Abstract
MS, and IR, 1H NMR and 13C NMR spectroscopy were employed to elucidate 4 novel evodiamine (EVO) derivatives with nitro, amino, and methoxy groups, namely 2-NO2-EVO (
Introduction
Cancer is the biggest threat to public health due to its increasing incidence and high mortality rate.
1
Chemotherapeutic drugs are the most popular treatment options, but they are also accompanied by many serious side effects, high toxicity, and drug resistance. In the past few decades, many plant products have demonstrated effective therapeutic effects against cancer.2,3 Evodiamine (
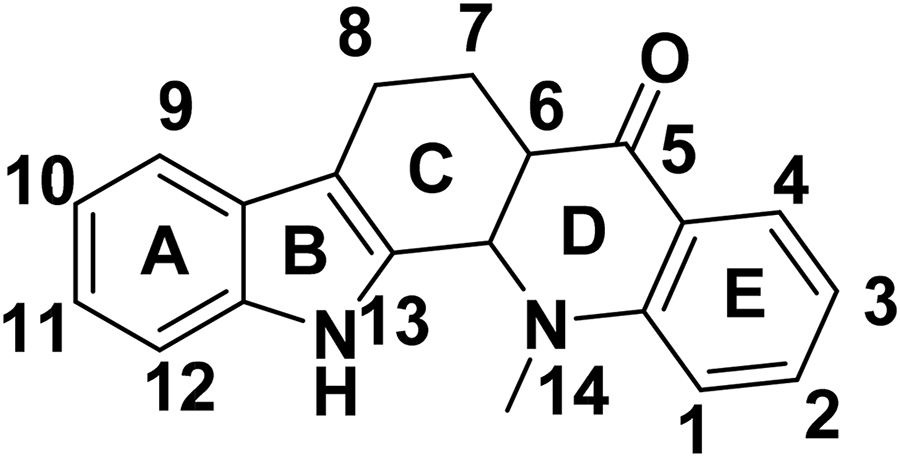
Structure of evodiamine (EVO).
In this work, MS, and IR, 1H NMR and 13C NMR spectroscopy were employed to elucidate 4 EVO derivatives with nitro and amino groups, namely 2-NO2-EVO (
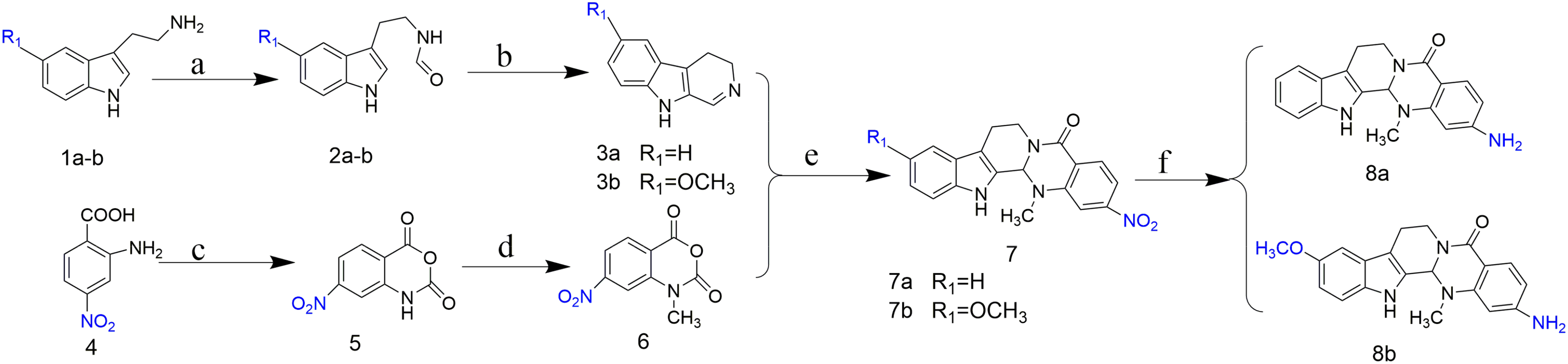
Synthesis of compounds
Results and Discussion
Synthesis and Characterization of Compounds
Synthesis of Compounds 7a, 7b, 8a, and 8b
The synthetic route is shown in Scheme 1. Compounds
NMR Spectra and HRMS of Compounds 7a, 7b, 8a, and 8b
The chemical structures of
In the infrared spectrum, peaks at about 3353 cm−1 in
In Vitro Cytotoxicity Assay
The antiproliferative activity of compounds (

In vitro cytotoxicity of evodiamine (EVO),
Cell Apoptosis Assay
To ascertain further that

In vitro cell apoptosis. Flow cytometry analysis of MDA-MB-231 cells incubated with culture medium as control (A) and 20.00 µM of
Materials and Methods
Materials
Tryptamine, 5-methoxytryptamine, 4-nitroanthranilic acid, and triphosgene were purchased from Aladdin Industrial Corporation. Sodium hydroxide (NaOH), phosphorus oxychloride (POCl3), iodomethane (CH3I), and stannous chloride dihydrate (SnCl2·(H2O)2) were procured from Energy Chemical; fetal bovine serum (FBS) and Dulbecco's modified Eagle's medium (DMEM) from Gibco BRL; and CCK-8 kit from Beyontime, China. Ultrapure water was prepared with a Milli-Q purification system from Millipore at 18.2 MΩ cm−1. The chemical reagents used were all analytically pure. ESI mass spectra were gathered on Aglient 7250 and JEOL-JMS-T100LP AccuTOF spectrometers.
Synthesis of the Compounds
3,4-Dihydro-β-carboline (3a)
Tryptamine (2.048 g, 12.8 mmol) was dissolved in ethyl formate (100 mL) and stirred under gentle reflux overnight at 60 °C. After solvent removal, a light yellow oily liquid was obtained. Using DCM (30 mL) for dissolution, POCl3 (5 mL) was added dropwise, while the temperature was maintained at 0 °C for 2 h. After the addition, the mixture was allowed to warm to room temperature and stirred for an additional 3 h. Solvent was removed and the residue was completely dissolved in aqueous acetic acid (10%). DCM (100 mL) was added and separated. The aqueous layer was neutralized with ammonia solution, and a large amount of precipitate was generated. After filtration, the residue was washed with cooled EtOAc to give 1.46 g (yield 67%) of a yellow solid. 24 1H NMR (400 MHz, DMSO-d6) δ 11.30 ppm (s, 1H), 8.36 ppm (t, J = 2.2 Hz, 1H), 7.56 ppm (d, J = 7.9 Hz, 1H), 7.4 ppm (d, J = 8.2 Hz, 1H), 7.167.21 ppm (m, 1H), 7.027.06 ppm (m, 1H), 3.763.81 ppm (m, 2H), and 2.772.82 ppm (m, 2H).
6-Methoxy-3,4-dihydro-β-carboline (3b)
5-Methoxy-tryptamine (4.05 g, 20 mmol) was dissolved in ethyl formate (100 mL) and stirred under gentle reflux overnight at 60 °C. After solvent removal, a light yellow oily liquid was obtained. This was dissolved in DCM (80 mL) before POCl3 (5 mL) was added dropwise, while the temperature was maintained at 0 °C for 2 h. After the addition, the mixture was allowed to warm to room temperature and stirred for an additional 3 h. Solvent was removed and 10% acetic acid aqueous solution was added until completely dissolved. After the addition of ammonia solution to adjust the alkalinity, a large amount of precipitate was generated. After filtration, the residue was washed with ethyl acetate, obtaining 3.5 g of white solid, with a yield of 87%. 24 1H NMR (DMSO-d6, 400 MHz) (ppm) δ 11.14 (1H, s), 8.31 (1H, t, J = 2.2 Hz), 7.30 (1H, d, J = 8.9 Hz), 7.01 (1H, d, J = 2.4 Hz), 6.83 (1H, dd, J = 8.9, 2.5 Hz), 3.74 to 3.78 (5H, m), and 2.74 to 2.79 (2H, m).
7-Nitro-isatoic anhydride (5)
2-Amino-4-nitrobenzoic acid (4.57 g, 25.1 mmol) was dissolved in anhydrous THF (100 mL). Then, triphosgene (2.96 g, 10 mmol) was added and the reaction refluxed at 70 °C for 5 h. On pouring the solution into ice water (500 mL), a large amount of precipitate was generated. A yellow solid (4.85 g) was obtained by filtration, with a yield of 93%. 1H NMR (400 MHz, DMSO-d6) δ 12.08 ppm (s, 1H), 8.16 ppm (d, J = 8.6 Hz, 1H), 7.97 ppm (dd, J = 8.6, 2.2 Hz, 1H), and 7.87 ppm (d, J = 2.1 Hz, 1H). 13C NMR (101 MHz, DMSO-d6) δ 159.2, 152.4, 147.1, 142.6, 131.4, 117.8, 115.9, 110.7 ppm.
N-methyl-7-nitro-isatoic anhydride (6)
7-Nitroastraphthalic anhydride (1.015 g, 4.88 mmol) was dissolved in 50 mL DMF, and cooled to 0 °C in an ice-water bath. NaH (0.3 g, 12.5 mmol) was added and the mixture was stirred at 0 °C for an additional 1 h. After that, MeI (3.142 g, 22.14 mmol) was added, dropwise. The reaction was allowed to warm up to room temperature and was stirred for an additional 4 h. The reaction solution was poured into ice water (300 mL), followed by filtration, and washing the precipitate with water to obtain a yellow solid product, 0.67 g (yield 62%). 1H NMR (400 MHz, DMSO-d6) δ 8.25 ppm (d, J = 8.6 Hz, 1H), 8.13 ppm (d, J = 1.3 Hz, 1H), 8.07 ppm (dd, J = 8.5, 2.0 Hz, 1H), and 3.56 ppm (s, 3H). 13C NMR (101 MHz, DMSO-d6) δ 158.4, 152.9, 147.9, 143.6, 131.6, 118.0, 117.1, 110.4, 32.6 ppm.
2-Nitro evodiamine (7a)
N-methyl-7-nitro-isatoic anhydride (1.11 g, 5 mmol) and 3,4-dihydro-β-carboline (0.85 g, 5 mmol) were dissolved in 100 mL anhydrous DCM and heating under reflux overnight. The solvent was removed and the residue washed with water. The precipitate was obtained as an orange solid product (1.16 g), with a yield of 66.5%. m.p. 260.5261.9 °C. 1H NMR (DMSO-d6, 400 MHz) δ:10.99 ppm (s, 1H), 7.96 ppm (d, J = 8.4 Hz, 1H), 7.68 ppm (d, J = 2.0 Hz, 1H), 7.62 ppm (dd, J = 8.5, 2.1 Hz, 1H), 7.46 ppm (d, J = 7.8 Hz, 1H), 7.36 ppm (d, J = 8.1 Hz, 1H), 7.12 ppm (t, J = 8.0 Hz, 1H), 7.01 ppm (t, J = 7.2 Hz, 1H), 6.35 ppm (s, 1H), 4.65 ppm (dd, J = 13.0, 5.4 Hz, 1H), 3.443.47 ppm (m, 1H), 3.24 ppm (s, 3H), 2.95 to 3.04 ppm (m, 1H), and 2.74 to 2.79 ppm (m, 1H). 13C NMR (101 MHz, DMSO-d6) δ 163.4, 151.4, 148.6, 136.8, 131.4, 130.1, 126.5, 122.5, 122.1, 119.5, 118.7, 113.3, 112.2, 112.0, 109.8, 71.1, 42.5, 36.9, 19.7 ppm. MS(ESI):349.12946 (C19H16N4O3, [M + H]). Anal. Calcd for C19H16N4O3: C 65.51, H 4.63, N 16.08; found C65.92, H 4.57, N 16.27.
10-Methoxy-2-nitroevodiamine (7b)
N-methyl-7-nitro-isatoic anhydride (2.22 g, 10 mmol) and 6-methoxy-3,4-dihydro-β-carboline (2 g, 10 mmol) were dissolved in 150 mL anhydrous DCM and stirred overnight under reflux. The solvent was removed and the residue was washed with water to give a yellow solid product (1.96 g), with a 52% yield, m.p. 269.5271.0 °C. 1H NMR (DMSO-d6, 400 MHz) δ 10.81 ppm (s, 1H), 7.94 ppm (d, J = 8.5 Hz, 1H), 7.65 ppm (d, J = 2.1 Hz, 1H), 7.61 ppm (dd, J = 8.5, 2.1 Hz, 1H), 7.23 ppm (d, J = 8.8 Hz, 1H), 6.94 ppm (d, J = 2.3 Hz, 1H), 6.76 ppm (dd, J = 8.8, 2.4 Hz, 1H), 6.31 ppm (s, 1H), 4.63 ppm (dd, J = 13.0, 5.5 Hz, 1H), 3.74 ppm (s, 3H), 3.36 ppm (m, 1H), 3.233.36 ppm (s, 3H), 2.97 ppm (m, 1H), and 2.74 ppm (m, 1H). 13C NMR (101 MHz, DMSO-d6) δ 163.4, 153.9, 151.4, 148.6, 132.0, 131.8, 130.1, 126.9, 121.9, 113.2, 112.9, 112.6, 111.8, 109.7, 100.4, 71.2, 55.8, 42.6, 36.9, 19.8 ppm. MS(ESI):379.13922 (C20H18N4O4, [M + H]). Anal. Calcd for C20H18N4O4: C 63.49, H 4.80, N 14.81; found C63. 83, H 4.53, N 14.68.
2-Amino evodiamine (8a)
2-Nitro evodiamine (1.164 g, 3.34 mmol) was suspended in DMF (50 mL), and HCl (5 mL) was added. SnCl2·(H2O)2 (4 g, 17.7 mmol) was dissolved in 30 mL DMF. The previously made Vilsmeier reagent was added dropwise and stirred overnight at 50 °C. Then, saturated sodium hydroxide solution was added until the pH reached 9. After filtering, water (20 mL) was added and the mixture extracted with EtOAc (3 × 100 mL).The combined organic layers were washed with brine (50 mL) and dried with Na2SO4. After solvent removal, the residue was purified on silica gel, eluting with EtOAc/hexanes (1:1→1:2, Rf = 0.17) to obtain a solid (100 mg) compound (
10-Methoxy-2-amino evodiamine (8b)
To 10-methoxy-2-nitro evodiamine (1.2 g, 3.17 mmol) dissolved in 50 mL DMF, 15 mL HCl was added. SnCl2·(H2O)2 (4 g, 17.7 mmol) was dissolved in 30 mL DMF. The previously made Vilsmeier reagent was added dropwise and stirred overnight at 50 °C. Then, saturated sodium hydroxide solution was added until the pH reached 9. After filtering, water (20 mL) was added and extracted with EtOAc (3 × 100 mL).The combined organic layers were washed with brine (50 mL) and dried with Na2SO4. After solvent removal, the residue was purified on silica gel, eluting with (DCM/Ethanol = 100:1, Rf = 0.23) to give compound
Characterization of Compounds 7a, 7b, 8a, and 8b
For elucidation of the chemical structures of
Culture of SW620 and MDA-MB-231 Cells
Human breast carcinoma MDA-MB-231 and human colon carcinoma SW620 cells, purchased from the Type Culture Collection of Chinese Academy of Sciences in Shanghai, China, were cultured in 75 cm2 flasks using DMEM medium containing 1% Pen-Strep (vol/vol) and 10% FBS under 5% CO2 at 37 °C. Medium replacement was implemented at 2-d intervals, and, following cellular confluence, the cells were carried on the passage with a ratio of 1:3. The fourth generation cells were used in this experiment.
In Vitro Cytotoxicity
CCK-8 assay was performed for evaluating the antiproliferative activity of the compounds against MDA-MB-231 and SW620 carcinoma cells.
27
For cell culturing, 5000 cells were seeded into each well of a 96-well plate containing 200 µL medium. After culturing for 24 h, the medium in each well was replaced with fresh medium (20 μL) containing samples in varying concentrations, in order to attain final sample concentrations of 0.015, 0.045, 0.137, 0.411, 1.234, 3.703, 11.11, 33.33, and 100 μM. After 48 h of incubation, the medium was removed and replaced with fresh culture medium containing CCK-8 reagent, which was incubated for another 4 h. The absorbance at 450 nm was read utilizing a microplate reader. The formula for cell viability was calculated as follows
28
:
Cell Apoptosis Assay
Dual-staining with PI and Annexin V-FITC was employed to determine whether the death of cancer cells incubated with 2-NH2-EVO (8a) was induced by apoptosis. After seeding 5 × 105 MDA-MB-231 cells into each well of a 24-well plate overnight, 20.00 µM of 8a was added and incubated for 48 h. The untreated cells were used as control. After 48 h, the cells were dual-stained using PI and annexin V-FITC. With the aid of FCM, the PI and/or annexin V-FITC-positive cell percentages were recorded inside the quadrants.
Statistical Analysis
Data, all of which were acquired from at least 3 separate trials (n = 3), were processed and expressed as means ± SDs. Statistical analysis was carried out through one-way analysis of variance using GraphPad Prism 8, and P values of <.05 were considered to indicate significant inter-mean differences. For calculation of IC50 values, GraphPad Prism 8 was used as well.
Conclusion
In this work, 4 novel EVO derivatives with nitro and amino groups were designed and synthesized. The antiproliferative activities of all compounds were tested against MDA-MB-231 and SW620 cancer cells for 48 h and compared to EVO. Among them,
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211059645 - Supplemental material for Synthesis and Antitumor Activity of Evodiamine Derivatives With Nitro, Amino, and Methoxy Groups
Supplemental material, sj-docx-1-npx-10.1177_1934578X211059645 for Synthesis and Antitumor Activity of Evodiamine Derivatives With Nitro, Amino, and Methoxy Groups by Ruolan Yang, Jingjing Ma, Hui Guo, Qinghua Meng, Yuwei Wang, Hao Yan, Ruyi Jin, Zhi Li and Lingjie Meng in Natural Product Communications
Footnotes
Authors’ Note
Ruolan Yang and Jingjing Ma contributed equally to this work. H.G. conceived and designed the experiments; R.L.Y. performed the experiments; J.J.M., Z.L., Q.H.M., and H.Y analyzed the data; R.Y.J finished calculation; L.J.M contributed reagents/materials/analysis tools; R.L.Y. and H.G. wrote the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (81001669), the Key Science Foundation of Shaanxi Province (2020JZ-55), the Subject Innovation Team of Shaanxi University of Chinese Medicine (2019-PY02), and the Key Laboratory Research Project of Shaanxi Province (20J5035).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

