ubiquitin-mediated degradation. On the other hand, IκB inhibition triggers NF-κB translocation to the nucleus, where it binds to the promoter region of iNOS. 25 Therefore, we explored the modulatory effect of MLG on the nuclear translocation of p65 in LPS-stimulated astrocytes using immunocytochemistry. The MLG treatment attenuated LPS-induced nuclear translocation of p65 (Figure 3A and B) and mitigated the LPS-induced upregulation of GFAP expression, a marker of astrocyte activation. 26 Furthermore, the ERK inhibitor, PD184352, exhibited a similar effect to MLG on p65, suggesting that MLG suppresses nitrite release by inhibiting p65 nuclear translocation in astrocytes during inflammation.
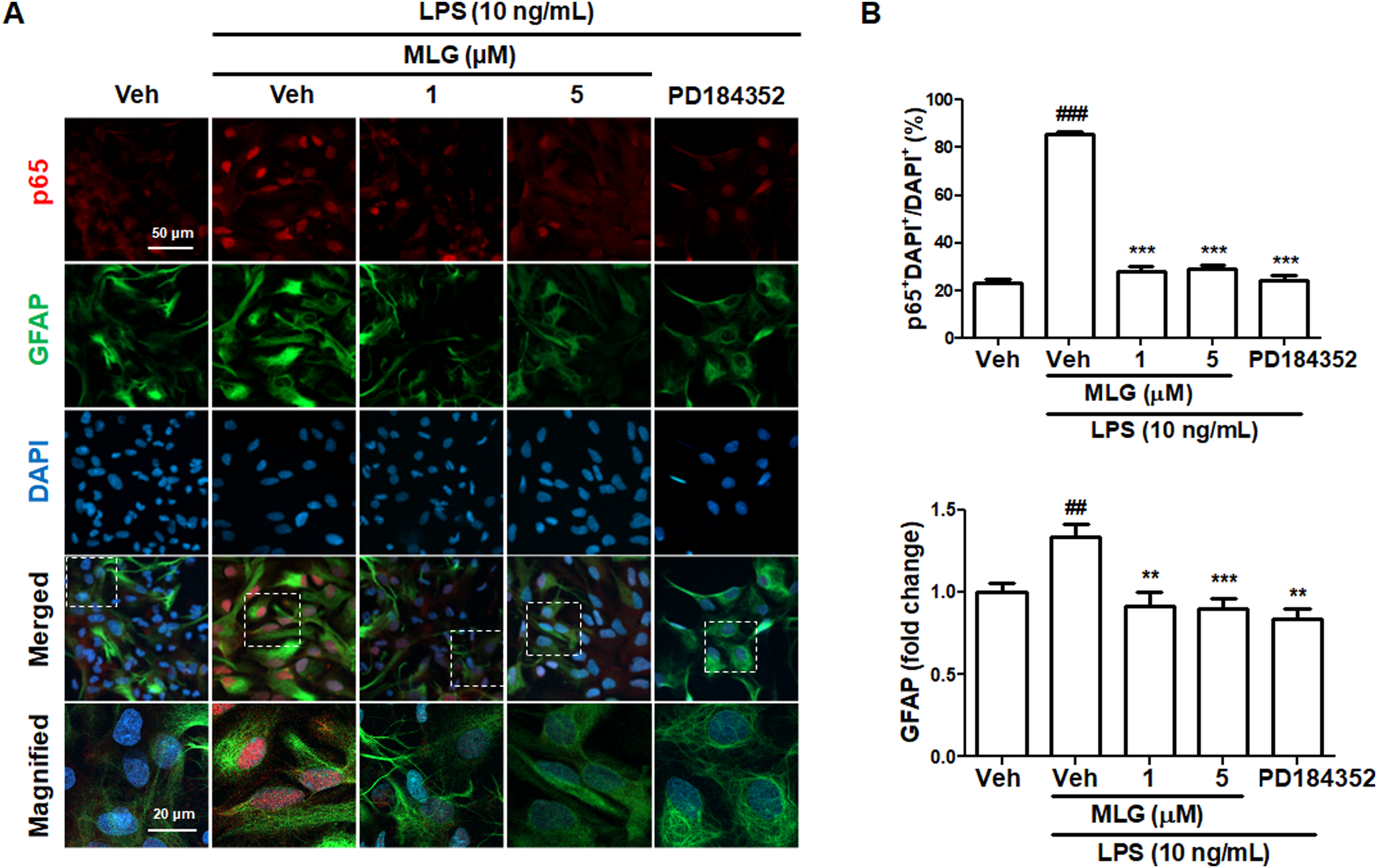
Effects of MLG on nuclear translocation of p65 in LPS-stimulated primary astrocytes. Astrocytes were preincubated with MLG or PD184352 and stimulated with LPS. Cells were fixed 2 h after LPS treatment and immunostained to visualize p65 and GFAP. (A) Representative images of immunocytochemistry. (B) Quantification of nuclear p65 and relative GFAP expression compared to Veh. Nuclear translocation of p65 was measured by counting p65-positive cells in DAPI-positive nuclei. ###P < .001 compared with Veh; **P < .01 compared with Veh + LPS; ***P < .001 compared with Veh + LPS.
Effects of MLG on NO Release and Nuclear Translocation of p65 in LPS-Stimulated BV-2 Cells
To further elucidate the regulatory roles of MLG on NO release in microglia, BV-2 cells were subjected to MLG and LPS treatments in a manner analogous to astrocyte treatments. The MLG treatment abrogated LPS-induced NO release (Figure 4A) without causing cytotoxicity in BV-2 cells (Figure 4B). Moreover, MLG treatment diminished LPS-induced nuclear translocation of p65 in BV-2 cells (Figure 4C). Collectively, these findings indicate that MLG inhibits NO release by impeding the nuclear translocation of p65 in LPS-stimulated glial cells.

Effects of MLG on nitrite release and nuclear translocation of p65 in BV-2 cells. BV-2 cells were pretreated with MLG at indicated concentrations. After 1 h of MLG pretreatment, cells were treated with LPS. (A) Nitrite levels were measured 24 h after LPS treatment. (B) Cytotoxicity of MLG measured by the MTT assay 24 h after LPS treatment. (C) Representative images and quantification of nuclear translocation of p65 in LPS-stimulated BV-2 cells. After 2 h of LPS treatment, BV-2 cells were fixed and immunostained to visualize p65. Nuclear translocation assessed by p65 signal in DAPI-positive nuclei. ###P < .001 compared with Veh; ***P < .001 compared with Veh + LPS.
Molecular Docking of ERK to MLG
The inhibitory effect of MLG on ERK (PDB 6RQ4) was investigated through in silico molecular docking studies. The structure of LY-3214996, cocrystallized with ERK, was found to be similar to that of MLG. Additionally, western blotting results demonstrated that MLG inhibited ERK phosphorylation. Consequently, ERK (PDB 6RQ4) was chosen for docking experiments, conducted using LeDock. The binding mode of MLG was compared to the corresponding cocrystallized molecule, LY-3214996, which was docked to the active site of the ERK protein (Figure 5A and B). A binding energy of −8.46 kcal/mol was recorded for the ligand (LY-3214996) and protein complex. The redocked ligand–protein complex exhibited interactions between the ligand (LY-3214996) and the ERK protein, involving amino acids such as IIE 31, TYR 36, VAL 39, ALA 52, MET 108, THR 110, LYS 114, and LEU 156 (Figure 5A and B). With a root mean square deviation (RMSD) value lower than the crystallographic resolution of 6RQ4, these results validated the molecular docking methodology in accordance with previous studies. 27

Mode of binding between LY-3214996 and the ERK protein.
The MLG–ERK complex displayed RMSD values below 1 (Figure 6A), indicating that MLG binds deeply within the cavity, resulting in a low binding energy of 7.23 kcal/mol through interactions with amino acids such as TYR 36, VAL 39, LYS 54, GLU 71, CYS 166, ASP 167, and LEU 156. The TYR 36 phenol ring exhibited an “out” conformation, consistent with other ERK-specific ATP-competitive inhibitor conformations. Moreover, several amino acid residues, including TYR 36, GLU 71, LYS 114, and ASP 169, have been reported to participate in the critical interaction domain of ERK.28–31 The 2-dimensional representation of MLG's binding mode predicted a crucial hydrogen bond with GLU 71 and van der Waals interactions involving MET 38, MET 108, PHE 168, GLY 169, and others (Figure 6B). Furthermore, amino acid residues such as TYR 36, VAL 39, and LEU 156 were found to be shared between the MLG–ERK and LY-3214996–ERK protein complexes, suggesting that both molecules bind within the same pocket.

Mode of binding between MLG and the ERK protein. (A) Overview of the potential binding pocket. Interacting residues are labeled with residues and ligands shown in stick figures. (B) Interactions between the ERK protein and MLG are shown in 2D.
Discussion
During neuroinflammation, glial cells release proinflammatory mediators, and an excessive or dysregulated release of these mediators can cause neural damage. NO has been implicated in neuroinflammation and neurodegenerative diseases due to the observed increase in inflammatory mediators and nitration of pathological inclusions in synucleinopathies and tauopathies.32–34 Elevated NO levels can disrupt mitochondrial function, increase free radical generation, and activate cyclooxygenase.35,36 Overproduction of NO appears to result from the hyperactivation of astrocytes and microglia, 15 with a strong correlation between NO overproduction, glial cell activation, and neuroinflammation in the human brain. 37 iNOS is rarely expressed in normal brain tissues but is strongly expressed in astrocytes and microglia under neuroinflammatory stimuli or neuropathological conditions. 38 In our study, we demonstrated that MLG attenuated LPS-induced NO release by regulating iNOS expression in glial cells. As astrocytes and microglia are the primary glial cells producing excessive NO through iNOS expression in the brain, we investigated the regulatory effect of MLG on NO production in astrocytes and BV-2 cells, which are representative microglial cells commonly used to identify bioactive molecules with antineuroinflammatory properties.39–41
The LPS treatment is a widely employed method for studying neuroinflammation mechanisms in glial cells and identifying novel bioactive compounds with anti-inflammatory properties.24,42 The LPS activates the MAPK signaling pathway and induces iNOS expression in primary astrocytes.43,44 Interestingly, anti-inflammatory compounds have been shown to exhibit differential effects on MAPK activation while successfully suppressing the expression of proinflammatory mediators in glial cells.45,46 For instance, the antidepressant tianeptine selectively inhibits the activation of ERK but not JNK or p38, yet it still reduces the expression of proinflammatory mediators and iNOS in LPS-stimulated microglial cells. 47
The ERK contributes to iNOS expression during NF-κB activation. Various stressors and signaling pathways have been implicated in the induction of iNOS expression in glial cells, including ROS, the Toll-like receptor/MyD88 pathway, the caspase pathway, and the Raf/MAPK kinase/ERK signaling pathway.17,48 In our current research, MLG demonstrated suppressive effects on ERK activation without influencing the phosphorylation of JNK or p38. Our previous work indicated that MLG represses glutamate-induced MAPK activation in HT22 cells, 18 a discrepancy that may arise from cell type-specific effects. Investigating the binding affinity of MLG and MAPK in various cell types could provide valuable insights.
Natural compounds have been proven to be valuable resources for drug discovery. 49 For instance, Camptothecin and Taxol, derived from Camptotheca acuminata and Taxus brevifolia, respectively, are renowned cancer treatment drugs. 50 Furthermore, natural products facilitate multitarget approaches in drug development. Curcumin exemplifies this, as it exhibits antioxidant, antiproliferative, and anti-inflammatory effects by targeting several molecules, such as cyclooxygenase-1, p53, and the NF-κB signaling pathway.51–53 A myriad of phytochemicals have been proposed as potential therapeutic agents for neuroinflammation and neurodegenerative diseases, as they modulate glial cell activation and inhibit the production of proinflammatory mediators.45,54 Despite the promising results of numerous natural products in drug discovery, the specific bioactive compounds responsible for these effects and the underlying mechanisms remain largely unexplored.
Pterocarpans, bioactive prenylated flavonoids, exhibit enhanced lipophilicity and membrane permeability due to the presence of a prenyl moiety.55,56 As pterocarpans, including MLG, have been shown to inhibit ROS formation and monoamine oxidase B,57–59 within cells, as well as disrupt cell membrane integrity, 60 it is likely that MLG can partially penetrate cell membranes. Pterocarpans comprise the second largest group of isoflavonoids and have ethnopharmacological significance as alternative medicine, with demonstrated anticancer, antifungal, and anti-inflammatory properties.61–63 Furthermore, these compounds have been found to inhibit NO production in macrophages.64,65 Our findings support the regulatory role of MLG in NO production in glial cells during inflammation. Given the role of NO and the NF-κB pathway in the expression of other inflammatory cytokines,66,67 MLG may also suppress additional proinflammatory proteins.
Conclusion
In this study, we found that MLG, a compound present in LB, exerts anti-inflammatory effects by inhibiting the ERK and NF-κB pathways in glial cells. Building on a previous study that highlighted MLG's protective role in glutamate-induced neurotoxicity, it is plausible that MLG may also protect against neuroinflammation in the brain. Collectively, these findings suggest that MLG could be a potential therapeutic compound for the treatment or management of neuroinflammation-induced neurological diseases. Future research exploring the protective effects of MLG against neuroinflammation in vivo would be of great interest.
Footnotes
Author Contributions
YMP conducted experiments with primary astrocytes and BV-2 cells. MV analyzed the data and supervised the study. NC performed molecular docking analyses of MLG. SHL oversaw the experiments and authored the manuscript. All authors have approved the submitted version of the manuscript for publication.
Data Availability
The data generated in this study are available upon reasonable request from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Administration Committee of the IACUC at Chung-Ang University.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the 2020 Chung-Ang University Young Scientist Scholarship (CAYSS) and the National Research Foundation (NRF) of Korea, funded by the Korean government (MSIT) under grant numbers NRF-2020R1C1C1008852 and NRF-2022R1C1C1009626.
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted in accordance with the guidelines set forth by the IACUC at Chung-Ang University (Approval number: 2017-00093), Seoul, Republic of Korea.
Statement of Informed Consent
As there were no human subjects involved in this study, informed consent is not applicable.
