Abstract
Epileptic seizures are a consequence of uncontrolled neuronal discharge and demand urgent and prolonged treatment. Due to a lack of potent and low-risk drugs that could prevent seizures, efforts are underway to discover new drugs, and plant metabolites are often considered valuable candidates in this search. Lemon balm (Melissa officinalis L., Lamiaceae) is a world-renowned medicinal plant affecting central nervous system function. In this study, we aimed to examine whether the results from in silico modeling of the interactions of major lemon balm essential oil constituents with GABAA and glycine receptors correspond to in vivo results of the convulsions in mice induced by the application of either pentylenetetrazol (GABAA antagonistic) or strychnine (glycine receptor antagonist). While molecular docking experiments of essential oil constituents to the two receptors demonstrated the possibility of ligand–receptor interactions, no binding poses were scored highly, and as the in vivo effect of the essential oil on convulsions seems to be only moderate, the results of the in silico study seem not to contradict results obtained by in vivo models. Thus, we can say that molecular docking might be a useful tool in initial screening for active constituents of natural product mixtures and the potential prediction of their activity in models in vivo.
Introduction
An epileptic seizure is an abnormal, unregulated electrical outburst that occurs within the gray matter of the cerebral cortex and transiently interferes with normal brain function. An epileptic seizure usually leads to disturbances of consciousness and sensations and causes involuntary movements or convulsions. Seizures could be localized in one part of the brain cortex (ie, partial), or general when they involve synchronous discharge in more cortical regions. The seizures could be in a form of tonic–clonic (grand mal) seizures, subtle jerks of some body part (eg, hand), or loss of postural tone. 1 One isolated epileptic seizure can be provoked in a normal brain with transient stress factors (eg, hypoxia and hypoglycemia) or by applying some neuroactive (neurotoxic) substance. 2
The convulsive poison strychnine (STR) can cause an epileptic seizure by competitive and reversible inhibition of the glycinergic inhibitory neurotransmission at the postsynaptic neuronal sites in the spinal cord and medulla. This results in unchecked reflex stimulation of motor neurons affecting all the striated muscles. 3 The onset of STR poisoning is rapid and ends with terminal medullary paralysis. 4 In some cases, due to prolonged convulsions preceding death, hemorrhages of the heart and lungs, as well as cyanotic congestion from anoxia may be observed. Treatment must be focused on detoxication and gastric lavage, prevention of asphyxiation, and supportive care. 5 Seizures in small animals which are induced by STR can be controlled with pentobarbital and/or muscle relaxants, that is, methocarbamol. In large animals, chloral hydrate, xylazine, or diazepam (DZP) can be used to control seizures.4,6
Pentylenetetrazol (PTZ) is a selective antagonist of the GABAA (gamma-amino buteric acid) receptor chloride ionophore complex, which can cause convulsions by single or repetitive exposure. Its application causes suppression of the function of inhibitory synapses, leading to an increase in neuronal activity. A single high dose of PTZ leads to dystonia, and generalized clonic and clonic–tonic convulsions in experimental animals. 7 Its administration affects numerous brain regions, from which hippocampal formation represents one of the major targets. 8 If administered chronically, it causes hippocampal neuronal atrophy and damage through the process of ferroptosis, apoptosis, and autophagy, followed by astrocytosis.9,10 Prolonged PTZ application causes an alteration in the density and sensitivity of different glutamate receptor sub-types in many parts of the brain. 11
Plant-based remedies are a part of the traditional medicines of many nations, and some originating from the Balkan Peninsula have a particularly long tradition; during the twentieth century, a collection of that knowledge was systematized into a monograph. 12 Numerous plant extracts are suggested to impact central nervous system (CNS) function,13,14 and among them, lemon balm (Melissa officinalis L., Lamiaceae) proved to be one of the most frequently utilized naturally based products. This plant contains a plethora of structurally diverse compounds, and especially valuable and unique are the components of the essential oil (aroma carriers), such as β-caryophyllene, citronellal, geranial, geraniol, and neral. 15 The need for plant remedies and new plant-derived CNS-acting drugs arises from limited availability of antiepileptics that are sufficiently effective and which do not lead to a high number of side effects, especially during prolonged application. 16 Herbal drugs have a significant advantage over synthetic ones since they are readily available and inexpensive.
Some studies investigated the antiagitation/anticonvulsive properties of lemon balm (M. officinalis), but, interestingly enough, the essential oil was only rarely tested. In some works, water or water/alcohol extracts of lemon balm of unknown composition were used, at doses higher than 100 mg/kg, 17 showing that the observed activity could potentially involve GABA transmission. Water/alcoholic extracts of lemon balm were also found to cause a significant dose-dependent reduction in both excitatory and inhibitory transmission, with a net depressant effect on neurotransmission that, in contrast to the classical GABA antagonist picrotoxin, evokes profound epileptiform burst firing. 18
Having in mind the previously mentioned findings, the present study aimed to examine the anticonvulsive potential of lemon balm (M. officinalis) essential oil and its main constituent citronellal in two models targeting the GABAergic and glycinergic neurotransmissions. In an effort to unveil potential connections of in vivo observed activities with events at the molecular level, an initial structure-based virtual screening for affinity toward the mentioned neurotransmitter receptors was performed on a library containing the most abundant essential oil constituents.
Materials and Methods
In Silico Docking Study
Molecular docking was performed using the GOLD software package (2020.3 CSD (Cambrige structural data base) release, Cambridge Crystallographic Data Center, Cambridge, UK).19,20 Docking was performed using default parameters for population size (100), selection pressure (1.1), number of operations (10.000), number of islands (1), niche size (2), operator weights for migrate (0), mutate (100), and crossover (100) were applied. Default genetic algorithm settings were used for all calculations, and the ChemPLP scoring function was employed. Three-dimensional structures of ligands at physiological pH were acquired from the ZINC database 21 as MOL2 files, while structures of the receptors were obtained as PDB files from the PDB.
Drugs and Chemicals
In this study, the administered essential oil of M. officinalis was obtained from a commercial source (Siempreviva, Niš, Serbia), as well as citronellal (Sigma-Aldrich, USA). Detailed analysis of the oil was made using GC and GC-MS employing a Hewlett-Packard 6890N gas chromatograph equipped with a fused silica capillary column Optima 5MS ((5%-diphenyl-) 95%-dimethylpolysiloxane, 30 m × 0.25 mm, film thickness, Macherey-Nagel, Germany) and coupled with a 5975B mass selective detector from the same company. The conditions of the analysis and constituent identification/quantification are presented in detail elsewhere. 22 Diazepam, an injection solution, used as the standard anxiolytic, was purchased from Galenika (Belgrade, Serbia). Both PTZ and STR were obtained from Sigma-Aldrich (St. Luis, USA).
Animals and Housing
Male BALB/c (Bagg and Albino c) mice aged 8 to 10 weeks, weighing 20 to 25 g, were used in this study. These animals were housed, nurtured, and fed in the vivarium at the Medical Faculty in Niš, Scientific Research Center for Biomedicine. Prior to and during the experiment, animals were kept under standard laboratory conditions of temperature (25 ± 2° C) and humidity (50 ± 15%), at equal, 12-hour, day–night intervals. During this period, food and water were freely available. The control of their health condition and welfare of the animal was performed daily. All efforts were made to minimalize the number of animals and to reduce their suffering during the experiment. The experiments were approved by the local Ethics Committee and by the Ethics Committee of the Republic of Serbia (323-07-06862/2016-05/1).
Pentylenetetrazol-Induced Convulsions
The anticonvulsant action mediated through GABAA receptors was studied using PTZ (70 mg/kg), which was administered intraperitoneally (i.p.) 1 hour after either the essential oil of M. officinalis (12.5 mg/kg) or citronellal (12.5 mg/kg). In the positive control group, animals received DZP at a dose of 1 mg/kg, while in the negative control group animals received the vehicle (olive oil) at a dose of 10 mL/kg. The onset of general convulsions (in seconds after PTZ injection) was noted to be the time when the animal's bodily muscles were rapidly and repeatedly contracting and relaxing, which is followed by a transitory loss of righting reflex. The occurrence of HLTE in seconds was also recorded. 23
Strychnine-Induced Convulsions
The protocol for the animal treatment was the same as in the previous experimental groups, apart from the convulsive agent, which, in this case, was STR applied i.p. at a dose of 2 mg/kg. 24 Immediately after the application of STR, the animals were monitored for the occurrence of general convulsions (in seconds).
Statistical Analysis
The obtained results from the in vivo anticonvulsive tests were presented as mean ± SD. Significant differences were determined by one-way analysis of variance followed by Tukey's post hoc test for multiple comparisons (Graphpad Prism version 8.0, San Diego, CA, USA). Probability values (P) of <.05 were considered to be statistically significant.
Results and Discussion
There is a global trend in the search for CNS-acting medicinal plants, and the investigation of ones with potential anticonvulsant properties is not uncommon. 25 Although some plants are considered to be of ethnomedicinal esteem, there is a strong need for biological evaluation and their scientific validation, and in past decades some efforts were made in this direction.26,27 Apart from beneficial properties, a number of plants, when ingested, have the potential of inducing generalized tonic–clonic convulsions, in both humans and laboratory animals. Such an example is the traditional Chinese herbal drug nux vomica, dried seeds of Strychnos nux-vomica L. (Loganiaceae), which contains STR and brucine. 28
Different plant-derived molecules (eg, bicuculline and picrotoxin) were used throughout the years to help understand the role of ionotropic GABAA receptor function and structure. 25 We are currently aware that various plants and food sources (vegetables, fruits, meat, and wine) possess brain–blood barrier permeable substances that could alter the activity of this receptor 25 and we refer to such food as a functional food.
Despite limitations and a level of caution needed when interpreting quantitative outputs, molecular docking remains a frequently used in silico method which offers the possibility to predict whether a small molecule (ie, ligand) is able to bind to a macromolecule (ie, receptor), and score such interactions so as to assess the relevance of predicted binding modes. 29 One of its obvious advantages is the ability to assess ligand–receptor interactions efficiently for entire compound libraries and is commonly a starting point when investigating biological activities of natural product mixtures. 30
Based on our previous findings, 31 a library of compounds containing the ten most abundant lemon balm essential oil constituents was prepared. Molecular docking experiments were performed in the software package GOLD, using the empirical ChemPLP scoring function.
A crystal structure of an α1β2γ2 form of GABAA bound to picrotoxinin was recently reported, 32 and could thus be of use as a template to probe the binding of the major essential oil constituents to this binding site. Considering their importance in hyperekplexia, epilepsy, and chronic pain, we identified glycine receptors (GlyR) as another possible biological target of the essential oil constituents. Similarly, we performed docking experiments to investigate the potential affinity of the major constituents toward this receptor, using the previously reported crystal structure of pentameric α3 GlyR bound to STR, a known competitive antagonist of this receptor (Protein Data Bank [PDB] No. 10.2210/pdb5CFB/pdb). The results of the docking experiments are presented in Table 1.
GOLD Docking Scores of the Essential Oil Constituents for α1β2γ2 GABAA and α3 GlyR.
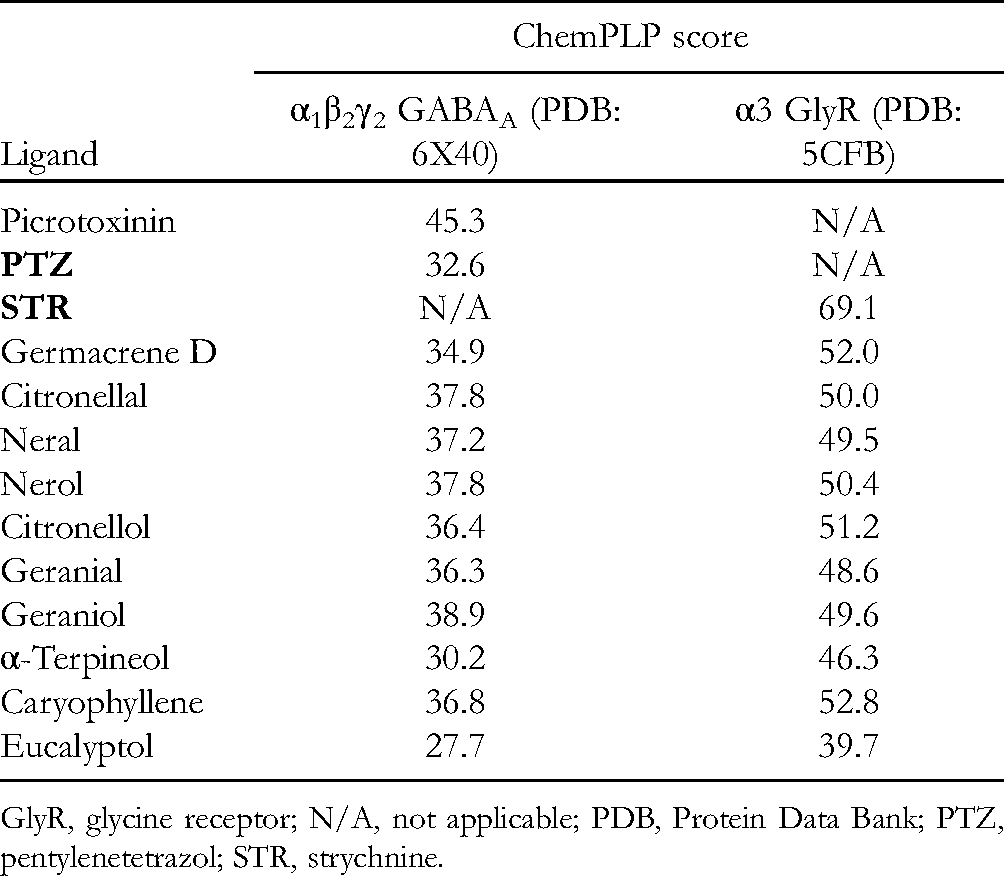
GlyR, glycine receptor; N/A, not applicable; PDB, Protein Data Bank; PTZ, pentylenetetrazol; STR, strychnine.
As expected, the ChemPLP score of picrotoxinin, a well-known receptor antagonist, re-docked to its binding site in α1β2γ2 GABAA was the highest. Nevertheless, ChemPLP scores for both PTZ and the essential oil constituents, although positive and indicating binding could occur, were significantly lower. Such a result for PTZ is in agreement with the previous findings that picrotoxinin and PTZ most probably interact with overlapping but distinct domains of the GABAA receptor. 33
In the case of docking to the active site of GlyR, the re-docking of STR was successful, indicated by its high ChemPLP score. Most of the essential oil constituents scored approximately 20 points below this value with no compound standing out in particular, implying that binding is possible, but the affinity of these molecules toward GlyR is not expected to be very high.
Having put forth the aim to examine the anticonvulsive potential of lemon balm essential oil and its main constituent citronellal in in vivo tests, we found it important to perform an initial detailed Gas chromatography–mass spectrometry (GC-MS) analysis of the utilized essential oil; for this purpose, a sample of the oil was obtained from a local producer (Niš, Serbia). The analysis of this oil revealed the presence of 81 essential oil constituents that represented 99.4% of the total detected GC-peak areas (Supplemental Table S1). The dominant constituents with more than 20% abundance in the sample were citronellal (22.2%) and geranial (23.8%). The chemical composition of the tested sample of M. officinalis essential oil was qualitatively and quantitatively comparable to the one used in our previous analyses and evaluated in several animal models.22,31
The model of PTZ-induced convulsions is believed to be adequate for studying convulsions that are similar to those observed in humans with generalized myoclonic and/or absence seizures. 34 Convulsions induced by PTZ were found to occur statistically significantly later in animals treated with M. officinalis essential oil (12.5 mg/kg) when compared to the vehicle-treated mice (Figure 1A). Interestingly, citronellal applied at the same dose did not influence the time of the PTZ-induced convulsion occurrence (Figure 1A). Both the essential oil and citronellal had no influence on the hindlimb tonic extension (HLTE) occurrence (Figure 1B). DZP (1 mg/kg), used as a positive control drug, prolonged the time of the PTZ-induced convulsion occurrence (Figure 1A), as well as the time of HTLE appearance (Figure 1B). When the effects of M. officinalis essential oil and citronellal were mutually compared, no significant differences in their effects were found (Figure 1A and B).
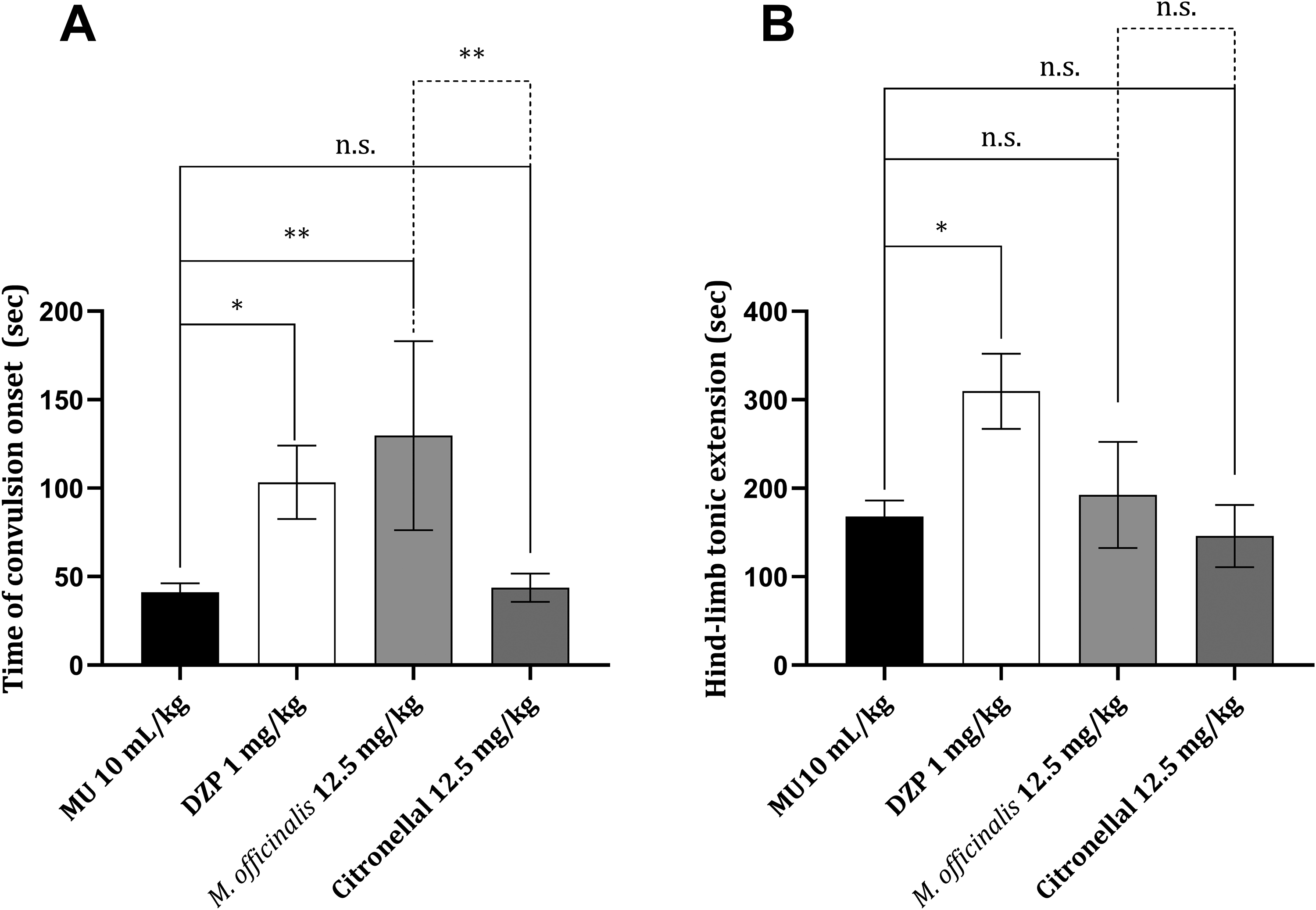
Effect of the vehicle, diazepam (1 mg/kg), Melissa officinalis (12.5 mg/kg) essential oil, and citronellal (12.5 mg/kg) on the onset of convulsions (A) and time needed for the onset of hind-limb tonic extension (B) induced by pentylenetetrazol (70 mg/kg). Data are given as mean ± SD; the comparison was made using one-way analysis of variance followed by Tukey's post hoc test. *P < .001, **P < .01 versus vehicle-treated animals; n.s., no significant difference.
It was previously shown that the water extract of M. officinalis applied at a wide and relatively high-dose range (>100 mg/kg) produced a reduction in PTZ-induced seizures, and at the same time reduced the time of seizure onset. 17 These results are difficult to compare with the findings of our study since the type of extract (essential oil vs water extract) and the doses applied (differing ca. 10 times compared to the lowest one of the water extract 17 ) are quite different. However, on a general notion, both the essential oil and the water extract 17 of M. officinalis were found to be capable of preventing PTZ-induced seizures. Certain studies demonstrated unclear and/or weak results regarding the impact of the whole plant methanolic and aqueous extract on GABAA receptor binding of flumazenil. 35 A recent study showed that the essential oil of M. officinalis reduces hippocampal neuron firing through the inhibition of the voltage-gated sodium channel. 36
The occurrence of HLTE is often used as an endpoint for the quantification of PTZ-induced convulsions and is described when an angle of the hindlimbs to the torso exceeds 90°. 37 In the case of DZP-treated mice, as well as those from the control-treated and citronellal-treated groups, the occurrence of HLTE was postponed from the initial convulsion for almost double the time needed for convulsions to occur (Figure 1B). Interestingly enough, the time of HLTE occurrence in mice treated with M. officinalis essential oil was only slightly postponed compared to the initial convulsion (Figure 1B).
Application of M. officinalis essential oil at a dose of 12.5 mg/kg statistically significantly reduced the time of the convulsion occurrence in mice treated with STR (Figure 2). On the other hand, the same dose of citronellal failed to significantly reduce the convulsion occurrence in STR-treated mice. Interestingly, although the effects of the oil and citronellal were different when compared to the vehicle-treated mice, there was no significant difference between these two groups (Figure 2). A standard drug DZP, at a dose of 1 mg/kg, significantly prolonged the occurrence of convulsions induced by STR application (Figure 2).

Effect of the vehicle, Melissa officinalis (12.5 mg/kg) essential oil, and citronellal (12.5 mg/kg) on the onset of convulsions induced by strychnine (2 mg/kg). The data are given as mean ± SD, and the comparison was made using one-way analysis of variance followed by Tukey's post hoc test. *P < .01 versus vehicle-treated animals; n.s., no significant difference.
The results of the molecular docking studies are in accordance with the results obtained during in vivo experiments, as the essential oil and citronellal had a poor impact on the convulsion occurrence (Figure 2).
Patients suffering from chronic epilepsy are known to develop anxiety disorders and insomnia 38 ; thus, M. officinalis, which is used for the treatment of such disorders by psychiatric patients from Serbia, 14 could be very useful. The usefulness of such drugs is known previously since DZP is widely used for the treatment of anxiety disorders and insomnia, and also possesses anticonvulsant properties. 39 One desirable effect of anticonvulsant drugs is myorelaxation, which DZP is capable of inducing; however, we showed that M. officinalis essential oil at doses from 25 to 100 mg/kg increases animal movement in the open field test, while citronellal at a dose of 12.5 mg/kg decreases animal movement in the same test. 22 Neither M. officinalis essential oil nor citronellal application produced muscle relaxation in the horizontal wire. 22 That is why M. officinalis remains a potential candidate for the treatment of various CNS disorders.
Conclusions
As with all highly valued medicinal plants, there is a clear need for objective evidence of various aspects of their biological activities. This work demonstrates evidence that M. officinalis essential oil moderately reduces the PTZ-induced and STR
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221139961 - Supplemental material for Lemon Balm (Melissa officinalis) Essential Oil Moderately Affects Strychnine-Induced and Pentylenetetrazol-Induced Convulsions —In Silico and In Vivo Studies
Supplemental material, sj-docx-1-npx-10.1177_1934578X221139961 for Lemon Balm (Melissa officinalis) Essential Oil Moderately Affects Strychnine-Induced and Pentylenetetrazol-Induced Convulsions —In Silico and In Vivo Studies by Nikola M. Stojanović, Ivan Rajković, Vidak Raičević, Milan N. Petković, Pavle J. Randjelović and Niko S. Radulović in Natural Product Communications
Footnotes
Acknowledgments
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (grant numbers 451-03-68/2022-14/200113 and 451-03-68/2022–14/200124). This publication is a part of Nikola M. Stojanović's Ph.D. thesis, under the supervision of Pavle J. Randjelović.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja (grant numbers 451-03-68/2022-14/200113 and 451-03-68/2022–14/200124)
Ethical Approval
The experiments were approved by the local Ethics Committee and by the Ethics Committee of the Republic of Serbia (323-07-06862/2016-05/1).
Statement of Human and Animal Rights
All experimental procedures with the animals were conducted in compliance with the Declaration of Helsinki and European Community guidelines for the ethical handling of laboratory animals (EU Directive of 2010; 2010/63/EU).
Supplemental Material
Supplemental material for this article is available online.
Statement of Informed Consent
Not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
