Abstract
Herbal medicine can present an alternative way of treating liver cancer. Here, we explored a matrine- and sophoridine-containing herbal compound medicine (AH-05) extracted from Adenophora capillaris, Sophora flavescens, Astragalus, and other plants. H22 and HepG2 cell models, as well as an H22 xenograft model, were established. Cell proliferation and apoptosis were measured in vitro, and tumor volume and weight were observed in vivo. The activation of AKT/mTOR and nuclear factor-κB (NF-κB) pathways in tumor cells and the polarization of CD4/CD8 T cells in the spleen were tested. To assess safety, hematological toxicity and pathology of the liver, kidney, spleen, and intestine were evaluated. AH-05 inhibited cell viability in a dose- and time-dependent manner. In vivo, tumor volume and weight were reduced, and the activation of NF-κB p50, NF-κB p65, AKT, p-AKT Ser473, and mTOR was suppressed. In addition, AH-05 promoted CD4+ T cell polarization in the spleen. With regard to safety, slight intestinal mucosa edema was observed, but no severe pathological or hematological toxicity was detected. AH-05 exhibited its therapeutic effects against liver cancer by regulating the AKT/mTOR and NF-κB signaling pathways, and the immune environment, by promoting CD4+ T cell polarization in the spleen. Thus, AH-05 represents a potential supplementary herbal compound medicine for liver cancer.
According to a report by the National Central Cancer Registry of China (NCCR), liver cancer is one of the most fatal cancers and its mortality rate presents an upward trend. 1,2 Effective treatments of liver cancer are limited by the complicated molecular mechanisms underlying its pathogenesis. Liver resection, interventional therapy, radiotherapy, biotherapy, and molecular target therapy have their advantages, but these do not always produce optimal patient outcomes. 3 -5 Many studies have been conducted on the use of herbal medicines (HMs) in patients with liver cancer. 6,7 The use of HMs is now widespread in many countries, where they are considered an initiative therapeutic option for liver cancer, because of their multilevel, multitarget, and coordinated intervention effects. 2,8
In clinical practice, HMs are prescribed for terminal-stage and maintenance-stage cancer patients following general treatment. After 12 years of tracking clinical cases, the Chinese University of Hong Kong revealed that HMs may be capable of inhibiting tumor progression, having an immunomodulatory effect and protecting cancer patients from suffering complications, increasing sensitivity and reducing side effects of chemoradiotherapy, and improving the quality of life and survival of patients. 9 -12 Proving the clinical success of HMs is expected to pave the way for the adoption of HM-based therapies for cancer treatment. 2 However, although a great deal of effort has gone into understanding the therapeutic principles and molecular targets of HM in cancer treatment, the mechanisms underlying the action of most HM formulas remain unclear due to the complex composition of these medicines. 13 -15
In addition, researchers have indicated that many HMs and the compounds extracted from them, such as alkaloids, phenylpropanoids, flavonoids, and saccharides, have considerable antitumor effects. 16,17 Alkaloids are useful for treating breast cancer, pancreatic cancer, colon cancer, and liver cancer, 18,19 and they have been receiving an increasing amount of attention for liver cancer treatment. Previous data have shown that alkaloids suppress cancer cell proliferation, migration, and invasion, in addition to them triggering cell cycle arrest and decreasing cytoskeletal stability by interacting with various pathways, such as the MAPK, AKT/mTOR, and nuclear factor-κB (NF-κB) pathways. 4,16 In this study, we explore the anticancer properties of AH-05, which is a matrine- and sophoridine-containing herbal compound medicine, in relation to treating liver cancer.
Material and Methods
Cell Lines and Culture
An H22 mouse liver cancer cell line and a HepG2 human liver cancer cell line were purchased from the Shanghai Institutes for Biological Sciences. Cells were cultured under a 5% CO2 environment at 37 °C in Dulbecco's Modified Eagle Medium (DMEM) high glucose (Thermo HyClone) supplemented with 10% fetal bovine serum and 1% penicillin and streptomycin.
Alkaloids From the Herbal Compound (AH-05)
AH-05 is an HM prescription for the treatment of liver cancer developed by the Pharmaceutical Department of Chengdu Fuxing Hospital. This HM is prepared mainly from the extracts of Sophora flavescens root (Chinese name: KU-SHENG; habitat: Inner Mongolia, China), Adenophora capillaris (Chinese name: NONG-DAN-CAO; habitat: Sichuan, China), Astragalus (Chinese name: Huang Qi; habitat: Gansu, China), and Viscum coloratum (Chinese name: HU-JI-SHENG; habitat: Hei Longjiang, China). The herbs were mixed and pulverized into a coarse powder, soaked in 0.5% hydrochloric acid at 26 °C for 2 hours and then extracted ultrasonically thrice (40 minutes each time; 20 kHz). The solution was concentrated and adjusted to a pH of 10 with strong ammonia water, and the precipitate was discarded by centrifugation at 3500 rpm for 25 minutes, before being repeatedly extracted thrice with dichloromethane (CH2Cl2). The extract was dried in rotary evaporators and a vacuum drying chamber; the dried fractions were stored at 26 °C in a dryer.
Alkaloid Analysis of AH-05
High-performance liquid chromatography (HPLC) was used to identify matrine, oxymatrine, sophoridine, sophocarpine, and oxysophocarpine in AH-05. Ten microliters of a sample of AH-05 (1 mg/mL) and standard samples were, respectively, injected into the HPLC system (Waters 2695 and Waters 2996 Diode Array Detector, United States) and separated on a C18 ODS column (Hypersil ODS2 4.6 × 250 mm, 5 µm, ELITE, China) with gradient elution. The flow rate was 1.0 mL/min, and the detection was performed at 220 nm for matrine, oxymatrine, and sophoridine and at 258 nm for sophocarpine and oxysophocarpine.
MTT Assay
HepG2 cell viability was quantitated using 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay (MTT assay, Sigma-Aldrich, United States). HepG2 cells (3 × 103 cells/well) were seeded in 96-well plates and exposed to different concentrations of AH-05 (0.5, 1.0, 1.5, 2.0, 2.5, 3.0, and 4.0 mg/mL). After 24 hours of incubation, 20 µL of 5 mg/mL MTT solution was added to each well and the plate was further incubated at 37 °C for 4 hours. Thereafter, the medium was aspirated, and 200 µL of DMSO was added to each well. The microtiter plate was placed on a shaker for 15 minutes in order to dissolve the dye. After the formazan crystals had dissolved, the absorbance was determined spectrophotometrically at 492 nm on an INFINITE F50 microplate reader (TECAN, Austria). This procedure was replicated thrice.
CCK-8 Assay
H22 cell viability was quantitated using Cell Counting Kit-8 (CCK-8 reagent, Solarbio, China). H22 cells (3 × 103 cells/well) were seeded in 96-well plates and exposed to different concentrations of AH-05 (0.5, 1.0, 1.5, 2.0, 2.5, 3.0, and 4.0 mg/mL). After 24 hours of incubation, 20 µL of 5 mg/mL CCK-8 solution was added to each well and the plate was further incubated at 37 °C for 4 hours. The absorbance of the samples was determined spectrophotometrically at 492 nm on an INFINITE F50 microplate reader (TECAN, Austria). This procedure was replicated thrice.
Colony Formation Assay
HepG2 cells were seeded in 24-well plates at a density of 2 × 104 cells per well and incubated for 24 hours. Then, the cells were exposed to different concentrations of AH-05 (0.5, 1.0, 1.5, 2.0, 2.5, 3.0, and 4.0 mg/mL) for 72 hours continuously. After fixation with 4% paraformaldehyde for 30 minutes, the cells were stained with crystal violet solution for 2 hours. Images were taken after washing the cells with PBS. This procedure was replicated thrice.
H22 Xenograft Model and Treatment
Male SPF-KunMing mice weighing 21.0 ± 1.2 g, purchased from the Institute of Experimental Animals of Sichuan People’s Hospital (Chengdu, China), were used. The animal study was approved by our Animal Care and Use Committee. H22 cells (1.0 × 106 cells/mouse) were transplanted into the right dorsal side of each mouse. The 10 tumor-bearing mice were divided into a control group and an AH-05 group (n = 5 each). Saline and AH-05 were administered to the mice in the control and AH-05 groups, respectively, by oral gavage for 21 days. The tumor volume and weight were recorded at 3-day intervals. Tumor volume was calculated using the following formula: volume (mm3) = width2 × length/2.
Flow Cytometry Assay
In vitro, H22 and HepG2 cells (5 × 105 cells/well) were seeded in 6-well plates. Cells were exposed to AH-05 (0.5 and 1.5 mg/mL) for 24 hours before being collected. Cells were stained with propidium iodide (PI) and an Annexin V kit (Keygen, China) for 20 minutes in the dark. In vivo, a spleen cell suspension was prepared, and the cells were stained with CD4-PE and CD8-FITC for 1 hour in the dark. Samples were tested using a Guava EasyCyte plus flow cytometer (Merck, United States). This procedure was replicated thrice.
Western Blotting Assay
Tumor tissues were lysed in lysis buffer and then centrifuged at 15 000 rpm for 15 minutes at 4 °C. Protein concentrations were determined using a BCA kit (Beyotime, China). Twenty micrograms of each protein sample was subjected to 6% to 10% SDS-PAGE; the resultant bands were transferred onto Poly(vinylidene fluoride) (PVDF) membranes (Merck Millipore, United States). The membranes were blocked for 1 hour at room temperature with 5% bovine serum albumin containing 0.1% Tween-20, and then treated with the following primary antibodies: NF-κB p50 (50 kDa), NF-κB p65 (65 kDa), AKT (60 kDa), p-AKT Ser473 (60 kDa), p-AKT Thr308 (60 kDa), extracellular signal regulated kinase (ERK)1/2 (44/42 kDa), p-ERK1/2 (44/42 kDa), mTOR (289 kDa), p70 S6 kinase (70/85 kDa), and MEK (45 kDa) and p-MEK (45 kDa) (Abcam, United Kingdom; CST, United States; Beyotime, China; diluted by 1:1000), overnight at 4 °C. The membranes were then washed with Tris-Buffered Saline and Tween 20 (TBST) 3 times and incubated with the corresponding secondary antibodies (1:5000) at 37 °C for 2 hours. The membranes were washed again and the proteins were visualized using an enhanced chemiluminescence assay kit (Beyotime, China). Images were captured using the BioRad XRS+ system (BioRad, United States). This procedure was replicated thrice.
Hematoxylin-Eosin Staining Assay
Liver, colon, spleen, and kidney tissues were dissected from the implanted mice, fixed in 10% buffered formalin for 24 hours, and embedded in paraffin. Paraffin-embedded blocks were prepared and 5-μm thick sections were cut and stained with hematoxylin-eosin (H&E) for histological examination. Images were acquired using an Olympus BX50 microscope (Olympus, Japan).
Hematology Toxicity Assay
Blood samples were collected from the implanted mice by eyeball extirpation and clear serum samples were prepared. Liver and kidney function, as well as routine blood tests, was performed using a BC-240VET Chemistry Analyzer and BC-5000 Blood Cell Analyzer (Mindray, China), respectively.
Statistical Analysis
Data are expressed as the means, and t-tests were performed for statistical analysis using Prism GraphPad 8.0. A P value of <.05 was considered to indicate a statistically significant difference.
Results
Matrine and Sophoridine Assay of AH-05
The alkaloid compounds identified in MH are reported in Table 1. The presence of matrine, sophoridine, and sophocarpine in AH-05 was proven according to the results of the HPLC assays. The concentrations of matrine, sophoridine, and sophocarpine in AH-05 were 60.2/100 mg (60.2%), 10.3/100 mg (10.3%), and 11.4/100 mg (11.4%), respectively.
CAS Number, Retention Time (min), Maximum Absorption Wavelengths (λ, nm), and Concentration (mg/100 mg of AH-05) for the Considered Alkaloid Compounds Determined in AH-05.
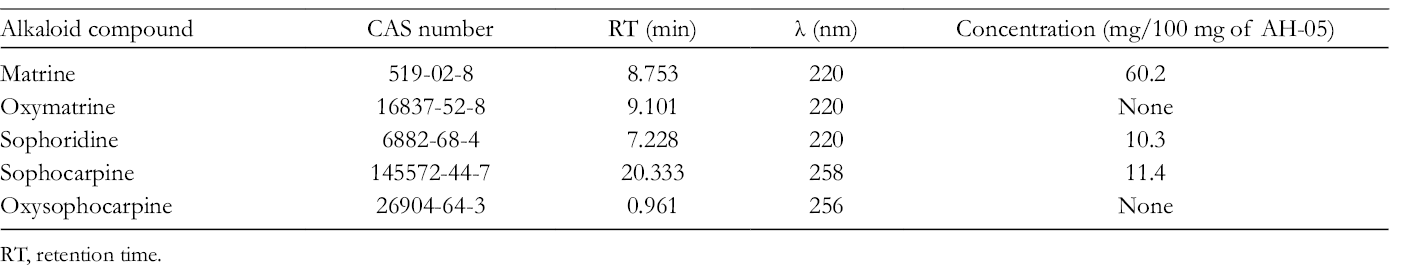
RT, retention time.
AH-05 Inhibits Cell Proliferation
MTT/CCK-8 and colony formation assays were performed for cell proliferation after exposure to AH-05 at different concentrations (0.5, 1.0, 1.5, 2.0, 2.5, 3.0, and 4.0 mg/mL). The results demonstrated that cell proliferation was inhibited in a concentration-dependent manner. As the AH-05 concentration increased, a greater inhibitory effect was exerted against HepG2 and H22 cell proliferation (P ≤ .05*; Figure 1(A)). The effect of AH-05 on cell proliferation within 24 hours was limited, with an inhibition rate of up to 83.4% ± 0.9% for H22 cells and 67.7% ± 2.0% for HepG2 cells. The IC50 of AH-05 against HepG2 and H22 cells was 2.55 and 1.58 mg/mL, respectively. Furthermore, the colony formation assay proved that cell viability was notably suppressed (Figure 1(B)).

Cell proliferation assay. (A) H22 cell viability was quantitated using CCK-8 reagent and HepG2 cell viability was quantitated using MTT assay: AH-05 inhibited the proliferation of (A1) H22 and (A2) HepG2 cells in a concentration-dependent manner. The proliferation inhibition rate was 83.4% ± 0.9% for H22 cells and 67.7% ± 2.0% for HepG2 cells (P ≤ .05*). (B) Colony formation assay. AH-05 notably reduced the numbers of HepG2 cell colonies at different concentrations from 1.0 to 4.0 mg/mL.
AH-05 Induced H22 and HepG2 Cell Apoptosis
HepG2 and H22 cells were exposed to AH-05 at concentrations of 0.5 and 1.5 mg/mL, respectively. Flow cytometry showed that the percentages of Annexin V-positive and Annexin V/PI-positive cells were significantly increased. In the HepG2 cell line group, the 0.5 and 1.5 mg/mL dose of AH-05 induced the Annexin V positive rate up to 41.2% ± 1.3% (P < .001***) and 10.8% ± 1.1% (P = .086), respectively, and the Annexin V/PI both positive rates were 13.9% ± 0.5% (P < .001***) and 63.5% ± 0.07% (P < .001***), respectively. In the H22 cell line group, the 0.5 and 1.5 mg/mL dose of AH-05 induced the Annexin V positive rate to 22.2% ± 2.0% (P < .01**) and 12.3% ± 0.4% (P < .01**), respectively, and the Annexin V/PI both positive rates were 10.5% ± 5.1% (P < .01**) and 30.3% ± 3.11% (P < .01**), respectively (P < .001***; Figure 2(A) and (B)). In addition, the low concentration of the medicine significantly induced early apoptosis (P < .01**), and the high concentration induced late apoptosis (P < .001***). Furthermore, examination of HepG2 and H22 cell morphology showed cell death after exposure to AH-05 for 48 hours (Figure 2(C) and (D)).

Flow cytometry assay showing that AH-05 induced H22 and HepG2 cell death. (A1, A2) Flow cytometry results for HepG2 cells. (B1, B2) Flow cytometry results for H22 cells. (C) HepG2 cell morphology. (D) H22 cell morphology. AH-05 induced an increase in the proportion of Annexin V-positive and Annexin V/PI-positive HepG2 and H22 cells. The 0.5 mg/mL dose of AH-05 mainly induced early apoptosis (P ≤ .001***) and the 1.5 mg/mL dose of AH-05 mainly induced late apoptosis (P ≤ .001***). In addition, HepG2 and H22 cell morphology changes and cell death were observed after exposure to AH-05 for 48 hours at 1.0 and 1.5 mg/mL concentrations.
Effect of AH-05 on Tumor Growth in H22 Tumor-Bearing Mice
The tumor weight and volume of the tumor-bearing mice in the AH-05 group (dose = 0.32 mg/g) were significantly reduced compared with the control group. The inhibition rates of tumor weight and volume were 55.8% (4.75 ± 2.5 vs 10.7 ± 3.1 g; P = .01**) and 52.5% (4699.3 ± 2135.3 vs 9893.5 ± 4457.9 mm3; P ≤ .05*), respectively (Figure 3(A) and (B)). In addition, the body weight of the mice in the AH-05 group was reduced by 21.2% compared with that of the control group (41.6 ± 5.3 vs 52.1 ± 4.7 g, respectively; P ≤ .05*), which perhaps was due to the lighter tumor weights (Figure 3(C)).

(A, B) Tumor volume and weight are shown for control and AH-05 groups. Tumor volume and weight in the AH-05 group were reduced compared with the control group (P = .047* and P = .01**, respectively). (C) The body weight of mice from the AH-05 group was reduced compared with that of those from the control group (P = .026*). (D) Western blot assay showed that the expression of NF-κB p50, NF-κB p65, AKT, p-AKT Ser473, and mTOR in the tumors from mice in the AH-05 group was downregulated compared with that in tumors from mice in the control group, whereas the ERK1/2, p-ERK1/2, and p-MEK expression was increased slightly. Meanwhile, p70 S6 kinase, MEK, and p-AKT Thr308 expression did not change.
AKT/mTOR, MAPK, and NF-κB pathways are involved in the regulatory network of cell carcinogenesis, including the processes of cell growth, metastasis, invasion, and apoptosis. To elucidate the mechanisms underlying the action of AH-05, we evaluated the levels of key proteins in the AKT/mTOR and NF-κB pathways by Western blotting. Based on the results, the expression of NF-κB p50, NF-κB p65, AKT, p-AKT Ser473, and mTOR was suppressed in the tumors of the mice from the AH-05 group, compared with those of mice from the control group. However, the expression level of ERK1/2, p-ERK1/2, and p-MEK protein in the experimental group increased slightly (Figure 3(D)); p70 S6 kinase, MEK, and p-AKT Thr308 expression levels were not affected. AH-05 affected the spleen function, and H&E staining was used to check for splenomegaly, showing white vs red pulp areas of the spleen (Figure 4(A)). The results showed that although the spleen weight reduction was not significant, the percentage of CD4+ T cells in the spleen was raised by 19.9% ± 2.3% compared with 4.5% ± 0.7% in the control group (P ≤ .05*; Figure 4(B) and (C)), and no pathological change was found in the spleen tissue.

(A) Hematoxylin-eosin stain of spleen. The result showed that no pathological change was found in the spleen tissue. (B1-2) The gross specimen and weight of the spleen. Spleen weight reduction did not show any significant difference between the AH-05 and control groups (P = .12). (C1-2) Flow cytometry assay. The percentage of CD4+ T cells in the spleen was increased, showing a significant difference between the AH-05 and control groups (P = .012*). However, the percentage of CD8+ T cells in the spleen was increased, but did not show a significant difference between the AH-05 and control groups (P = .108).
Toxicity Assessment of AH-05 Treatment in Tumor-Bearing Mice
The hematology and pathology analyses were performed according to the Common Terminology Criteria for Adverse Events (CTCAE) 3.0. In the hematology assay, the serum levels of alanine aminotransferase and aspartate aminotransferase were measured as indexes of hepatic function, the levels of blood urea nitrogen and creatinine served as indexes of renal function, and the levels of white blood cells, red blood cells, and blood platelets (PLT), as well as the lymphocyte ratio (LYM%), served as indexes of the routine blood test. Based on the results, no hematological toxicity over Grade II was observed compared with the control group (P ≥ .05; Figure 5(A) and (B)). No kidney or liver pathology was observed, except for slight intestinal mucosa edema (Figure 5(C)). Thus, the data demonstrated the safety of AH-05 treatment for tumor-bearing mice.
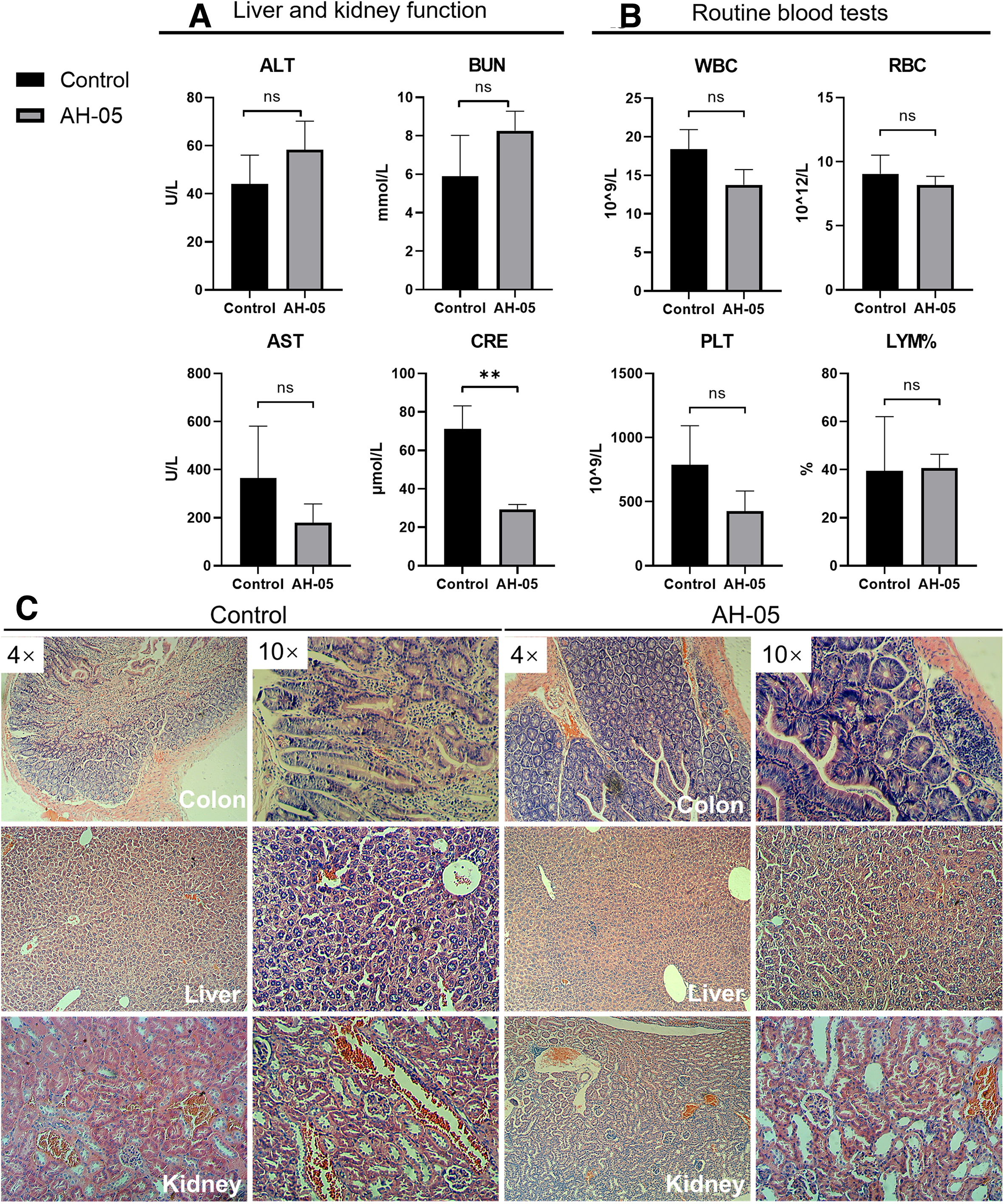
Hematological toxicity and pathology. (A) Liver and kidney function. AST: 178.7 ± 63.8 U/L (0-80 U/L; P ≥ .05); ALT: 58.3 ± 9.7 U/L (0-77 U/L; P ≥ .05); BUN: 8.26 ± 0.83 mmol/L (2.8-11.9 mmol/L; P ≥ .05); creatinine: 29.3 ± 2.0 μmol/L (10-80 μmol/L; P ≤ .05*) in the AH-05 group; AST: 364.3 ± 176.8 U/L; ALT: 44.0 ± 9.9 U/L; BUN: 5.91 ± 1.73 mmol/L; creatinine: 71 ± 9.9 μmol/L in the control group. (B) Routine blood tests. WBC: 13.8 ± 1.6 × 109/L (0.8-6.8 × 109/L; P ≥ .05); RBC: 8.28 ± 0.56 × 1012/L (6.36-9.42 × 1012/L; P ≥ .05); PLT: 426.7 ± 127.9 × 109/L (450-1590 × 109/L; P ≥ .05); LYM%: 40.7% ± 4.6% (55.8%-90.6%; P ≥ .05) in the AH-05 group; WBC: 18.4 ± 2.1 × 109/L; RBC: 9.06 ± 1.19 × 1012/L; PLT: 788.7 ± 247.2 × 109/L; LYM%: 39.4% ± 18.5% in the control group. Note that these P values represent the statistical difference between the AH-05 and control groups. (C) Hematoxylin-eosin staining of colon, liver, and kidney tissues. Slight intestinal mucosal edema was found. No pathological change was found in the kidney or liver. ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; LYM%, lymphocyte ratio; PLT, blood platelets; RBC, red blood cell; WBC, white blood cell.
Discussion
Liver cancer is a common malignant cancer and is the third leading cause of cancer-related deaths worldwide. 2 According to clinical statistics, about 13 million patients suffer from liver cancer in China each year, accounting for about 45% of the global liver cancer mortality. 20,21 Recently, the recurrence and metastasis of liver cancer have been identified as the main therapeutic challenges associated with this disease. However, complicated factors lead to the invasion and metastasis of liver cancer, including tumor cell migration, tumor angiogenesis, tumor cell proliferation, and metastasis. 21,22 With a long history of clinical use, although not without criticism, 23 HMs are now being recognized as agents with an important role in cancer prevention, and are a notable choice due to their multilevel, multitarget, and coordinated intervention effects against liver cancer. 24,25 Indeed, HMs have been shown to act on angiogenesis and vascular normalization in tumor-derived endothelial cells, cancer cell differentiation and apoptosis, cytotoxic function, and immunity regulation 26 -28 by the activation of NF-κB pathway 29 and ERK pathway and the expression of epidermal growth factor and vascular endothelial growth factor. 21,30 These functions can prevent disease progression and improve overall survival with low toxicity. 31 Signaling pathways that control multiple processes are commonly dysregulated in the pathological progression of liver cancer. 32,33 However, it is the complexity of HM constituents that enables them to treat disease via the synergistic effects of multiple components, unlike the case for when a single chemical drug is used. 34 That being said, the inability to assure the quality and consistency of herbal extracts has been a major obstacle in their development. 35
Alkaloids, including piperidine, isoquinolines, indoles, terpenoids, and steroidal and quinoline alkaloids, have been reported as potential ingredients for liver cancer treatment. 36 Some previous studies indicated that matrine and sophoridine have effective biological activities, including apoptosis, antiproliferation, and maintenance of mitochondrial function at nontoxic concentrations. Matrine is extracted from Radix Sophorae flavescentis, 37 and sophoridine is a quinolizidine alkaloid found in the stems and leaves of Sophora alopecuroides L. and Euchresta japonica Benth and the roots of S. alopecuroides Ait. 38,39
Here, we demonstrated that the novel herbal compound medicine, AH-05, contains both matrine and sophoridine. However, the HPLC assay showed that besides these alkaloids, AH-05 contains other active elements, highlighting the need for further studies to confirm its effects. Our results also indicate that AH-05 is a potential antitumor agent for liver-related malignancies. AH-05 was extracted from Radix S. flavescentis (Chinese name: KU-SHENG), A. capillaris (Chinese name: NONG-DAN-CAO), and Astragalus (Chinese name: Huang Qi) using CH2Cl2. After identifying matrine and sophoridine by TLC and HPLC, the curative effects of AH-05 on liver cancer in vitro and in vivo were investigated. In vitro, AH-05 significantly inhibited liver cancer cell viability and colony formation ability in a dose-dependent manner. In vivo, AH-05 suppressed tumor growth and promoted CD4+ T cell polarization in the spleen. Importantly, the expression levels of NF-κB p50, NF-κB p65, AKT, p-AKT Ser473, and mTOR were significantly inhibited, whereas ERK1/2, p-ERK1/2, and p-MEK expression increased slightly. In addition, the data demonstrated the safety of AH-05 treatment in tumor-bearing mice, as this treatment did not exacerbate adverse hematological and pathological effects.
Conclusion
This study indicated that AH-05 exhibits its prominent therapeutic effects on liver cancer mainly via the regulation of cell proliferation, apoptosis, and the tumor microenvironment through the AKT/mTOR and NF-κB signaling pathways, as well as the immune environment by promoting CD4+ T cell polarization in the spleen. Moreover, AH-05 has been shown to be safe. Thus, AH-05 represents a useful supplementary medicine for liver cancer, and the potential therapeutic effects of AH-05 may be further ascertained in further studies involving clinical trials on liver cancer patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
