Abstract
In the present study, we aimed to evaluate the therapeutic efficacy of β-
β-
Materials and Methods
M2000 Preparation
β-
Animal Selection and Grouping
In the current experiment, 30 male Wistar rats, weighing 250-300 g and aged 10 weeks, were used. They were divided into 3 groups randomly (10 rats in each group): I—normal group (no kindled seizures induction and no treatment), II—control group (kindled seizures induction only), and III—M2000 group (kindled seizures induction and intraperitoneal injection of M2000). The rats in the M2000 group were intraperitoneally pretreated with a daily dose of 50 mg/kg M2000 before the study. During the study period, the same dose of M2000 was injected 30 minutes before each pentylenetetrazole (PTZ) injection. Rats were injected with an equal volume of normal saline in the control group. Final equal volume of saline and PTZ as the vehicle were used for the injection of rats in the normal group. In all groups, injections were performed for 14 times and at 30-minute intervals. For adaptation, all rats were housed in cages under 12 hours light-dark cycles for 2 weeks, and the same meal plan was used for all rats. All experiments were performed between 10.00 and 12.00 a.m. in a silent room, at a temperature of 22-24°C. The experimental procedures were approved by the ethical committee of Alborz University of Medical Sciences (IR.ABZUMS.REC.1397.077) based on international guidelines on the use of laboratory animals.
Induction of Kindled Seizures
PTZ (Sigma) was dissolved in sterile isotonic saline. Kindling was induced by a total of 14 injections with 37.5 mg/kg PTZ intraperitoneally on every second day. Rats were observed for 30 minutes after the last PTZ administration; after an additional 30 minutes, the rats were observed for lethality before returning to the cage. Seizure intensity was evaluated using the following scale 15 : stage 0, no response; stage 1, hyperactivity and vibrissae twitching; stage 2, head nodding, head clonus, and myoclonic jerk; stage 3, unilateral forelimb clonus; stage 4, rearing with bilateral forelimb clonus; and stage 5, generalized tonic-clonic seizure with loss of postural control. The animals were considered to be kindled after having received 14 PTZ injections and having reached at least 3 consecutive stage 4 or 5 seizures.
Sample Preparation
The animals were sacrificed and the brains were removed and cleaned with ice-cold (4°C) saline. Each brain tissue samples were divided into 2 lobes. One lobe was stored at −70°C and used for gene expression analysis. Another lobe was stored at 10% formalin and used for histopathology.
Histological Assessment
Rats were sacrificed following anesthesia, and the hippocampus from normal, kindled seizures induction, and M2000-treated rats were removed and fixed in neutral 10% formalin, embedded in paraffin, sectioned (8 µm thick), and then stained with hematoxylin-eosin (H&E). For histopathological evaluation, calcification, dead neurons, degeneration, perivascular cuffing, hypercellularity, spongiosis, and counts of meningeal and parenchymal inflammatory cell infiltration were examined. A histopathologist analyzed all sections.
RNA Analysis for Cytokines
For gene expression analysis, brain tissues were removed and stored in RNAlater (Qiagen). Total ribonucleic acid (RNA) was isolated using RNeasy Lipid Tissue Mini Kits (Qiagen, Hilden, Germany) following a standard protocol. Total RNA was reverse transcribed into complementary deoxyribonucleic acid (DNA) by Takara kits (Takara, Japan) according to the manufacturer’s instructions. The expression levels of IL-6, TNF, chemokine (C-C motif) ligand 2 (CCL-2), cyclooxygenase-2 (COX-2), IL-10, interferon-gamma (IFN-γ), and IL-1β were measured by quantitative reverse transcription-polymerase chain reaction (RT-PCR) using SYBR Green PCR Master Mix (Takara, Japan) with specific primers (Table 1) and were normalized relative to the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) level.
Primer Sequences.

IL, interleukin; CCL-2, chemokine (C-C motif) ligand 2; COX-2, cyclooxygenase-2; IFN-γ, interferon gamma; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; TNF-α, tumor necrosis factor-alpha.
Statistical Analysis
Statistical analysis was performed using a commercially available software package (SPSS Statistics 22.0.0, SPSS, Chicago, IL, USA). A 1 sample Kolmogorov-Smirnov test was applied to determine whether data followed a normal distribution. Parametric and non-parametric analyses were performed based on the findings of this evaluation. A P-value <0.05 was considered statistically significant.
Results
Clinical Findings
PTZ-induced kindling intensity
To evaluate the effect of M2000 on seizures and epilepsy, we used the PTZ-induced kindling model on Wistar rats. Our findings showed that M2000 pretreatment prone rats to epilepsy immediately following the first PTZ injection (mean ± standard deviation [SD] for seizure stage in the M2000 group was 3.12 ± 1.55 vs 0.75 ± 1.03 for control, P = 0.004). As illustrated in Figure 1, M2000 at doses of 50 mg/kg were able to increase significantly PTZ-induced seizures. Statistical analysis of results from the first to the fourth injection indicates that there were significant differences in the seizure intensity between the M2000 group and the control group (P < 0.05).

Effect of M2000 pretreatment on the pentylenetetrazole-induced kindling intensity. n = 10 in each group. *P < 0.05 and **P < 0.01 indicate significant differences as compared with control group. To compare significant differences, independent-samples t-test was performed.
As illustrated in Figure 2, pretreatment of animals with M2000 (50 mg/kg) has a significant effect on the latency that the rats reach phase 2 and 5 seizures. The mean (±SD) on the latency phase 2 and 5 seizures was lower in the M2000 group compared with the control group (79.4 ± 30.7 vs 133.0 ± 38.4, P < 0.001, and 126.1 ± 27.6 vs 233.3 ± 142.8, P = 0.001, respectively). The mean (±SD) days needed to reach the kindled state was lower in the M2000 group compared with the control group (3.25 ± 0.86 vs 7.62 ± 0.90, P = 0.003) (Figure 3). Moreover, the mean (±SD) duration of phase 5 seizures was significantly higher in the M2000 pretreated group compared with the control rats (344 ± 54 vs 197 ± 94, P < 0.001) (Figure 4).

Effect of M2000 (50 mg/kg) on the latency of arriving to phase 2 and 5 of seizure. n = 10 in each group. *P < 0.01 was statistically significant. To compare significant differences, independent-samples t-test was performed.

Effect of M2000 (50 mg/kg) on the days needed to reach the kindled state. n = 10 in each group. *P < 0.01 was statistically significant.
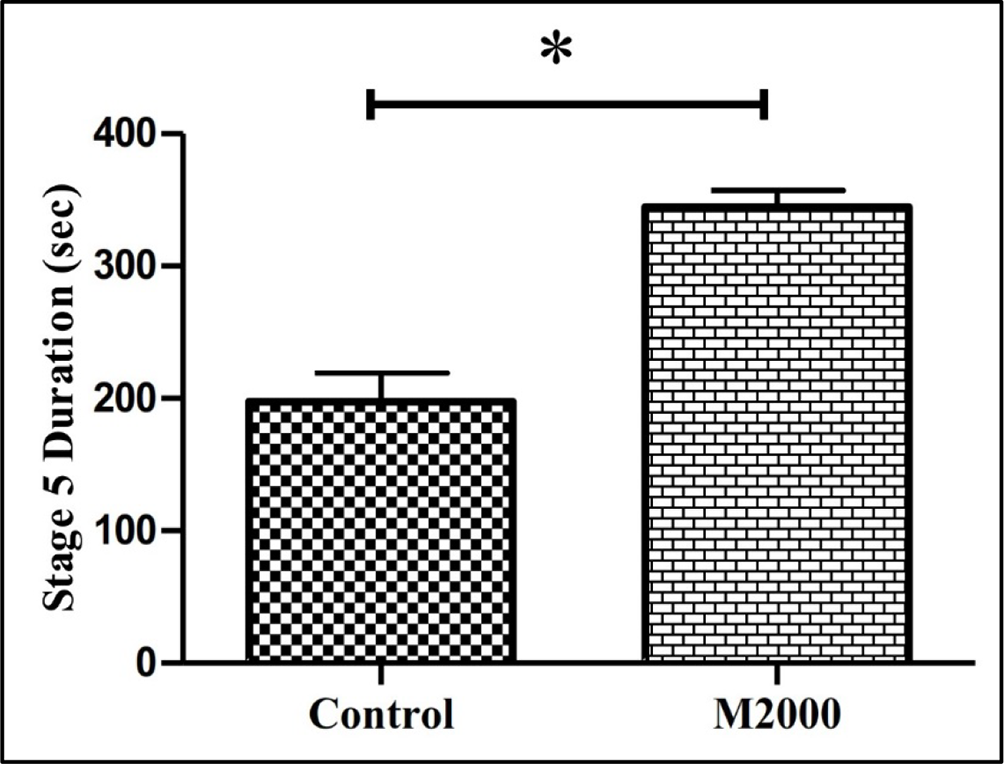
Effect of M2000 (50 mg/kg) on the remaining time in the phase 5 of seizure. n = 10 in each group. *P < 0.001 was statistically significant. To compare significant differences, independent-samples t-test was performed.
Histological Findings
Histological analysis was performed by H&E staining on the hippocampus. For each tissue sample, at least 5 sections were prepared, and for each section, 10 microscopic fields were assessed based on conventional criteria in literature. An expert pathologist scored all sections by light microscopy, and his report illustrated that cystic degeneration, perivascular cuffing congestion, and dead neurons in PTZ-induced kindling rats treated by M2000 were equal to the control group. However, these findings were not seen in the normal group (Figure 4).
The Messenger RNA Expression of Cytokines, Chemokine, and COX-2
The transcription levels of cytokine, chemokine, and COX-2 genes in the brain of rats were evaluated with RT-PCR. The expression of IL-1β, IL-6, TNF-α, IFN-γ, and COX-2 genes was higher in the PTZ-induced kindling than in the M2000 and normal group; however, the differences were not statistically significant. Moreover, the expression level of IL-10 was higher in the brains of rats in M2000 vs control and normal groups; however, the differences were not statistically significant (Figure 5).

Representative optical microscopy images (×400) of the brain slices (hippocampus) in different groups. (a) Normal group displaying normal histological structure of the hippocampus. (b and c) Control and M2000 groups displaying neuronal necrosis and vacuolation (arrow) of the hippocampus.
Discussion
To investigate antiepileptic drugs, kindling is a widely used animal model for the development of seizures either by chemical or electrical subconvulsive stimulus. In this context, chemical-induced kindling seizures produced by PTZ agent is a useful approach for stimulating central nervous system and as previously described, injection of PTZ as a non-competitive antagonist of GABA receptor channels develops epileptic seizures. 16,17 In the present study, the treatment of rats with M2000 decreases latency with increasing the intensity and duration of PTZ-induced seizure. In the M2000 treated group, reduced latency was observed to reach phase 2 and 5 seizure while phase 5 seizure significantly lasted longer. Although the effect of M2000 in PTZ-induced seizure model or its impact on GABA receptor channels has not been previously studied, the epileptogenesis property of M2000 might be due to the synergistic effect of PTZ in blocking GABA receptor channels or the consequence of its individual effect on neuronal ion channels and glutamate receptors. Taking into account that administration of M2000 prior to PTZ injection might change the kindling protocol by enhancing PTZ effect, the addition of experimental group including treatment of rats with M2000 after observing insult from PTZ-induced kindling also deserves attention and further strongly is suggested while this is a major limitation of our study.
A growing body of research has indicated that brain inflammation plays an important role in epilepsy. 5 Stroke, traumatic brain injury, and infections in the central nervous system may lead to epilepsy through inflammatory responses, neuronal loss, or plasticity. 18 During epilepsy, the permeability of affected BBB stimulates invasion of proinflammatory cytokines into the brain while this further progresses epileptogenesis. 19 In epileptic brain tissue, microglia activation changes their function including modification of glutamine/glutamate cycle or alteration of glutamate receptor expression. 20,21 Further, the inflammatory response caused by the activation of the glial cells in brain tissue alters neuronal excitability through stimulation of the intracellular signaling pathway. Various studies have proved that this alteration perturb the interaction between neurons and glial cells and could result in neuronal death or seizures exacerbation. 22 -24 Hence, targeting inflammatory responses in the brain with anti-inflammatory molecules can be a potential therapeutic approach in an epileptic seizure. In our laboratory, the anti-inflammatory and immunosuppressive properties of M2000 has been investigated. Briefly, in a clinical trial, treatment with M2000 effectively reduced expression levels of COX-1/COX-2 genes in ankylosing spondylitis (AS) patients. This protein is a crucial enzyme in the initiation of inflammatory pathways in AS patients. 25 In other clinical studies, oral administration of M2000 significantly decreased IL-17) and RORγt levels in patients with rheumatoid arthritis. 26 The IL-17 family cytokines are potential inducers for inflammation and contribute to the tissue damage in various chronic inflammatory and autoimmune disorders. 27 In this study, the effect of M2000 administration was assessed by histopathological observation of brain tissues for inflammatory cell infiltration and neuronal death in the PTZ-induced kindling model. Histopathological observation on the hippocampus as the major component of the brain that plays important roles in episodic memory and spatial cognition revealed no differences in inflammatory cells infiltration among control, normal, and M2000 treated groups. Similar histopathological findings including cystic degeneration, perivascular cuffing congestion, and dead neurons were observed in all groups. Furthermore, the gene expression levels of inflammatory cytokines (IL-1β, IL-6, TNF-α, and IL-10), CCL-2, and COX-2 were measured (Figure 6). Slightly elevated expression levels of IL-1β, IL-6, TNF-α, IFN-γ, and COX-2 were seen in the PTZ-induced kindling group comparing with M2000 treated and normal groups, while the slightly elevated expression level of IL-10 was observed in M2000 treated group vs control and normal groups. However, the observed differences among the expression levels of the immune mediators were not statistically significant. These findings have shown that our PTZ-induced kindling model could not induce adequate inflammation response in the tested groups. Further, due to the limited volume of the samples, gene expression analysis was performed in whole 1 lobe of the animal brain while examining the specific brain region like the hippocampus could have increased the value of our study. Despite several previous studies that showed M2000 has anti-inflammatory and immunosuppressive properties, our results demonstrate the potential proconvulsant effect of M2000 treatment prior to PTZ injection may be masking the beneficial effects of this NSAID in our animal model of epileptogenesis. In addition, we realize that M2000 might even exacerbate epilepsy, presumably by blocking the GABA receptor channels or excessive glutamate release. Further experimental study including behavior experiments and assessment of long-term potentiation will be required to unveil the molecular details of epileptogenesis property of M2000.

The messenger ribonucleic acid expression of cytokines, chemokine, and cyclooxygenase-2 (COX-2) in control, M2000, and normal groups. Slightly elevated expression levels of interleukin (IL)-1β, IL-6, tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ) and COX-2 were seen in the pentylenetetrazole-induced kindling group comparing with M2000 treated and normal groups. Slightly elevated expression level of IL-10 was observed in M2000 treated group vs control and normal groups. The measured differences were not statistically significant. To compare significant differences, Mann–Whitney U test was performed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by CinnaGen Medical Biotechnology company, under Grant No. FNP0245010092.
