Abstract
Chromatographic purification of the ethyl acetate extract of the entomopathogenic fungus Aspergillus tamarii NL3 culture broth led to the isolation of griseofulvin (
Keywords
Introduction
Fungi are one of the most important sources of bioactive components for drug discovery and development. Literature surveys indicate that about 15 600 bioactive compounds are derived from fungi. The discovery rate of fungal-derived new compounds has increased significantly in the past 2 decades. 1 Aspergillus is a diverse genus with 339 accepted species; many of them are considered as decomposers of organic materials and cause destructive rots in agricultural products. 2 The genus Aspergillus is rich in alkaloids, terpenes, steroids, and polyketones. 3
Aspergillus tamarii has produced structurally diverse compounds with a wide range of bioactivities, including an antibacterial pentacyclic indole alkaloid, 4 anti-inflammatory dipyrrolobenzoquinones, 5 antibiotic and cytotoxic cyclic peptides, 6 butenolides, 7 and anti-phytopathogenic indolyl diketopiperazines. 8 In a continuing search for new antimicrobial and antioxidant agents, we report herein the isolation and structural determination of 5 secondary metabolites from the ethyl acetate (EtOAc) extract of the entomopathogenic fungus A tamarii NL3 culture broth. All isolates were further evaluated for their antimicrobial activity against 6 reference bacterial strains (Escherichia coli, Salmonella enterica, Pseudomonas aeruginosa, Enterococcus faecalis, Staphylococcus aureus, and Bacillus subtilis) and 4 reference fungal strains (Candida albicans, Aspergillus niger, Fusarium oxysporum, and Pythium ultimum), as well as their 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonate) (ABTS) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activities.
Results and Discussion
Five products were obtained from the culture broth of the entomopathogenic fungus Aspergillus tamarii NL3. Their chemical structures (Figure 1) were determined as griseofulvin (

The structures of
The obtained compounds
Antimicrobial Activities of Isolated Compounds

Abbreviations: Cip, ciprofloxacin; Mic, miconazole.
The radical scavenging ability of these compounds on ABTS and DPPH radicals was evaluated (Table 2). Unfortunately, all isolated compounds (
Antioxidant Activities of Isolated Compounds
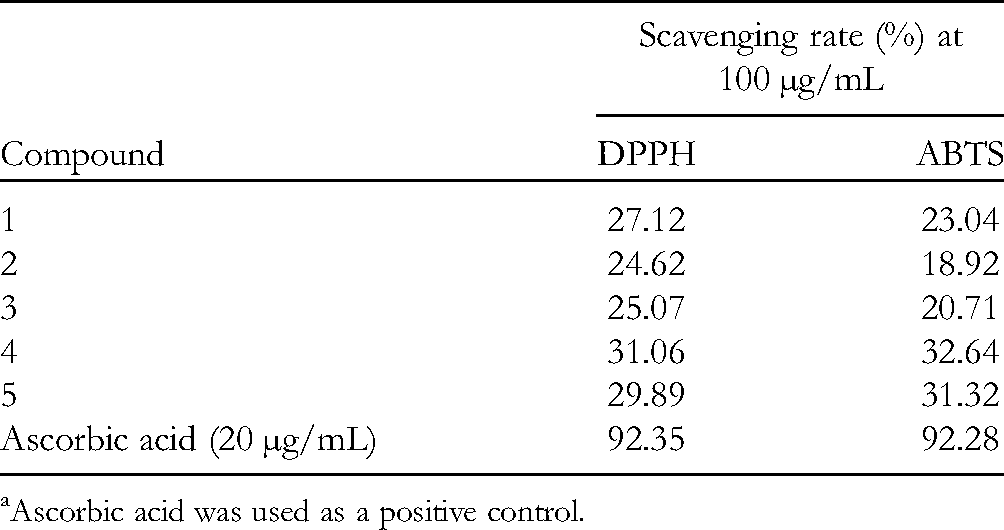
Ascorbic acid was used as a positive control.
Conclusion
Five known compounds, griseofulvin (
Materials and Methods
General Experimental Procedures
The HRESIMS were obtained from a MicroQ-TOF III mass spectrometer (Bruker Daltonics, 255,748 Germany). All NMR spectra were recorded on either a Bruker AM500 FT-NMR spectrometer (500 MHz for 1H-NMR and 125 MHz for 13C-NMR) or a Bruker Avance NEO600 spectrometer (600 MHz for 1H-NMR and 150 MHz for 13C-NMR). IR spectra were obtained from KBr pellets on an IR Prestige-21 spectrometer (Shimadzu). Optical rotations were measured on a JASCO P-2000 polarimeter. The stationary phases for column chromatography (CC) included silica gel (Kieselgel 60, 70-230, 230-400 mesh, Merck), reverse phase resins (150 µm, Fuji Silysia Chemical Ltd), and Sephadex LH-20 (Pharmacia Co.). For thin layer chromatography (TLC), precoated silica gel 60 F254 (0.25 mm, Merck) and RP-18 F254S plates (0.25 mm, Merck) were used. Compounds were detected on TLC either under UV light or by heating after spraying with 10% H2SO4 in C2H5OH (vol/vol).
Fungal Materials
Aspergillus tamarii NL3 was isolated from the grasshopper Oxya chinensis collected from rice fields in Thua Thien Hue province, Vietnam, in July 2020. The strain NL3 was identified as A tamarii using the ITS gene sequence with primers ITS4 and ITS5. 14 The ITS gene sequence of the NL3 strain showed 100% identity to A tamarii L1 (accession number MT340979) in the NCBI GeneBank database.
The fungus was cultivated in 125 mL Potato Dextrose Broth (PDB, Himedia) in a shaking incubator at 25 °C for 72 h. Following that, the culture was then inoculated in a 5 L fermentor (BioFlo 120, Eppendorf) containing 2.5 L Glucose Peptone Yeast Broth (GPYB, Himedia) at an inoculum of 5% (vol/vol). The fermentation was performed at 25 °C for 72 h with an agitation speed of 150 rpm. Afterward, the culture was moved to a 100 L bioreactor (BioFlo 610, Eppendorf) containing 50 L GPYB at an inoculum of 5% (vol/vol). The bioreactor was maintained at 25 °C for 15 days at an agitation speed of 150 rpm.
Extraction and Isolation
The culture solution of A tamarri (50 L) was extracted with EtOAc (50 L × 3 times). The EtOAc residue (APE, 16.3 g) was separated by RP-18 CC using an increasing ratio of methanol (MeOH) (10%-100%) in water as mobile phases to obtain six fractions, APE1-APE6. Fraction APE5 (2.1 g) was separated by silica gel CC using CH2Cl2/EtOAc (10/1, vol/vol) as the mobile phase to yield 5 subfractions, APE5.1-APE5.5. Compound
Antimicrobial Assay
Antimicrobial activity of the isolated compounds was evaluated against reference microorganisms obtained from Mientrung Institute for Scientific Research, Thua Thien Hue province, Vietnam, including Gram-negative bacteria (ie, E coli ATCC 25922, S enterica ATCC 13076, P aeruginosa ATCC 27853), Gram-positive bacteria (ie, E faecalis ATCC 29212, S aureus ATCC 25923, and B subtilis ATCC 6633), and fungi (C albicans ATCC 10231, A niger MISR 11215, F oxysporum MISR 20415, and Rhizoctonia solani MISR 11115). Minimum inhibitory concentrations (MICs) of the compounds against the reference microorganisms were determined using the microdilution method, as described by Dat et al. 15 The antibiotics ciprofloxacin and miconazole were used as positive controls.
Antioxidant Assay
The ABTS free radical scavenging activity of each sample was determined using the method described by Pellegrini et al.
16
A quantity of 0.1 mL sample (concentrations ranging from 25 to 100 µg/mL) was mixed with 3.9 mL of ABTS (7 mM) solution, and then the absorbance was measured at 734 nm. Ascorbic acid was employed as a positive control. The scavenging capability toward ABTS was calculated as follows:

Supplemental Material
sj-doc-1-npx-10.1177_1934578X221141548 - Supplemental material for Secondary Metabolites From the Grasshopper-Derived Entomopathogenic Fungus Aspergillus Tamarii NL3 and Their Biological Activities
Supplemental material, sj-doc-1-npx-10.1177_1934578X221141548 for Secondary Metabolites From the Grasshopper-Derived Entomopathogenic Fungus Aspergillus Tamarii NL3 and Their Biological Activities by Dat Ton That Huu, Ha Tran Phuong, Phan Thi Diem Tran, Bakeo Souvannalath, Hieu Le Trung, Duc Viet Ho and Cuong Le Canh Viet in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vietnam Academy of Science and Technology, (grant number QTLA01.01/20-21).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
