Abstract
Platycodin D (PD) is a naturally occurring, biologically active triterpenoid saponin isolated from a medicinal food homology plant called Platycodon grandiflorus (Jacq.) A. DC. It is involved in the processing of various biological activities. While investigating the anti-inflammatory property of PD using lipopolysaccharide (LPS)-stimulated murine RAW 264.7 macrophage cells, we unexpectedly found that PD exhibited toxicity to RAW 264.7 cells. In this study, the toxic effect of PD on RAW 264.7 cells was systematically evaluated for the first time. The results showed that PD (12.5−200 µM) significantly reduced cell viability and inhibited cell proliferation in a dose-dependent manner. At a concentration of 20 µM, PD significantly increased lactate dehydrogenase activity and the mRNA and protein expression of Bax, p53, Casp3, IL-1β, and TNF-α. Interestingly, PD (0.8−20 µM) inhibited the expression of inflammatory cytokines in LPS-stimulated RAW 264.7 cells. PD (20 µM) also significantly increased reactive oxygen species (ROS) levels and the expression of oxidative stress-related genes and proteins. This study revealed that PD exhibited immunotoxicity to RAW 264.7 cells, with possible mechanisms including oxidative stress-mediated apoptosis resulting in activation of the mitochondrial apoptosis pathway and dysregulated expression of inflammatory cytokines. This study evaluated the impact of PD on immunity and provided guidelines for its future biological application.
Introduction
Platycodon grandiflorus (Jacq.) A. DC (Figure 1), a traditional Chinese medicine, is widely used in clinical settings to treat respiratory diseases such as cough, phlegm, and sore throat. 1 Modern pharmacological research has shown that P grandiflorus has antitumor, anti-inflammatory, antibacterial, antioxidative, immunoregulatory, and other broad pharmacological properties. 2 P grandiflorus is not only an important medicinal herb but also a nutrient-rich food. It is frequently used to create delectable table foods, such as pickles and cold dishes. 3 It has recently shown great potential in the market of health products, cosmetics, functional foods, and immunostimulatory agents. 4 Previous research reported that P grandiflorus extract induced macrophage cell proliferation 5 and relieved spleen and thymic damage in cyclophosphamide-induced rats. 6 Polysaccharides extracted from P grandiflorus produced a large amount of nitric oxide (NO) and stimulated the release of TNF-α and IL-6 in macrophages, which exhibited a great immune-stimulating effect. 7 Polysaccharides are also specific activators of B cells and macrophages.8,9 Additionally, P grandiflorus total saponins are frequently employed to boost their hemolytic and adjuvant properties. 10 For instance, platycodin D2 (PD2) elicited a mixed Th1/Th2 immune response and significantly enhanced splenocyte proliferation. 11 The serum hepatitis B surface antigen (HBsAg)-specific antibody titers and the expression of Th1 and Th2 cytokines were increased by platycodin D (PD), which may be a candidate hepatitis B vaccine adjuvant. 12

(A) P. grandifloras. 15 (B) The medicinal part of P. grandifloras. (C) Pickles of P. grandifloras. (D) Molecular formula of Platycodin D (PD).
Macrophages are the first line of defense against pathogens and immune activation coordinators and are critical for the health of the immune system. 13 PD is the characteristic constituent of P grandiflorus, which exhibits antitumor effects through multiple mechanisms. 14 While studying the anti-inflammatory effects of PD, its toxicity toward RAW 264.7 cells was unexpectedly observed. Previous reports have mainly focused on the potential role of PD as a vaccine adjuvant, and little research has been done to explore the effect of PD on macrophages. To investigate the immune functions influenced by PD, we studied the potential properties of PD on RAW 264.7 macrophages. Cell proliferation assays demonstrated that PD significantly reduced cell viability in a dose-dependent manner. The mRNA expression of apoptosis-associated genes and inflammatory cytokines was detected after cells were treated with PD or PD + lipopolysaccharide (LPS). In addition, we measured reactive oxygen species (ROS) levels and the protein content and mRNA expression of oxidative stress-related cytokines in RAW 264.7 cells. These preliminary experimental data may help elucidate the immunity of PD on macrophages and provide health risk assessment in clinics. This further promotes the development and application of PD.
Results
PD Inhibited Proliferation and Induced Apoptosis in RAW 264.7 Cells
As presented in Figure 2A and B, the cell survival rate of PD (12.5−200 µM) was significantly lower than that of the control after 24 h of culture. In particular, the cell survival rate remained approximately 25% when the cells were treated with PD (20 µM) (Supplemental Table S2). More serious damage occurred in cells treated with PD for 48 h. After treatment with PD (20 and 100 µM), proliferating cells were barely observed (Figure 2D). Apoptosis often causes many changes in morphological and biochemical processes. 16 Intracellular components were severely shrunken and destroyed with 100 µM PD (Figure 2E). Lactate dehydrogenase (LDH) studies further revealed that LDH activity at 20 µM PD was approximately 4.5-fold greater than that in the control (Figure 2C and Supplemental Table S3). However, the significant decline in LDH activity observed in the 100 µM PD group was probably due to severe changes in cell morphology.

(A) Relative cell viability of RAW 264.7 cells after treatment with PD (1.56−200 µM) for 24 h or 48 h. (B) Relative cell viability of RAW 264.7 cells after treatment with PD (0.16−100 µM) for 24 h or 48 h. (C) Relative LDH activity in RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) for 24 h. (D) After treatment with PD (0.8, 4, and 20 µM) for 24 h, cell proliferation was observed using fluorescence microscopy (bar = 200 µm). (E) Cell morphology was observed using fluorescence microscopy (bar = 200 µm). *P <0.05, **P < 0.01, compared to the control. Abbreviations: PD, Platycodin D; LDH, lactate dehydrogenase.
PD Regulated the mRNA Expression of Apoptosis-Related Genes in RAW 264.7 Cells
To further explore the mechanism of PD-induced cell apoptosis, the expression of Bax, p53, and caspase-3 was determined. Figure 3 reveals that PD (0.8 µM) induced a 1.64-, 1.65-, and 1.27-fold increase in Bax, p53, and caspase-3 mRNA levels, respectively, compared to the control. Furthermore, the relative mRNA levels of Bax and p53 were significantly increased (P <0.01) by treatment with 20 µM PD, resulting in approximately 2.15- and 1.56-fold increases in mRNA levels, respectively (Supplemental Table S4).
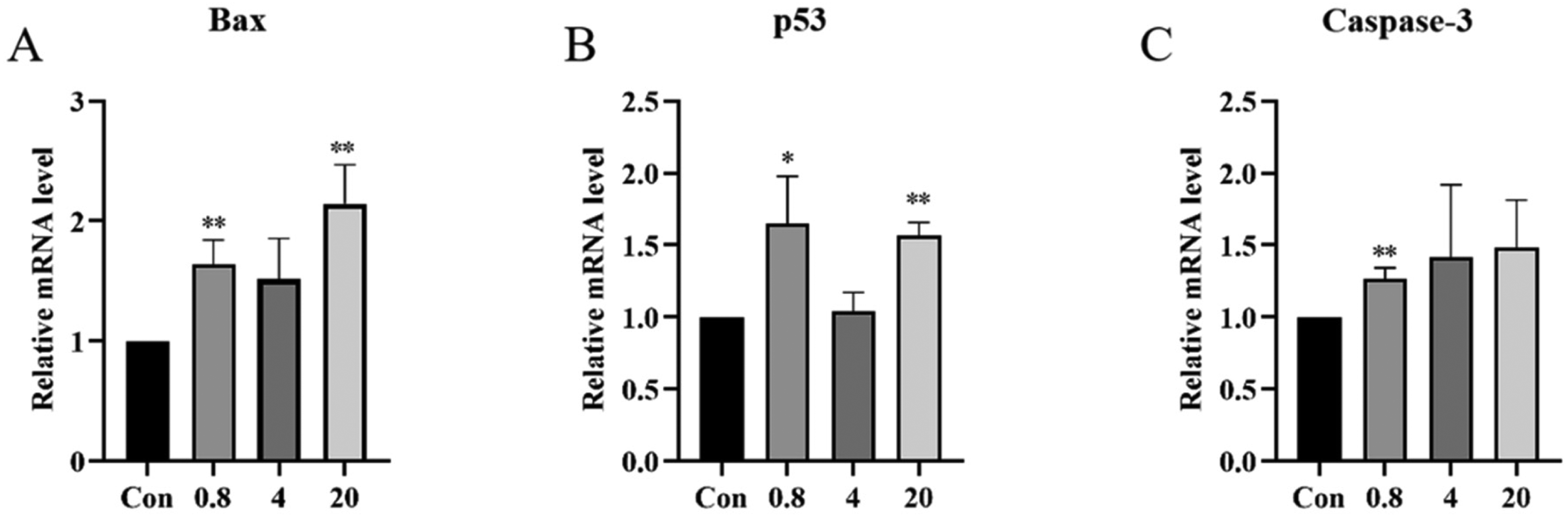
The relative mRNA expression levels of Bax, p53, and caspase-3 in RAW 264.7 cells after treatment with Platycodin D (PD) (0.8, 4, and 20 µM) for 24 h. *P < 0.05, **P < 0.01, compared to the control.
PD Regulated the mRNA and Protein Expression of Inflammatory Cytokines in RAW 264.7 Cells
In LPS-stimulated RAW 264.7 cells, the LPS group promoted cell proliferation, while treatment with PD (20 and 100 µM) for 24 h significantly decreased the cell survival rate (Figure 4A and Supplemental Table S5). Figure 4B demonstrates that PD significantly regulated the protein content of IL-1β (Supplemental Table S6). The mRNA expression of IL-1β and TNF-α was increased by 17.68- and 3.40-fold, respectively, after treatment with 20 µM PD compared to the control (Figure 4C and Supplemental Table S7). The mRNA expression of inflammatory factors was significantly increased in cells treated with LPS (1 µg/mL) for 24 h. In coculture conditions of PD and LPS, treatment with various concentrations of PD reduced IL-1β and IL-6 levels (Figure 4D and Supplemental Table S8).

(A) Relative cell viability of RAW 264.7 cells after treatment with PD (0.8, 4, 20, and 100 µM) and LPS (1 µg/mL) for 24 h.
PD Increased ROS Levels in RAW 264.7 Cells
Excessive ROS is an important factor in mitochondria-mediated apoptosis. 17 Intracellular ROS levels can be reflected by the fluorescence intensity produced by 2',7'-dichlorofluorescein (DCF). Figure 5A and Supplemental Table S9 show that treatment of cells with 20 µM PD induced an approximately 1.5-fold increase in ROS levels compared to the control, whereas ROS levels hardly changed in the PD (0.8 and 4 µM) groups. Except for the 20 µM PD group, green fluorescence was rarely observed under the fluorescence microscope (Figure 5B).

(A) Relative fluorescence intensity of RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) for 24 h.
PD Regulated the mRNA and Protein Expression of Oxidative Stress-Related Cytokines in RAW 264.7 Cells
The mRNA expression of oxidative stress-related genes was measured 24 h after PD therapy. Real-time quantitative polymerase chain reaction (RT-qPCR) analysis revealed that PD increased the mRNA expression of oxidative stress-related genes, with different doses of PD causing dose-dependent increases in their expression levels (Figure 6 and Supplemental Table S10). Treatment of cells with PD at 4 and 20 µM concentrations caused 1.39- and 1.84-fold increases in the mRNA level of Sod2 and 1.75- and 2.06-fold increases in the mRNA level of Gpx2, respectively, compared to the control. After treatment with 20 µM PD, the mRNA levels of Sod1 and Gpx1 increased 2.38- and 3.64-fold and the mRNA levels of Cat and Gsta1 increased 6.67- and 9.28-fold, respectively, compared to the control. Furthermore, exposure to PD (20 µM) significantly increased Sod content from 1.44 to 5.5 ng/mL, and Cat content from 1313 to 2424 ng/mL compared to the respective control levels (Figure 6 and Supplemental Table S11).

(A) and (B) Protein content of Sod and Cat after treatment with PD (0.8, 4, and 20 µM) for 24 h.
Conclusions and Discussions
The immune system is usually activated by innate immune cells (macrophages and neutrophils). Numerous environmental toxins and chemicals have been documented to be immunotoxic to macrophages.18,19 According to previous reports, fermented P grandiflorus extract did not induce toxicity at 0.8−500 µg/mL concentration. It promoted cell proliferation at high concentrations of 100 to 500 µg/mL. 5 In accordance with earlier findings, our investigation demonstrated that macrophages treated with 3 µM PD did not exhibit toxicity. However, we found that cell viability significantly decreased at 20 µM PD, and LDH activity, commonly used as an indicator of cell death, 20 also increased. Cell viability reduction is commonly linked to apoptosis. Mitochondria are the main site of ROS production, with excessive ROS causing mitochondrial dysfunction and activating the mitochondria-mediated apoptosis pathway. 21 The Bcl-2 family is the core regulator of this pathway, controlling the permeabilization of the mitochondrial outer membrane and releasing proapoptotic factors to activate the caspase cascade. 22 Furthermore, p53 is a crucial transcription factor that activates the transcription of hundreds of genes, including Bcl-2 and caspase family genes, which are involved in apoptosis.22,23 In this study, 20 µM PD increased the level of ROS in RAW 264.7 cells. Meanwhile, the mRNA expression of p53 increased, leading to an increase in the expression of the downstream apoptotic genes Bax and caspase-3. This indicated that PD activated the oxidative stress-induced apoptotic pathway in cells. Research indicated that the activity of Cat and Gpx increased in larvae exposed to 2,4-D herbicide, which may endanger fish growth. 24 Ceramide increased ROS levels by downregulating the Sod1 protein and upregulating the Sod2 protein. 25 This study demonstrated that 20 µM PD increased the levels of oxidative stress-related genes and proteins, including Sod1, Sod2, Cat, Gpx1, and Gpx2. Additionally, it also elevated the expression of Gsta1, which is considered a marker of serious hepatocyte injury. 26 These oxidative stress-related cytokines further indicate that PD (20 µM)-induced cell apoptosis was due to the oxidative stress-activated mitochondrial apoptosis pathway.
When the immune system is compromised, activated macrophages release IL-6 to combat pathogenic microorganisms and stimulate other immune cells to respond to pathogens. 27 However, the excessive and unregulated release of inflammatory cytokines by immune cells is the cause of many diseases. 28 Even though members of the interleukin family are frequently immune activators, unregulated release can be detrimental to the host. 29 Platycodon grandifloras and polysaccharides exert immunostimulatory activity, stimulating macrophages to secrete TNF-α, IL-6, and other inflammatory cytokines.5,7 Notably, oxidative stress can also damage cellular structures and activate various transcription factors, further promoting the expression of growth factors and inflammatory cytokines.30,31 In our study, PD (20 µM) caused the uncontrolled release of many inflammatory cytokines, including IL-1β and TNF-α. We also found that PD (0.8, 4, and 20 µM) downregulated the expression of selected cytokines upon LPS stimulation. Research has shown that immunotoxic chemicals possess the ability to dysregulate expression levels of cytokines, thus impairing immune function. 18
The data presented in this study demonstrated that PD (20 µM) is immunotoxic to macrophages; it significantly affected the viability and dysregulated the expression of cytokines in RAW 264.7 macrophages, possibly through oxidative-mediated apoptosis (Figure 7). Understanding the immunotoxicity of PD on RAW 264.7 cells will help reduce the adverse reactions of its biological application and improve its therapeutic effects. The impact of PD on other immune cells should be investigated further in future studies. In addition, PD has traits of poor permeability and hemolysis; therefore, in vivo experiments can be performed to gather information regarding the effect of PD on the immune system. This research provides a valuable reference for developing and implementing PD, which is widely employed in food and health goods.

Platycodin D (PD) exhibited immunotoxicity to RAW 264.7 cells, with possible mechanisms including oxidative stress-mediated apoptosis resulting in activation of the mitochondrial apoptosis pathway and release of inflammatory cytokines.
Materials and Methods
Chemicals and Cell Culture
PD, purchased from Shanghai Yuanye Biological Technology Company, was dissolved in phosphate-buffered saline (PBS) to a stock concentration of 10 mM and stored at −20 °C. LPS from Escherichia coli O55: B5 (Sigma-Aldrich) was dissolved in PBS to a stock concentration of 1 mg/mL and stored at −20 °C. Dimethyl sulfoxide (DMSO) was purchased from Sinopharm Chemical Reagent Company, MTT cell proliferation and cytotoxicity assay kits and LDH activity detection kits from Beijing Solarbio Science & Technology Company, ROS assay kits and the BeyoClick™ EdU Cell Proliferation Kit with Alexa Fluor 488 from Beyotime Biotechnology, enzyme-linked immunosorbent assay (ELISA) kits for IL-1β, Cat and Sod from Jianglai Biological, and the SPARKeasy Improved Tissue/Cell RNA Kit, SPARKscript II RT Plus Kit, and 2x SYBR Green qPCR Mix from Sparkjade. Primers were synthesized by WeiLai Biotechnology Co., Ltd.
RAW 264.7 cells were purchased from the National Collection of Authenticated Cell Cultures. They were cultured in mixed media (DMEM, 10% fetal bovine serum, penicillin, and streptomycin mixed solution) at 37 °C in 5% CO2.
Cell Proliferation Assay
RAW 264.7 cells in the logarithmic growth phase were cultured at a density of 5 × 103/well in 96-well plates(Corning). After 12 h of culture, the cells were treated with various concentrations of PD (0.16-200 µM) for either 24 h or 48 h. After removing the culture medium, MTT solution was added to each well, and the cells were incubated at 37 °C for 4 h. The formazan crystals were then solubilized with 110 µL of DMSO and shaken for 10 min. Finally, the optical density (OD) value of each well was measured at 490 nm using a microplate reader (Bio-TEK). The relative cell viability was calculated as follows: OD (PD group)/OD (control group) x 100%. In addition, RAW 264.7 cells were incubated at the same density in 96-well plates. After 12 h of culture, the cells were pretreated with various concentrations of PD (0.8, 4, 20, and 100 µM) for 0.5 h and then cotreated with PD and LPS (1 µg/mL) for 24 h. The cell viability assay was performed as described previously.
In the EdU proliferation assay, RAW 264.7 cells in the logarithmic growth phase were cultured at 4 × 104/well in 24-well plates (Corning). After 12 h of culture, the cells were treated with various concentrations of PD (0.8, 4, 20, and 100 µM) for an additional 24 h. The cells were then treated according to the manufacturer's instructions and assessed by fluorescence microscopy (Bio-TEK).
LDH Activity Assay
RAW 264.7 cells were incubated at a density of 5 × 105 cells/well in 6-well plates (Corning). After 12 h of culture, the cells were exposed to various concentrations of PD (0.8, 4, 20, and 100 µM). According to the manufacturer's instructions for the LDH activity detection kit, cells were collected and added to the extraction solution (500 million cells/1 mL). The supernatant was harvested by centrifugation at 8000 × g for 10 min at 4 °C. The LDH in the supernatant catalyzed the oxidation of lactic acid to pyruvic acid, which then reacted with 2,4-dinitrophenylhydrazine to yield a brown product. The LDH activity was then measured at 450 nm.
Intracellular ROS Measurement
RAW 264.7 cells were cultured in 6-well plates and exposed to various concentrations of PD (0.8, 4, and 20 µM) for 24 h. Following the kit's instructions, the cell culture supernatant was removed, and 1 mL of 10 µM DCFH-DA was added to each well before incubating the plate for 20 min at 37 °C. The cells were resuspended in PBS after being washed 3 times with serum-free cell culture media. Fluorescence was observed using a flow cytometer (BECKMAN COULTER) and fluorescence microscopy.
ELISA Measurement
RAW 264.7 cells were exposed to PD (0.8, 4, and 20 µM) for 24 h. According to the manufacturer's instructions, the supernatants were collected and centrifuged at 1000 × g for 20 min to measure the content of IL-1β. The cells were collected and washed 3 times with PBS and then lysed by ultrasonication at 200 W. Cell debris was removed by centrifugation at 1500 × g for 10 min, and the supernatant was collected and analyzed to detect the contents of Sod and Cat.
RT-qPCR Analysis
RAW 264.7 cells were cultured in 6-well plates and exposed to various concentrations of PD (0.8, 4, and 20 µM) for 24 h to measure the mRNA expression of apoptosis- and oxidative stress-related genes. RAW 264.7 cells were cotreated with PD (0.8, 4, and 20 µM PD) and LPS (1 µg/mL) for 24 h to measure the mRNA expression of inflammatory cytokines. Total RNA was extracted and purified according to the manufacturer's instructions using the SPARKeasy Improved Tissue/Cell RNA Kit. cDNA was then synthesized using the SPARKscript III RT Plus Kit. The cDNA was subjected to RT-qPCR using the 2 × SYBR Green qPCR Mix kit. Data were collected using qTower3 G (Analytik Jena AG). The β-actin gene was used as the internal reference. The primer sequences are given in Supplemental Table S11.
Statistical Analysis
All experiments were performed in triplicate. Data were analyzed using Prism version 8.0.1 software (GraphPad) and are presented as the mean. The differences between groups were evaluated by the unpaired t test, with the significance level set at #P <0.05 and ##P <0.01 or *P <0.05 and **P <0.01.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221150366 - Supplemental material for Platycodin D-Induced Immunotoxicity in RAW 264.7 Macrophages via Oxidative Stress-Mediated Apoptosis
Supplemental material, sj-docx-1-npx-10.1177_1934578X221150366 for Platycodin D-Induced Immunotoxicity in RAW 264.7 Macrophages via Oxidative Stress-Mediated Apoptosis by Xinying Du, Xinhai Cui, Xiaowen Sun, Hui Li, Kuo Xu and Xianjun Fu in Natural Product Communications
Footnotes
Authors’ Contributions
This work was written by Xinying Du and Xinhai Cui, and they contributed equally to this work. Kuo Xu and Xianjun Fu approved the final version of the manuscript. All authors have reviewed and agreed to the published version of the manuscript.
Availability of Data and Materials
Supplemental data for this article can be found online at http://///, Supplemental Table S1: The relative cell viability of RAW 264.7 cells after treatment with PD (1.56−200 µM) for 24 h or 48 h; Supplemental Table S2: The relative cell viability of RAW 264.7 cells after treatment with PD (0.16−100 µM) for 24 h or 48 h; Supplemental Table S3: Relative LDH activity in RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) for 24 h; Supplemental Table S4: The relative mRNA expression of Bax, p53, and caspase-3 in RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) for 24 h; Supplemental Table S5: The relative cell viability of RAW 264.7 cells after treatment with PD (0.8, 4, 20, and 100 µM) and LPS (1 µg/mL) for 24 h; Supplemental Table S6: The protein content of IL-1β in RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) for 24 h; Supplemental Table S7: The relative mRNA expression of inflammatory cytokines in RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) for 24 h; Supplemental Table S8: The relative mRNA expression of inflammatory cytokines in RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) and LPS (1 µg/mL) for 24 h; Supplemental Table S9: The relative fluorescent intensity of RAW 264.7 cells after treatment with PD (0.8, 4, and 20 µM) for 24 h; Supplemental Table S10: The relative mRNA expression of Sod1, Sod2, Gpx1, Gpx2, Cat, Gsta1 after treatment with PD (0.8, 4 and 20 µM) for 24 h; Supplemental Table S11: The protein content of Sod and Cat after treatment with PD (0.8, 4 and 20 µM) for 24 h.; ![]() : The primers sequences used in the RT-PCR.
: The primers sequences used in the RT-PCR.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Medical College of Yangzhou University, Jiangsu Province, China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Shandong Provincial Natural Science Foundation (NOs. 2021CXGC010510, ZR2022YQ71, and ZR2022LZY026), Shandong Province Key Discipline Construction Project of Traditional Chinese Medicine (Marine Traditional Chinese Medicine), Shandong Province Traditional Chinese Medicine High-level Talent Cultivation Project, and Youth Innovation Team of Shandong University of Traditional Chinese Medicine.
Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
