Abstract
Gouty arthritis (GA) is an inflammatory disease caused by the deposition of monosodium urate in the synovial membrane and cartilage due to a high concentration of uric acid in the blood. Dioscorea nipponica Makino is widely used in the clinic to treat GA. Total saponins are its main components and showed an anti-inflammatory effect on GA in a previous study. The mitogen-activated protein kinase (MAPK)-peroxisome proliferator-activated receptor γ (PPARγ) signaling pathway plays a key role during the onset of GA; however, little is known about its potential mechanism. Based on in vitro experiments, this study aims to determine the mechanism of total saponins from Dioscorea nipponica Makino (TDN) in treating GA by regulating the MAPK-PPARγ signaling pathway. Fibroblast-like synoviocytes were divided into 3 groups: the model group, which was given 10 µg/L IL-1β to induce proliferation; TDN group (10 µg/L IL-1β + 100 µg/L TDN); and indomethacin group (10 µg/L IL-1β + 100 µg/L indomethacin). Seventy-two hours after treatment, the real-time PCR method was used to detect the mRNA expression levels of extracellular signal regulated kinase 1/2 (ERK1/2), p-38, c-Jun N-terminal kinase (JNK), IKKα, c-Jun, MAPK phosphatase (MKP), vascular cell adhesion molecule 1 (VCAM1), intercellular adhesion molecule 1 (ICAM1), C-X-C motif chemokine ligand 1 (CXCL1), PPARγ, and Adipor2. The Western blot method was used to detect the protein expression of ERK1/2, p-ERK1/2, p-38, p-P38, JNK, p-JNK, IKKα, p-IKKα, c-Jun, p-c-Jun, MKP, p-MKP, VCAM1, ICAM1, CXCL1, PPARγ, and Adipor2. Compared with the model group, the TDN group displayed significantly increased mRNA expression of c-Jun, MKP, CXCL1, PPARγ, and Adipor2 and significantly decreased mRNA expression of ICAM1. Compared with the model group, the TDN group exhibited significantly increased protein expression of MKP and significantly decreased protein expression of p-ERK1/2, p-38, p-JNK, p-IKKα, p-c-Jun, VCAM1, ICAM1, and PPARγ. Our results indicated that TDN could treat GA by influencing the MAPK-PPARγ signaling pathway.

Gouty arthritis (GA) is a kind of rheumatoid lesion that is caused by the deposition of uric acid. 1 It will develop to tophi, nephrolithiasis, and urate nephropathy. 2 Its clinical manifestations include obvious joint swelling with heat and pain. If it is not treated in a timely manner, joints will deform and the activities of patients will be restrained. It will bring immense pain and influence quality of life. Thus, it is sensible to explore Chinese medicine to treat GA since it has little adverse effects.
Mitogen-activated protein kinases (MAPKs) facilitate signaling pathways that are widely distributed in all kinds of animal cells. The pathways are composed of a series of Ser and Thr protein kinases that are activated in a cascade. By successively activating protein kinases, MAPKs amplify signals outside the cells and transduce them into the cell nucleus, thus controlling cell proliferation, development, apoptosis, and gene expression in the cells. 3,4 Their downstream molecules include protein kinases, phosphatases, and nuclear transcription factors. Mitogen-activated protein kinases that participate in GA attacks are mainly extracellular signal regulated kinase 1/2 (ERK1/2), c-Jun N-terminal kinase (JNK), and p-38 mitogen-activated protein kinase. 5 When ERK1/2 are activated, they act on nuclear factor kappa B (NF-κB) and regulate target gene expression. The inhibitor of nuclear factor kappa-B kinase (IKK) complex is an inhibitor of NF-κB kinase. Its activation leads to phosphorylation of IκB and activation of NF-κB. IKK is composed of IKKα, IKKβ, and IKKγ. IKKα is the catalytic component. 6 c-Jun N-terminal kinase acts by phosphorylating the transcription factor c-Jun, while MAPK phosphatase (MKP) has a negative effect on this signaling pathway.
Monocyte and neutrophil granulocytes are in the peripheral blood under normal conditions. When monosodium urate (MSU) induces these cells, some adhesion molecules, such as vascular cell adhesion molecule 1 (VCAM-1), can activate them, and these activated cells will enter tissues around the vasculature after adhering to vascular endothelial cells under the action of chemokines, such as C-X-C motif chemokine ligand 1 (CXCL1). The adhesion of neutrophils to vascular cells, translocation across endothelial cells, and migration toward inflammatory sites is a basic feature of acute inflammation. 7 Translocation of inflammatory cells across the membrane requires intercellular adhesion molecule 1 (ICAM1).
Peroxisome proliferator-activated receptors (PPARs) belong to the nuclear factor family, which is activated by ligands. There are 3 types: PPARα, PPARβ, and PPARγ. Peroxisome proliferator-activated receptors can bind to retinoid X receptor (RXR), form a dimer after binding to ligands, and then recruit a series of cofactors, thus regulating the transcription of target genes. 8,9 The PPARγ signaling pathway is related to inflammation, and it can competitively inhibit the action of NF-κB. Adiponectin is a multifunctional hormone protein that is secreted by adipocytes. Its receptors include AdipoR1, AdipoR2, and T-cadherin. Agonists of PPARγ can promote increased expression of ADP and its receptor AdipoR2, thus making AdipoR2 a new target to treat GA. 10
Dioscorea nipponica Makino has been recorded to relax muscles and tendons as well as invigorate the blood circulation and anti-inflammation. It is widely used in the clinic to treat GA; however, its mechanism needs to be further elucidated. Our previous study showed that total saponins from D. nipponica Makino (TDN) could regulate the TLR2/4-IL1R signaling pathway and decrease the expression of IL-1β and other inflammatory factors to treat GA. 11 This study was designed to reveal whether TDN treats GA by influencing the MAPK-PPARγ signaling pathway, thus regulating the expression of vascular cell adhesion molecule 1 (VCAM1) and ICAM1. The mechanism of the MAPK-PPARγ signaling pathway during the pathogenesis of GA is shown in Figure 1.
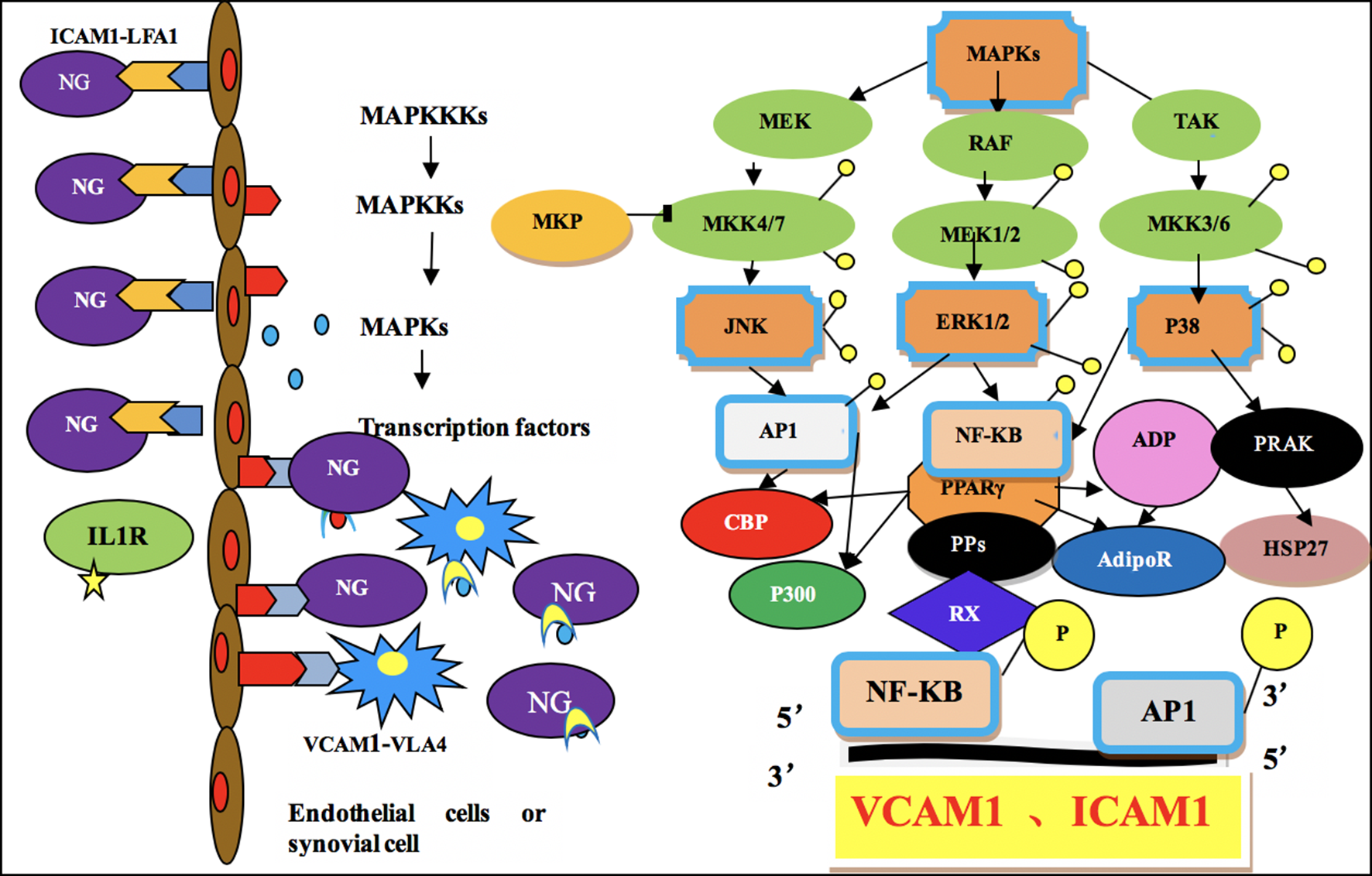
Mechanism of the mitogen-activated protein kinase-peroxisome proliferator-activated receptor γ signaling pathway during the pathogenesis of gouty arthritis.
In this study, we tested the mRNA and protein levels of the key factors in this signaling pathway by real-time PCR and Western blot methods, respectively. Our results showed that TDN could influence this signaling pathway to treat GA.
Experimental Approach
Reagents
Total saponins from D. nipponica Makino crude drugs were purchased from the Harbin World Electuary Factory. Indomethacin (purity >99%) was purchased from Sigma (United States). An MTT kit was purchased from Beyotime Biotechnology (China). TRIzol was purchased from Invitrogen (United States). A RevertAid H minus first-strand cDNA synthesis kit, Deoxyribonuclease I, and RiboLock ribonuclease inhibitor were all purchased from Fermentas (United States). Synergy brands (SYBR ) Green PCR master mix was purchased from ABI (United States).Radio-immunoprecipitation assay( RIPA )protein extraction and BCA protein quantification kits were purchased from Beijing Kangwei Century Biotechnology. Phenylmethanesulfonyl fluoride (PMSF) mother liquor and BSA were purchased from American Amresco. The nitrocellulose filter ( NC) membrane was purchased from American Millipore.
Plant Material and Extraction
Dioscorea nipponica Makino was purchased from the Heilongjiang Province Drug Company. Voucher specimens (hlj-201104) of the herb were authenticated by Prof. Ke Fu, Institute of Traditional Chinese Medicine. One gram of crude drug was extracted 3 times with 6 mL of 50% ethanol for 1.5 hours and separated and purified by D-101 microporous adsorption resin (polarity: nonpolar, particle diameter: 0.3-1.25 mm, surface area: 480-520 m2/g, average pore diameter: 25-28 nm, and moisture content: 60%-75%). The purification process was as follows: concentration of the sample solution, 500 mg/mL; loading quantities, 1 g sample/2 g resin; adsorption flow rate, 2 BV/h (bed volume/hour); eluent, 50% ethanol; eluent volume, 14 BV; and elution flow rate, 2 BV/h. The ethanol phase was evaporated under a vacuum and oven dried at 60°C to obtain the extracts. The drug extraction ratio was 4.97% (w/w).
Determination of the Content of TDN
The content of TDN was calculated according to the content of dioscin. The calculated content of TDN in the extract was approximately 55.9%. 12
UPLC/MS Analysis
To standardize the herbal extract chemically, ultra performance liquid chromatography/mass spectrometry(UPLC/MS) analysis was performed. Three major compounds in the TDN extract were identified. They were dioscin, protodioscin, and pseudo protodioscin. 12
Animals
Approximately 10 male Wistar rats (200 ± 20 g) were purchased from Vital River Laboratory Animal Technology Co., Ltd., China. They were allowed at least 1 week to adapt to their environment before being used for experiments. Animals were housed 5 per cage (600 mm ×450 × 280 mm) under a normal 12 hours/12 hours light/dark schedule with the lights turned on at 07:00
Preparation of Fibroblast-like Synoviocytes (FLSs)
Synovial membrane samples were obtained from the knee joint of rats. Fibroblast-like synoviocytes (FLSs) were prepared according to Lu et al . IL-1β (10 µg/L) was used to induce the proliferation of FLSs. 13
Cytotoxicity Assay
According to Lu et al , compared with that in the control group (P < 0.05), the cell viability in 10, 100, and 1000 µg/L TDN groups was decreased. The cell viability in the 0.01, 0.1, and 1 µg/L TDN groups was also less than that in the control group, but there was no significant difference. Hence, 100 µg/L TDN was chosen for this experiment.
A cytotoxicity assay was used to also determine the concentration of indomethacin. Indomethacin concentrations of 0.01, 0.1, 1, 10, 100, and 1000 µg/L were used. Synovial cells were seeded in a 96-well plate (10 000 cells/well). Incubation was carried out at 37°C in an atmosphere of 5% CO2 for 24 hours. Various concentrations of indomethacin were added to each well and then incubated at 37°C in a 5% CO2 incubator for 72 hours. MTT was added to each well and further incubated for 4 hours. The MTT solution was aspirated off, and the cell crystals were dissolved using 150 mL dimethyl sulfoxide. Finally, color intensity was measured using a Tecan Infinite M200 microplate reader (Tecan Inc., Maennedorf, Switzerland) at 570 nm. A concentration of 100 µg/L indomethacin was ultimately chosen for this experiment.
Molecular Analysis of the MAPK-PPARγ Pathway
Real-time PCR of ERK1/2, P-38, JNK, IKKα, c-Jun, MKP, VCAM1, ICAM1, CXCL1, PPARγ, and Adipor2
RNA was extracted using the classic TRIzol reagent method. Reverse transcription was performed using reverse transcriptase and 1 µg RNA to synthesize the cDNA. PCR amplification was carried out using gene-specific PCR primers synthesized by Kingsley Biotechnology (China). The sequences of the gene-specific PCR primers and the lengths of products as well as the melting temperatrues (TM) are summarized in Table 1.Actin beta (ACTB) was used as a reference gene. The amplification was performed in 25 µL reactions containing 1 µL cDNA, 100 nM forward and reverse primers, 12.5 µL SYBR Premix Ex TaqTM, and sterilized ddH2O to 25 µL. Each reaction was carried out for 40 cycles of denaturation at 95°C for 5 seconds and annealing.
Summary of Gene-Specific Real-Time PCR Primer Sequences.

CXCL1, C-X-C motif chemokine ligand 1; ERK1, extracellular signal regulated kinase 1; ERK2, extracellular signal regulated kinase 2; ICAM1, intercellular adhesion molecule 1; JNK, c-Jun N-terminal kinase; MKP, MAPK phosphatase; PPARγ, peroxisome proliferator-activated receptor γ; VCAM1, vascular cell adhesion molecule 1.
Western Blot Analysis of ERK1/2, p-ERK1/2, P-38, p-P38, JNK, p-JNK, IKKα, p-IKKα, c-Jun, p-c-Jun, MKP, p-MKP, VCAM1, ICAM1, CXCL1, PPARγ, and Adipor2
RIPA protein extraction reagent was precooled, and a protease inhibitor was added. PMSF mother liquor (0.01 M) was added before protein extraction, and the final concentration was 1 mM. Synovial tissues were weighed. The amount of RIPA added was 9 times the weight of the synovial tissue. A Fluka electric tissue homogenizer was used to homogenize the tissues at a speed of 15 000 rpm every 10 seconds for 10 seconds, and this procedure was repeated 2 times. The centrifuge tube was placed into a mixture of ice and water to lower the temperature after homogenization. After homogenization, the mixture was incubated for 20 minutes on ice and then centrifuged at 4°C at a speed of 13 000×g for 20 minutes. After centrifugation, the supernatant was obtained and preserved for further testing. The bicinchoninic acid(BCA) solution was prepared with a ratio of solution A to solution B of 50 to 1. All of the BSA standard substances were diluted. The samples were diluted with PBS. The ratio of the sample to the BCA solution was 1 to 8. After mixing, the mixture was incubated at 37°C for 30 minutes. An enzyme assay instrument was used to test the OD value at 570 nm. The concentrations of each protein were calculated, and RIPA buffer was used to adjust them. After addition of 5× reducing sample buffer, the final concentration of the sample was 4.0 mg/mL. The exact concentrations were confirmed according to different samples. The sample was boiled for 5 minutes to denature the proteins. Separation gels were made at concentrations of 12% and 8% according to the molecular weights of the proteins of interest. The concentration of the spacer gel was 5%. The loading quantity of the sample was 20 µg per well. The proteins were electrophoretically transferred for 1 hour to an nitrocellulose filter (NC )membrane (0.45 µm), which was completely immersed into 3% BSA-TBST, with shaking for 30 minutes at room temperature.Bovine serum albumin (BSA)-tris buffer saline tween (TBST) (3%) was used to dilute primary antibodies and incubated for 10 minutes at room temperature and then at 4°C overnight. The specific antibodies used were anti-ERK1/2 (1: 5000), anti-p-ERK1/2 (1: 1000), anti-p-38 (1:2000), anti-p-P38 (1:500), anti-JNK (1:2500), anti-p-JNK (1:2000), anti-IKKα (1:2000), anti-p-IKKα (1:2000), anti-c-Jun (1:10 000), anti-p-c-Jun (1:2500), anti-MKP (1:500), anti-p-MKP (1:1000), anti-VCAM1 (1:5000), anti-ICAM1 (1:2000), anti-PPARγ (1: 500), and anti-Adipor2 (1:2000) (Table 2). Immunoreactive bands were detected using an horseradish peroxidase (HRP)-conjugated goat anti-mouse or goat anti-rabbit IgG as the secondary antibody that was 1:4000 diluted in TBST (Table 2). The samples were shaken gently at room temperature for 40 minutes. TBST was used to wash the membrane 6 times for 3 minutes each.Electrochemi luminescence ECL was added to the membrane to react for 3 to 5 minutes. Film exposure occurred for 10 seconds to 5 minutes. The film was developed for 2 minutes and fixed. The contents of the target proteins were analyzed via densitometry using Total Lab Quant software (Image Quant) and normalized by the respective blotting signal from β-actin.
Summary of Antibodies Used in Western Blotting Analysis.

CXCL1, C-X-C motif chemokine ligand 1; ERK1/2, extracellular signal regulated kinase 1/2; ICAM1, intercellular adhesion molecule 1; JNK, c-Jun N-terminal kinase; MKP, MAPK phosphatase; PPARγ, peroxisome proliferator-activated receptor γ; VCAM1, vascular cell adhesion molecule 1.
Statistical Analysis
All the data were expressed as the mean ± standard error of the mean, and the statistical analysis was performed using independent sample t-tests to determine the levels of significance. A value of P < 0.05 was considered statistically significant.
Results
Cytotoxicity of Indomethacin
Compared with that in the control group (P < 0.05), the cell viability in the 0.1, 1, 10, 100, and 1000 µg/L indomethacin groups was decreased. The cell viability in the 0.01 µg/L indomethacin group was also less than that in the control group, but there was no significant difference (Figure 2).

Cytotoxicity of indomethacin. Synovial cells were seeded in a 96-well plate (10 000 cells/well). Cells were treated with various concentrations of total saponins from Dioscorea nipponica Makino for 72 hours. MTT was added to each well and incubated for an additional 4 hours. The MTT solution was aspirated, and the cell crystals were dissolved in 150 µL dimethyl sulfoxide. Finally, color intensity was measured using a Tecan Infinite M200 microplate reader at 570 nm. The data represent the mean ± standard error of the mean of each group (n = 10). *P < 0.05 vs control group (independent sample t-test).
Effects of TDN on mRNA Expression of ERK1/2, P-38, JNK, IKKα, c-Jun, MKP, PPARγ, VCAM1, ICAM1, CXCL1, and Adipor2
As shown in Figures 3(f) and 4(a-f), TDN significantly increased the mRNA expression of c-Jun, MKP, PPARγ, ICAM1, CXCL1, and Adipor2 compared with that in the control group (P < 0.05, P < 0.01, P < 0.01, P < 0.01, P < 0.01, and P < 0.01, respectively). Indomethacin also significantly increased the mRNA expression of c-Jun, MKP, PPARγ, ICAM1, CXCL1, and Adipor2 (P < 0.05, P < 0.01, P < 0.01, P < 0.01, P < 0.01, and P < 0.01, respectively). For MKP and CXCL1, there were significant differences between the TDN and indomethacin groups (P < 0.05 and P < 0.01, respectively). However, for VCAM1 and CXCL1, there was a significant difference between the TDN and indomethacin groups (P < 0.05 and P < 0.01, respectively).

Effects of total saponins from Dioscorea nipponica Makino and indomethacin on the mRNA levels of extracellular signal regulated kinase 1/2, Jun N-terminal kinase, p-38, IKKα, and c-Jun. The data represent the values of the mean ± standard error of the mean of each group. *P < 0.05 and **P < 0.01 vs control group (independent sample t-test).

Effects of total saponins from Dioscorea nipponica Makino and indomethacin on mRNA levels of MAPK phosphatase, peroxisome proliferator-activated receptor γ, vascular cell adhesion molecule 1, intercellular adhesion molecule 1, C-X-C motif chemokine ligand 1, and Adipor2. The data represent the values of the mean ± standard error of the mean of each group. *P < 0.05 and **P < 0.01 vs control group; # P < 0.05 and ## P < 0.01 vs total saponins from Dioscorea nipponica Makino group (independent sample t-test).
Effects of TDN on the Protein Expression of ERK1/2, p-ERK1/2, p-38, p-P38, JNK, p-JNK, IKKα, p-IKKα, c-Jun, p-c-Jun, MKP, p-MKP, VCAM1, ICAM1, CXCL1, PPARγ, and Adipor2
As shown in Tables 3 -5, and Figures 5(b,d,f) -9, 7, 8, compared with the control group, the TDN group exhibited significantly decreased protein expressions of p-ERK1/2, p-JNK, p-P-38, p-IKKα and p-c-Jun (P < 0.01, P < 0.01, P < 0.05, P < 0.01, and P < 0.01, respectively). Indomethacin also significantly decreased the protein expression of p-ERK1/2, p-JNK, p-IKKα, and p-c-Jun (P < 0.01, P < 0.01, P < 0.01, and P < 0.01, respectively).
Protein Expression Change of Extracellular Signal Regulated Kinase 1/2, p-ERK1/2, Jun N-Terminal Kinase, and p-JNK.
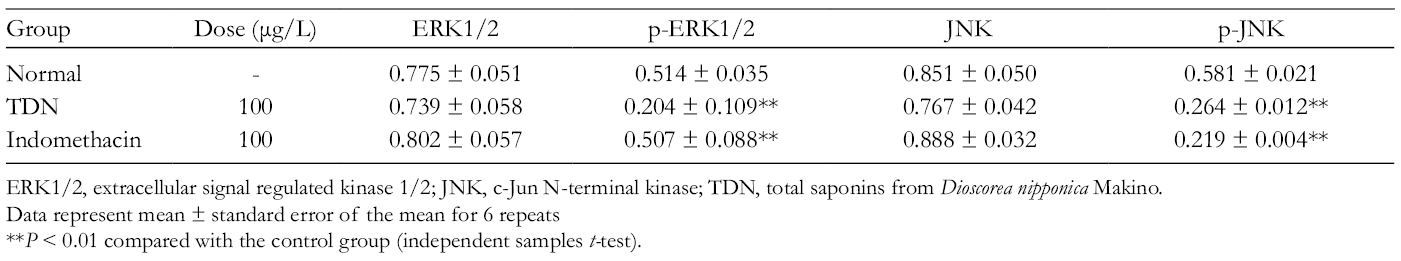
ERK1/2, extracellular signal regulated kinase 1/2; JNK, c-Jun N-terminal kinase; TDN, total saponins from Dioscorea nipponica Makino.
Data represent mean ± standard error of the mean for 6 repeats
**P < 0.01 compared with the control group (independent samples t-test).
Protein Expression Change of P38, p-P38, IKKα, and p-IKKα.
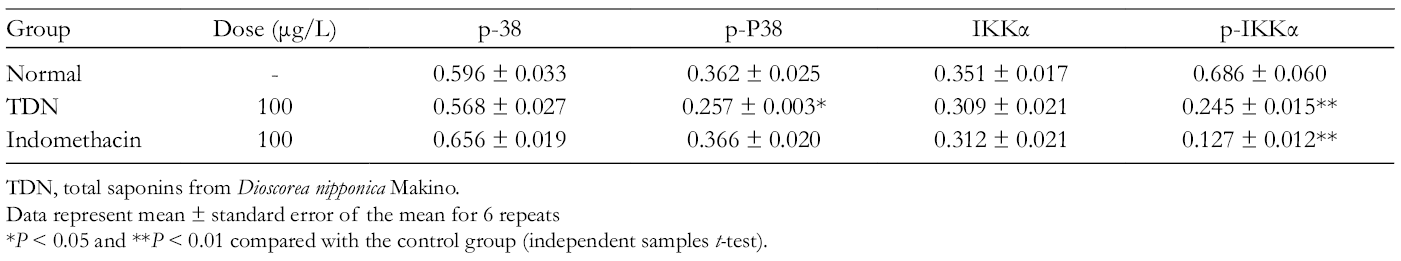
TDN, total saponins from Dioscorea nipponica Makino.
Data represent mean ± standard error of the mean for 6 repeats
*P < 0.05 and **P < 0.01 compared with the control group (independent samples t-test).
Protein Expression Change of c-Jun, p-c-Jun, MAPK Phosphatase, and p-MKP.

MKP, MAPK phosphatase; TDN, total saponins from Dioscorea nipponica Makino.
Data represent mean ± standard error of the mean for 6 repeats .
**P < 0.01 compared with the control group (independent samples t-test).

Effects of total saponins from Dioscorea nipponica Makino and indomethacin on the protein levels of extracellular signal regulated kinase 1/2, p-ERK1/2, Jun N-terminal kinase, p-JNK, p-38, and p-P38. The data represent the values of the mean ± standard error of the mean of each group. *P < 0.05 and **P < 0.01 vs control group (independent sample t-test).

Effects of total saponins from Dioscorea nipponica Makino and indomethacin on the protein levels of IKKα, p-IKKα, c-Jun, p-c-Jun, MAPK phosphatase, and p-MKP. The data represent the values of the mean ± standard error of the mean of each group. *P < 0.05 and **P < 0.01 vs control group (independent sample t-test).
Effects of total saponins from Dioscorea nipponica Makino and indomethacin on the protein levels of extracellular signal regulated kinase 1/2, p-ERK1/2, Jun N-terminal kinase, p-JNK, p-38, p-P38, IKKα, and p-IKKα. All the groups were induced with IL-1 (10 µg/L), while both the total saponins from Dioscorea nipponica Makino (100 µg/L) and indomethacin (100 µg/L) groups were incubated for 72 hours.

Effects of total saponins from Dioscorea nipponica Makino and indomethacin on the protein levels of c-Jun, p-c-Jun, MAPK phosphatase, p-MKP, vascular cell adhesion molecule 1, intercellular adhesion molecule 1, C-X-C motif chemokine ligand 1, peroxisome proliferator-activated receptor γ, and Adipor2. All the groups were induced with IL-1β (10 µg/L), while both the total saponins from Dioscorea nipponica Makino (100 µg/L) and indomethacin (100 µg/L) groups were incubated for 72 hours.
As shown in Table 5 and Figures 6 (e,f),8, compared with the control group, the TDN group displayed significantly increased MKP protein expression (P < 0.01), and indomethacin significantly increased MKP protein expression (P < 0.01).
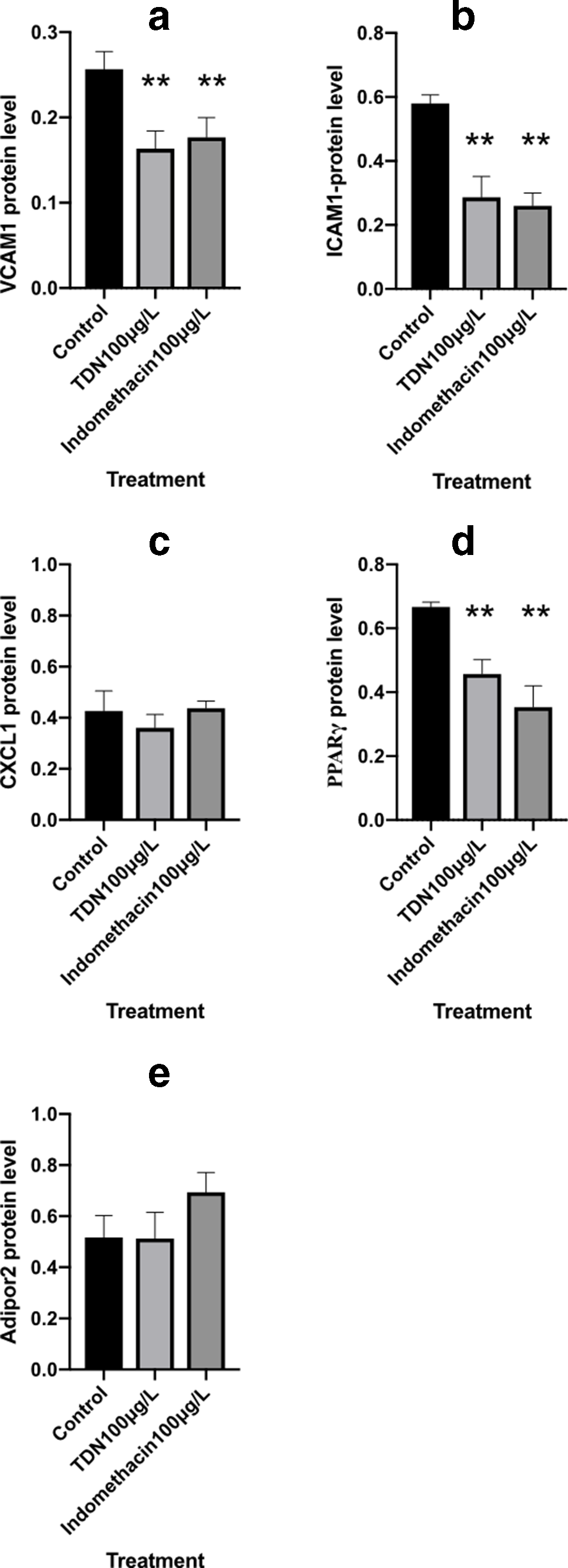
As shown in Table 6 and Figures 8 (a,b,d),9, TDN significantly decreased the protein expression of VCAM1, ICAM1, and PPARγ compared with that in the control group (P < 0.01, P < 0.01, and P < 0.01, respectively). Indomethacin also significantly decreased the protein expression of VCAM1, ICAM1, and PPARγ (P < 0.01, P < 0.01, and P < 0.01, respectively).
Protein Expression Change of Vascular Cell Adhesion Molecule 1, Intercellular Adhesion Molecule 1, C-X-C Motif Chemokine Ligand 1, Peroxisome Proliferator-Activated Receptor γ, and Adipor2.

CXCL1, C-X-C motif chemokine ligand 1; ICAM1, intercellular adhesion molecule 1; PPARγ, peroxisome proliferator-activated receptor γ; TDN, total saponins from Dioscorea nipponica Makino; VCAM1, vascular cell adhesion molecule 1.
Data represent mean ± standard error of the mean for 6 repeats.
**P < 0.01 compared with the control group (independent samples t-test).

Effects of total saponins from Dioscorea nipponica Makino and indomethacin on the protein levels of vascular cell adhesion molecule 1, intercellular adhesion molecule 1, C-X-C motif chemokine ligand 1, peroxisome proliferator-activated receptor γ, and Adipor2. The data represent the values of the mean ± standard error of the mean of each group. *P < 0.05 and **P < 0.01 vs control group (independent sample t-test).
Discussion
Gout is a prime example of uric acid disturbance. It is the most well understood and described type of arthritis. Newer drugs in the pharmacological armamentarium are proving successful and can supplement older ones. 14 The MAPK-PPARγ signaling pathway plays an important role during the pathogenesis of GA. However, drugs targeting this pathway have not been explored.
Dioscorea nipponica Makino is a kind of traditional Chinese medicine that is widely used in the clinic to treat GA. 15 Its mechanism to treat this disease needs to be clarified further to employ it as a new drug. Total saponins from Dioscorea nipponica Makino are the principal components that show antiinflammatory effects. 13,16 Our previous study showed that the content of TDN was 55%, and the UPLC-MS method was used to verify that its main components were dioscin, protodioscin, and pseudo protodioscin. The MTT method was also used to test the active component of TDN. 12 This study aimed to reveal the mechanism of TDN in treating GA by influencing the MAPK-PPARγ signaling pathway.
Total saponins from Dioscorea nipponica Makino significantly increased the mRNA expression of c-Jun, MKP, PPARγ, ICAM1, CXCL1, and Adipor2. It also significantly increased the protein expression of MKP and significantly decreased the protein expression of p-ERK1/2, p-JNK, p-P-38, p-IKKα, and p-c-Jun. However, it did not show obvious effects on either the mRNA or protein expression of ERK1/2, JNK, p-38, IKKα, or VCAM1 or the protein expression of CXCL1 and Adipor2. This finding was an interesting result to be discussed below.
The ERK1/ERK2, JNK, and p-38 cascades have overlapping and cross-regulatory effects but differ in their predisposition to activation by specific receptor-mediated and nonreceptor-mediated stimuli. Each of these 3 MAPK cascades utilizes distinct dual-specificity MAPK kinases (MEK) that phosphorylate the TEY domain on each serine/threonine MAPK. 17 IKKα is not active under normal conditions; once it is activated, it becomes p-IKKα and phosphorylates the NF-κB inhibitor IκB, resulting in IκB degradation and the release and translocation of active NF-κB to the nucleus. 11 c-Jun is a transcription factor, and the protein expression of its active form, phosphor-c-Jun, was enhanced in response to MSU crystals. 18 In this study, TDN did not influence the mRNA and protein expression of ERK1/2, JNK, p-38, and IKKα. Total saponins from Dioscorea nipponica Makino increased the mRNA expression of c-Jun and had no effect on its protein expression. However, TDN significantly decreased the expression of p-ERK1/2, p-JNK, p-P-38, p-IKKα, and p-c-Jun. This result is different from our previous study in which TDN decreased the expression of p-38 in a rat model of GA. 19 This difference may be due to both different time periods of treatment and drug concentrations. At the same time, it may also be because one is an in vivo study and the other is an in vitro study. Total saponins from Dioscorea nipponica Makino increased the mRNA expression of c-Jun but had no effect on its protein expression.
MAPK phosphatase is a negative regulator of this signaling pathway. In this study, TDN increased both the mRNA and protein expression of MKP but had no obvious effects on p-MKP. A study showed that the half-life period of MKP is very short, which may be due to MAPK phosphorylation. This phosphorylation may inhibit the degradation of MKP, thus increasing its stability. 20
Peroxisome proliferator-activated receptor γ plays an important role in lipid metabolism and glucose homeostasis, modulates metabolism and inflammation in immune cells, and controls cell proliferation. The regulation of PPAR-γ-dependent gene expression depends on the formation of ADP-ribose, which is synthesized by poly (ADP-ribose) polymerase (PARP). Intracellular ADP-ribosylation is catalyzed by PARPs, which are activated by cytokine inducible SH2-containing protein (CIS). 21 Adipor2 is a negative regulator of GA. When the Adipor2 signaling pathway is blocked, the concentration of IL-β is increased, which is a main pathogenesis pathway of GA. 22 In this study, TDN increased the mRNA expression of both PPARγ and Adipor2.
Intercellular adhesion molecule 1 is a member of the immunoglobulin superfamily of receptors and is important in mediating cell-cell interactions and inflammatory responses. Monosodium urate stimulation increased the expression of ICAM-1. 23 C-X-C motif chemokine ligand 1 induces immune cells toward inflammatory sites during GA attack. In this study, TDN increased the mRNA expression of ICAM1 and CXCL1 but had no effect on their protein expression. This result is different from the study of Marchetti in which the NLRP3 inflammasome inhibitor OLT1177 suppressed joint inflammation in murine models of acute arthritis by decreasing CXCL1. 21 This difference may be due to different testing methods.
Our results indicated that TDN influenced the MAPK-PPARγ signaling pathway to treat GA. It also points to the MAPK-PPARγ signaling pathway as a promising target to explore new drugs to treat GA. However, this hypothesis is based on only an in vitro study. Our animal model is on the way to verify the above result.
Total saponins from Dioscorea nipponica Makino has anti-inflammation effects to treat GA by regulating both mRNA and protein expression of some key genes of the MAPK-PPARγ signaling pathway. This study provides further pharmacology evidence to explore TDN as a new drug for GA.
Footnotes
Author Contributions
Qi Zhou designed the experiment and wrote this paper. Dong Hua Yu and Na Zhang cultured the cells, Xiao-Yang Wang and Ning Zhang performed the WB, Fang-Fang Lin performed the PCR, and Shu-Min Liu provided the facilities for the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Innovative Talent Support Programs of Colleges and Universities atthe Heilongjiang Education Department (051599).
