Abstract
Objective
For centuries, some mullein species (species of the genus Verbascum L.) have been used to treat respiratory diseases and internal and external infections and as excellent skin softeners. The healing properties of Verbascum species are the basis for their use in both traditional and conventional medicine. The present work aimed to identify and quantify the bioactive compounds of the flower extracts of V. phlomoides, V. speciosum, and V. niveum, and to determine their anti-inflammatory, antioxidant, and antimicrobial activities.
Methods
After spectrophotometric determination of the total phenolic content, the bioactive compounds were identified and quantified by HPLC, and the biological activities were assessed by in vitro methods.
Results
According to the obtained results, the major compounds in most of the tested extracts of Verbascum species were p-hydroxybenzoic acid, vanillic acid, verbascoside, aucubin, and catalpol. In both methods (DPPH and BCBT), the ethanol extracts showed stronger antioxidant activity than the water and propylene glycol extracts. All tested samples showed an anti-inflammatory effect in the BSA assay with an inhibition of protein denaturation of over 70%. The tested extracts were more active against Gram-positive than Gram-negative bacteria and showed the highest antimicrobial activity against Cutibacterium acnes and Streptococcus pneumoniae bacteria. Based on the results obtained, the tested species (V. phlomoides, V. niveum, and V. speciosum) are rich in polyphenolic acids, iridoids, and verbascoside and show comparable antioxidant, anti-inflammatory, and antimicrobial activity.
Conclusion
In conclusion, the studied Verbascum species can be considered a natural source of bioactive ingredients with great potential for the pharmaceutical industry.
This is a visual representation of the abstract.
Highlights
Extracts obtained with different solvents were analyzed.
Twenty-three compounds were identified in the extracts of the analyzed species.
Propylene glycol extracts had the highest anti-inflammatory activity (BSA test).
The assessed extracts showed antioxidant (DPPH, BCBT) and antimicrobial activities (MIC).
Studied Verbascum species showed high potential for further use in phytotherapy.
Introduction
The Scrophulariaceae family comprises approximately 200 genera and over 2500 species globally. The genus Verbascum L., known as “mullein,” comprises approximately 450 species, making it one of the largest genera in the family.1,2 The species of this genus are native to Europe and Asia, mainly wild and not yet cultivated. 1 Verbascum species are tall, annual, biennial, and perennial plants that flower in summer.2,3
Most pharmacopeias mention the flowers of this plant for their medicinal properties. The mullein flowers’ active ingredients have expectorant, demulcent and mucolytic properties and are used in the treatment of respiratory disorders, such as dry coughs, bronchitis, pertussis, asthma, and tuberculosis.4,5 The Committee on Herbal Medicinal Products at the European Medicines Agency (HMPC/EMA) recommends the use of Verbasci flos derived from V. thapsus L., V. densiflorum Bertol. (V. thapsiforme Schrad) and V. phlomoides L. flowers in respiratory disorders. 6 Studies indicate that Verbascum songaricum Schrenk. flowers are used for menstrual and fertility issues in Türkiye. 7 In the Himalayas, V. thapsus L. flowers treat ear infections. 8 V. phlomoides L. is a potential source of metabolites with acetylcholinesterase and tyrosinase inhibitory effects. 9 The flowers are effective against earache, fungal infections, wounds, eczema and inflammatory skin diseases. 4 In Turkish traditional medicine, Verbascum L. species’ leaves, flowers, and aerial parts manage eczema and act as desiccants for wounds. 10
The medicinal properties of Verbascum species have led to their use in both traditional and conventional medicine. 11 Several Verbascum species have been shown to possess various biological properties, including antioxidant, anti-inflammatory, antimicrobial, antiviral, antinociceptive, antitussive, and immunomodulatory activities. These activities are associated with the chemical constituents of the plants, and numerous compounds have been identified in Verbascum species. 11
The EMA assessment report states that aucubin and verbascoside have anti-inflammatory effects. 6 There are also studies on the anti-inflammatory effect of various Verbascum species: V. pterocalycinum var. mutense Hub.-Mor., 12 V. chionophyllum Hub.-Mor., 13 V. pycnostachyium Boiss. & Held. V. salviifolium Boiss., V. latisepalum Hub.-Mor.,13,14 V. mucronatum, V. lasianthum Boiss. ex Benth., 14 V. xanthophoeniceum Griseb., 15 Verbascum cheiranthifolium Boiss. var. cheiranthifolium Boiss. 16 and V. nubicum Murb. 17 In a recently published study, a formulation containing mullein oil macerate (V. phlomoides L.) (10%) showed a positive effect on human skin pre-irritated with sodium lauryl sulfate, as it was shown to effectively hydrate and regenerate the skin barrier. 18
In addition, several studies have investigated the antimicrobial properties of different Verbascum species and their extracts, 19 suggesting that various species have shown antimicrobial (both antibacterial and antifungal) activity.19-21 In addition, flowers of V. thapsus L. have shown antiviral effects against HCov-229E, HBV, and HSV II. 22
In our previous works, we determined the phenolic content and antioxidant potential of V. phlomoides L. in vitro, 23 and the antimicrobial activity of various extracts of V. niveum Ten. 24 Therefore, we continued the study on Verbascum species and in this work, we conducted a comparative study on three Verbascum species, one of which is the officially approved medicinal species according to the EMA monograph for the drug Verbasci flos (V. phlomoides L.), on the second one we found very few data (V. speciosum Schrad.) while there is no data in the literature on the third one (V. niveum Ten.). The objectives of our study were: i) to identify and quantify bioactive compounds of flower extracts prepared with different solvents; ii) to determine their content of total polyphenols, total tannins, phenylpropanoid derivatives, and total flavonoids; and iii) to compare their biological activities: antioxidant, anti-inflammatory and antimicrobial. To our knowledge, this is the first study on the chemical composition and biological activities of the species V. niveum Ten., and it also provides a systematic comparison of chemical profiles and activities of selected three species.
Materials and Methods
Plant Material and Extracts
The plant material (flowers of V. phlomoides L., V. speciosum Schrad., and V. niveum Ten.) was collected during flowering in 2021 in Bosilegrad and the surrounding area. Prof. Bojan Zlatković from the Faculty of Science and Mathematics, University of Niš identified the species. The voucher specimens (Herbarium codes: V. phlomoides L.—14506, V. niveum Ten.—14615, V. speciosum Schrad.—14616) are deposited in the herbarium of the Faculty of Science and Mathematics, University of Niš.
The flowers were dried in a dark, well-ventilated area at room temperature. Once dried, the plant material was ground using a laboratory mill and stored in glass containers until further analysis. Extracts were prepared using three different solvents (50% ethanol, distilled water, and 80% propylene glycol) by percolation (
Chemical Reagents and Instrumentation
All reagents and solvents used in this study were of analytical grade or better and were purchased from Sigma-Aldrich Co. (Sigma-Aldrich Co., St Louis, MO), or Extrasynthese (Lyon, France). Standards for HPLC determination (apigenin, apigenin-7-O-glucoside, luteolin, luteolin-7-O-glucoside, diosmin, chrysoeriol, kaempferol, rutin, quercetin, aucubin, catalpol, verbascoside, ferulic acid, caffeic acid, rosmarinic acid, vanillic acid, p-hydroxybenzoic acid, p-cumaric acid, protokatechuic acid, neochlorogenic acid, chlorogenic acid, trans-cinnamic acid, gallic acid) were of HPLC grade.
For spectrophotometric measurements, an Evolution 60 Thermo scientific spectrophotometer (Fisher Scientific, Loughborough, UK) and Multiskan Ascent No354 (Thermo Labsystems, Finland) ELISA microplate reader was used. Incubation was done with Incuterm Raypa® trade (Catalonia, Spain). All experimental procedures were conducted in triplicate.
HPLC analysis was done on Agilent Technologies 1200 HPLC (Santa Clara, CA, USA), equipped with a Lichrospher RP 18e column (250 × 4.6 mm, 5 µm, Agilent Technologies, Santa Clara, CA, USA), with photodiode-array (PDA) detection.
Chemical Characterization
Total Polyphenols and Total Tannins
The content of total polyphenols was determined by the Folin–Ciocalteu colorimetric method. 26 To 0.02 mL of diluted extract was added 0.25 mL Folin-Ciocalteu reagent and 1.25 mL 20% Na2CO3 and the tube was shaken. After 40 min, the absorbances of the samples and standards were measured at 725 nm, and the results were expressed as gallic acid equivalents per g of extract.
The total tannin content was determined by the same Folin-Ciocalteu procedure 26 after the removal of tannins using an insoluble binder (polyvinylpolypyrrolidone, PVPP). To the tube was added 100 mg of PVPP, 1 mL of distilled water, and 1 mL of tested extract, and the tube was shaken. After 15 min in the fridge, the mixture was shaken again and then centrifuged at 3000 rpm for 10 min. The test was performed with a clear supernatant, and the results were also expressed as gallic acid equivalents per g of extract (mg GA/g extract).
Phenylpropanoid Derivates
The content of total phenylpropanoid derivatives was determined spectrophotometrically, according to the slightly modified Arnow method. 27 This method is based on the reaction of derivatives of o-dihydroxycinnamic acid with sodium molybdate. Absorbance was recorded at a wavelength of 525 nm, and the phenylpropanoid content was expressed as the content of the hydroxycinnamic acid derivative (% HA).
Total Flavonoids
The total flavonoids content in the extracts was determined spectrophotometrically at 430 nm by the AlCl3 method. 28 To 1 mL of extract dissolved in methanol was added 1 mL of methanolic AlCl3 • 6H2O (2%), the mixture was shaken, and the absorbance was measured after 10 min. The results were expressed as mg rutin (R)/g of dry extract after quantification based on a standard curve with rutin.
HPLC
The determinations were performed in extracts dissolved in 70% alcohol. Chemical characterization of the prepared extracts was carried out by HPLC, for identification and quantification of flavonoids (apigenin, apigenin-7-O-glucoside, luteolin, luteolin-7-O-glucoside, diosmin, chrysoeriol, kaempferol, quercetin), iridoid glucoside (aucubin, catalpol), phenylethanoid glycoside (verbascoside), and polyphenolic acids (ferulic, caffeic, rosmarinic, vanillic, p-hydroxibenzoic, p-cumaric, neochlorogenic, chlorogenic, trans-cinnamic, and gallic acid).
“Fingerprinting” of the investigated phenolic compounds was achieved by two methods, depending on the type of secondary metabolites we have been looking for. Namely flavonoids and polyphenolic acids were identified using an HPLC, applying gradient elution of two mobile phases, ie “A/B” (“A"-0.2 M phosphoric acid solution, and “B"-being a pure acetonitrile) at flow-rates 1 mL/min, with photodiode-array (PDA) detection (UV at 325 nm), always within 70 min. The gradient program was as follows: 11–25% B (35 min), 25–40% B (20 min), 40–65% B (5 min), 65–100% B (10 min), injection volume 10 μL, and column temperature 25 °C. The described qualitative and quantitative analysis represented the modified HPLC method given in the paper Tadic et al 29
For the profiling of iridoid compounds in the investigated extracts, method presented by Liu et al, with the modification, was used to enable the complete identification of this group of secondary metabolites. 30 They were identified employing an HPLC, applying gradient elution of two mobile phases, ie “A/B” (“A"-0.1 M solution of phosphoric acid, and “B"-being pure acetonitrile) at flow-rates 1 mL/min, with photodiode-array (PDA) detection (UV at 210 nm), always within 45 min. The gradient program was as follows: 5–15% B (18 min), 15–25% B (7 min), 25–60% B (6 min), 60–100% B (9 min), and 100% B (5 min). The injection volume was 10 μL, and the column temperature was 25 °C.
The concentrations of investigated samples were VNE = 36.55 mg/mL; VNH = 37.06 mg/mL; VNP = 516.18 mg/mL; VSE = 39.89 mg/mL; VSH = 26.19 mg/mL; VSP = 503.88 mg/mL; VPE = 43.80 mg/mL; VPH = 37.43 mg/mL; VPP = 504.94 mg/mL. Before injection, all investigated extracts were filtered through a PTFE membrane filter. Compound identification was based on the comparison of retention times and UV-VIS spectra with those of standards. Upon successful spectral matching, results were further confirmed by spiking the samples with the corresponding standards to verify peak purity and achieve definitive identification. Peaks that did not meet these criteria were excluded from quantification. Quantification was carried out using external calibration with reference standards. For standards used in the investigation, the concentrations were: gallic acid = 0.43 mg/mL; neochlorogenic acid = 0.30 mg/mL; protocatechuic acid = 0.47 mg/mL; chlorogenic acid = 0.43 mg/mL; p-hydroxybenzoic acid = 0.43 mg/mL; vanillic acid = 0.30 mg/mL; caffeic acid = 0.52 mg/mL; p-coumaric acid = 0.50 mg/mL; ferulic acid = 0.43 mg/mL; rutin = 0.44 mg/mL; verbascoside = 0.74 mg/mL; luteolin-7-O-glucoside = 0.34 mg/mL; apigenin-7-O-glucoside = 0.49 mg/mL; diosmin DMSO = 0.47 mg/mL; rosmarinic acid = 0.43 mg/mL; trans-cinnamic acid = 0.58 mg/mL; luteolin = 0.49 mg/mL; quercetin = 0.43 mg/mL; apigenin = 0.54 mg/mL; chrysoeriol = 0.41 mg/mL; kaempferol = 0.49 mg/mL; aucubin = 0.45 mg/mL; catalpol = 0.51 mg/mL. A volume of 10 µL was injected for both the standard solutions and the sample extracts.
Antioxidant Activity
DPPH Test
Radical scavenging activity was evaluated using the stable free radical 1,1-diphenyl-2-picrylhydrazyl (DPPH) according to the method described by Chang et al (2010),
31
with minor modifications. The extracts were prepared at various concentrations. In 96-well microtiter plates, 40 μL of each extract dilution was mixed with 120 μL of ethanol, followed by the addition of 40 μL of ethanolic DPPH solution (0.2 mg/L). The plate was briefly shaken and then left in the dark for 30 min. Absorbance was recorded at 550 nm (using an ELISA reader). The DPPH radical inhibition percentage was calculated using the formula:
β-Carotene Bleaching Test (BCBT)
β-Carotene bleaching test (BCBT) was done according to the method described by Koleva et al (2002) with slight modifications.
32
β-carotene (1 mg) was dissolved in 5 mL of chloroform (HPLC grade) and 1 mL of this solution was mixed with 25 μL linolic acid and 200 mg Tween 20. Chloroform was removed in a rotary vacuum evaporator under pressure, 50 mL of fresh bubbled water was added, and the mixture was shaken until a clear emulsion. In the 96-well microtiter plates, 25 μl of each diluted sample and 200 μl of prepared emulsion were added. The microtiter plates were gently shaken on a microplate shaker and the zero-time absorbance (A0) was measured at a wavelength of 450 nm on an ELISA reader. The plates were incubated at 47 °C for 120 min, and then the absorbance was measured again (A120). The ability to inhibit β-carotene decolorization is expressed as a percentage of inhibition according to the formula:
The IC50 value (concentration required to inhibit β-carotene bleaching to 50%) was calculated from the concentration/% inhibition curve and expressed as the average value of three independent experiments in μg/mL.
In Vitro Anti-Inflammatory Activity
The inhibition of protein denaturation assay was performed by the protocol of Lavanya et al 2010,
33
with slight modifications. Bovine serum albumin (BSA, 5% w/v aqueous solution) was used. As a test solution 100 µg/mL extract was used while the test control solution contained distilled water instead of extract and the product control solution consisted of distilled water and 100 µg/mL extract. The samples were incubated at 37 °C for 20 min and the temperature was increased to keep the samples at 57 °C for 3 min. After cooling, 0.2 mL of phosphate buffer saline (pH = 6.3) was added to the above solutions, and the absorbance was measured at 340 nm. The inhibition (%) of protein denaturation was calculated by applying the following equation:
The control represents 100% protein denaturation. The results were compared with diclofenac sodium (100 µg/mL) effects.
Antimicrobial Activity
To evaluate antimicrobial activity, dry extracts (ethanol and distilled water) were dissolved in dimethyl sulfoxide (DMSO), while propylene glycol extracts were used without further modification.
Test Organisms
The antimicrobial bioassays were done with one yeast: Candida albicans (CNM-CL F8555a), and six bacterial strains (all from the American Type Culture Collection—ATCC): Gram-negative strains (Escherichia coli (ATCC 25922) and Pseudomonas aeruginosa (ATCC 27853)), and Gram-positive strains (Staphylococcus aureus (ATCC 29213), Enterococcus faecalis (ATCC 29212), Streptococcus pneumoniae (ATCC 49619) and Cutibacterium acnes (ATCC 6919)), while Clostridium perfringens (ATCC 13124) was a control for C. acnes.
Experimental Procedure
Antimicrobial activity was assessed using control strains, following the guidelines set by the European Committee on Antimicrobial Susceptibility Testing (EUCAST).34,35 The antimicrobial activity was determined using the broth microdilution method, the reference standard for evaluating the antimicrobial susceptibility of rapidly growing aerobic bacteria. In accordance with EUCAST guidelines, testing was performed following the International Standard ISO 20776-1. 36
Bacterial and fungal inoculates were prepared by suspending isolated colonies, grown for 18 to 24 h on blood agar plates, directly into broth. The turbidity of the suspension was adjusted to match a 0.5 McFarland standard, corresponding to approximately 107–108 colony forming units CFU/mL for bacteria (depending on the genus) and 0.4 × 105 to 5 × 105 spores/mL for fungal strains. Within 15 min of preparation, the inoculum was further diluted 1:20 in broth, yielding a final concentration of 5 × 106 CFU/mL.
Ten serial two-fold dilutions of each extract (ranging from 0.20 mg/mL to 100 mg/mL) were prepared in 96-well microtiter plates. Each well contained 0.1 mL of the diluted extract, to which 0.01 mL of the inoculum was added, resulting in a final bacterial concentration of approximately 5 × 105 CFU/mL. Plates were incubated at 37 °C for 24 h for bacterial strains, and at 25 °C for 48 h for fungal strains and C. acnes. Antimicrobial activity was expressed as the minimum inhibitory concentration (MIC), defined as the lowest concentration of extract (mg/mL) that completely inhibited visible microbial growth. Colistin (for E. coli and P. aeruginosa), vancomycin (for S. aureus and E. faecalis), fluconazole (for C. albicans), and ampicillin (for S. pneumoniae and C. perfringens) served as positive controls. DMSO (used to dissolve dry extracts) and 80% propylene glycol were included as negative controls. All experiments were conducted in triplicate on separate plates.
Statistical Analysis
All experimental measurements were conducted in triplicate and reported as the mean ± standard deviation. The normality of the distribution of the continuous variants was determined using the Shapiro-Wilk test. To compare variables between groups, ANOVA analysis of variance followed by Tukey's post hoc test was used if the distribution was normal, or the Kruskal-Wallis test with post hoc Mann-Whitney test if the distribution deviated from the normal distribution. Pearson's linear correlation coefficient was used for the correlation analysis. A significance level of p < 0.05 is considered statistically significant. The statistical analyses were conducted using SPSS v. 20.0 software. The heatmap was created using OriginLab graphing and data analysis software.
Results and Discussion
Verbascum species have been used in traditional medicine for centuries due to their numerous biological activities. Since different species of this genus can be used as herbal medicine, it would be very useful to distinguish between the species. 37
In the present report, we investigated flower extracts made with different solvents of three Verbascum species: V. phlomoides L., V. speciosum Schrad., and V. niveum Ten. for their secondary metabolites and biological activities.
The DER values were between 1.48 and 7.06 (presented in Table 1).
DER Values for Dry Extracts.

Chemical Characterization
Total Polyphenols, Tannins, and Flavonoids
Table 2 shows the contents of total polyphenols, total tannins, and total flavonoids in the tested extracts. Data are expressed as mean ± SD and were compared using One-Way ANOVA followed by Tukey's post hoc test with a significance level of p < 0.05.
Contents of Total Polyphenols, Total Tannins, Phenylpropanoid Derivates, and Total Flavonoids in Tested Extracts.

As for polyphenols, ethanol extracts were richer than water and propylene glycol, with the highest values obtained for VNE and VSE (77.16 ± 4.02 and 79.84 ± 1.38 mg GA/g extract respectively). The results for V. phlomoides ethanol extracts were previously published. 23 According to Mihailovic et al, 38 methanol extracts of all tested species (V. nigrum L., V. phlomoides L. and V. thapsus L.) had a higher polyphenol content than water extracts. The tannins content was in line with the polyphenols content. In all the extracts assessed, the ethanol extracts were with higher content of total polyphenols, total tannins, and total flavonoids than water and propylene glycol extracts.
Chemical Characterization of the Prepared Extracts
Table 3 shows the results of the detailed chemical characterization of the different extracts assessed.
Chemical Characterization of Different Extracts of Tested Verbascum Species (in mg/g of Dry Extracts).

Figures S1–S6 (Supplemental material) shows the HPLC chromatograms of investigated samples.
Secondary metabolites are usually considered as bioactive molecules—compounds that have no nutritional value but exert biochemical effects in the human body. These effects may help explain some of the properties attributed to medicinal plants by their users. 39 We used HPLC in conjunction with a photodiode-array (PDA) detection to identify and quantify the secondary metabolites. The results are shown in Table 3. Some of the secondary metabolites were present in all extracts assessed, while some were only detected in extracts of specific species or in extracts prepared with specific solvents. Verbascoside was detected in varying amounts in all samples assessed (except in the VNH extract). It was first isolated from mullein (Verbascum sinuatum L.) in 1963 and named verbascoside. 40 The highest content was found in VPE extract (3.56 mg/g dry extract), while propylene glycol extracts had the lowest content. These results are consistent with previous studies that have found that various Verbascum species are rich in verbascoside.38,41-43 All tested extracts were rich in iridoids (aucubin and catalpol; although aucubin was not detected in the VNE extract). The extract of V. mucronatum Lam., which is rich in iridoid glucosides, is said to have anti-inflammatory, antinociceptive, and wound-healing properties. 44 In addition, aucubin as an isolated compound has been found to have significant antinociceptive and anti-inflammatory effects.45,46 In terms of flavonoid content, rutin was detected in all extracts except VNH. Rutin together with chlorogenic acid and p-coumaric acid was found to be the major phenolic compounds in V. flavidum (Boiss.) Freyn & Bornm. 47 Apigenin, luteolin, and luteolin-7-O-glucoside were detected in most of the extracts. The flavonoids luteolin and apigenin were also detected in methanol extracts of V. phlomoides L. 38 Kaempferol was present in extracts of V. speciosum Schrad. and V. phlomoides L. (except VPH), apigenin-7-O-glucoside was only present in V. niveum extracts (VNP and VNH), while luteolin-7-O-glucoside was found only in propylene glycol extracts of V. speciosum Schrad. and V. phlomoides L. In general, the tested samples were rich in phenolic acids (protocatechuic acid and p-hydroxybenzoic acid were detected in all samples; gallic acid, neochlorogenic acid, vanillic acid, and caffeic acid were present in most of them, while the remaining phenolic acids were present only in a few extracts). Rosmarinic acid in V. phlomoides L. has been identified as one of the major phenolic compounds. 14 In our study, it was found in extracts of V. phlomoides L., but also V. speciosum Schrad. (except VSH extract). Chlorogenic acid, and p-coumaric acid as the major phenolic compounds in V. flavidum (Boiss.) Freyn & Bornm. 47 Based on the results obtained, we can say that most of the components identified were present in the ethanol and propylene glycol extracts, while the water extracts had a poorer chemical composition.
Antioxidant Activity
Table 4 shows the results of the in vitro antioxidant activities of different extracts of Verbascum species assessed in two complementary test systems. Data are expressed as mean ± SD and were compared using One-Way ANOVA followed by Tukey's post hoc test with a significance level p < 0.05.
The Results of the in Vitro Antioxidant Activities of Different Extracts of Verbascum Species.

The strong antioxidant activity of phenols is primarily due to the presence of hydroxyl groups in their molecular structure, which enable them to neutralize reactive oxygen species (ROS), particularly superoxide. Antioxidants play a crucial role in protecting cells from oxidative damage and in reducing the risk of disease by neutralizing excess free radicals. 48 The antioxidant activity of natural products can be tested using several different methods, but none of the methods is universal and cannot accurately and qualitatively measure the exact antioxidant capacity, as there are different types of free radicals and different mechanisms involved in the antioxidant capacity of antioxidants. 38 In our work, we used two complementary in vitro methods to measure antioxidant capacity: the DPPH assay and BCBT.
The results of the in vitro antioxidant activities determined in the DPPH assay show that ethanol extracts exhibited stronger activity than water and propylene glycol extracts. The activity of the tested extracts was ethanol > propylene glycol > water. The results of the antioxidant activity of V. phlomoides ethanol extracts were previously published. 23 According to the results, the ethanol extracts showed the best activity, and their content of phenols and flavonoids was the highest. Surprisingly, the propylene glycol extracts showed better antioxidant activity, although the polyphenol content was higher in the water extracts.
In the β-carotene bleaching test, there was a correlation between the content of polyphenols and flavonoids and the expressed antioxidant activity; higher polyphenol and flavonoid content was associated with stronger antioxidant activity. Ethanol extracts showed the best results, while propylene glycol extracts showed the weakest activity. Flavonoids have the highest antioxidant activity among phenolic compounds, and the polyphenolic nature of flavonoids causes the collection of free radicals such as superoxides.5 Water extracts also showed lower antioxidant activity compared to methanol extracts of V. nigrum L., V. phlomoides L., and V. thapsus L. 38
In Vitro Anti-Inflammatory Activity
Table 5 shows the results of the in vitro anti-inflammatory activity of the tested extracts. Data are expressed as mean ± SD and were compared using One-Way ANOVA followed by Tukey's post hoc test with a significance level of p < 0.05.
Inhibition of Protein Denaturation Induced by Different Tested Extracts (Mean ± SD) in BSA Assay.

All the extracts showed significant anti-inflammatory activity in vitro, with the highest percentage of inhibition of BSA denaturation (87.22 ± 0.006%) observed in the propylene glycol Ten. extract of V. phlomoides L., while the lowest value was observed in the ethanol extract of V. niveum Ten. (70.95 ± 0.004%). Table 4 shows that all extracts assessed had lower values than the standard value for diclofenac sodium (95.60 ± 0.001%), although the difference was not great considering that all samples had a percentage inhibition of protein denaturation of over 70. Protein denaturation inhibition values above 80 were obtained with two extracts from the species V. phlomoides L. (VPH and VPP). For all species assessed, the best results were obtained with propylene glycol flowers extracts. For the extracts of V. niveum Ten. and V. phlomoides L., the lowest inhibition was obtained with ethanolic extracts, while for V. speciosum Schrad. the weakest result was obtained with water extract. Other researchers have also demonstrated the anti-inflammatory effect of different types of Verbascum species using different methods.12,16 Phenolic compounds, especially flavonoids, and phenylethanoids, are mainly held responsible for anti-inflammatory and antimicrobial activities.49,50 However, the study by Grigore et al (2013) showed a weak correlation between the polyphenol content and the anti-inflammatory activity of V. phlomoides L., so it was hypothesized that the anti-inflammatory activity is a consequence of the presence of iridoids and phenylethanoids. 14 In addition, an iridoid glucoside, aucubin was found to possess significant antinociceptive and anti-inflammatory activities. 45 Our extracts were rich in phenolic compounds, and aucubin and catalpol were present in high amounts in almost all extracts. The strong anti-inflammatory effect is probably the result of the synergistic action of all the compounds present.
Antimicrobial Activity
Table 6 and Table 7 show the results of the in vitro antimicrobial activities of the tested extracts against selected bacterial species.
The Obtained Results of the in Vitro Antimicrobial Activities of Different Extracts of V. speciosum and V. phlomoides (in mg/mL).

The Obtained Results of the in Vitro Antimicrobial Activities of Different Extracts of V. niveum (in mg/mL).

Since both propylene glycol (PG) and dimethyl sulfoxide (DMSO) possess inherent antimicrobial properties, only minimum inhibitory concentration (MIC) values lower than those of the solvents were considered indicative of true antimicrobial effects. In general, the MIC values of all tested extracts against bacterial strains were lower than those of PG and DMSO, suggesting authentic antimicrobial activity. The antimicrobial activity of Verbascum niveum extracts against E. coli, S. aureus, P. aeruginosa, E. faecalis, and C. albicans has already been reported. 24 In general, the extracts were more effective against Gram-positive bacteria (S. aureus, E. faecalis, S. pneumoniae), with particularly strong activity observed against S. pneumoniae, compared to Gram-negative bacteria (E. coli, P. aeruginosa). Similar results were reported by Dulger et al, 51 who tested 80% methanol extracts of three Verbascum species (V. olympicum Boiss., V. prusianum Boiss., and V. bombyciferum Boiss.) using the agar disk diffusion method. They observed antimicrobial activity mainly against Gram-positive bacteria and yeasts, with Staphylococcus aureus and Micrococcus luteus being the most sensitive strains. 51 The EMA Assessment report reports that the herbal drug Verbasci flos shows antibacterial activity, including against Klebsiella pneumoniae. 6 Both S. pneumoniae and K. pneumoniae are common pathogens of pneumonia, and the antibacterial activity of mullein against these pathogens is partly the reason for the efficacy of mullein against respiratory diseases. In terms of antifungal activity (effect on C. albicans), the MIC values of the extracts of V. phlomoides L. and V. speciosum Schrad. were lower than our previously published results of the extracts of V. niveum Ten. 24 Very good results of antimicrobial activity were also obtained against C. acnes. While the MIC values of DMSO and PG were 12.5 mg/mL (like the MIC values against other strains assessed), the MIC values for our extracts were below 3.12 mg/mL. This may be partly a reason for the traditional use of Verbascum species for dermatological problems (in the Kurdish traditional medicine, V. calvum Boiss.& Kotschy is recommended for the treatment of burns and other skin diseases 52 ) and may be beneficial for the potential use of Verbascum extracts for various skin problems (some products from Verbascum species are already listed in the Cosmetic ingredient database (CosIng53R 53 )). Various studies reported that extracts of different Verbascum species showed better activity against Gram-positive than Gram-negative strains.54,55 The best results were obtained against S. pneumoniae and C. acnes, with most MIC values below 1 mg/mL (bold in Table 6 and Table 7).
Statistical Analysis
The total content of phenolic compounds, and tested antioxidant and anti-inflammatory activities did not differ between the studied species (p < 0.05) (Figure 1a, b, c, d, e). However, there was a statistically significant difference in antimicrobial activity between studied species (E. faecalis, S. pneumoniae and C. albicans between species V. niveum Ten. and V. specious Schrad. and E. faecalis and C. albicans between species V. niveum Ten. and V. phlomoides L.) (p < 0.05) (Figure 1f).

The Effect of Three Different Verbascum Species (VN, VS, and VP) on the Content of Total Polyphenols and Total Tannins (7a), the Content of Total Flavonoids (7b), the Content of Phenylpropanoid Derivatives (7c), Antioxidant Activity (7d), Anti-Inflammatory Activity (7e) and Antimicrobial Activity (7f). Data are Presented as Mean ± SD and Further Compared Using the ANOVA (Tukey's Post hoc Test) or Kruskal—Wallis Test (Mann–Whitney Post hoc Test), a: p < 0.05.
Three different solvents (50% v/v ethanol, distilled water, and 80% propylene glycol), showed an influence on the content of total polyphenols and total tannins (Figure 2a), but there was no statistically significant difference in the content of phenylpropanoid derivates (Figure 2c). A statistically significant difference was found in the content of total flavonoids between 50% v/v ethanol and distilled water and 50% v/v ethanol and 80% v/v propylene glycol (Figure 2b). In the DPPH test there was a statistically significant difference between 50% v/v ethanol and distilled water and distilled water and 80% v/v propylene glycol (Figure 2d). In the BCBT there was a statistically significant difference between 50% v/v ethanol and 80% v/v propylene glycol and distilled water and 80% v/v propylene glycol (Figure 2e). A statistically significant difference between 50% v/v ethanol and 80% v/v propylene glycol was found in the BSA test. A statistically significant difference between used solvents was found in antimicrobial activity (C. acnes 50% v/v ethanol and distilled water, C. acnes and C. albicans between 50% v/v ethanol and 80% v/v propylene glycol and S. aureus between distilled water and 80% v/v propylene glycol) (Figure 2f).
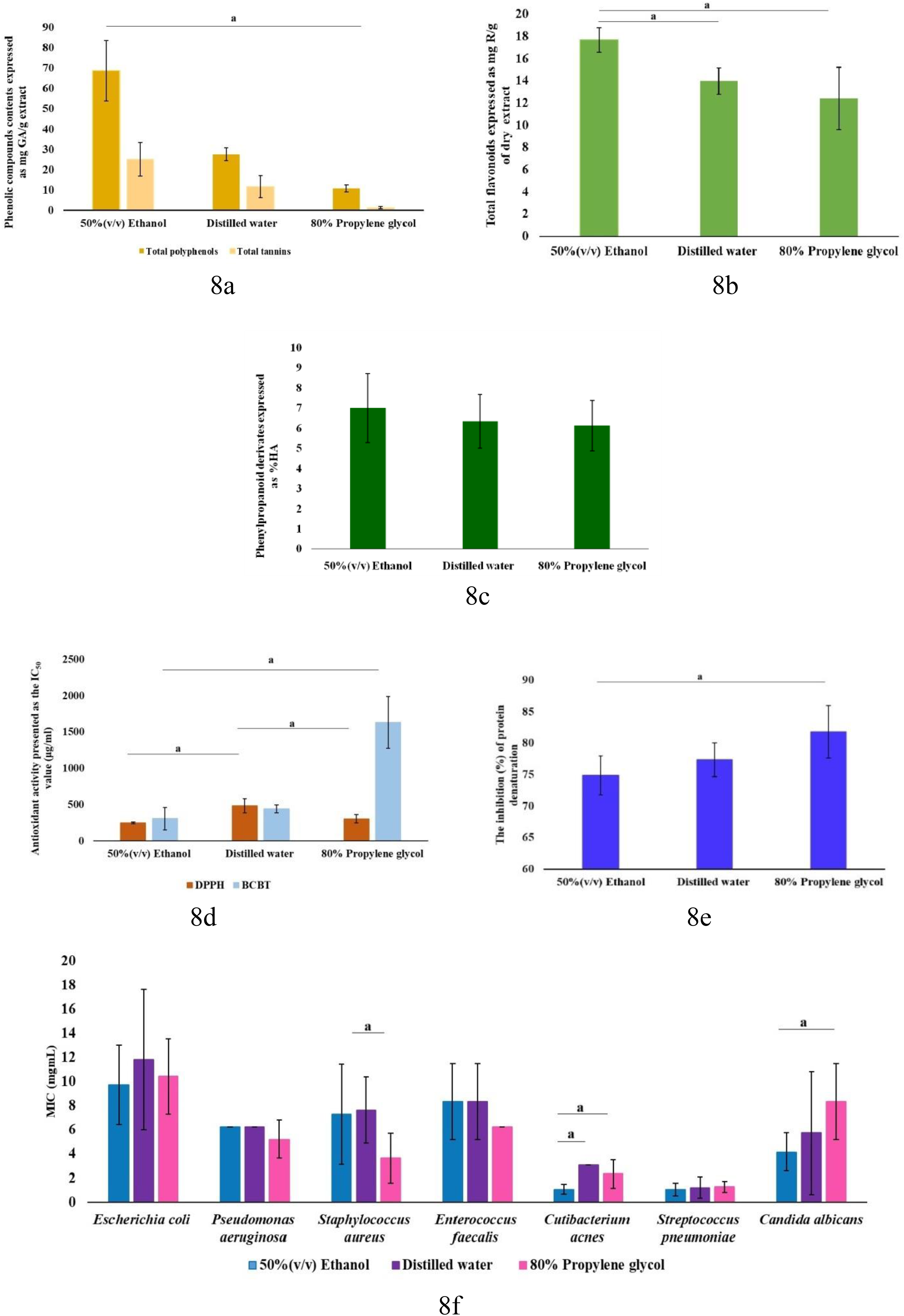
The Effect of Three Different Extracts Solvents (50% v/v Ethanol, Distilled Water, and 80% Propylene Glycol) on the Content of Total Polyphenols and Total Tannins (8a), the Content of Total Flavonoids (8b), the Content of Phenylpropanoid Derivatives (8c), Antioxidant Activity (8d), Anti-inflammatory Activity (8e) and Antimicrobial Activity (8f). Data are Presented as Mean ± SD and Further Compared Using the ANOVA (Tukey's Post hoc test) or Kruskal—Wallis Test (Mann–Whitney Post hoc test), a: p < 0.05.
Heatmap of the correlation matrix for the phenolic compounds and antioxidant activity (inhibition of DPPH free radical and inhibition of β-carotene decolorization) and phenolic compounds and anti-inflammatory activity for flower extract from each Verbascum species and extraction solvent studied. The scale ranges from −1 (green) to +1 (red) (Figure 3).

Heatmap of the Correlation Matrix for the Phenolic Compounds and Antioxidant Activity (Inhibition of DPPH Free Radical and Inhibition of β-Carotene Decolorization) and Phenolic Compounds and Anti-Inflammatory Activity for Flower Extract from Each Verbascum species and Extraction Solvent Studied. the Scale Ranges from −1 (Green) to +1 (red).
In our work, we conducted a detailed investigation of the chemical composition and biological activities of different extracts of three Verbascum species. One of them, V. niveum Ten., is a species for which we have not found any data in the literature, so this work represents the first study on the chemical composition and biological activities of this species.
Limitations of the Study
The results of this study provide information on the chemical composition and biological activities of three Verbascum species. In addition, the study addresses their potential antioxidant, anti-inflammatory and antimicrobial activities. As mentioned above, the Verbascum species were collected from a single geographical location (Bosilegrad, Serbia), which may lead to regional variations in the composition of the extracts influenced by environmental factors. In addition, only two (although complementary) antioxidant assays (DPPH and BCBT) were used. The inclusion of additional methods such as FRAP or ABTS could provide a more comprehensive understanding of the antioxidant potential of the extracts assessed. The antibacterial activities of extracts vary according to their constituents and usually involve the synergistic action of a few constituents, although some constituents may have independent effects. Notwithstanding that antibacterial efficacy was assessed, the active ingredients and underlying mechanisms were not investigated. In vivo studies are also desirable to confirm the results obtained.
Conclusion
The interest in exploring the untapped potential of medicinal plants is growing worldwide. We investigated three Verbascum species (V. phlomoides L., V. niveum Ten. and V. speciosum Schrad.) for their bioactive compounds and biological activities. This paper is the first detailed study on the composition and activities of the species V. niveum Ten. Generally, our study showed that all three species possess considerable amounts of polyphenols and flavonoids. Polyphenolic acids, iridoids, and verbascoside were the most important secondary metabolites detected. Based on the results obtained, ethanol proved to be the best of the three solvents used. With strong anti-inflammatory activity and solid antioxidant and antimicrobial activities, better against Gram-positive bacteria than Gram-negative bacteria, investigated Verbascum species can be considered a natural source of bioactive ingredients with great potential in the pharmaceutical industry.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251372422 - Supplemental material for Comparative Study of Three Verbascum Species (V. phlomoides, V. niveum and V. speciosum)—Chemical Characterization and Biological Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X251372422 for Comparative Study of Three Verbascum Species (V. phlomoides, V. niveum and V. speciosum)—Chemical Characterization and Biological Activities by Maja Grigorov, Dragana Pavlović, Bojan Zlatković, Anđela Dragićević, Vanja Tadić, Jelena Matejić, Snežana Mladenović Antić, Dušan Ilić and Ivana Nešić in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251372422 - Supplemental material for Comparative Study of Three Verbascum Species (V. phlomoides, V. niveum and V. speciosum)—Chemical Characterization and Biological Activities
Supplemental material, sj-docx-2-npx-10.1177_1934578X251372422 for Comparative Study of Three Verbascum Species (V. phlomoides, V. niveum and V. speciosum)—Chemical Characterization and Biological Activities by Maja Grigorov, Dragana Pavlović, Bojan Zlatković, Anđela Dragićević, Vanja Tadić, Jelena Matejić, Snežana Mladenović Antić, Dušan Ilić and Ivana Nešić in Natural Product Communications
Footnotes
Acknowledgments
The authors express gratitude for the financial support from the Ministry of Science, Technological Development and Innovation of the Republic of Serbia (No. 451-03-136/2025-03/200113 and 451-03-137/2025-03/200113) and an internal scientific project (No. 15) of the Faculty of Medicine, University of Niš.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia (No. 451-03-136/2025-03/200113 and 451-03-137/2025-03/200113) and an internal scientific project (No. 15) of the Faculty of Medicine, University of Niš.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Right
This article does not contain any studies with human or animal participants.
Statement of Informed Consent
There are no human subjects in this article so informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



