Abstract
Background
Cancer is one of the most serious diseases worldwide that threatens human health and leads to death. Chemotherapy is the main clinical method to treat tumors, but, despite the development of new chemotherapeutic drugs, the multidrug resistance (MDR) of cancer cells to conventional chemotherapeutic drugs remains a major cause of failure in cancer prevention and treatment. Therefore, overcoming this resistance has become a major challenge in cancer prevention and treatment.
Method
With the in-depth study of traditional Chinese medicines (TCMs) for the treatment of tumors, many such medicines have been found that can reverse MDR and enhance the sensitivity of chemotherapy. ZJW is a famous traditional medicine formula from China, recorded first in an ancient medicine book named Danxi Xinfa. It is composed of Huanglian and Wuzhuyu in a ratio of 6:1 by mass.
Conclusion
ZJW can inhibit proliferation, induce apoptosis, inhibit invasion and metastasis, and reverse MDR of tumor cells through multiple pathways and multiple targets. In this paper, we briefly review recent research on ZJW and its main components, evodiamine and berberine, in the anti-tumor mechanism and reversal of multidrug resistance.
According to the International Agency for Research on Cancer (IARC), 17 million cases of malignant tumors and 9.5 million-related cases were reported worldwide in 2018, 1 suggesting that cancer has critically endangered people's lives and health. At present, the prevention and treatment methods for cancer in clinical practice typically include surgery, radiotherapy, chemotherapy, and biological therapy. Among these, chemotherapy significantly improves the survival of these patients in clinical settings.2,3 However, multidrug resistance (MDR) of tumor cells to chemotherapeutic agents has emerged as the main cause of tumor treatment failure.4–6 Currently, most Western drugs reverse drug resistance of tumor cells through a single underlying mechanism, and some co-existing mechanisms result in a poor effect of single-target drugs, leading to more adverse reactions. Therefore, there are some limitations in their clinical application. Chinese traditional herbal medicines have the advantages of high potency, low toxicity, multiple binding sites, high specificity, and reversal of MDR of tumor cells through multiple mechanisms simultaneously, attracting strong attention from scientists.7–9 Numerous studies have shown that many herbal medicines can reverse tumor MDR.10,11
ZJW is a traditional medicine formula from China, recorded first in an ancient medicine book, Danxi Xinfa. It is composed of Huang Lian and Wuzhuyu in a ratio of 6:1 by mass and is mainly used in treating “liver fire in the stomach.” 12 Modern studies have confirmed that ZJW has a wide range of pharmacological effects, including the treatment of gastric ulcers, hyperlipidemia, and diabetes, as well as antibacterial and anti-inflammatory effects. ZJW can also be used in the treatment of tumors because it can inhibit proliferation, invasion, and metastasis of cancer cells and induce apoptosis, as well as reverse MDR.13,14
Wuzhuyu is the dried, near-ripe fruit of Cornus officinalis, ie, Shihu or Cornus officinalis, belonging to the family Rutaceae. It has the properties of dispersing cold and relieving pain, lowering rebelliousness, and stopping vomiting. It is commonly used in the treatment of syncopal headaches, abdominal pain from cold diseases, vomiting, and acid swallowing, 15 and is classified as a member of the liver, spleen, and stomach meridians. 16 The main active ingredient in Wuzhuyu is evodiamine (EVO). A pharmacological study showed significant inhibitory effects of EVO on the proliferation of several cancer cells, including those of the lung, colon, breast, ovaries, gastric cancer (GC), and leukemia.17,18 Several reviews on the mechanism of the antitumor effects of EVO have been published.19–21 EVO mainly acts on the PI3K/Akt/Caspase, MAPK, and Fas-L/NF-κB signaling pathways to arrest the tumor cell cycle in the G2/M phase and induce apoptosis, 22 along with decreasing the level of polymeric microtubule proteins in cells and hindering the apoptosis of tumor cells. 23 The drug prevents the mitosis of tumor cells by reducing the level of polymeric microtubule proteins in cells and impeding the formation of spindles. EVO can induce apoptosis by upregulating the phosphorylation of Bcl-2 and enhancing the expression of Bax and aMase proteins. 24 Recent studies have shown that EVO in combination with other chemotherapeutic agents (eg, cisplatin, oxaliplatin, erlotinib, adriamycin, paclitaxel, gemcitabine, etc) has the potential to increase chemotherapeutic sensitivity and reverse drug resistance.
Berberine (BBR), an isoquinoline alkaloid isolated from the Chinese herb, Huanglian, and other Berberis plants, has a wide range of pharmacological properties, including anti-inflammatory, 25 anti-infection, anti-tumor, 26 and anti-arrhythmic functions, along with the regulation of blood lipid and glucose levels.27,28 Numerous experiments have shown that BBR has significant antitumor effects in liver cancer, breast cancer, gastric cancer, non-small cell lung cancer, ovarian cancer, melanoma, and other tumors.29,30 BBR can promote drug-resistant tumor cell death by promoting cycle arrest of resistant tumor cells, inhibiting migration and invasion, suppressing tumor cell proliferation, promoting sensitivity to chemotherapy or radiotherapy, and promoting autophagy and apoptosis31–34 in an attempt to solve the problem of drug resistance in tumor cells after long-term chemotherapy or radiotherapy (Figure 1).

Chemical structural formula (A) Evodiamine (B) Berberine.
Gastric Cancer (GC)
Gastrocolic cancer is a common tumor characterized by high morbidity and mortality. 35 To date, surgery remains the only treatment option. However, due to the manifestations in advanced stages, GC is detected in most patients with advanced disease when the tumor cells usually migrate, leading to an extremely low 5-year survival rate. 36 Chemotherapy is the treatment of choice for advanced GC.
Cisplatin (DDP) is a commonly used agent for the treatment of GC. 37 The use of DDP-based adjuvant chemotherapy was approved to increase survival after gastrectomy. Unfortunately, intrinsic or acquired drug resistance severely limits the therapeutic efficacy of cisplatin. 38 Therefore, developing effective strategies to improve the sensitivity of cisplatin in GC treatment is necessitated. Q.F. Tang et al. 39 found overexpression of phosphorylated cofilin-1 (p-cofilin-1) in the DDP-resistant human GC cell line, SGC7901/DDP relative to SGC-7901 cells and DDP could induce p-cofilin-1 dephosphorylation in the parental cell line, but not in DDP-resistant cell lines. However, the ZJW-induced dephosphorylation of cofilin-1 in SGC7901/DDP cells promoted the translocation of cofilin-1 from the cytoplasm to the mitochondria, which in turn induced the mitochondrial apoptotic pathway. Moreover, the ZJW extract could increase the sensitivity of SGC-7901/DDP cells to DDP by its concentration-dependent increase in the cytoplasm. It also enhanced the pro-apoptotic effect of cisplatin by promoting JNK activation and Bcl-2 expression, inhibiting Bax expression and subsequently increasing the release of cytochrome C from mitochondria to the cytoplasm, thus ultimately activating the cysteine cascade response. 40 Treatment with PP1 and PP2A inhibitors or cofilin-1-specific siRNA transfection reversed the resistance developed by cells toward DDP. Sun et al. 41 found that when DDP was combined with ZJW, the PI3K/Akt pathway mediated by ROCK1/PTEN significantly enhanced PP1 and PP2A expression and promoted p-cofilin-1 dephosphorylation and cofilin-1-mediated mitochondrial potential, in turn enhancing the chemosensitivity of DDP-resistant cells. Sun et al. 42 investigated the inhibitory effect of ZJW on the proliferation and glycolysis of GC-resistant cells, SGC-7901/DDP. The cell survival rate in each concentration group of the ZJW exposure was reduced significantly at all time points of the assay in a time- and concentration-dependent manner compared to the control group. After 24 h of drug intervention, the glucose uptake rate and lactate content of cells in each ZJW concentration group reduced significantly compared to the control group, suggesting the ZJW could significantly inhibit SGC-7901/DDP glycolysis. It was concluded that ZJW could significantly inhibit the proliferation of the GC-resistant cells, SGC-7901/DDP, and this process may be related to the inhibition of glycolysis, with c-myc, hypoxia-inducible factor-1α (HIF-1α), GLUT1, LDHA, and HK II as its targets of action. Subsequently, the effects of ZJW on SGC-7901/DDP resistance and related protein expressions were specifically investigated. Either EVO or BBR alone and their combination significantly enhanced the sensitivity of SCC-7901/DDP cells to DDP in vitro, and these effects were associated with elevated expressions of apoptotic proteins, caspase-3, and caspase-9, along with the downregulation of resistance-related proteins, P-gp and MRP. 31
Recent epidemiological findings suggest a lower risk of GC in diabetic patients with long-term metformin use. 43 Typically, GC development is related to energy metabolism, and blocking these metabolic alterations is an emerging therapeutic approach for cancer. Some metabolic enzymes involved in glycolytic pathways are considered new therapeutic targets. 44 BBR regulates lipid metabolism and glucose homeostasis by modulating the expression of the hepatocyte nuclear factor-4α (HNF4α) in metabolic diseases like hepatic metabolism disorders. 45 Hu et al. 46 found that BBR not only downregulated HNF4α and upregulated p-AMPK levels in the treatment of GC but also induced cell cycle G0/1 phase arrest by downregulating cyclin D1 expression, thereby inhibiting the proliferation ability of tumor cells. BBR attenuated tumor cell migration and invasion by downregulating matrix metalloproteinase-3 (MMP-3). Li et al. 47 found that BBR induced fatty acid accumulation and inhibited the expression of fatty acid-binding proteins (FABPs) in the human GC cell line, MGC803, in turn inducing apoptosis in GC cells. In addition to the induction of apoptosis, stimulating cancer cell autophagy is also effective in the treatment of cancer. Cellular autophagy is a major self-degradation process characterized by the formation of double-membrane autophagosomes. 48 Zhang et al. 49 found that BBR induced autophagy in GC BGC-823 cells by inhibiting the MAPK/mTOR/p70S6K and Akt pathways, thereby inhibiting the growth of human GC cells in vivo and in vitro. Recent studies suggest that BBR may also affect cancer-related pathways by regulating the expression of cyclic RNAs and their corresponding target genes in GC cells, 50 thereby inhibiting cancer cell growth.
GC stem cells (GCSCs) promote tumorigenesis, aggressive growth, recurrence, metastasis, and drug resistance, and thus new therapeutic strategies targeting GCSCs are urgently needed in clinical settings. Cai et al. 51 demonstrated that the levels of β-catenin, c-myc, cyclin D1, and axin 2 expression in GCSCs were essential for the self-renewal of cancer stem-like cells in human GC by measuring the expression of proteins in the Wnt/β-catenin pathway, which is essential for the self-renewal of cancer stem-like cells in human GC. Wen et al. 52 examined the effect of EVO on the self-renewal of GCSCs by altering the sphere-forming ability of GC cells, inducing pluripotent stem cell factor expression, epithelial-mesenchymal transition (EMT) factor expression, and oxaliplatin resistance. EVO inhibited the proliferation of GCSCs, promoted Bax:B-cell lymphoma 2, and altered the expression of caspase-3 activity in GCSCs. EVO inhibited the sphere-forming ability of GC cells and reversed the resistance of GCCS to oxaliplatin by decreasing the expressions of Sox2, KLF4, Bmi-1, Oct4, and other proteins. EVO decreased the expression of Slug, Twist, Zeb1, and vimentin, suggesting an inhibitory effect on EMT. The expression of β-catenin, c-Myc, and cyclin D1 reduced significantly in EVO-treated GC cell spheres. EVO inhibited EMT by inhibiting the Wnt/ β-catenin signaling pathway, which in turn suppressed the proliferation and stem cell properties of GCSCs (Table 1).
Effects of ZJW, EVO, and BBR on gastric cancer.

Non-Small Cell Lung Cancer (NSCLC)
Lung cancer is a common malignancy with a high incidence worldwide and is the leading cause of cancer death in the United States. In recent years, the incidence of lung cancer in China has been on an annual rise. According to its pathological type, lung cancer can be divided into small cell lung cancer (SCLC) and NSCLC, with the latter accounting for more than 80% of all lung cancer patients. 53
Zheng et al. 54 found that BBR inhibited the growth and induced apoptosis of NSCLC cells through a p38α MAPK-mediated increase in protein levels of p53 and the transcription factor, FOXO3a. Phosphatidylinositol-dependent kinase 1 (PDPK1) is a proximal signaling molecule of phosphatidylinositol 3 kinase (PI3-K) among the AGC kinases, and the activation of PDPK1 promotes cancer cell metabolism and enhances their metastatic potential. 55 DNA methyltransferase 1 (DNMT1) can attenuate tumor suppressor gene activity by initiating DNA hypermethylation. 56 Zheng et al. 57 further showed that BBR reduced tumor cell migration and invasion by inhibiting the protein expression of transcription factors, SP1 and PDPK1. BBR also blocked the cell cycle by inhibiting DNMT1 expression; the interaction between PDPK1 and SP1 could enhance the DNMT1 inhibitory activity of BBR. Systemic diseases often share common features, including high expression of sterol regulatory element-binding protein 1 (SREBP1) and strong lipogenic activity in tumor cells from patients with gefitinib-resistant NSCLC (G-R NSCLC) and synovial fibroblasts (RASFs) from patients with rheumatoid arthritis. Fan et al. 58 found that BBR regulated SREBP1 through the ROS/AMPK pathway, in turn regulating SREBP1 and selectively inhibiting the growth of G-R NSCLC cells and RASFs, further suppressing NSCLC proliferation. In terms of cellular pathways, Li et al. 59 found that BBR could inhibit NSCLC cell proliferation and promote tumor cell apoptosis by downregulating MMP-2 expression, thus reducing tumor cell invasion, decreasing the Blc-2/Bax ratio and inhibiting the Jak2/VEGF/NF-κB/AP-1 signaling pathway. Chen et al. 60 found that BBR could also induce apoptosis in NSCLC cells by upregulating miR-19a expression and downregulating transcription factors, in turn activating the MAPK signaling pathway. BBR could inhibit the growth of tumor cells in multiple ways during the anti-NSCLC cellular process.
NOTCH3 signaling is mainly activated in NSCLC and is considered a therapeutic target. Inhibition of NOTCH3 can suppress cell proliferation and induce apoptosis. 24 Su et al. evaluated the role of NOTCH3 signaling on tumor cells using EVO against a mouse lung cancer model and in two NSCLC cell lines, A549 and H1299. 61 EVO effectively reduced the size and number of tumors in mice and inhibited NOTCH3 expression in tumors. These findings provided new insights into the anti-NSCLC effects of EVO and confirmed its potential role in lung cancer prevention and treatment. Li et al. 62 assessed the reversal of MDR in NSCLC cells in vitro using EVO and its possible mechanism of action. EVO could effectively reverse cisplatin resistance in lung cancer-resistant strain A549/DDP cells and induce cellular apoptosis. Simultaneously, when EVO was combined with cisplatin, EVO improved the chemosensitivity toward cisplatin, inhibited the mRNA and protein expressions of NF-κB subtype p65 (NF-κB p65), and down-regulated the expression of MDR1 in A549/DDP cells, providing a theoretical basis for the reversal of drug resistance. Erlotinib, an oral epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI), is effective in NSCLC patients carrying known drug-sensitizing EGFR mutations.63,64 but those with wild-type EGFR show resistance. 65 Several studies have confirmed the synergistic effects of erlotinib in combination with standard chemotherapy in patients with NSCLC carrying wild-type EGFR.66–68 Li et al. 69 found that EVO induced anti-proliferation and apoptosis in four wild-type EGFR NSCLC cell lines (A549, NCI-H460, NCI-H1299, and NCI-H1650). The combination of EVO with erlotinib successfully inhibited the proliferation and survival of wild-type EGFR NSCLC cells. Mechanistic studies showed that the expression of PARP and caspase-3 increased significantly in wild-type EGFR-NSCLC cells after the combination treatment of EVO with erlotinib, and the PI3K-AKT-mTOR signaling pathway was blocked, as evidenced by the simultaneous decrease in the levels of p-4E-BP-1 (Ser-65), p-p70S6K1 (Ser-371) and p70S6K1. Thus, the combination treatment of EVO and erlotinib synergistically exerted anti-cancer effects by blocking the PI3K-AKT-mTOR pathway (Table 2).
Effects of EVO and BBR on non-small cell lung cancer.

Colon Cancer
A high-fat diet is one of the main risk factors for disrupting the balance of intestinal flora, eventually inducing colorectal cancer (CRC). In the case of CRC patients, the main cause of death is the invasion and metastasis of tumor cells. 70
In a recent study, Wen et al. 71 in an ex vivo experiment found that ZJW effectively inhibited the proliferation and telomerase activity of HT29 cells, thus hindering the development and progression of early cancer. Xu et al. 72 found that ZJW significantly inhibited the proliferation of HCT-116 cells in a dose-dependent manner. Compared to the blank group, high doses of ZJW resulted in the upregulation of Bax and Bak proteins and downregulation of Bcl-2 and Bcl-xL proteins, along with enhanced caspase-3 and caspase-9 protease activities, indicating that ZJW inhibited tumor cell proliferation and apoptosis through the activation of the mitochondrial pathway. In HCT116/L-OHP cells, Sui et al. 10 detected significant enhancement of phosphorylated AKT and NF-κB activity with upregulation of P-gp. The levels of phosphorylated AKT and NF-κB reduced significantly in HCT116/L-OHP after ZJW intervention. However, the inhibition of P-gp expression, AKT phosphorylation, and the NF-κB pathway by ZJW was attenuated after treatment with PI3K/Akt activator in these cells. Thus, ZJW exerted an inhibitory effect on the expression of P-gp by inhibiting the PI3K/Akt/NF-κB pathway and down-regulating the protein expression of ABCB1/P-gp. ZJW could reduce tumor volume and induce apoptosis in combination with chemotherapeutic drugs. Studies on MDR cell lines were conducted and the effects of ZJW on the protein expressions of ABCB1/P-gp, ABCC-1/2, ABCG2/BCRP, and LRP were examined. ABCB1/P-gp, ABCC-2, and LRP were substantially upregulated in HCT116/L-OHP cells compared to HCT116 cells. Further analysis showed that the reversal of MDR by ZJW was accompanied by the down-regulation of P-gp and was dependent on ABCB1/P-gp inhibition.
Ruan et al. 73 showed that BBR could bind to 9-cis-retinoic acid (9-cis-RA) and synergistically activate retinoid X receptor α (RXR-α) to inhibit c-Cbl-mediated degradation of the β-catenin signaling pathway, thereby suppressing colon cancer proliferation. Excessive glucose uptake and glycolytic metabolism are a hallmark of cancer. Mao et al. 74 found that BBR inhibited excessive glucose metabolism in colon cancer cells by suppressing mTOR-dependent HIF-1α protein synthesis, thereby attenuating cancer cell proliferation. Based on this, the relationship between colon cancer cell proliferation and adipogenesis was investigated. Liu et al. 75 found that BBR inhibited cell proliferation by inducing G0/G1 phase cell cycle arrest and the Wnt/β-catenin pathway in colon cancer cells, while also suppressing sterol regulatory element-binding protein-1 (SREBP-1) activation and SREBP cleavage. BBR can inhibit tumor growth in colon cancer in part by targeting the SCAP/SREBP-1 pathway, leading to the downregulation of lipogenic enzymes and resulting in the inhibition of lipid synthesis along with the suppression of colon cancer xenograft growth and adipogenesis. Stress markers, including glucose-regulated protein 78 (GRP78) are induced by ER stress. 76 Gong et al. 77 found that BBR could inhibit proliferation and migration and induce apoptosis in SW480 colon cancer cells by downregulating GRP78 expression, hinting at GRP78 as a potential target protein for cancer therapy.
EVO can inhibit apoptosis, migration, and invasion of cancer cells and shows a good anti-colorectal cancer effect. Zhou et al. 78 investigated the effect of EVO on CRC cell metastasis in vitro and in vivo and found that it significantly inhibited migration and invasion of HT-29 and HCT-116 CRC cells and reduced tumor cell metastasis. The mechanism may be related to the reduction of acyl-NF-κB p65 levels. EVO inhibited tumor microangiogenesis by downregulating vascular endothelial growth factor (VEGF) expression, 79 making the treatment of CRC with EVO a highly effective multifunctional therapeutic strategy. Sui et al. 80 examined the effect of EVO on drug resistance in CRC along with the underlying molecular mechanisms and therapeutic targets. EVO significantly counteracted resistance and promoted chemotherapeutic drug-induced apoptosis against oxaliplatin-resistant HCT-116/L-OHP cells in a time- and dose-dependent manner. EVO inhibited the growth of CRC xenografted tumors and downregulated p-NF-κB levels in vivo. This study provided the first direct evidence for attenuating MDR by EVO in CRC by blocking the p-NF-κB signaling pathway (Table 3).
Effects of ZJW, EVO, and BBR on colon cancer.

Breast Cancer
Breast cancer is the leading cancer among women and the second most common cancer worldwide. 81 Chemotherapy is the main treatment for breast cancer, and the commonly used chemotherapeutic agents include camptothecin (PTX), cisplatin, lapatinib, doxorubicin (DOX), and tamoxifen. However, their efficacy is reduced due to side effects and drug resistance.
Jia et al. 82 found that ZJW-containing serum inhibited the proliferation of MCF-7 human breast cancer cells. The drug-containing serum significantly down-regulated Cyclin D1 and Cymiln E expressions, along with up-regulation of p21, p27, p53, and Bax expressions. Cyclin D1, Cymiln E, p21, and p27 are the key proteins regulating the G1/S cell cycle barrier, while p53 and Bax factors play important roles in apoptosis induction. The inhibitory effects of ZJW on MCF-7 human breast cancer cells are related to the blocking of G1/S cell cycle progression and induction of apoptosis.
BBR directly binds to DNA and increases lung and esophageal cancer cells’ radiosensitivity and the sensitivity of ovarian cancer cells to cisplatin and PARP inhibitors 83,84; however, its effect on cellular DNA repair and the chemotherapeutic drug treatment remains unclear. Gao et al. 85 found that BBR arrested the cell cycle of breast cancer cells in the S phase and induced DNA breaks. It affected DNA repair in tumor cells by interfering with XRCC1-mediated base excision repair, thereby sensitizing cancer cells to the chemotherapeutic drugs, cisplatin, and cyclosporin. The chemotherapeutic drug, tamoxifen, is widely used for preventing and treating breast cancer; however, the emergence of resistant cells has led to a decrease in its therapeutic effects. 86 Wen et al. 87 found that when BBR and tamoxifen were combined, BBR induced G1 phase arrest and increased the sensitivity of drug-resistant MCF-7 cells to tamoxifen by upregulating p21 expression, along with enhanced tumor cell apoptosis by downregulation of the Bcl-2/Bax ratio, thereby promoting the anticancer effects of tamoxifen. The chemotherapeutic drug, lapatinib, a novel tyrosine kinase inhibitor of HER2/EGFR, is used to treat HER2-positive breast cancer. 88 However, acquired drug resistance limits the efficacy of lapatinib. BBR induces oxidative stress and mitochondria-related apoptotic pathways in human breast cancer cells. 89 Zhang et al. 90 evaluated the combination of BBR and lapatinib. JabbarZadeh et al. 91 found that BBR greatly reduced the phosphorylation of EGFR and AKT and inhibited the activity of P38 MARK and ERK1/2 compared to lapatinib alone, resulting in the attenuation of tumor cell proliferation while preventing acquired drug resistance. BBR may serve as a possible alternative to lapatinib. A hypoxic microenvironment is one of the causes of MDR in tumor cells, and the upregulation of HIF-1α-activated P-gp protein expression is responsible for the development of hypoxic resistance. 92 Hypoxia promotes HIF-1α expression to inhibit p53 activity, thus evading the apoptotic mechanism. 93 Pan et al.94,95 found that during AMPK activation, hypoxia increased breast cancer cell DOX resistance. On the one hand, low doses of BBR could increase the sensitivity of drug-resistant breast cancer cells by inhibiting the AMPK-HIF-1α-P-gp pathway. On the other hand, high doses of BBR induced apoptosis directly by downregulating AMPK- and HIF-1α- induced p53 activation, thus making BBR a sensitizer and a chemotherapeutic agent with an important role in reversing MD. Qian et al. 96 found that BBR could directly induce apoptosis by downregulating P-gp and MDR protein (MRP1). BBR also inhibited K+ efflux by suppressing the efflux of the ATP-binding cassette transporter to attenuate cellular proliferation. Mitochondria play a role in most cellular metabolic processes, 97 and drug delivery from mitochondria is a promising and effective strategy for cancer therapy. The efficacy of PTX, a broad-spectrum anticancer drug, decreases due to MD. Cheng and Ji 98 linked BBR and PTX via disulfide bonds (PTX-ss-BBR) and the combined drug, in A549 cells, showed good efficacy, dissipation of mitochondrial membrane potential, increased ROS levels in cancer cells, and led to cell cycle arrest in the G2/M phase, eventually causing apoptosis of cancer cells and inhibition of tumor growth. The combination of BBR and PTX enhanced the therapeutic effect of conventional chemotherapeutic drugs in A549.
Liao et al. 99 found that EVO inhibited Adriamycin-resistant human breast cancer NCI/ADR-RES cells and assessed the underlying mechanisms. EVO inhibited the proliferation of NCI/ADR-RES cells in a concentration-dependent manner, blocked the G2/M phase transition, and increased microtubule protein polymerization while promoting the phosphorylation of Raf-1 kinase and Bcl-2 proteins. EVO's anti-proliferative and apoptotic activities against several drug-resistant breast cancer cells have been demonstrated in vitro and in vivo. Wang et al. 100 found that EVO induced apoptosis in a caspase-dependent manner in Adriamycin-sensitive MCF-7 and Adriamycin-resistant MCF-7/ADR cells. The reversal of apoptosis resistance by Augustine might be related to its ability to inhibit the expression of the Ras/MEK/ERK pathway and inhibitors of apoptosis (IAPs). EVO potentiated the apoptotic effect of Adriamycin (DOX) by inhibiting the protein expression of the Ras/MEK/ERK cascade and IAPs, but not the levels and activity of P-gp. This study provided a new approach for the use of the natural product, EVO alone or in combination with DOX for the treatment of drug-resistant breast cancer (Table 4).
Effects of ZJW, EVO, and BBR on breast cancer.

Ovarian Cancer
Zhao et al. 101 found that treatment with the chemotherapeutic drug, etoposide (VP16), triggered the AA pathway, leading to elevation in PGE2 levels, ultimately enhancing the regeneration of ovarian cancer cells. BBR inhibited the reaggregation of tumor cells caused by the caspase3-iPLA2-AA-COX-2-PGE2 pathway by suppressing the expressions of iPLA2 and COX-2. BBR also reversed the elevated levels of FAK phosphorylation caused by abnormal PGE2 levels, thus reversing the regeneration of ovarian cancer cells after VP16 treatment. Recently, amplification and overexpression of the epidermal growth factor receptor (EGFR) and ErbB2 were confirmed in different cancers 102 ; these have been implicated in tumor cell proliferation and metastasis. Chuang et al. 103 found that BBR inhibited the activation of cyclin D1, MMPs, and VEGF, downstream of EGFR and ErbB2 by suppressing the EGFR-ErbB2/PI3K/Akt signaling pathway, thus reducing the production of EGFR and ErbB2, while BBR degraded ErbB2 through a ubiquitin-mediated proteasomal pathway, in turn inhibiting the proliferation and migration of tumor cells. MicroRNAs (miRNAs) have an important role in cancer cell proliferation, and miR-145 is the target gene of a large number of oncogenic factors. miR-145 levels are usually downregulated in cancer cells,104,105 and MMPs play an important role in tumor cell invasion and metastasis. 106 Li et al. 107 found that BBR could inhibit the proliferation, migration, and invasion of SKOV3 tumor cells. It promoted the expression of miR-145 and decreased that of MMP16. Zhi et al. 108 found that BBR inhibited the ability of ovarian cancer SKOV3 cells to proliferate, migrate, and invade by inhibiting the hERG1 channels, and thus, BBR could serve as a potential therapeutic agent against platinum-resistant ovarian cancer cells, in which miR-93 is overexpressed. Chen et al. 109 found that BBR could increase the sensitivity of ovarian cancer cells to cisplatin by downregulating miR-93 levels and increasing those of PTEN. The regulation of the PTEN/AKT pathway by BBR has an important role in inhibiting drug-resistant cells. Liu et al. 110 used the combination of BBR and DDP on ovarian cancer cells which induced the G0/G1 cell cycle block along with the downregulation of Cyclin D1. BBR and DDP inhibited PCNA and Ki67 expression while enhancing those of caspase-3 and caspase-8, thus promoting apoptosis. Enhanced expressions of RIPK3 and MLKL promoted cell necrosis.
Chemotherapeutic resistance is a major factor contributing to the poor prognosis of patients with ovarian cancer. Zhong et al. 111 investigated the effects and mechanisms of action of the Chinese herbal extract, EVO, on chemotherapy-sensitive and resistant human epithelial ovarian cancers. EVO was found to inhibit the proliferation of human ovarian epithelial carcinoma A2780 and related paclitaxel-resistant cell lines along with the induction of cell cycle arrest in a dose- and time-dependent manner. Further studies on the mechanism underlying the block revealed that this may be mediated by the combined action of Cyclin B1 and cell division cycle protein 2 (Cdc2), including the upregulation of Cyclin B1, p27, and p21, and the failure of Cdc2 and pRb activation, together with the regulation of the MAPK signaling pathway. EVO also enhanced the chemosensitivity of paclitaxel-resistant A2780/PTX cells through the downregulation of MDR-1 expression, accompanied by inhibition of MDR-1 function. This study preliminarily demonstrated that EVO exerted anti-proliferative effects on human ovarian epithelial cancer cells, A2780/WT and A2780/PTX, induced G2/M phase cell cycle arrest, and had treatment potential against ovarian cancer chemoresistance (Table 5).
Effects of EVO and BBR on Ovarian Cancer.

Liver Cancer
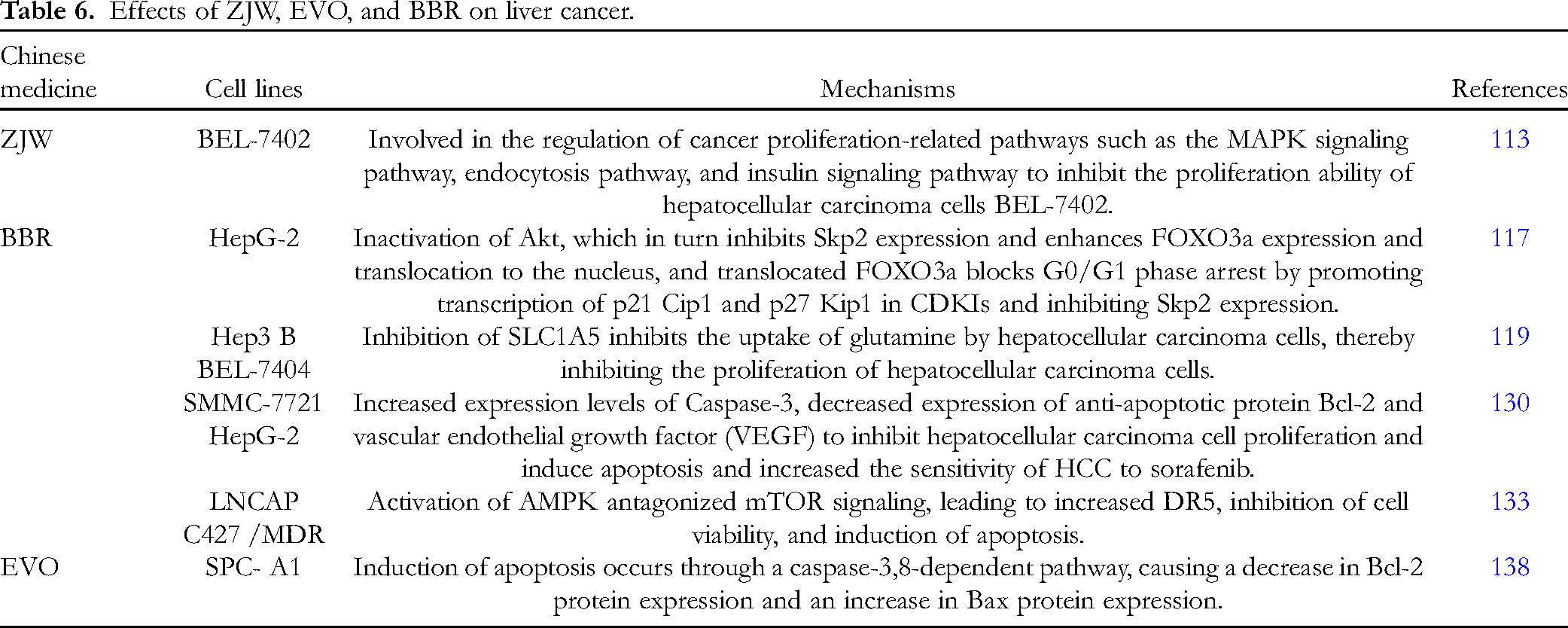
Globally, liver cancer is the most common fatal malignancy. Chemotherapy and immunotherapy are the best treatment options. 112 Yin et al. 113 found that the ZJW significantly inhibited the proliferative capacity of hepatocellular carcinoma (HCC) cells, BEL-7402, mainly through the regulation of cancer proliferation-related pathways, including the MAPK, endocytosis, and insulin signaling pathways. The specific target genes affected by ZJW cover the three types of membrane receptors on the cell membrane upstream of the pathway and are involved in the transduction of downstream cascades. NF-κB signaling is a major pathway regulating the cell cycle. NF-κB can upregulate the expression of the cyclin D1 gene and antagonize the activity of P53 (oncogene), thus promoting tumor cell proliferation. 114 Chou et al. 115 found that ZJW inhibited the proliferation of human HCC HepG2 cells by suppressing c-myc. Xu et al. 116 showed that caspase-3 and-9 activities increased significantly in cells treated with ZJW; Bax and Bak protein levels increased, while those of Bcl-2 and Bcl-xl decreased. Cell cycle arrest in the G0/G1 phase inhibited the proliferation of HCC cells.
Li et al. 117 found that BBR enhanced the expression and nuclear translocation of FOXO3a by inactivating Akt, which in turn inhibited the expression of Skp2, and the translocated FOXO3a blocked the G0/G1 phase arrest by promoting the transcription of p21 Cip1 and p27 Kip1 in CDKIs, thus inhibiting the expression of Skp2. For cancer cell proliferation, glutamine is essential and is mainly transported as Na+ through the plasma membrane by the transporter protein, SLC1A5. 118 Zhang et al. 119 found that BBR could inhibit the uptake of glutamine by HCC cells by suppressing SLC1A5, thus inhibiting the proliferation of HCC cells. Li et al. 120 found that BBR inhibited the proliferation of HCC cells by reducing the level of NF-κB p65, thus promoting apoptosis. A combination of BBR and a Taspine derivative is a new strategy for postoperative cancer treatment, and the Taspine derivative, HMQ1611, showed inhibitory activity against breast cancer and NSCLC. 121 Dai et al. 122 combined HMQ1611 and BBR in HCC cells and found that the latter caused cell cycle arrest in the G1 phase by inhibiting the expressions of cyclinD1, cyclin E, and cdc2 and inhibited the phosphorylation of Akt, mTOR, and ERK, effectively suppressing cell proliferation. BBR negatively regulated the Wnt/β-catenin signaling pathway by upregulating Axin and inducing the degradation of β-catenin, which in turn inhibited the tumor cell migration ability. Thus, BBR can act on HCC cells through multiple pathways in combination. Radiotherapy is a treatment strategy for cancer, but the lack of radiosensitivity in HCC leads to low therapeutic efficacy in clinical settings. The Nrf2-mediated antioxidant defense system is the mainstay against oxidative stress in the liver,123,124 and You et al. 125 found that BBR inhibited proteins related to Nrf2 signaling (Nrf2, HO-1, and NQO-1) in Huh7 and HepG2 cells, thereby increasing the sensitivity toward radiation, making BBR an effective adjuvant in HCC radiotherapy. Wang et al. 126 found that BBR could recruit Skp, Cullin, and F-box complex-β-transduced repeat protein (SCF (β-TCP))-containing HCC cells to induce cyclin D1 degradation, thereby increasing sensitivity to BBR. DOX is a chemotherapeutic agent commonly used in the treatment of HCC. However, DOX, like other chemotherapeutic agents, leads to suppression of the immune microenvironment, 127 thus facilitating tumor growth and inhibiting or even eliminating the immune rejection of cancer cells. Zhang et al. 128 found that in the combination of BBR and DOX, the latter phenotype can be retained by maintaining the phenotype of neutrophil N1, thereby maintaining immune surveillance and sensitivity of HCC HL-60 cells to DOX while promoting tumor cell apoptosis by blocking JAK/STAT3 to regulate neutrophil N2 cell function. Sorafenib is a multi-kinase inhibitor that suppresses multicellular surface tyrosine kinases and intracellular serine/threonine kinases; therefore, sorafenib is well suited to improving the survival and treatment of patients with advanced HCC, 129 but most patients develop resistance to sorafenib after long-term use. Huang et al. 130 found that in the combination of BBR and sorafenib, BBR inhibited HCC cell proliferation and induced apoptosis by increasing the expression of caspase-3 and decreasing those of anti-apoptotic proteins Bcl-2 and VEGF, thus increasing the sensitivity of HCC cells to sorafenib. The tumor necrosis factor-related apoptosis-inducing ligand (TRAIL/Apo2L) has been widely used in cancer chemotherapy because of its low toxicity to normal tissues 131 ; however, the resistance of cancer cells to TRAIL through various mechanisms limits its utility. 132 Ke et al. 133 reported that when TRAIL was combined with BBR, BBR-activated AMPK antagonized mTOR signaling, causing an increase in DR5, inhibition of cell viability, and induction of apoptosis, thus improving TRAIL resistance involving DR5 in patients with advanced HCC. 10-HCPT is a topoisomerase I inhibitor effective for the treatment of HCC, and upregulation of HIF-1α expression in HCC tissues is directly implicated in the development of drug resistance.134,135 BBR can promote ROS production and is a HIF-1α inhibitor. 136 Qi et al. 137 showed greater antiproliferative capacity against HCC cells with the 10-HCPT and BBR combination, exhibiting a longer half-life, and similarly showed inhibition of tumor Hep-3B cells in immunodeficient nude mice, suggesting that BBR in 10-HCPT-resistant cells exhibited good activity.
Yu et al. 138 found that EVO exerted good antitumor effects both in vivo and in vitro in whole animal tumor suppression and drug in vitro tumor suppression experiments, respectively. Its anti-tumor mechanism was as follows: EVO blocked tumor cells in the G2/M phase in a time-dependent manner; it increased the expression and activities of caspases-3 and 8, which in turn gradually decreased the protein levels of the downstream cleaved PARP 1196KD fragment and increased the protein expression of the cleaved 85KD fragment, further decreasing the Bcl-2 protein expression while increasing that of Bax. This suggests that EVO induces the mechanism of apoptosis onset in a caspases-3 and 8-dependent manner, subsequently eliciting the whole process in tumor cells. Lai and Ju 139 found that EVO induced apoptosis through the MAPK pathway by upregulating cleaved PARP protein expression and downregulating the Bcl-2 protein level (Table 6).
Effects of ZJW, EVO, and BBR on liver cancer.
Leukemia
Chronic myeloid leukemia (CML) is the result of the transformation of primitive hematopoietic cells by break point aggregation region protein (BCR) and the oncogene homolog of the Abelson murine leukemia virus (ABL) (BCR-ABL). Imatinib, a tyrosine kinase inhibitor, binds to the ATP-binding site of ABL 140 but its long-term use has led to imatinib resistance in many patients. Yin et al. 141 found that BBR could bind to the protein tyrosine kinase (PTK) structural domain of BCR-ABL, thus inhibiting its activity based on the T315I mutation, and recruited the E3 ubiquitin-protein ligase, LRSAM1. Degradation of these proteins via the autophagy-lysosomal pathway inhibited the viability and colony formation of CML cells, thereby increasing the sensitivity of drug-resistant cells to imatinib.
Acute myeloid leukemia (AML) is a hematological disorder, wherein healthy hematopoietic cells produce blocked and immature adult myeloid cells that proliferate abnormally. 142 Although chemotherapy results in some improvement in AML, its poor prognosis results in the longer survival of tumor cells. 143 Li et al. 144 further elucidated the mechanism of reversal of MDR in K562/ADR cells by investigating the effects of EVO bases on cellular proliferation, cell cycle, and MDR in leukemia K562 cells and K562/ADR-resistant cells. The proliferation of K562 and K562/ADR cells was significantly inhibited by EVO and ursodiol (DNR), which correlated positively with drug dose and administration time. The protein and mRNA levels of BCRP and MDR1 were significantly down-regulated. EVO was effective in reversing DNR resistance in leukemic K562/ADR cells, and this effect might be related to a reduction in the expression of MDR1 on the cell membrane (Table 7).
Effects of EVO and BBR on Leukemia.

Discussion and Conclusions
MDR is a common form of drug resistance, which can be divided into natural and secondary types. With advances in herbal research, several of these have shown a reversal of MDR. EVO and BBR, main active ingredients of a traditional Chinese medicine, have good therapeutic effects on several tumors (Tables 8 and 9).
Abbreviated list of common cancer molecular targets.

Cancer cell line thumbnails.

ZJW can not only inhibit the proliferation of tumor cells, but also reverse multi-drug resistance. Its mechanism of tumor inhibition may be related to activation of mitochondrial pathway, upregulation of Bax and Bak, downregulation of Bcl-2 and Bcl-xL proteins; inhibition of the PI3 K/Akt/NF-κB pathway, and downregulation of ABCB1/P-gp protein expression. In addition, ZJW enhanced JNK activation and Bcl-2 expression while inhibiting Bax expression, thus promoting the pro-apoptotic effects of cisplatin and significantly improving the chemosensitivity of DDP in drug-resistant cells. BBR induced cell cycle G0/1 phase arrest by downregulating HNF4α and cyclin D1 expression; inhibited MAPK/mTOR/p70S6 K and AKT, induced cellular static autophagy, and inhibited the protein expressions of transcription factors SP1 and PDPK1, reduced tumor cell migration and invasion; selective inhibition of G-R NSCLC cells and RASFs growth through the ROS/AMPK pathway; inhibition of Wnt/β-catenin pathway, downregulation of GRP78 expression, and inhibition of AMPK-HIF-1α-P-gp pathway to improve the sensitivity of drug-resistant breast cancer cells; and enhance the activities of caspase-3 and caspase-8 through various ways, and to inhibit the proliferation of ovarian cancer cells. EVO inhibited the Wnt/β-catenin signaling pathway to hinder cell proliferation; inhibited the NOTCH3 signaling pathway, which affected the NF-κB signaling pathway to promote apoptosis and inhibit cell proliferation. The pro-apoptotic effect of Adriamycin was enhanced by inhibiting the protein expression of Ras/MEK/ERK Cascade and IAPs, and down-regulated Cdc2 and pRb levels by upregulating Cyclin B1, p27, and p21, induced cell cycle arrest and apoptosis in G2/M phase. EVO downregulates the protein and mRNA expressions of BCRP and MDR1, increases sensitivity of K562/ADR to DNR; and down regulates mTOR/ s6k1-mediated Mcl-1, enhancing erlotinib-induced apoptosis of NSCLC cells, while reversing drug resistance.
ZJW, EVO, and BBR can reverse drug resistance to some extent, but this work is still in a preclinical research stage because of lack of in-depth research on the mechanism of reversing drug resistance and relatively simple research methods.
Cancer is a complex disease that changes at multiple molecular levels. These molecules belong to different omics and have their own special and complex roles in the body, including proteomics, metabolomics, transcriptomics, genomics, and epigenomics, which are collectively referred to as “multiomics.” In recent years, the study of tumor drug resistance has developed rapidly from single-group to multi-group combination. 145 Spatial transcriptome sequencing is an emerging sequencing technology, which can provide “tissue spatial information” of gene expression and understand the relative positional relationship between specific cells and tissue slices, and thus provides a new perspective for tumor research. 146 This new technology can be used to detect the reverse drug resistance of ZJW, EVO, and BBR, and further study the mechanism of reverse drug resistance through bioinformatics analysis and cell biology experiments.
In addition, studies have shown that the combination of TCM and chemotherapy can not only regulate multiple signaling pathways of abnormal cells, significantly enhance the anti-tumor effect of these therapies, and achieve the purpose of killing tumor cells at different stages of tumor, but also can inhibit the toxic side effects and complications caused by chemotherapy. Therefore, combination therapy is a new treatment option, which is becoming more and more prominent in the treatment of cancer. Shen et al. 147 combined TCM with the chemotherapy drug cisplatin to observe the patient's condition. The results showed that chemotherapy combined with TCM was effective with less adverse reactions, stable control of the disease, and good prognosis. Wu et al. 148 combined Sijunzi Decoction with cisplatin to treat gastric cancer patients. The results showed that the combined therapy could prolong the overall survival period and improve the quality of life of gastric cancer patients. Zhang et al. 149 combined Duhuo Jisheng Decoction with doxorubicin to treat advanced gastric cancer patients with spleen deficiency syndrome. The results found that compared with the chemotherapy group and the single administration group, the TCM combined with chemotherapy could prolong the overall survival time, improve the life quality of patients, reduce the toxicity, and enhance the efficacy of tumor treatment.
The anti-tumor effects of Huanglian, Wuzhuyu, and their main active components, BBR and EVO, have been deeply studied, and it was found that ZJW composed of Huanglian and Wuzhuyu had stronger effects than a single medicine (Huanglian, Wuzhuyu, BBR, or EVO) in anti-tumor cell growth and for promoting tumor cell apoptosis, indicating that Huanglian and Wuzhuyu have synergistic effects. Therefore, in future, more advanced methods, such as spatial transcriptome technology and multiomics technology, will be used to study Huanglian combined with Wuzhuyu, EVO in combination with BBR, and ZJW combined with other chemotherapy drugs, to find more drug targets, and to determine the reversing drug resistance mechanism of Zuojinwan, the synergistic antitumor mechanism of EVO in combination with BBR, and Huanglian combined with Wuzhuyu.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was financially supported by the National Natural Science Foundation of China (81001669, 82003653), the Nature Science Foundation of Shaanxi Province (2020JZ-55), the Subject Innovation Team of Shaanxi University of Chinese Medicine (2019-PY02), and the Key Laboratory Research Project of Shaanxi Province (20J5035), Shaanxi Provincial Health Research Fund project (2022A018)
