Abstract
Introduction
The genus Salvia L. is one of the famous aromatic genera of the Lamiaceae, represented by more than 900 species distributed worldwide in the temperate, subtropical, and tropical zones.1,2 Salvia officinalis subsp. lavandulifolia (Vahl) Gams (synonym: S. lavandulifolia Vahl) is a small woody perennial shrub that grows in the Americas, Europe, and Asia. 1 It is also found in the mountains of eastern Spain, southern France, and the northern part of Africa and is cultivated widely in the Mediterranean area. 3 The plant can reach up to 100 cm in height and is characterized by its dense branches, wiry stems, opposite green or gray-white leaves, and vivid blue-purple flowers. 4
Similar to other Salvia species, S. lavandulifolia is recognized for its medicinal and economic value. Traditionally, S. lavandulifolia was used as gargles for mouth ulcers, common cold, sore throat, premenstrual syndrome (PMS), and menopause. 1 It was also utilized as a food preservative and a flavoring spice. The leaves were used orally as a choleretic and topically as an antiseptic drug. 5 The aqueous extract was hypoglycemic when taken orally. 3 Due to its medicinal value, Spanish sage was subjected to several phytochemical and pharmacological studies. The essential oil (EO) of S. lavandulifolia is responsible for its distinctive flavor and aroma and is usually obtained by distillation. 6 Flavonoids, monoterpenoids, and triterpenes were reported from the aerial parts of the plant. 1 S. lavandulifolia EO demonstrated moderate anti-inflammatory activity (IC50 = 30 μg/mL) 1 ; moderate cytotoxicity against human leukemia (HL60), myelogenous leukemia (K562), breast (MCF7), and ovarian (A2780) cancer cell lines 7 ; antioxidant 4 ; in vitro acetylcholinesterase inhibitory activity 8 ; antiseptic, analgesic and sedative properties 9 ; weak antimalarial activity against Plasmodium falciparum 1 ; acaricidal toxicity against synanthropic mites 10 ; strong antifeedant effects against Leptinotarsa decemlineata, Spodoptera littoralis, Myzus persicae, and Rhopalosiphum padi 11 ; phytotoxic activity against Lactuca sativa and Lolium perenne 11 ; and antifungal activity against Fusarium sp. 11 and Candida albicans. 12 In a placebo-controlled, double-blind trial, acute administration of a standardized EO extract of S lavandulifolia improved the immediate word recall scores in healthy young adult volunteers. 9 Another placebo-controlled, double-blind demonstrated positive acute mood and cognition modulation of a standardized EO extract of S. lavandulifolia in 24 healthy young adults. 6 In a recent randomized cross-over, placebo-controlled, and double-blind study, acute administration of Salvia mixture (400 mg of S. officinalis aqueous extract and 200 mg S. lavandulaefolia EO) encapsulated with acacia gum improved cognitive functions (perceived exertion, working memory, and reaction time) in athletes performing a fatiguing cycling task. 13
There are great variations in the reported major components of S. lavandulifolia EO and their percentages in the literature, which can account for the variations in the biological activities of the EO. 1 These variations also suggest the presence of multiple chemotypes. Therefore, the current study aimed to investigate the composition and enantiomeric distribution of S.lavandulifolia EOs from Spain over the course of 5 years. A hierarchical cluster analysis based on the EO compositions from this study and from previous reports was also carried out. This study also reports the enantiomeric ratios of the chiral components of Spanish sage EO.
Results and Discussion
S. lavandulifolia EO Composition
Eleven hand-harvested S. lavandulifolia samples were steam distilled for 3 to 4 h. The average yield was 2.0% to 2.5% (volume per mass fresh plant material). The aroma could be described as camphoraceous, sharp, slightly sweet, 1,8-cineole-like, and slightly herbaceous. The EOs were analyzed by gas chromatography–mass spectrometry (GC-MS) and gas chromatography–flame ionization detection (GC-FID). A total of 89 compounds were identified representing 99.8% to 100.0% of the total EO compositions. Monoterpenoid hydrocarbons, oxygenated monoterpenoids, sesquiterpene hydrocarbons, and oxygenated sesquiterpenoids accounted for 22.3% to 29.1%, 69.1% to 75.8%, 1.5% to 2.6%, and 0.2% to 0.5%, respectively. S. lavandulifolia EOs were mainly made of 1,8-cineole (24.3%-34.0%), camphor (23.5%-28.8%) followed by camphene (4.9%-6.4%), and α-pinene (4.3%-6.6%) (major components, Table 1; complete composition, Supplementary Table). The tested samples were in agreement with the ISO 3526 standard for Spanish sage oil. 14
Major Components in the Essential Oils (EOs) of Salvia lavandulifolia (Spanish Sage) Cultivated in Murcia, Spain.

The major components are the top 16 based on percent composition (average > 0.8%).
Abbreviation: RI, retention index.
The EO compositions in this study show similarities and differences from previous reports on Spanish sage EOs. Cutillas et al reported camphor, 1,8-cineole, camphene, and α-pinene as the main components of the EO of S. lavandulifolia cultivated in Murcia (Spain). 15 S. lavandulifolia EOs from France were mainly made of 1,8-cineole (25.5%), camphor (39.0%) and linalyl acetate (10.2%). 7 Usano-Alemany et al. compared the composition of the steam-distilled S. lavandulifolia EOs in different seasons and revealed an important seasonal variation. The major compounds of the EOs were 1,8-cineole (21.4%--33.8%), limonene (5.6%-10.4%), α-pinene (10.5%-17.5%), β-pinene (6.0%-17.3%), myrcene (trace-10.0%), camphor (6.1%-9.4%), and (E)-β-caryophyllene (4.0%-8.5%). 16 The main components of S. lavandulifolia EO from Castilla–La Mancha, Spain were α-pinene (23.2%), β-pinene (19.2%), limonene (16.6%), (E)-β-caryophyllene (8.1%), caryophyllene oxide (5.6%) and viridiflorol (9.7%). 17 On the other hand, β-phellandrene (9.3%), terpineol (12%), and ledol (11%) were the main components of the EO from Solsona, Lerida, Spain. 18 Interestingly, 1,8-cineole (15.5%-55.1%) was the most abundant compound in S. lavandulifolia EO cultivated in Cuenca, Spain. 19 The air-dried aerial parts of wild S lavandulifolia collected from Morocco had camphor (16%-31%), 1,8–cineole (13%-19%), β-pinene (8%-13%), camphene (5%-11%), and viridiflorol (0%-12%) as the major components. 5 Similarly, wild S. lavandulifolia EOs have shown camphor (15.3%), 1,8-cineole (15.0%), α-pinene (11.3%), β-pinene (8.5%), and limonene (7.5%) as the main constituents. 20 The EO obtained from S. lavandulifolia from Lublin, Poland contained 1,8-cineole (21.33%), camphor (19.03%), β-pinene (16.15%), thujone (13.08%), and α-pinene (7.22%). 2 (E)-β-Caryophyllene (11.87%), spathulenol (8.13%), neomenthol (7.75%), pulegone (6.97%), hexadecanoic acid (6.85%), and germacrene-D (5.70%) were the major constituents of Spanish sage EO from Peru. 1 The EO with acetylcholinesterase inhibitory activity was made of camphor (27%), 1,8-cineole (13%), α- and β-pinene (10%--15%) and bornyl acetate (10%). 8 S. lavandulifolia EO from Brazil contained β-thujone (20.0%), camphor (19.0%), α-thujone (19.0%), 1,8-cineole (8.1%), and β-pinene (4.0%). 21 Commercial Spanish sage EO from Germany had camphor (30.5%), 1,8-cineole (24.8%), α-pinene (6.5%), linalool (4.0%), and linalyl acetate (3.5%) as the main components. 22
Enantiomeric Distribution
The enantiomeric distributions of chiral components of S. lavandulifolia EO are shown in Table 2. The results show a total of 17 chiral compounds, namely α-thujene, α-pinene, camphene, sabinene, β-pinene, limonene, cis-sabinene hydrate, linalool, camphor, terpinen-4-ol, linalyl acetate, borneol, bornyl acetate, α-terpineol, α-terpinyl acetate, (E)-β-caryophyllene, and δ-cadinene. (–)-Linalyl acetate, (–)-bornyl acetate, (+)-α-terpinyl acetate, (–)-(E)-β-caryophyllene, and (–)-δ-cadinene were detected as pure enantiomers. Cutillas et al. reported no pure enantiomers and a prevalence of (+)-limonene, (+)-sabinene hydrate, ( )-camphor, (+)-terpinen-4-ol, (+)-α-terpineol and (+)-α-terpinyl acetate, (–)-camphene, (–)-β-pinene, (–)-linalool, (–)-bornyl acetate, (–)-borneol, and (–)-β-caryophyllene. 15 These identified chiral compounds and their enantiomeric distributions may serve as a benchmark for S. lavandulifolia EO authentication or adulteration detection.
Enantiomeric Distributions of Chiral Compounds Present in the Essential Oils (EOs) of Salvia lavandulifolia (Spanish Sage) Cultivated in Murcia, Spain.

Hierarchical Cluster Analysis
Based on S. lavandulifolia EO compositions, a hierarchical cluster analysis of the EOs from literature4,5,7,16,17,22–27 and from this work was carried out. The dissimilarity index is very small indicating no significant differences in the EO compositions of the tested samples in this work (Figure 1). The cluster analysis including compositions reported in the literature revealed 3 different clusters (Figure 2): Cluster 1 (camphor/1,8-cineole), Cluster 2 (1,8-cineole/α-pinene/β-pinene), and Cluster 3 (1,8-cineole). All samples reported in this work are in the camphor/1,8-cineole chemotype.
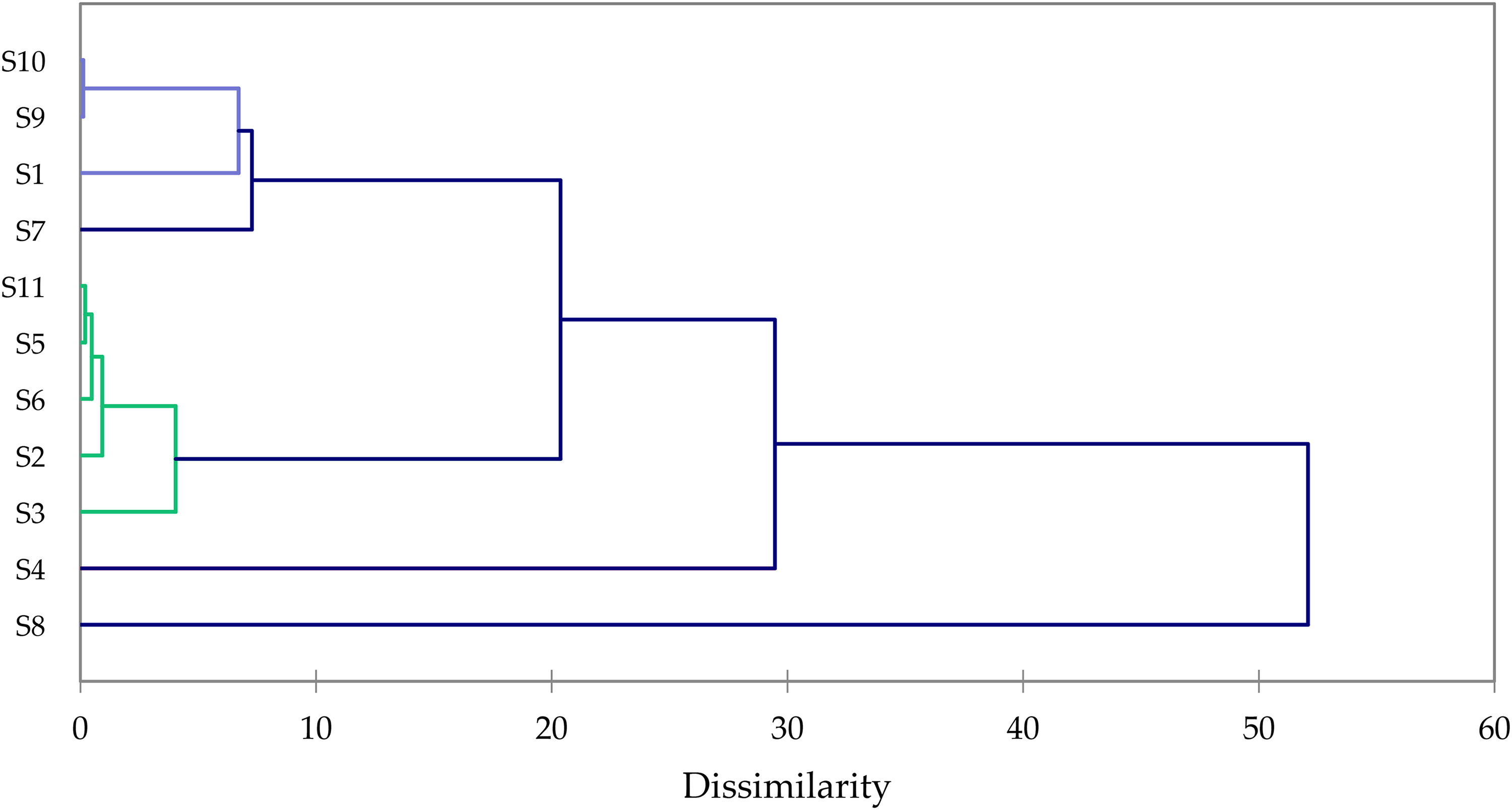
Agglomerative hierarchical cluster (AHC) analysis of Salvia lavandulifolia (Spanish sage) essential oils (EOs) cultivated in Murcia, Spain.

Agglomerative hierarchical cluster (AHC) analysis of Salvia lavandulifolia essential oil (EO) compositions from this work and the literature.
Conclusions
Steam-distilled S. lavandulifolia EOs cultivated in southern Spain showed very similar chemical compositions, with 1,8-cineole (24.3%-34.0%), camphor (23.5%-28.8%) followed by camphene (4.9%-6.4%), and α-pinene (4.3%-–6.6%) as the major components. The enantiomeric distributions of chiral components of S lavandulifolia EO show a total of 17 chiral compounds that can be used in the Spanish sage EO authentication. There was no significant difference in the EO compositions of the tested samples in this work. The cluster analysis revealed 3 different chemotypes for S lavandulifolia: camphor/1,8-cineole chemotype, 1,8-cineole/α-pinene/β-pinene chemotype, and 1,8-cineole chemotype. All samples reported in this work belong to the camphor/1,8-cineole chemotype.
Experimental
EO Extraction
Cultivated Spanish sage samples were collected from the hills and mountains around Totana, Murcia, in southern Spain about 600 m above sea level from August to November of 2016 to 2021. The plant samples were handpicked and identified by Mr. Juan Lorente (Figure 3). The fresh aerial parts of Spanish sage samples were steam distilled for 3 to 4 h in industrial scale stills that take up to 1000 kg of plant material. A separate steam generator connected by ducts was used to generate the steam. The steam reached a pressure of 0.4 to 0.7 Mpa and a temperature of 150 to 170 °C. EO samples were numbered from S1 to S11. S1 and S2 are samples collected between August and November 2017, respectively. S3 and S4 are samples collected between August and November 2018, respectively. S5 and S6 are samples collected between August and November 2019, respectively. S7 and S8 are samples collected between August and November 2020, respectively. S9 to S11 are samples collected between August and November 2021, respectively.

Spanish sage (Salvia lavandulifolia) plants in the field in Murcia, southern Spain.
GC-MS Analysis
S. lavandulifolia EOs were analyzed by GC-MS as previously described by DeCarlo et al. 30 Shimadzu GC-MS-QP2010 Ultra, electron impact (EI) mode (70 eV, 40-400 m/z scan range, 3.0 scan/sec scan rate). ZB-5 ms capillary column (30 m × 0.25 mm, 0.25 μm film thickness); temperature program (50 °C for 2 min, increased 2 °C/min to 260 °C); He carrier gas, column head pressure 552 kPa, flow rate 1.37 mL/min; injector temperature 260 °C, ion source temperature 200 °C. For each sample, 0.3 μL of a 1:10 v/v dilution in dichloromethane (DCM) was injected, split ratio of 1:30. The chemical components were identified by comparing mass spectral fragmentation patterns (> 80% similarity match) and retention indices (RIs) (calculated using a series of homologous C8-C20 n-alkanes) with those reported in databases (NIST database, and our in-house library), Lab Solutions GC-MS software (version 4.45).
GC-FID Analysis
Analysis of Spanish sage EOs was carried out as previously described by DeCarlo et al. 30 : Shimadzu GC 2010 FID, ZB-5 capillary column under the same operating conditions as above for GC-MS.
Enantiomeric Analysis by Chiral Gas Chromatography–Mass Spectrometry (CGC-MS)
The enantiomeric analysis of Spanish sage EO was carried out by CGC-MS as previously described by Poudel et al. 31 : Shimadzu GC-MS-QP2010S, EI mode (70 eV), B-Dex 325 chiral capillary GC column, 40 to 400 m/z scan range, 3.0 scan/sec scan rate; column temperature program (50 °C initial temperature, increased 1.5 °C/min to 120 °C, then 2 °C/min to 200 °C, held constant at 200 °C final temperature); He carrier gas, flow rate 1.8 mL/min. For each EO sample, 0.1 μL of 3% w/v solution was injected, split ratio of 1:45. The enantiomer ratios were determined from peak areas. Enantiomer identification was carried out by comparison of retention times and mass spectral fragmentation patterns with authentic samples obtained from Sigma-Aldrich.
Hierarchical Cluster Analysis
Compositions of S. lavandulifolia EOs from the published literature as well as those obtained from this work were used in the cluster analysis. The operational taxonomic units (OTUs) were the EO compositions; the major components (α-pinene, camphene, sabinene, β-pinene, myrcene, p-cymene, limonene, 1,8-cineole, γ-terpinene, linalool, trans-sabinol, camphor, borneol, terpinen-4-ol, α-terpineol, linalyl acetate, bornyl acetate, trans-sabinyl acetate, α-terpinyl acetate, and (E)-β-caryophyllene) were used for agglomerative hierarchical cluster (AHC) analysis (XLSTAT Premium, version 2018.5.53172, Addinsoft). Dissimilarity was determined using Euclidean distance, and clustering was defined using Ward's method.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221139402 - Supplemental material for Volatile Composition and Enantiomeric Distribution of Spanish Sage (Salvia officinalis Subsp. lavandulifolia [Vahl] Gams) From Spain
Supplemental material, sj-docx-1-npx-10.1177_1934578X221139402 for Volatile Composition and Enantiomeric Distribution of Spanish Sage (Salvia officinalis Subsp. lavandulifolia [Vahl] Gams) From Spain by Noura S. Dosoky, Ambika Poudel, Prabodh Satyal and William N. Setzer in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank Juan Lorente for plant identification and Jared Moon for valuable discussions. We would also like to thank Russel Osguthorpe for supporting this research project.
Author’s Contribution
Conceptualization: N.S.D and P.S.; Methodology: A.P.; Software: W.N.S.; Validation: P.S. and A.P.; Formal analysis: A.P. and P.S.; Investigation: N.S.D and P.S.; Data curation: P.S. and W.N. S.; Writing—original draft preparation: N.SD; Writing—review and editing: N.S.D, P.S., and W.N.S.; Supervision: W.N.S. All the authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Data Availability Statement
Data from this investigation are available in the manuscript and Supplemental material.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
