Abstract
Keywords
Introduction
Big sagebrush, Artemisia tridentata Nutt., Asteraceae, is a conspicuous shrub growing in much of the western United States (Figure 1). The plant is an essential food source and cover for the pygmy rabbit, Brachylagus idahoensis 1 ; the Gunnison sage-grouse, Centrocercus minimus; and the greater sage-grouse, Centrocercus urophasianus,2,3 which rely on this plant for up to 99% of their winter forage. In addition, several wild ungulates, including elk, Cervus canadensis; mule deer, Odocoileus hemionus 4 ; and pronghorn, Antilocapra americana, 5 forage on big sagebrush.

Range of Artemisia tridentata in North America.
The essential oil compositions of A. tridentata have been previously reported, 6 and the plant has been characterized by the major components camphor (20%-45%), camphene (3%-21%), 1,8-cineole (12%-30%), thujone (6%), and borneol (5%).7-12 Additionally, a number of irregular monoterpenoids have been isolated and characterized.13-17 Sesquiterpene lactones have been characterized in the non-volatile extracts of A. tridentata. 18
There is debate regarding the varieties and subspecies of A. tridentata. Nevertheless, there are three widely recognized subspecies: A. tridentata subsp. tridentata Nutt., A. tridentata subsp. wyomingensis Beetle & A.L.Young, and A. tridentata subsp. vaseyana (Rydb.) Beetle.19,20 It has been observed that the three different sagebrush subspecies have different palatability attributes with respect to herbivory. The vaseyana and wyomingensis subspecies are generally more palatable to herbivores, while the tridentata subspecies is the least preferable. 21 The chemical constituents of the three subspecies most likely play a role in the observed herbivory.21-23
To complement our previous examination of A. tridentata subsp. tridentata, 6 the purpose of this study was to characterize the essential oils of A. tridentata subsp. wyomingensis and A. tridentata subsp. vaseyana growing in southwestern Idaho using gas chromatographic techniques and to compare and contrast the three subspecies using multivariate analyses.
Results and Discussion
Essential Oil Composition
A. tridentata subsp. tridentata
The essential oil composition of A. tridentata subsp. tridentata from southwestern Idaho has been previously reported. 6 The major components were yomogi alcohol (5.8%-30.8%), santolina epoxide (1.7%-10.5%), camphor (5.2%-20.1%), and (Z)-tagetone (0.9%-8.9%).
A. tridentata subsp. wyomingensis
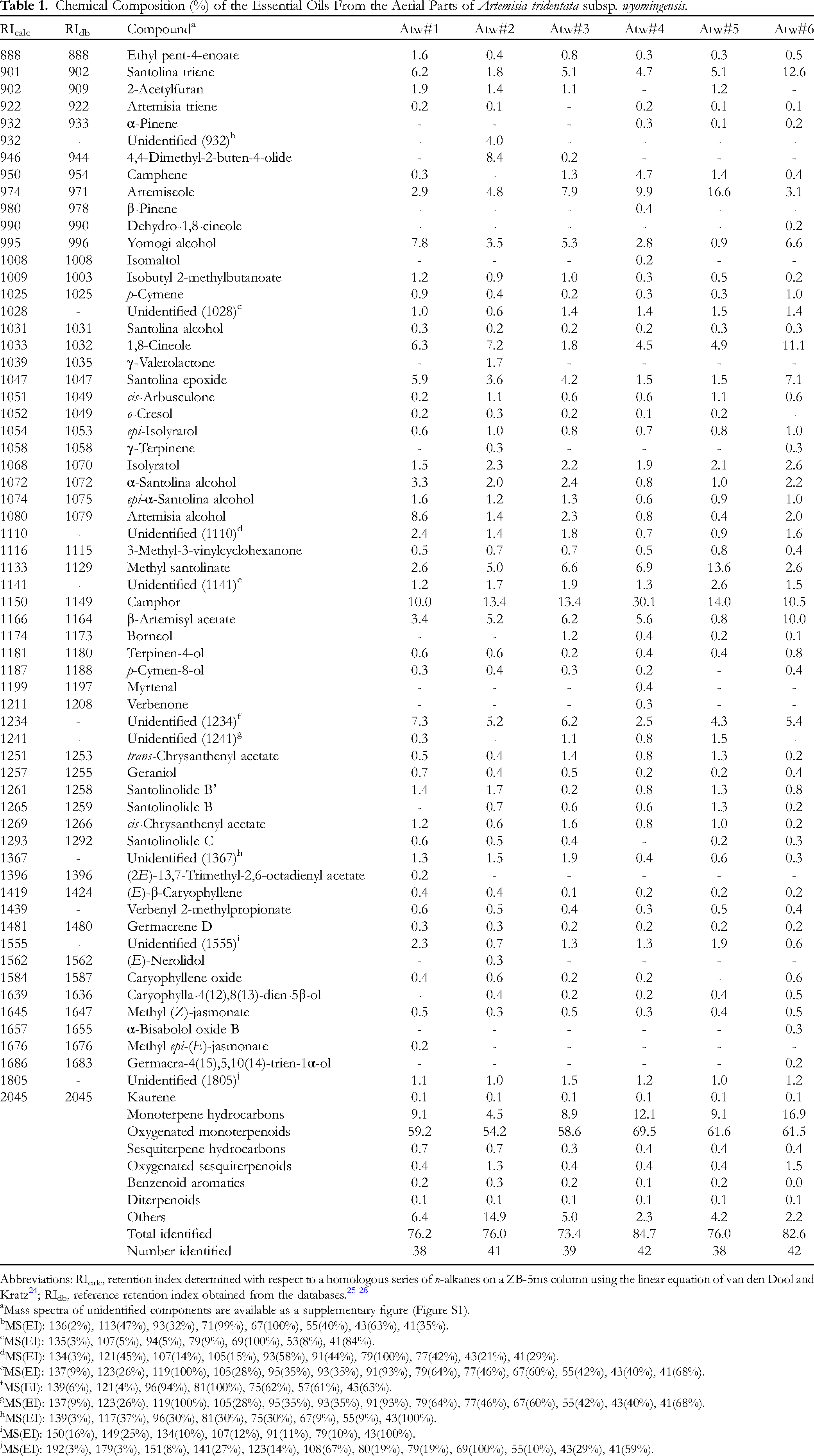
The aerial parts of A. tridentata subsp. wyomingensis were hydrodistilled to give pale yellow essential oils in 2.34% to 4.45% yield. Gas chromatographic analysis of the six essential oil samples led to identification of a total of 53 compounds, which accounted for 73.4%-84.7% of the total compositions. The essential oil compositions of A. tridentata subsp. wyomingensis are compiled in Table 1.
Chemical Composition (%) of the Essential Oils From the Aerial Parts of Artemisia tridentata subsp. wyomingensis.
Abbreviations: RIcalc, retention index determined with respect to a homologous series of n-alkanes on a ZB-5ms column using the linear equation of van den Dool and Kratz 24 ; RIdb, reference retention index obtained from the databases.25-28
aMass spectra of unidentified components are available as a supplementary figure (Figure S1).
bMS(EI): 136(2%), 113(47%), 93(32%), 71(99%), 67(100%), 55(40%), 43(63%), 41(35%).
cMS(EI): 135(3%), 107(5%), 94(5%), 79(9%), 69(100%), 53(8%), 41(84%).
dMS(EI): 134(3%), 121(45%), 107(14%), 105(15%), 93(58%), 91(44%), 79(100%), 77(42%), 43(21%), 41(29%).
eMS(EI): 137(9%), 123(26%), 119(100%), 105(28%), 95(35%), 93(35%), 91(93%), 79(64%), 77(46%), 67(60%), 55(42%), 43(40%), 41(68%).
fMS(EI): 139(6%), 121(4%), 96(94%), 81(100%), 75(62%), 57(61%), 43(63%).
gMS(EI): 137(9%), 123(26%), 119(100%), 105(28%), 95(35%), 93(35%), 91(93%), 79(64%), 77(46%), 67(60%), 55(42%), 43(40%), 41(68%).
hMS(EI): 139(3%), 117(37%), 96(30%), 81(30%), 75(30%), 67(9%), 55(9%), 43(100%).
iMS(EI): 150(16%), 149(25%), 134(10%), 107(12%), 91(11%), 79(10%), 43(100%).
jMS(EI): 192(3%), 179(3%), 151(8%), 141(27%), 123(14%), 108(67%), 80(19%), 79(19%), 69(100%), 55(10%), 43(29%), 41(59%).
The major components found in the essential oil of A. tridentata subsp. wyomingensis were camphor (10.0%-30.1%), artemiseole (2.9%-16.6%), methyl santolinate (2.6%-13.6%), 1,8-cineole (1.8%-11.1%), santolina triene (1.8%-12.6%), β-artemisyl acetate (0.8%-10.0%), and an unidentified component (retention index [RI] = 1234, 2.5%-7.3%).
A. tridentata subsp. vaseyana
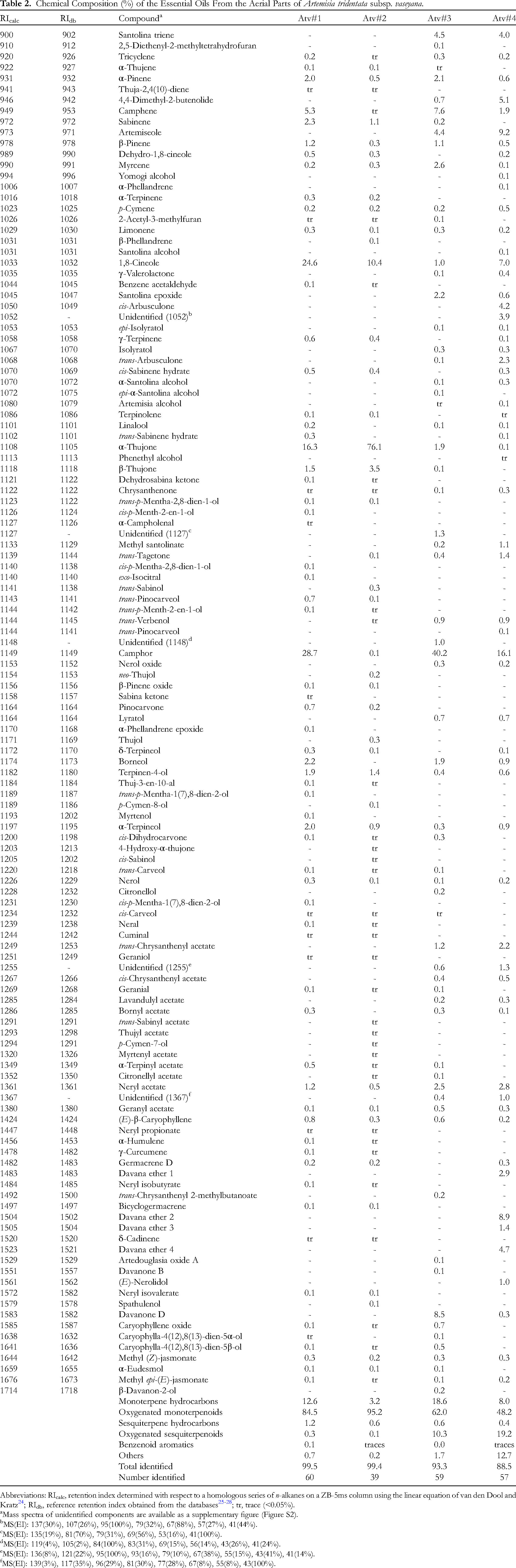
Hydrodistillation of A. tridentata subsp. vaseyana gave yellow essential oils (0.89%-1.73% yield). An aqueous leaf extract of each A. tridentata subsp. vaseyana sample gave a bright blue-white fluorescence under illumination with UV light indicating the presence of coumarins (eg, isoscopoletin, scopoletin, and esculetin) in the leaves. 9 Coumarins are known to be abundant in A. tridentata subsp. vaseyana, but are present in only trace amounts in subsp. tridentata or subsp. wyomingensis. 19 The chemical components of A. tridentata subsp. vaseyana essential oils are listed in Table 2.
Chemical Composition (%) of the Essential Oils From the Aerial Parts of Artemisia tridentata subsp. vaseyana.
Abbreviations: RIcalc, retention index determined with respect to a homologous series of n-alkanes on a ZB-5ms column using the linear equation of van den Dool and Kratz 24 ; RIdb, reference retention index obtained from the databases25-28; tr, trace (<0.05%).
aMass spectra of unidentified components are available as a supplementary figure (Figure S2).
bMS(EI): 137(30%), 107(26%), 95(100%), 79(32%), 67(88%), 57(27%), 41(44%).
cMS(EI): 135(19%), 81(70%), 79(31%), 69(56%), 53(16%), 41(100%).
dMS(EI): 119(4%), 105(2%), 84(100%), 83(31%), 69(15%), 56(14%), 43(26%), 41(24%).
eMS(EI): 136(8%), 121(22%), 95(100%), 93(16%), 79(10%), 67(38%), 55(15%), 43(41%), 41(14%).
fMS(EI): 139(3%), 117(35%), 96(29%), 81(30%), 77(28%), 67(8%), 55(8%), 43(100%).
The essential oils of A. tridentata subsp. vaseyana showed wide variation in composition, but major components included α-thujone (0.1%-76.1%), camphor (0.1%-40.2%), and 1,8-cineole (1.0%-24.6%).
Multivariate Analysis
To provide insight into possible chemical differentiation between the three subspecies of A. tridentata collected in southwestern Idaho, both agglomerative hierarchical cluster analysis (HCA) and principal component analysis (PCA) were carried out using the percent compositions of the major components. The compositions of A. tridentata subsp. tridentata 6 are also included in the analyses. The HCA is shown in Figure 2, and the PCA is shown in Figure 3.
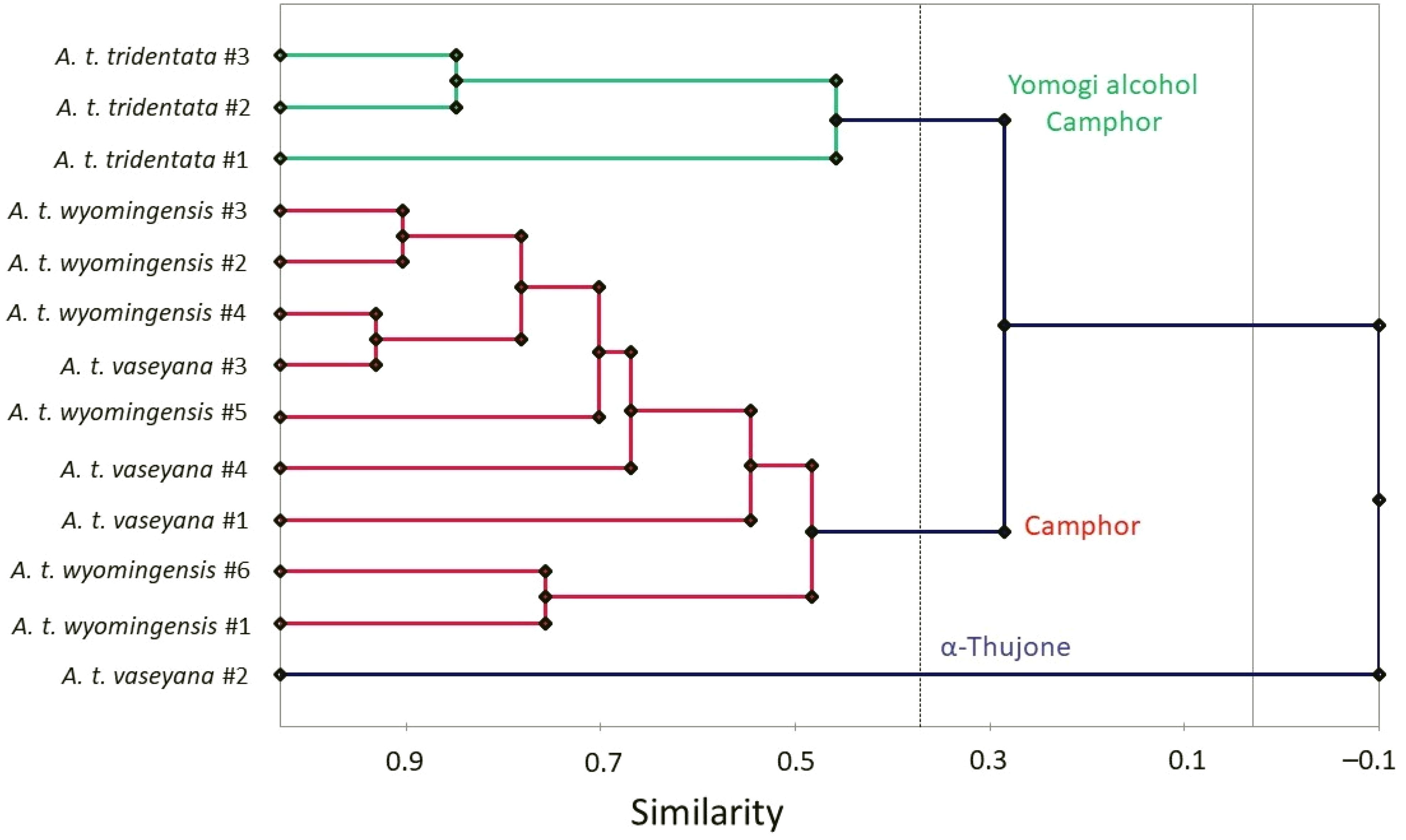
Dendrogram based on hierarchical cluster analysis of Artemisia tridentata chemical compositions.

Biplot based on principal component analysis of Artemisia tridentata chemical compositions.
The HCA revealed three well-defined clusters: (a) A cluster defined by relatively high concentrations of yomogi alcohol and camphor and made up of the three A. tridentata subsp. tridentata samples, 6 (b) a cluster dominated by camphor and comprised of all six A. tridentata subsp. wyomingensis samples as well as three A. tridentata subsp. vaseyana samples, and (c) a single A. tridentata subsp. vaseyana sample dominated by α-thujone. The PCA corroborates the HCA with the A. tridentata subsp. tridentata group positively correlating with yomogi alcohol and camphor. There is large grouping that strongly correlates with camphor and corresponds to the camphor-rich cluster from the HCA. Finally, A. tridentata subsp. vaseyana sample #2 strongly correlates with α-thujone.
To further probe the volatile components that differentiate the three A. tridentata subspecies, an analysis of variance (ANOVA) was carried out on each of the major components in A. tridentata essential oils. The chemical components that exhibited statistical differences between the subspecies are illustrated in Figure 4.

Percentage differences of volatile chemical components in the essential oils of three subspecies of Artemisia tridentata from Southwest Idaho, USA. Bars with same letter are not significantly different at P ≤ 0.05.
As observed in the HCA and PCA, the high concentration of yomogi alcohol in A. tridentata subsp. tridentata differentiates that subspecies from the wyomingensis and vaseyana subspecies. Furthermore, the (Z)-tagetone concentration differentiates A. t. tridentata from A. t. wyomingensis and A. t. vaseyana. On the other hand, the concentration of unidentified component (RI = 1234) in A. t. wyomingensis is significantly higher than those in A. t. tridentata or A. t. vaseyana. At this point, it is difficult to explain the chemical differences between the subspecies. There are likely genetic differences, but the collection sites were also different, so there may be environmental effects.
The essential oils of A. tridentata subsp. tridentata, A. tridentata subsp. wyomingensis, and A. tridentata subsp. vaseyana from Wyoming have been published. 23 A comparison of the major components of A. tridentata subsp. wyomingensis and A. tridentata subsp. vaseyana from Idaho and from Wyoming is summarized in Table 3.
Major Essential oil Components of Artemisia tridentata Subsp. wyomingensis and Artemisia tridentata Subsp. vaseyana from Southwestern Idaho and North-Central (Bighorn Mountains) Wyoming.

Abbreviation: nd, not detected.
aThe concentrations were significantly greater (ANOVA, P < 0.05) in the Wyoming samples than the Idaho samples.
bThe concentrations were significantly greater (ANOVA, P < 0.05) in the Idaho samples than the Wyoming samples.
There are some notable differences between the concentrations of the major components of the samples from Idaho and the samples from Wyoming. A perusal of Table 3 reveals that both the Idaho samples and the Wyoming samples of A. tridentata subsp. wyomingensis were rich in santolina triene, artemiseole, 1,8-cineole, and camphor. However, the Wyoming samples had significantly higher concentrations of artemiseole and 1,8-cineole. The camphor concentrations were statistically comparable. In addition, the Wyoming samples of A. tridentata subsp. wyomingensis showed significantly higher percentages of α-pinene, camphene, cis-arbusculone, trans-arbusculone, chrysanthenone, and borneol. Conversely, the concentrations of yomogi alcohol, santolina epoxide, methyl santolinate, and β-artemisyl acetate were significantly higher in the Idaho samples.
There are wide variations in the concentrations of the major components in A. tridentata subsp. vaseyana, in both the samples from Idaho and the samples from Wyoming. Nevertheless, the concentrations of 1,8-cineole and camphor were generally high in samples from both locations. α-Thujone was particularly abundant in A. tridentata subsp. vaseyana from Idaho, while significantly lower in samples from Wyoming. In addition, santolina epoxide and methyl santolinate concentrations were significantly higher in the A. tridentata subsp. vaseyana samples from Idaho. In a common garden experiment, Welch and McArthur 9 found α-thujone content to be significantly higher in A. tridentata susbp. vaseyana while camphor concentration was higher in A. tridentata subsp. tridentata. There were, however, large variations in monoterpenoid concentrations.
Interestingly, there were three compounds that were found in high concentrations in several samples of A. tridentata subsp. vaseyana from Wyoming 23 that were not detected in this subspecies from Idaho: Fragranol, trans-α-necrodyl acetate, and grandisol. Fragranol was first isolated and characterized from Artemisia fragrans. 29 It has been subsequently identified in several essential oils of the Asteraceae, including Tanacetum dumosum, 30 Achillea falcata, 31 Achillea ligustica, 32 and Achillea lingulata. 33 trans-α-Necrodyl acetate is a major component of Lavandula luisieri,34,35 but it has also been found in the essential oils of Cladanthus mixtus (Asteraceae) 36 and Achillea ageratifolia (Asteraceae). 37 Grandisol is a sex pheromone of the boll weevil (Anthonomus grandis) and the pine weevil (Pissodes castaneus). 18 It was also found in high concentration in Achillea falcata (Asteraceae) 31 and Artemisia vestita (Asteraceae). 38
Terpenoid Enantiomeric Distributions
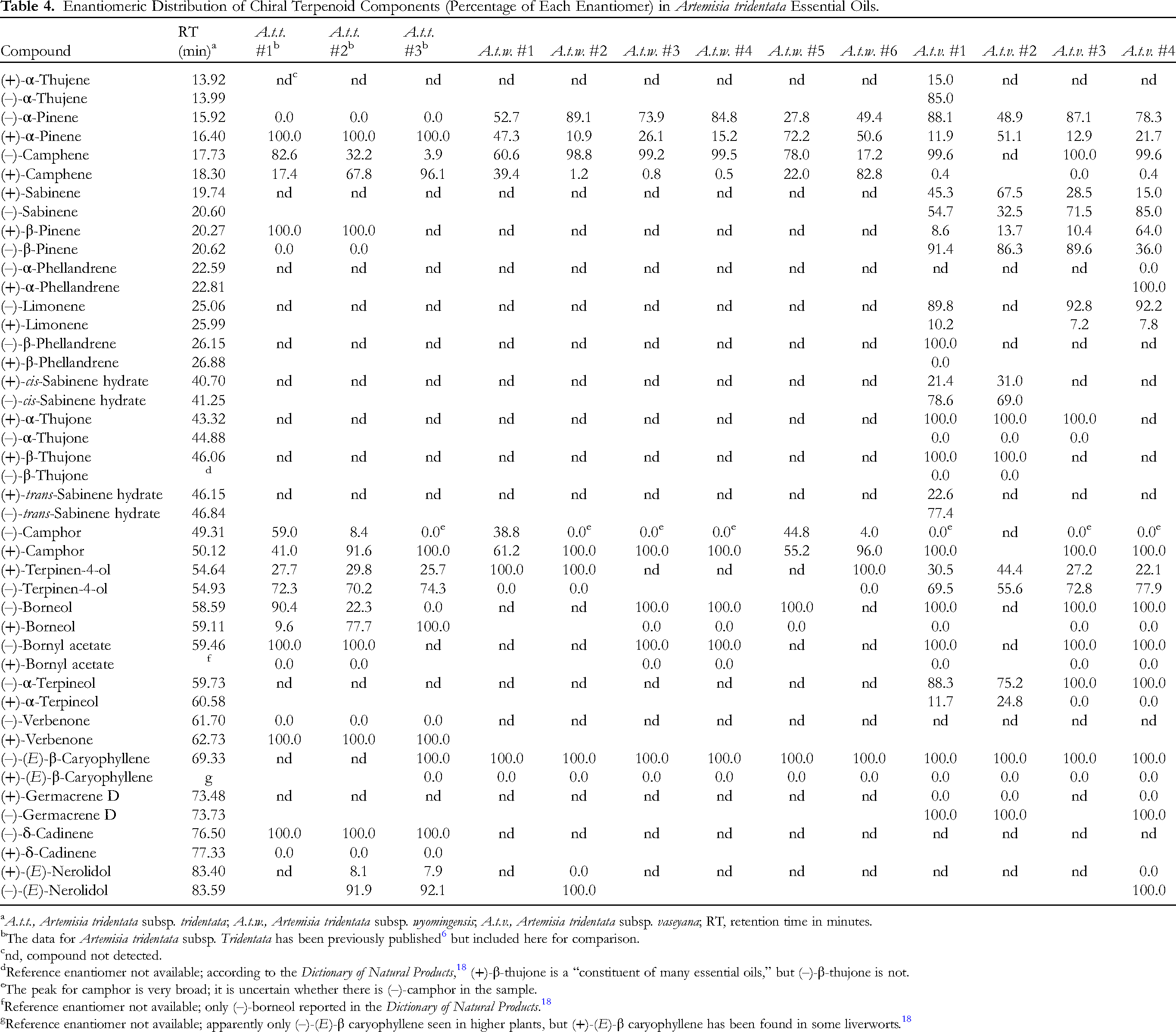
To further characterize the volatile phytochemistry of A. tridentata, chiral GC-MS was carried out on the essential oils of A. t. tridentata, 6 A. t. wyomingensis, and A. t. vaseyana (see Table 4).
Enantiomeric Distribution of Chiral Terpenoid Components (Percentage of Each Enantiomer) in Artemisia tridentata Essential Oils.
aA.t.t., Artemisia tridentata subsp. tridentata; A.t.w., Artemisia tridentata subsp. wyomingensis; A.t.v., Artemisia tridentata subsp. vaseyana; RT, retention time in minutes.
bThe data for Artemisia tridentata subsp. Tridentata has been previously published 6 but included here for comparison.
cnd, compound not detected.
dReference enantiomer not available; according to the Dictionary of Natural Products, 18 (+)-β-thujone is a “constituent of many essential oils,” but (–)-β-thujone is not.
eThe peak for camphor is very broad; it is uncertain whether there is (–)-camphor in the sample.
fReference enantiomer not available; only (–)-borneol reported in the Dictionary of Natural Products. 18
gReference enantiomer not available; apparently only (–)-(E)-β caryophyllene seen in higher plants, but (+)-(E)-β caryophyllene has been found in some liverworts. 18
A perusal of Table 4 reveals a few trends in enantiomeric distribution of terpenoids in A. tridentata essential oils, so differentiation between subspecies based on enantiomers does not seem possible. (+)-α-Pinene was the exclusive enantiomer in A. tridentata subsp. tridentata, but the enantiomeric distribution varied widely in the wyomingensis and vaseyana subspecies. The enantiomeric distribution of camphene varied widely within subspecies. Sabinene was only detected in A. tridentata subsp. vaseyana, but the enantiomeric distribution was variable. Limonene was only detected in A. tridentata subsp. vaseyana and (–)-limonene was the major enantiomer. Only the (+)-enantiomers of α- and β-thujone were observed in A. tridentata subsp. vaseyana. (+)-Camphor generally dominated the essential oils of A. tridentata. (–)-Terpinen-4-ol was the major enantiomer in A. tridentata subsp. tridentata and A. tridentata subsp. vaseyana. When observed, only (+)-terpinen-4-ol was found in A. tridentata subsp. wyomingensis. (–)-Borneol was the major enantiomer in A. tridentata subsp. wyomingensis and A. tridentata subsp. vaseyana. Only (–)-bornyl acetate was found in A. tridentata essential oils. α-Terpineol was only found in A. tridentata subsp. vaseyana and the (–)-enantiomer predominated. Only (–)-(E)-β-caryophyllene was detected in A. tridentata essential oils.
Conclusions
The essential oil compositions of three subspecies of big sagebrush, A. tridentata subsp. tridentata, A. tridentata subsp. wyomingensis, and A. tridentata subsp. vaseyana have shown chemical profiles that may be useful in differentiating these subspecies. However, there is much variation in essential oil compositions both within subspecies and between geographical locations. In this work, the essential oils were examined in different stages of development, which likely affects the compositions. It is apparent that much additional investigation is necessary to more fully characterize the volatile phytochemistry of A. tridentata, and we plan future studies to include collections in the same calendar year, at several different months, and different geographical locations, to examine seasonal and geographical variations as well as subspecies variations.
Materials and Methods
Plant Material
Aerial parts from six individuals of A. tridentata subsp. wyomingensis were collected on August 1, 2022 (flowering stage) from the Snake River Birds of Prey National Conservation Area, Idaho (see Table 5). Hydrodistillation of A. tridentata subsp. wyomingensis gave pale-yellow essential oils (Table 5). Note that an aqueous leaf extract of A. tridentata subsp. wyomingensis was not fluorescent under UV light, indicating the absence of coumarins.
Artemisia tridentata Collection and Hydrodistillation Details.
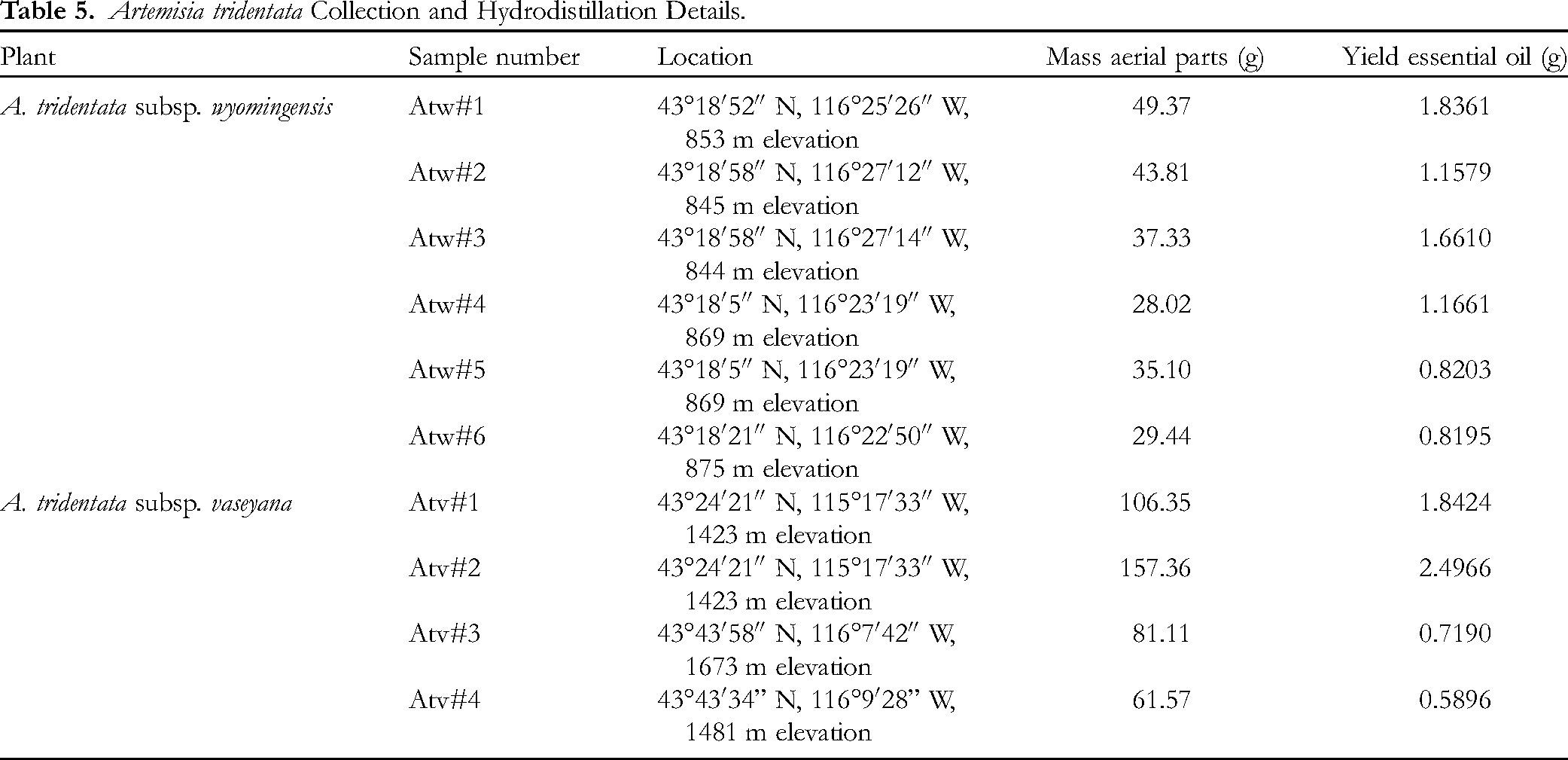
Two individuals of A. tridentata subsp. vaseyana were collected near Pine, Idaho on June 28, 2022 (pre-flowering) and two individuals were collected near Bogus Basin Ski Resort, Idaho on July 7, 2022 (pre-flowering) (see Table 5). Hydrodistillation of the four samples gave yellow essential oils. Note that aqueous leaf extracts of each A. tridentata subsp. vaseyana showed bright blue-white fluorescence under UV light, indicating the presence of coumarins.
Plants were identified by Setzer by consulting the botanical descriptions,21,39 and by comparison with samples from the New York Botanical Garden Virtual Herbarium (https://sweetgum.nybg.org/science/vh/, accessed on August 1, 2022). Voucher specimens (WNS-Atw-5745, WNS-Atv-5680, and WNS-Atv-5714) have been deposited in the University of Alabama in Huntsville herbarium. For each specimen, the aerial parts were fresh frozen and stored at −20 °C until distilled. The plant material was hydrodistilled using a Likens-Nickerson apparatus for 4 h to give the essential oils (Table 5).
Gas Chromatography Coupled with Flame Ionization Detection and Gas Chromatography–Mass Spectrometry Analyses
The essential oils of A. tridentata subsp. wyomingensis and A. tridentata subsp. vaseyana were analyzed by gas chromatography coupled with flame ionization detection, gas chromatography–mass spectrometry (GC/MS), and chiral GC/MS as previously described. 6 The essential oil components were identified by comparison of the mass spectral fragmentation patters and by comparison of RI values available in the Adams, 25 FFNSC 3, 26 NIST20, 27 and our own in-house database. 28 The identification of enantiomers was determined by the comparison of retention times with authentic samples obtained from Sigma-Aldrich (Milwaukee, WI, USA).
Statistical Analysis
For the HCA, the 13 A. tridentata essential oil compositions were treated as operational taxonomic units, and the percentages of the 22 most abundant essential oil components, namely, santolina triene, 4,4-dimethyl-2-buten-4-olide, camphene, artemiseole, yomogi alcohol, 1,8-cineole, santolina epoxide, cis-arbusculone, artemisia ketone, trans-arbusculone, α-santolina alcohol, artemisia alcohol, α-thujone, methyl santolinate, camphor, (Z)-tagetone, β-artemisyl acetate, unidentified (1234), davana ether 2, davana ether 4, unidentified (1554), and davanone D, were used to delineate the chemical associations between the A. tridentata essential oil samples. Pearson correlation was used to measure similarity, and the unweighted pair group method with arithmetic average was used for cluster definition. PCA was performed for the visual verification of the essential oil interrelationships of the different A. tridentata subspecies using the 13 major components as variables with a Pearson correlation matrix. The HCA and PCA analyses were performed using XLSTAT v. 2018.1.1.62926 (Addinsoft, Paris, France). ANOVA was conducted by one-way ANOVA followed by the Tukey test using Minitab® 18 (Minitab Inc., State College, PA, USA). Differences at P < 0.05 were considered to be statistically significant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231154965 - Supplemental material for Chemical Characterization of Three Artemisia tridentata Essential Oils and Multivariate Analyses: A Preliminary Investigation
Supplemental material, sj-docx-1-npx-10.1177_1934578X231154965 for Chemical Characterization of Three Artemisia tridentata Essential Oils and Multivariate Analyses: A Preliminary Investigation by Kathy Swor, Prabodh Satyal, Ambika Poudel and William N Setzer in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X231154965 - Supplemental material for Chemical Characterization of Three Artemisia tridentata Essential Oils and Multivariate Analyses: A Preliminary Investigation
Supplemental material, sj-docx-2-npx-10.1177_1934578X231154965 for Chemical Characterization of Three Artemisia tridentata Essential Oils and Multivariate Analyses: A Preliminary Investigation by Kathy Swor, Prabodh Satyal, Ambika Poudel and William N Setzer in Natural Product Communications
Footnotes
Acknowledgement
This work was carried out as a part of the activities of the Aromatic Plant Research Center (https://aromaticplant.org/).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Data Availability Statement
The data presented in this study are available upon reasonable request from the corresponding author.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
