Abstract
Abnormal angiogenesis plays a key role in cancer progression. In recent years, anti-angiogenic therapy has attracted increasing attention. Berberine (BBR), the main component extracted from Coptis (Ranunculaceae) rhizome, has an anti-angiogenic effect. However, the underlying mechanisms remain to be elucidated. Endothelial cell proliferation is a pivotal process in angiogenesis. In our research, we observed that BBR specifically downregulated the expression of the extracellular signal-regulated kinase 1/2 (ERK1/2) protein in human umbilical vein endothelial cells (HUVECs). The role of BBR in HUVEC proliferation was then assessed using methylthiazolyldiphenyl-tetrazolium bromide and cell counting Kit-8 (CCK-8) assays. The effect of BBR on the ERK1/2 signaling pathway was evaluated using Western blotting. BBR decreased HUVEC proliferation in a dose-dependent manner and inhibited the expression of phospho-ERK1/2 in HUVECs. PD98059, a specific inhibitor of ERK1/2 signaling, attenuated the BBR-induced decrease in the proliferation of HUVECs. Phorbol 12-myristate 13-acetate, a natural activator of ERK1/2 signaling, did not alter BBR-induced proliferation. In conclusion, BBR inhibited endothelial cell proliferation by suppressing ERK1/2 signaling. These findings may provide a potential therapeutic strategy for suppressing tumor growth.
Introduction
Angiogenesis occurs in small blood vessels and endothelial cells. 1 It involves multiple processes, including initiation of hypoxia or inflammation, inflammatory cell gathering, vascular endothelial growth factor generation, endothelial cell proliferation, migration, and differentiation. 2 Generally, the blood vessels of adults remain stable and seldom form fresh branches under physiological conditions. In fact, angiogenesis occurs only during either disease conditions or tissue repair. 3 During physiological angiogenic growth, the proliferative endothelial cells support the functional and structural integrality of budding capillaries. 3 However, an abnormal proliferation could also lead to disease progression. Endothelial cell proliferation, a pivotal process in angiogenesis, is tightly regulated by the extracellular signal-regulated kinase 1/2 (ERK1/2) pathway. 4
ERK1/2 signaling, belonging to the mitogen-activated protein kinase (MAPK) family, is closely related to cell proliferation.5,6 In proliferating cells, the Ras-Raf-MEK-ERK pathway directly controls the G1- to S-stage transition in the cell cycle. 5 ERK1/2 also induces the production of promoting factors in the cell cycle. 7 In addition, ERK1/2 phosphorylates and activates downstream proteins, including the p90 ribosomal S6 kinases (RSKs), mitogen- and stress-activated kinases (MSKs), and MAPK-interacting kinases (MNKs), to extend the MAPK pathway. 4 In this study, we focus on MAPK/ERK-stimulated proliferation.
The anti-angiogenic properties of Chinese herbal medicines have attracted research attention in recent years. Wei and Liu revealed that artemisinin and its derivatives could repress angiogenesis. 8 Meanwhile, the underlying molecular mechanisms have also been demonstrated.9‐11 Berberine (BBR) has been extracted from Chinese herbal medicine. The anti-angiogenic activity of BBR has been studied in vitro 12 and has been applied for treating cancer, 13 and digestive, 14 metabolic, 15 cardiovascular, 16 and neurological diseases in recent years.17‐19 Some studies have revealed that BBR regulates ERK signaling in various cell types. Furthermore, BBR weakens vascular smooth muscle cell proliferation by attenuating ERK activation after mechanical injury in vitro. 20 However, it is unclear whether the antiproliferative effect of BBR exists in endothelial cells.
The present study aimed to explore the antiproliferative action of BBR and its mechanism of action in endothelial cells. We hypothesized that BBR inhibits endothelial cell proliferation via the ERK1/2 pathway. Cell proliferation assays were performed on human umbilical vein endothelial cells (HUVECs) treated with BBR, and the phosphorylation of ERK1/2 was examined using Western blot analysis. In addition, the inhibitor and activator of ERK1/2 signaling were added to verify the signaling pathway involved.
Materials and Methods
Cell Culture
HUVECs were obtained from the ATCC Biosource Center (Manassas, Virginia, USA) and grown in Dulbecco's modified Eagle's medium (DMEM), 10% (v/v) fetal bovine serum (FBS), and 1% (v/v) penicillin–streptomycin (Gibco, Thermo Fisher Scientific). The cells were cultured in a humid environment. The medium was refreshed every other day. Dimethyl sulfoxide (DMSO) was used as a control. BBR (CAS No.-633-65-8, Selleck Chemical LLC), 2-(2′-amino-3′-methoxyphenyl)-oxanaphthalen-4-one (PD98059; CAS No.-167869-21-8, MedChemExpress), and phorbol 12-myristate 13-acetate (PMA) (CAS No.-16561-29-8, MedChemExpress) were dissolved in DMSO.
Cell Viability Assessment Using MTT Assay
Briefly, well-grown cells were seeded in a 96-well dish, and each well contained approximately 5 × 103 cells. 21 The next day, BBR (0, 25, 50, 75, 100, and 200 µM) was mixed with the fresh culture medium. After 24 h, the methylthiazolyldiphenyl-tetrazolium bromide (MTT) kit reagent (10 µL) was added to each well for 4 h. Afterward, the old culture medium was discarded, and DMSO (150 µL) was added to each well for 15 min. Eventually, the viable cells stained blue and formed formazan crystals. The colorimetric value was determined spectrophotometrically at 490 nm.
Cell Viability Assessment Using Cell Counting Kit-8 (CCK-8) Assay
HUVECs were seeded in a 96-well cell culture plate at approximately 5 × 103 cells/well. The next day, the medium was discarded and supplemented with fresh medium containing DMSO, BBR, PD98059, or PMA. Twenty-four hours later, cell viability was examined by adding CCK-8 solution (Cell Counting Kit-8, Lot No. K101818133EF5E, APExBIO), following the manufacturer's instructions. Absorbance was measured at 450 nm after 2 h of incubation.
Western Blot Analysis
Cells were extracted and lysed using radio immunoprecipitation assay (RIPA) lysis buffer. Equal amounts of protein were used for electrophoresis, as previously described. 22 Immunoreactivity was determined using chemiluminescence. The primary antibodies used were phospho-ERK1/2 (Thr202/Tyr204) (D13.14.4E) (1:1000; #4370; Cell Signaling Technology), ERK1/2(137F5) (1:1000; #4695; Cell Signaling Technology), and α-tubulin (1:8000; 11224-1-AP; Proteintech). Goat anti-rabbit IgG (1:8000; cat. No. ab150077; Abcam) was used as the secondary antibody.
Statistical Analysis
Either Student's t-test or one-way analysis of variance, followed by Tukey's multiple comparisons test was used for statistical analysis. Densitometric analysis of Western blots was performed using ImageJ software 1.4.3 (National Institutes of Health). GraphPad Prism software (version 5.0) was used for statistical analysis. Data were obtained from at least three independent experiments and are presented as mean ± standard error (SE). Differences were considered statistically significant at P < .05.
Results
BBR Decreased HUVEC Proliferation in a Dose-Dependent Manner
Endothelial cell proliferation, an important part of angiogenesis, was tested using MTT and CCK-8 assays. BBR significantly decreased HUVEC proliferation at 25 µM concentration (P < .01 and P < .001; Figure 1A-B). Therefore, 25 µM BBR was used to study its antiproliferative mechanism in further experiments. A previous study found that, in humans, a dosage of 2.97 g/kg was safe, which was 100 times higher than the common dosages in clinical research.23,24 The plasma concentration of BBR in humans is extremely low after oral administration. 25

BBR decreases endothelial cell proliferation in a dose-dependent manner. (A) Cell viability, as determined using MTT assay, in HUVECs treated with increasing concentrations of BBR for 24 h. n = 3; **P < .01. (B) Cell viability, as determined using CCK-8 assay, in HUVECs treated with increasing concentrations of BBR for 24 h. n = 3; ***P < .001.
BBR Inhibited Phospho-ERK1/2 in HUVECs
The ERK pathway is activated during cell proliferation, which plays a crucial role in tumor growth. 26 To elucidate the regulatory mechanism of BBR-induced inhibition, we treated HUVECs with BBR (25 µM) for 24 h. We found that the levels of phospho-ERK1/2 visibly decreased (Figure 2A), while total ERK1/2 was unchanged. As shown in Figure 2B, analysis of the percentage of phospho-ERK1/2 to total ERK1/2 suggested inhibition with 25 µM BBR (P <.01).

BBR inhibits the level of phospho-p44/42 (phospho-ERK1/2) in HUVECs. (A) Corresponding protein bands of phospho-p44/42, total p44/42, and tubulin from HUVECs treated with BBR for 24 h. (B) Quantitative analysis of the phospho-p44/42 and total p44/42 blots. n = 3; **P < .01.
Inhibitors of the ERK1/2 Pathway Abrogated the BBR-Induced Decrease in HUVEC Proliferation
PD98059 has been identified as a synthetic inhibitor of MEK but does not influence MAPK. 27 Inhibition of MEK by PD98059 also prevents activation of the ERK pathway in vitro. 28 To verify the influence of the ERK1/2 pathway in controlling the anti-proliferation effect of BBR, HUVECs were preincubated with 50 µM PD98059 for 45 min and then treated with BBR (25 µM) for 24 h. MTT and CCK-8 assays were performed to verify HUVEC proliferation (Figure 3A-B). The protein levels of phospho-ERK1/2 were determined using Western blotting (Figure 3C-F). We found that BBR did not induce a significant decrease in HUVEC proliferation after pretreatment with PD98059. We concluded that endothelial cell proliferation was decreased by BBR via suppression of ERK1/2 signaling.

PD98059 abrogates BBR-induced decrease in HUVEC proliferation. (A) Cell viability, as determined using MTT assay, in HUVECs treated with BBR and PD98059. n = 3; **P < .01; ns, nonsignificant; (B) Cell viability, as determined using CCK-8 assay, in HUVECs treated with BBR and PD98059. n = 3; **P < .01; ns, nonsignificant; (C) Corresponding protein bands from protein samples of HUVECs treated only with PD98059 for 24 h. (D) Quantitative analysis of the phospho-p44/42 and total p44/42 blots. n = 3; **P < .01. (E) Corresponding protein bands from HUVECs treated with PD98059 and BBR for 24 h. (F) Densitometric analysis of the phospho-p44/42 and total p44/42 blots. n = 3; *P < .05.
PMA did not Stimulate the BBR-Induced Decrease in HUVEC Proliferation
PMA is a natural PKC activator. To clarify further the effect of ERK1/2 signaling on BBR-induced reduction of endothelial cell proliferation, HUVECs were incubated with 25 µM BBR for 24 h and then 100 nM PMA was added for 15 min. Cell proliferation was determined using MTT and CCK-8 assays (Figure 4A-B). The influences of BBR and PMA on the ERK1/2 pathway were examined using Western blotting (Figure 4C-F). We found that PMA did not promote a BBR-induced decrease in HUVEC proliferation. Therefore, we conclude that the antiproliferative effect of BBR is mediated by ERK1/2 signaling.
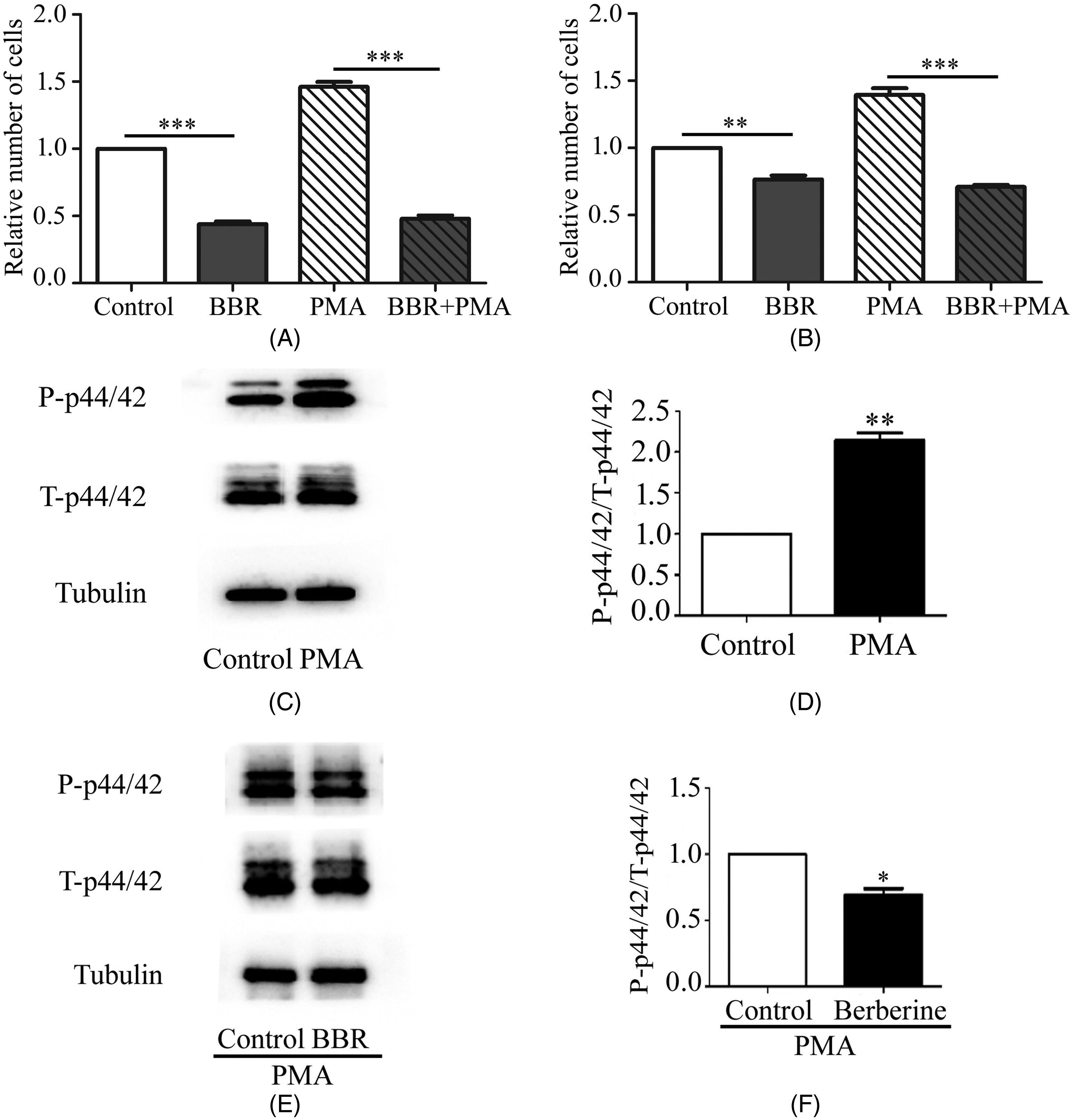
PMA does not stimulate the BBR-induced decrease in HUVEC proliferation. (A) Cell viability, as determined using MTT assay, in HUVECs treated with BBR and PMA. n = 3; **P < .01; (B) Cell viability, as determined using CCK-8 assay, in HUVECs treated with BBR and PMA. n = 3; ** P < .01; *P < .05; (C) Corresponding protein bands from HUVECs treated with PMA only for 15 min. (D) Densitometric analysis of the phospho-p44/42 and total p44/42 blots. n = 3; **P < .01. (E) Corresponding protein bands from HUVECs treated with BBR and PMA. (F) Quantitative analysis of the phospho-p44/42 and total p44/42 blots. n = 3; *P < .05.
Discussion
The occurrence and development of tumors are inseparable from abnormal angiogenesis, which is disorganized, immature, and permeable. 3 In addition, tumor angiogenesis can also be activated by tumor cells, which secrete extracellular vesicles containing regulatory factors into vascular endothelial cells, thereby, initiating a series of responses. 29 Therefore, anti-angiogenic therapy, involving monoclonal antibodies and tyrosine kinase inhibitors, is gradually receiving more attention. 30 BBR is the main available component extracted from the rhizome of Coptis species (Ranunculaceae). The anti-angiogenic properties of BBR have not yet been fully demonstrated. The intrinsic mechanism of the anti-angiogenesis characteristics of BBR was investigated in this study. Given that endothelial cell proliferation is a crucial process in angiogenesis, we explored whether BBR represses the proliferation of endothelial cells.
The influence of the MAPK pathway has been highlighted in the process of cellular activities. 6 There are three main MAPKs, ERK, JNK, and p38. The JNK and p38 families are mainly triggered by environmental stress, whereas ERK is closely related to cell proliferation.22,31 ERK is involved in fundamental signal transmission through the Ras/Raf/MEK/ERK signaling pathway. 32 Among these, Ras works as a promoting protein, Raf works as MAP3K, MEK works as MAP2K, while ERK serves as MAPK. 32 ERK is generally localized in the cytoplasm. When activated by cytokines, growth factors, and serum, ERK enters the nucleus and adjusts cytokine activity to influence cell proliferation by activating Ras, PKC-mediated, and G protein-coupled receptor responses.6,33
The ERK pathway has been shown to control cell proliferation in diverse cell types. In human non-small-cell lung cancer cells, chaetoglobosin G inhibits proliferation by attenuating the level of phospho-ERK, 34 which is similar to the effects of scutellarin on human retinal endothelial cells. 35 Similarly, some researchers have reverse-verified the above points. Danhong injection reportedly promotes the proliferation of hypoxic HUVECs by activating the ERK pathway. 36
BBR, the main bioactive substance, has been used in clinical treatment to alleviate dampness, clear heat, detoxify, and reduce fire for a long time. 37 BBR has also been used to treat bacterial diarrhea in China since the last century. 38 As previously reported, BBR is avirulent in normal cells. 38 Thus, BBR is an effective, safe, and natural drug. Thus, an increasing number of studies have focused on the metabolites and biological activities of BBR to strengthen its applicability. Preclinical studies have demonstrated its potent antioxidant, hepatoprotective, neuroprotective, hypolipidemic, hypoglycemic, antitumor, antimicrobial, and anti-inflammatory activities.37,38 In this study, we expanded the existing information about the biological activities of BBR and presented a stronger basis for further clinical applications of BBR.
Conclusion
In brief, BBR reduced the proliferation of HUVECs in a dose-dependent manner. The negative regulation of the ERK1/2 pathway was involved in the process. These findings present a potential therapeutic agent for suppressing angiogenesis and provide a new research direction for antitumor drugs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
