Abstract
The aerial parts of Аrtemisia annua L. were collected from Varzob, Rudaki, and Hisor regions of Tajikistan. The essential oil was obtained by hydrodistillation and analyzed by gas chromatography–mass spectrometry. The essential oils of aerial parts of А. annua were dominated by the monoterpenoids like camphor (32.5%-58.9%), 1,8-cineole (13.7%-17.8%), camphene (4.5%-8.4%), and α-pinene (1.9%-7.3%). Hierarchical cluster analysis of A. annua essential oils indicated the existence of 3 A. annua chemotypes: camphor/1,8-cineole, camphor, and artemisia ketone. The essential oils of А. annua show weak antioxidant activity and average antibacterial activity. In our opinion, the antibacterial activity of А. annua essential oils is related to the presence of 1,8-cineole. To our best knowledge, this is the first report concerning the chemical composition, chemotypic variation, antioxidant, and antimicrobial activities of the essential oils obtained from the aerial parts of А. annua, growing wild in Tajikistan.
Introduction
Artemisia annua L., also known as Qinghao, Sweet Wormwood, Sweet Annie, Sweet Sagewort, Annual Wormwood, is an annual herb in the Asteraceae family and is widely distributed in Asia, Europe, and North America. 1 Artemisia annua has been used as an important ethnomedicinal herb for many years, especially in China against fever and malaria for over 100 years. It has been described for the treatment of malaria, cough, and cold in ancient pharmacopeias of various Asian and European countries. This herb is described as a dietary food and used as a traditional herbal medicine against inflammatory diseases, infections by fungi, bacteria and viruses, gastric ulcer, and cancer among others. 2 It is also used for the treatment for diarrhea and has antihelmintic, antipyretic, antiseptic, antispasmodic, carminative, stimulant, tonic, and stomachic properties. 3 -6 Recently, studies are more focused to evaluate its anticancer and antiviral potential against human cancer cells and human immunodeficiency virus, respectively. 7,8 The genus Artemisia occupies a top position for its bioprospection. 9 The genus Artemisia is known to possess a rich phytochemical diversity. 10 -12 Artemisia annua is a rich source of biologically active artemisinin, which is widely used in the treatment of malaria. 13,14 Artemisia annua also represents great interest in the production of its essential oil.
The essential oil composition of A. annua has been extensively investigated by gas chromatography (GC) and GC–mass spectroscopy (MS) methods. 1,8-Cineole, artemisia ketone, camphor, α-pinene, β-caryophyllene, germacrene D, and artemisia alcohol have been commonly reported as principal components in the essential oils of A. annua. Due to the presence of valuable components such as 1,8-cineol, linalool, p-cymene, thujone, and camphor in the essential oil of A. annua, its essential oil is useful for its beneficial pharmacological properties.
More than 100 Artemisia species, including A. annua, grow in Tajikistan. In this respect, there is a great interest in assessing the potential of Tajik Artemisia species, including A. annua, for new sources of bioactive phytochemicals. Due to the rich and diverse composition of biologically active substances, it has great importance in food and pharmacy. To our best knowledge, there is no previous work of the chemical composition and biological activity of A. annua growing in Central Asia, especially in Tajikistan. In addition, in order to delineate the chemotypes of A. annua essential oils, a cluster analysis has been carried out using the reported chemical compositions of A. annua essential oils from numerous geographical locations around the world.
Results and Discussion
The aerial parts of А. annua were collected from the Varzob, Rudaki, and Hisor regions of Tajikistan. The essential oils were obtained by hydrodistillation and analyzed by GC–MS. The essential oils of aerial parts of А. annua were dominated by the monoterpene hydrocarbons and their oxygenated derivatives. The major components of the essential oil were camphor (32.5%-58.9%), 1,8-cineole (13.7%-17.8%), camphene (4.5%-8.4%), and α-pinene (1.9%-7.3%). The chemical composition of the essential oil of A. annua is represented in Table 1.
Essential Oil Composition of Аrtemisia annua L. From Tajikistan.
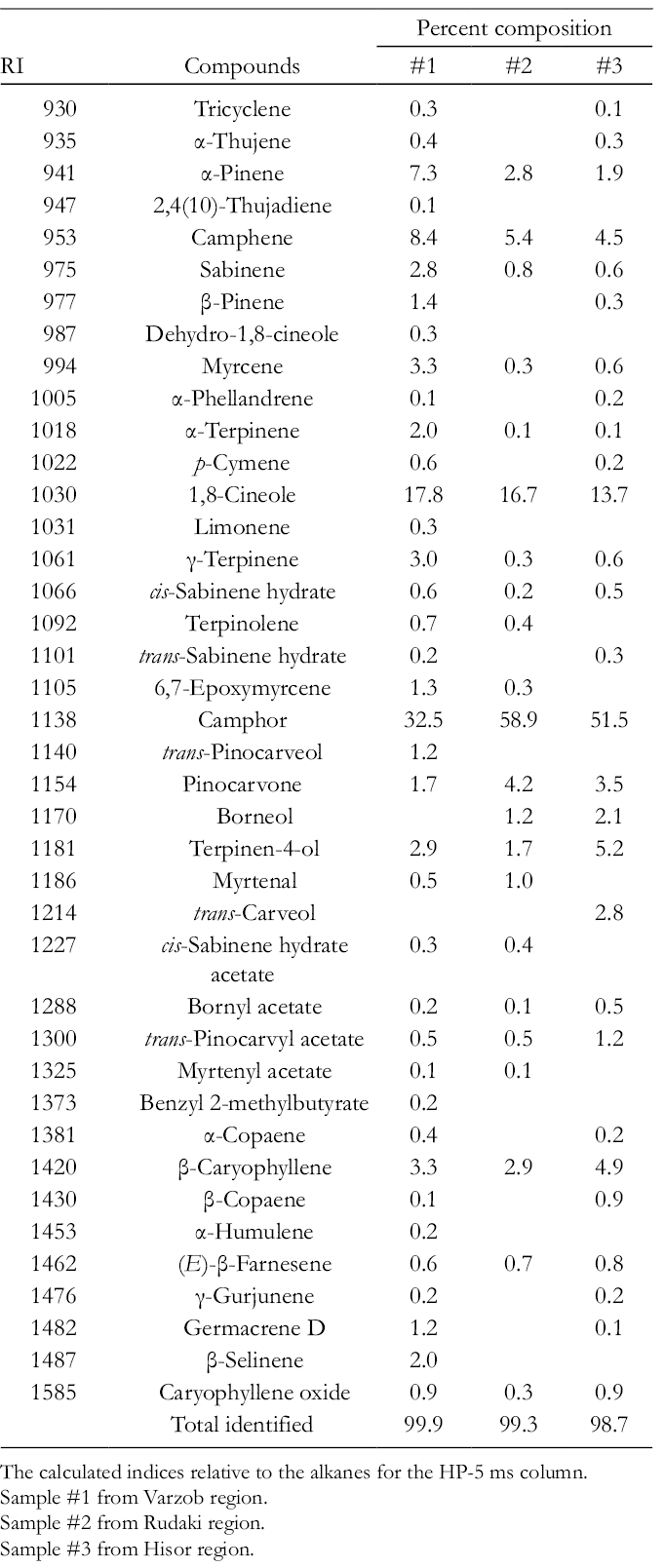
The calculated indices relative to the alkanes for the HP-5 ms column.
Sample #1 from Varzob region.
Sample #2 from Rudaki region.
Sample #3 from Hisor region.
Artemisia ketone, camphor, 1,8-cineole, α-pinene, β-caryophyllene, germacrene-D, p-cymene, o-cymol, and artemisia alcohol were reported as the major components (≥10%) of the essential oils of А. annua and are different from species growing in other geographic locations. 1,2,13,15 -54 Also, bisabolol, bisabolol oxide B26,27, and α-cubebene 18 were found as major components of the A. annua essential oils. Analyses in the published research data showed that many species of the Artemisia genus, including A. annua, A. vulgaris, A. diffusa, A. santonicum, A. spicigera, A. afra, A. asiatica, A. austriaca, and A. pedemontana, contain bornane derivatives (camphor, borneol and bornyl acetate) and 1,8-cineole as the major components. 55
A dendrogram of the hierarchical cluster analysis of A. annua essential oil compositions on the global phytogeographic origin is represented in Figure 1. In the present work, the hierarchical cluster analysis of A. annua essential oils indicates the existence of 3 A. annua chemotypes: camphor/1,8-cineole (cluster I), camphor (cluster II), and artemisia ketone (cluster III). Cluster I can be divided into 2 subgroups: Ia (high in β-caryophyllene, artemisia ketone, and germacrene D) and Ib (low in β-caryophyllene, artemisia ketone, and germacrene D). There are also 2 subgroups for cluster II: IIa (high in artemisia ketone, germacrene D, and β-caryophyllene), IIb (high in 1,8-cineole); and 2 sub-groups for cluster III: IIIa (high in camphor), IIIb (high in 1,8-cineole). Radulovic and co-authors had previously performed an agglomerative hierarchical cluster analysis of A. annua essential oils indicating the existence of 5 different A. annua chemotypes: camphor and camphor/1,8-cineole types, artemisia ketone/α-pinene/1,8-cineole and artemisia ketone/camphor/1,8-cineole types, camphor/artemisia ketone/germacrene D type, germacrene D/β-caryophyllene, and germacrene D/β-caryophyllene/1,8-cineole/artemisia ketone types. 13 Bilia and co-authors have classified A. annua essential oils on the basis of the concentration of 3 main components (artemisia ketone, 1,8-cineole, and camphor) and defined 6 groups: (1) Vietnamese oil with 3.3%-21.8% camphor and 0.3%-18.9% germacrene D, (2) Chinese oil with high content of artemisia ketone (64%), (3) Indian oil with 11.5%-58.8% of artemisia ketone, (iv) French oil with 2.8%-55% artemisia ketone, 1.2%-11.6% 1,8-cineole, and 15% germacrene D, (v) North American oil with 35.7%-68% artemisia ketone and 22.8%-31.5% 1,8-cineole, (vi) Iranian oil with 48% camphor and 9.4% 1,8-cineole. 22 Our results are in agreement with previous data on the chemotypification of A. annua based on its oil composition. 13,22 In the present work, A. annua essential oils were classified into 3 convenient chemotypes: camphor/1,8-cineole, camphor, and artemisia ketone. The essential oil from Varzob in this work belongs to the camphor/1,8-cineole chemotype, while the essential oils from Hisor and Rudaki are in the chemotype dominated by camphor. Thus, there is significant chemical variation in essential oil compositions within Tajikistan.

Hierarchical cluster analysis of Artemisia annua essential oils. Cluster I (purple and green) is a camphor/1,8-cineole mixed chemotype; cluster II (red), the largest cluster, is dominated by camphor; and cluster III (blue) has artemisia ketone as the major component.
The essential oils of А. annua have demonstrated good antioxidant capacity, in particular, as radical scavengers. Antioxidant capacities were 7.7-10.9 μmol/mL Trolox equivalents in the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay and 25.9-38.1 μmol/mL Trolox equivalents in the 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulfonic acid) (ABTS) assay for the essential oil of A. annua. 2
In the present work, the essential oils of А. annua has shown weak antioxidant activity compared with the positive control which was evaluated by DPPH and ABTS methods. The half-maximal inhibitory concentration (IC50) values for the essential oils were 5-6.5 mg/mL for DPPH and 2.4-3.5 mg/mL for ABTS, respectively. The IC50 values of the positive control (ascorbic acid) were 0.003 mg/mL for DPPH and 0.007 mg/mL for ABTS, respectively.
Antibacterial activity of A. annua essential oil was tested against both Gram-positive (Staphylococcus aureus) and Gram-negative (Pseudomonas aeruginosa and Escherichia coli) bacteria. Its activity was very similar to the bactericidal activity of Origanum vulgare essential oil and gentamicin (0.12 mg/mL) (positive control). Results of antimicrobial activity of essential oil of А. annua are represented in Table 2.
The Antimicrobial Activity of Essential Oil of Аrtemisia annua.
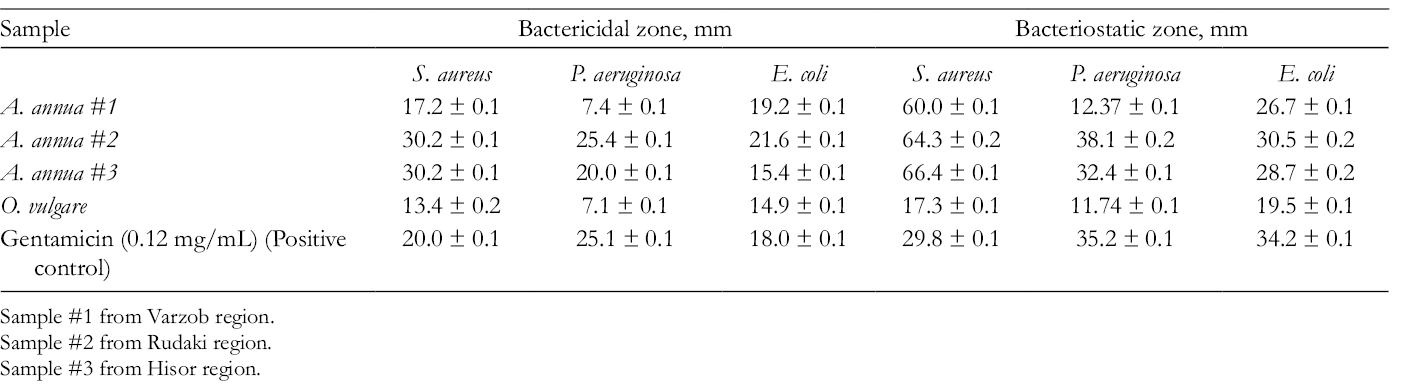
Sample #1 from Varzob region.
Sample #2 from Rudaki region.
Sample #3 from Hisor region.
The essential oil isolated from A. annua was previously found to have antimicrobial effects against E. coli, S. aureus, Streptococcus faecalis, P. aeruginosa, Klebsiella pneumoniae, Bacillus subtilis, Bacillus licheniformis, Candida albicans, and Saccharomyces cerevisiae. Microbial inhibition zone ranged from 10 to 45 mm at 10 µL for various concentrations (1/4 and 1/8 dilution) of the oils for 24-48 hours. 19 According to the literature data, 1,8-cineole is responsible for the antibacterial activity in essential oils; it exhibited comparatively strong activity against Gram-negative and Gram-positive bacteria. 56,57
In conclusion, camphor (32.5%), 1,8-cineole (17.8%), camphene (8.4%), and α-pinene (7.3%) were the major components of the essential oil obtained from the aerial parts of А. annua, growing wild in Tajikistan. The hierarchical cluster analysis of A. annua essential oils on the global phytogeographic origin indicates the existence of 3 A. annua chemotypes: camphor/1,8-cineole, camphor, and artemisia ketone. Present investigated essential oils of А. annua from Tajikistan belong to the camphor/1,8-cineole (Varzob) and camphor (Hisor and Rudaki) chemotypic variations. The essential oil of А. annua showed weak antioxidant activity and average antibacterial activity. Although neither camphor 58 nor 1,8-cineole 59 show pronounced antibacterial activity, and they may accentuate the activities of minor essential oil components in a synergistic manner.
Experimental Section
Plant Material
The aerial parts of A. annua L. were collected from 3 regions of Tajikistan: sample #1 from the Varzob region on July 2017, during the start of the flowering stage; sample #2 from the Rudaki region on August 2019, during the period of plant flowering; and sample #3 from the Hisor region on July 2019, during the start of the flowering stage. Raw material was identified with regards to specimens (accession number 360) in the Herbarium of the Institute of Botany, Plant Physiology and Genetics of the Tajikistan Academy of Sciences.
Isolation of the Essential Oil
The aerial parts (3 × 300 g) of A. annua were dried at 25°C in the shade and subjected to hydrodistillation, using a Clevenger-type apparatus for 3 hours. Yield of essential oils was 0.4%-0.6%.
GC–MS Analysis
GC–MS analysis was performed on the essential oil of A. annua (3 analysis of each essential oil) using an Agilent 6890 GC with Agilent 5973 MSD and HP-5ms capillary column as described previously by us. 60,61 Identification of the essential oil components was based on retention indices and mass spectral fragmentation patterns with those reported in the literature 62 and our own in-house database.
Antioxidant Activity
The antioxidant activity of the essential oils of A. annua was evaluated by DPPH and ABTS assays. DPPH and ABTS assays were performed as described earlier by us. 57
Antimicrobial Activity
The antimicrobial activities of the essential oils were determined against S. aureus (ATCC 23235), P. aeruginosa (ATCC 27853), and E. coli (ATCC 25922). Bacterial strains were tested on agar. Sterilized paper disks were loaded with 10 µL of A. annua L. essential oil or positive control and applied on the surface of agar plates. Inhibition zones (mm) of bacterial strains were defined after the incubation period for 24 hours at 37°C.
Hierarchical Cluster Analysis
The A. annua leaf essential oil compositions from this work and those from the literature were treated as operational taxonomic units. The percentage composition of 39 major essential oil components was used to determine the chemical relationship between the various A. annua essential oil samples by agglomerative hierarchical cluster analysis using the XLSTAT software, version 2018.1.1.6097 (Addinsoft, Paris, France). Euclidean distance was used to measure dissimilarity, and Ward’s method was used for cluster definition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the authors are grateful for financial support to the Central Asian Drug Discovery & Development Center of Chinese Academy of Sciences (Grant No. CAM 201808), Foreign young scholar (Grant No. 2018FYB0004) Chinese Academy of Sciences PIFI (Grant No. 2019PB0043) and the National Natural Science Foundation of China (Grant No. U1703235)
