Abstract
Introduction
Over the past 10 years, the incidence of leukemia has increased by 2% and its incidence and mortality are among the top 5 among all tumors. The five-year survival rate of chronic myelogenous leukemia (CML), the most common type of chronic leukemia in Asian countries, is 69%. 1 Currently, hematopoietic stem cell transplantation represents a possible cure for leukemia. Effective treatment with chemotherapeutic drugs is a prerequisite for the implementation of hematopoietic stem cell transplantation or other technologies that may cure leukemia. 2 Clinically, drug resistance and acute lesions are the main factors reducing the therapeutic effect on leukemia. Therefore, new treatments and targets must be identified to improve patient compliance and quality of life.
Cell cycle regulation is related to both cell proliferation and apoptosis, and abnormal regulation of the cell cycle is an important mechanism underlying tumor development. In cell cycle regulation, two key control points, G2/M and G1/S, are important for intracellular and extracellular signal transmission and integration. Programed death or the stationary G0 phase is key to activating cell cycle regulation. Most of the cytotoxic drugs currently in clinical use are not specific to the cell cycle, such as alkylating agents, cisplatin, and nitrosourea, which have serious toxic reactions. Therefore, research and development of targeted cell cycle-specific drugs are of great significance.
The balance between cell proliferation, differentiation, and apoptosis is closely related to the occurrence and development of tumors. The mitochondrial pathway plays an important role in the process of cell apoptosis. 3 Stress- or apoptosis-inducing agents can cause mitochondrial destruction and reduce mitochondrial membrane permeability, which manifests as a decrease in mitochondrial membrane potential (MMP). Once cytochrome C and apoptosis-inducing factors are released into the cytoplasm, caspase-3 is activated, thus leading to apoptosis. The balance between anti-apoptotic molecules and pro-apoptotic molecules in the Bcl-2 family is of great importance in maintaining MMP stability, especially the “molecular switch” role of Bcl-2/Bax in cell apoptosis. 4 After translocating from the cytoplasm to the mitochondrial membrane, Bax can open molecular channels to increase the release of pro-apoptotic factors. Bcl-2 is located on the outer mitochondrial membrane, which stabilizes the mitochondrial membrane and inhibits the release of proapoptotic factors. By forming a dimer with Bax, Bcl-2 can reduce the permeability of the mitochondrial membrane to exert an anti-apoptotic effect. A variety of small molecule inhibitors targeting Bcl-2 have been developed, although most of them are still in clinical trials and have not been officially launched. 5 AT101 is the first compound that can inhibit Bcl-2, Bcl-XL, and Mcl-1, and its anti-leukemia effects, which are associated with Bcl-2 protein inhibition, have been verified by experiments in vitro. 6
Convallatoxin is a strong cardiac glycoside isolated from
In this study, we evaluated the antitumor effect of convallatoxin on the erythroleukemia cell line K562. Convallatoxin inhibits the proliferation of K562 cells, promotes mitochondria-related apoptosis, and induces cell cycle arrest in the S and G2/M phases, which are related to the downregulation of Akt-E2F1 signal transduction. As mentioned above, convallatoxin may be a novel drug for leukemia treatment.
Results and Discussion
Convallatoxin Inhibits K562 Cell Proliferation
To identify the antitumor activity of convallatoxin, we performed a CCK-8 assay to detect the effect of convallatoxin on the proliferation of K562 cells (Figure 1). The cell viability of K562 cells treated with convallatoxin (0, 3, 10, and 30 μM) was reduced by 0%, 20%, 60%, and 65% compared with that of the non-convallatoxin group (IC50 = 4.88 μM, Figure 1A and B). Also, the cell viability decreased significantly over the treatment time, which decreased by 12%, 48%, and 51%, at 12, 24, and 48 h, respectively (Figure 1C). These results indicated that convallatoxin has a significant inhibitory effect on K562 cell proliferation in a dose- and time-dependent manner.

Convallatoxin inhibits cell proliferation in K562 cells. (A) K562 cells were treated with different concentrations of convallatoxin for 24 h, and cell viability was detected by CCK-8 assay; **
Convallatoxin Inhibits the Cell Cycle in K562 Cells
To explore the mechanism underlying the ability of convallatoxin to inhibit K562 cell proliferation, we examined the effect of the compound on the K562 cell cycle (Figure 2). The proportion of cells in phase G1 after treatment with 10 and 30 μM convallatoxin decreased by 5% and 12% compared with that of the control group, respectively, while the proportion of cells in phases S and G2/M increased by 6% and 12% after treatment with 30 μM convallatoxin, respectively (Figure 2). Tumor cell G2/M phase arrest is an important chemotherapeutic pathway; therefore, this finding suggested that convallatoxin could inhibit the proliferation of chronic myeloid leukemia cells through cell cycle arrest at phases S and G2/M.

Convallatoxin inhibits the cell cycle in K562 cells. (A) K562 cells were treated with different concentrations of convallatoxin for 24 h, and the cell cycle was detected by flow cytometry. (B-D) Proportions of each phase (phase G0/G1, S, and G2/M) were calculated. *
Convallatoxin Induces K562 Cell Apoptosis
Proliferation inhibition and apoptosis induction in tumor cells are important strategies for anti-cancer drug research. Therefore, Annexin V-FITC/PI staining was used to detect the effect of convallatoxin on K562 cell apoptosis by flow cytometry (Figure 3A and B). The proportion of apoptotic cells after 24 h of convallatoxin treatment increased in a dose-dependent manner. Specifically, the number of apoptotic cells in the 10 and 30 μM convallatoxin groups increased significantly by 18% and 35% compared to the control group. This suggests that convallatoxin significantly induced apoptosis in K562 cells.
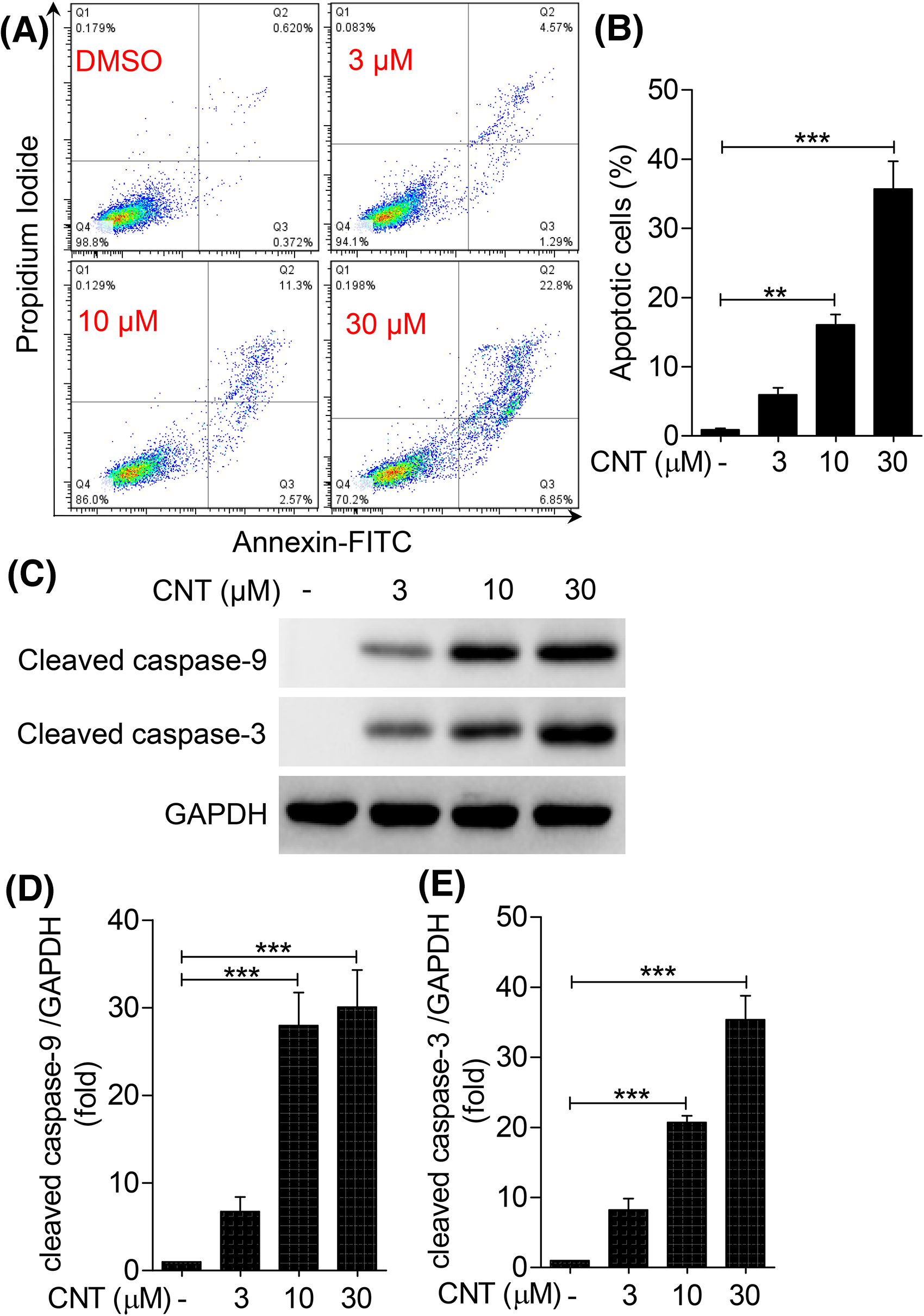
Convallatoxin induces cell apoptosis in K562 cells. (A) K562 cells were treated with different concentrations of convallatoxin for 24 h, and cell apoptosis was detected by Annexin V-FITC/PI staining. Results in the four quadrants are as follows: early apoptotic cells (lower right); late apoptotic cells (top right); normal cells (bottom left); and dead cells (top left). (B) Apoptosis rate is calculated as follows: number of apoptotic cells/total number of cells × 100%. (C) K562 cells were treated with different concentrations of convallatoxin for 24 h, and the protein expression of apoptosis-related proteins cleaved caspase-3 and cleaved caspase-9 was detected by Western blot. (D-E) Gray value analysis was performed for each band. *
To confirm the effect of convallatoxin on K562 cell apoptosis, we used Western blotting to detect the expression of apoptosis-related proteins, such as cleaved caspase-3 and cleaved caspase-9 (Figure 3C to E). The results showed that after treatment with 10 and 30 μM of convallatoxin, pro-apoptotic protein cleaved caspase-3 was upregulated by 28% and 30% in K562 cells, respectively, while cleaved caspase-9 was upregulated by 20% and 35%, respectively. These results suggest that convallatoxin promotes the apoptosis of K562 cells in a dose-dependent manner (Figure 4).
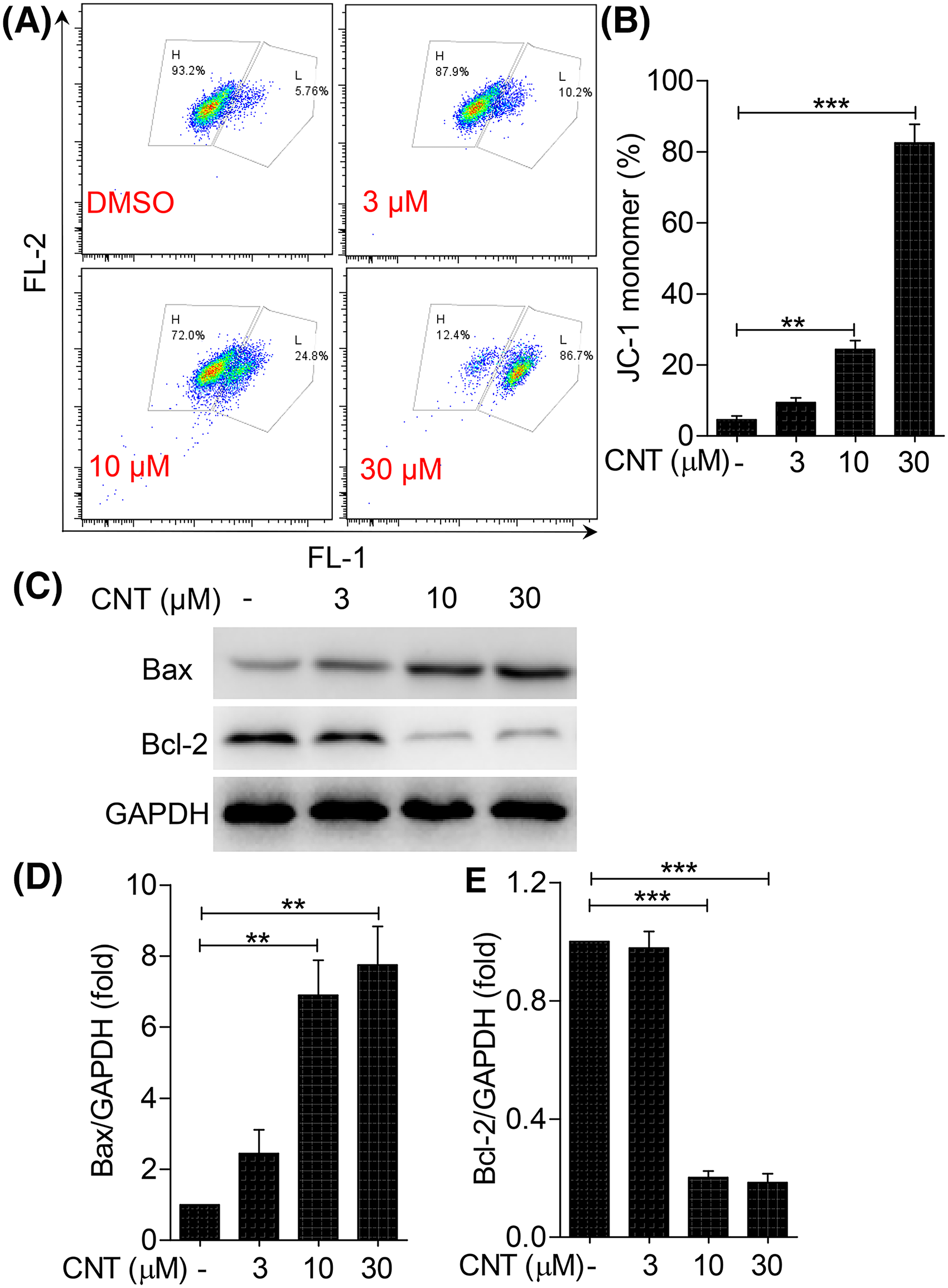
Convallatoxin induces cell apoptosis through the mitochondrial pathway in K562 cells. (A) JC-1 fluorescent probe was used to detect MMP changes in K562 cells. (B) Percentages of JC-1 monomer were calculated. (C) After stimulation with different concentrations of convallatoxin for 24 h, Western blot was used to detect the expression of apoptosis-related proteins Bax and Bcl-2. (D-E) Gray value analysis was performed for each band. *
Convallatoxin Induces Cell Apoptosis Through the Mitochondrial Pathway in K562 Cells
MMP is considered a hallmark event in early cell apoptosis. As a lipophilic cationic fluorescent dye, JC-1 is widely used for the detection of MMP. When MMP is high, JC-1 aggregates in the matrix of the mitochondria to form a polymer that produces red fluorescence; and when MMP is low, JC-1 exists as a monomer that produces green fluorescence. In this experiment, a JC-1 fluorescent probe and flow cytometry were used to evaluate the early apoptosis of K562 cells. The ratio of JC-1 monomers after treatment with 10 and 30 μM convallatoxin for 24 h increased by 21% and 80%, respectively, indicating that convallatoxin improved cell membrane permeability and enhanced early apoptosis of K562 cells (Figure 5 A and B).
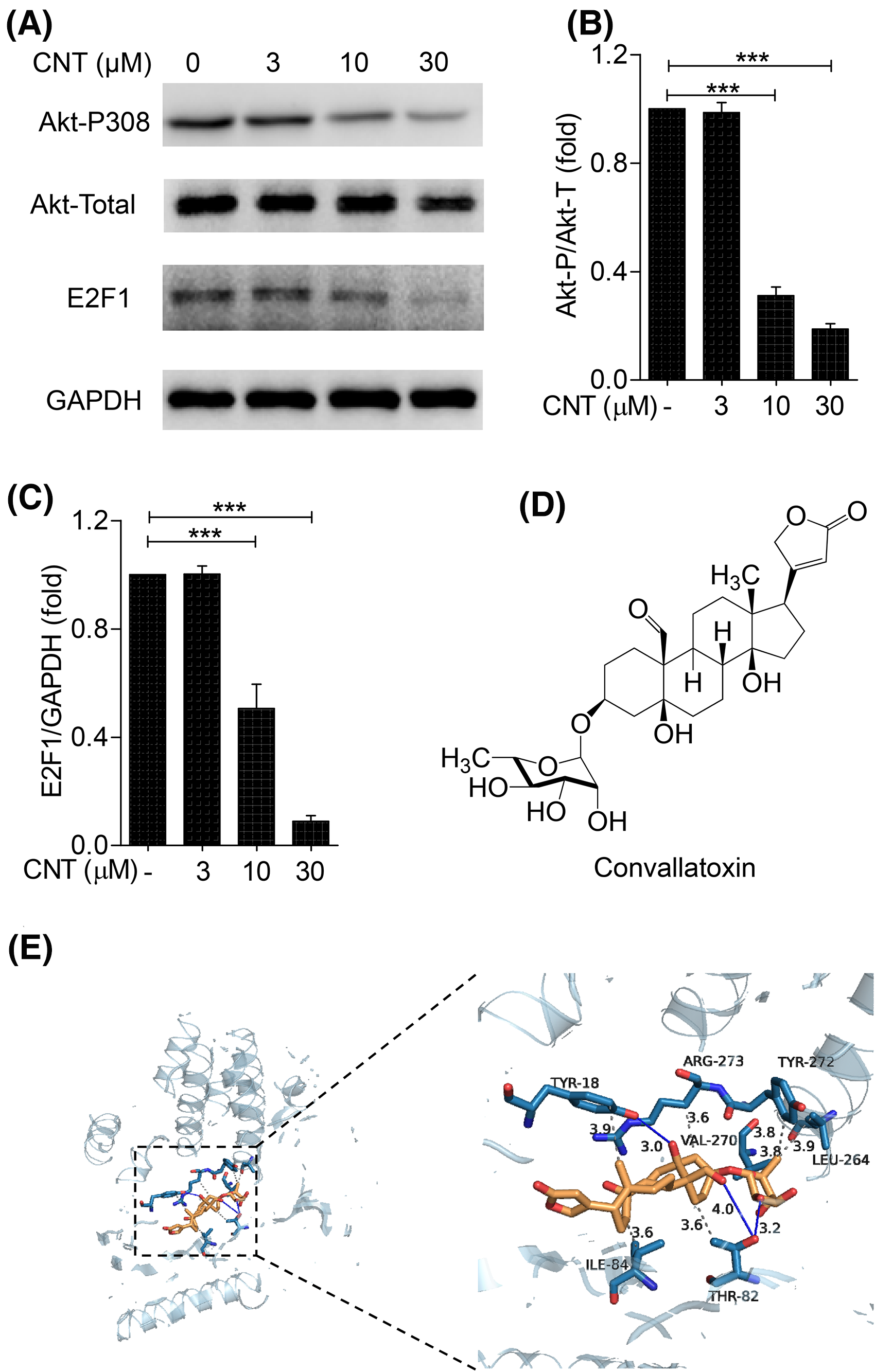
Convallatoxin inhibits the akt-E2F1 signaling pathway in K562 cells. (A) K562 cells were treated with different concentrations of convallatoxin for 24 h, and Western blot was performed to detect the expression of Akt-P308, total Akt and E2F1. (B-C) Gray value analysis was performed for each band. (D) Molecular structure of convallatoxin. (E) Proposed binding mode of convallatoxin (gold) in Akt1 (blue). Akt1 is shown in blue, while hydrogen bonds and hydrophobic force interactions are shown as blue solid lines and gray dashed lines, respectively. *
The Bcl-2 protein family is involved in mitochondrial membrane permeability and represents important regulatory proteins released by cytochrome C from the mitochondria. The ratio of the pro-apoptotic protein Bax to the anti-apoptotic protein Bcl-2 is closely related to the sensitivity of the mitochondrial membrane. 4 After treatment with 10 and 30 μM of convallatoxin for 24 h, the expression of Bax was upregulated by 64% and 70% while the expression of Bcl-2 was downregulated by 68% and 70%, respectively, compared with the control group, indicating that convallatoxin induces apoptosis of K562 cells in a dose-dependent manner by increasing the permeability of the mitochondrial cell membrane (Figure 5C and D).
Convallatoxin Inhibits Akt-E2F1 Signaling Pathway in K562 Cells
Cell cycle-related transcription factor E2F1 (E2F1) is a member of the E2F family involved in a variety of cellular processes, including the cell cycle, DNA repair, DNA replication, and cell differentiation, proliferation, and apoptosis. To study further the specific mechanism of convallatoxin action, Western blotting was performed to detect the expression of p-Akt (Ser 308), t-Akt, and E2F1 (Figure 5A). The results showed that Akt phosphorylation and E2F1 expression in K562 cells were significantly decreased after treatment with 10 and 30 μM convallatoxin (Figure 5B and C). These results indicate that convallatoxin induces K562 cell apoptosis by inhibiting Akt phosphorylation and downregulating E2F1.
To confirm further the interaction between convallatoxin and Akt1, we performed a molecular docking assay using the molecular structure of convallatoxin (Figure 5D) and the crystal structure of Akt1, which contains a critical structural motif for the interactions. The results showed that convallatoxin is directly bound to Akt1 (Figure 5E) with a binding energy of −10.4 kcal/mol, suggesting that convallatoxin has a strong affinity with the target protein Akt1. The docked convallatoxin showed extensive interactions with Akt1, including those with the Tyr18, Thr82, Ile84, Leu264, Val270, and Tyr272 residues, and Agr273 side chain with 3.9, 3.6, 3.6, 3.9, 3.8, 3.8, and 3.6 Å hydrophobic bonds, respectively. In addition, convallatoxin interacted with Tyr18 through 3.0 Å hydrogen bonds and with the Thr82 residue through 3.2 Å and 4.0Å hydrogen bonds. Thus, our data demonstrate that convallatoxin directly targets the Akt-E2F1 signaling pathway by binding with Akt1.
E2F1 Overexpression Rescues the Effects of Convallatoxin in K562 Cells
To study further the role of E2F1 in convallatoxin effects, K562 cells overexpressing E2F1 were constructed. The expression of E2F1 was verified using Western blotting (Figure 6). After treatment with 10 μM convallatoxin, the viability of K562 cells was significantly reduced by 55%, while the viability of K562 cells overexpressing E2F1 was 20% higher compared with that of the 0 μM convallatoxin group (Figure 6A and B). Similarly, after treatment with 10 μM convallatoxin, the percentage of apoptotic cells was increased by 15% while the percentage of apoptotic cells with E2F1 overexpression was 10% lower compared with that after treatment with 0 μM convallatoxin (Figure 6C and D). These results suggest that E2F1 overexpression can rescue the effects of convallatoxin.

E2F1 overexpression rescues the effects of convallatoxin in K562 cells. (A) Protein expression of E2F1 in K562 cells overexpressing the E2F1 gene was verified by Western blot. (B) K562 cells overexpressing E2F1 were treated with 10 μM of convallatoxin for 24 h, and cell viability was detected by CCK-8 assay. (C) Cell apoptosis was detected by Annexin V-FITC/PI staining. (D) Apoptosis rate is calculated as follows: number of apoptotic cells/total number of cells × 100%. **
Discussion
In this study, convallatoxin inhibited the proliferation of K562 cells in a time- and concentration-dependent manner. Concurrently, convallatoxin induced cell cycle arrest at the S and G2/M phases and promoted apoptosis in a mitochondrial-dependent manner. Furthermore, the anti-leukemic effect of convallatoxin was related to the attenuation of the Akt-E2F1 signaling pathway.
In recent years, drugs that target different signal transduction pathways of leukemia cells, including proliferation, differentiation, and apoptosis, have attracted much attention. 10 Representative anti-cancer drugs that specifically block the cell cycle include 5-Fu, pemetrexed, cytarabine, and tigeo, which act on the S phase; vinorelbine, paclitaxel, and etoposide, which act on the M phase; and bleomycin, which acts on the G2 phase. As an antimetabolite of pyrimidine, which acts on the S stage, cytarabine (CY) can inhibit DNA synthesis and interfere with the proliferation of tumor cells, and is the preferred chemotherapy drug for the treatment of CML. 11 However, CY is not a targeted therapy drug, and different myeloid leukemia cells respond differently to CY. In clinical treatment, high-dose CY is used to treat CML. However, high concentrations not only have neurotoxic effects but also increase the susceptibility to relapse after drug withdrawal; moreover, drug resistance leads to reduced sensitivity to CY.12,13 In sequential chemotherapy, tumors with rapid proliferation, such as choriocarcinoma and leukemia, have more cells in the proliferative phase. Cell cycle-specific drugs are usually used to kill cycle-sensitive cells first, and then cell cycle non-specific drugs are used to kill tumor cells at other stages, which makes the tumor treatment process more complicated. 14 Our study showed that different convallatoxin treatments at different concentrations caused cell cycle arrest of K562 cells in both the S and G2/M phases, suggesting that convallatoxin may serve as a potential anti-leukemia drug.
Convallatoxin presents various pharmacological activities and has been reported to inhibit NF-κB activity, which decreases the expression of pro-inflammatory cytokines in macrophages, intestinal cells, and other immune cells through the activation of PPARγ, thereby reducing mucosal inflammation and improving DSS-induced colitis. 15 Moreover, convallatoxin inhibits the migration and invasion of lung cancer cells by inhibiting the expression of MMP-2, MMP-9, and P-FAK. 16 The anti-tumor effect of convallatoxin has also been verified in this study, which mainly affects cell proliferation and apoptosis.
The proliferation and apoptosis of leukemia cells are regulated by abnormal activation of various signaling pathways. Among them, the PI3K-Akt signaling pathway has been shown to regulate the occurrence, development, and prognosis of leukemia.17‐19 As a proto-oncogene, Akt plays an important role in regulating cell metabolism, growth, proliferation, survival, and transcription and protein synthesis. 20 In tumor cells, over-activated Akt activates the NF-κB and mTOR pathways, thus leading to an anti-apoptotic effect. 21 E2F1 is a member of the E2F family and presents high expression in a variety of tumor tissues and cells, and its upregulation is closely related to tumor occurrence, development, metastasis, and prognosis.14,22,23 Reports have shown that E2F1 upregulates Akt activity through a transcription-dependent mechanism,23‐25 suggesting the existence of a negative feedback loop involving E2F and Akt related to cell apoptosis. 23 As the target of convallatoxin, the Akt-E2F1 signaling pathway was demonstrated to be related to the anti-leukemia mechanism.
In summary, this study confirmed that convallatoxin significantly inhibits the proliferation of K562 cells by cell cycle arrest, induces mitochondrial-dependent cell apoptosis, and exerts an anti-leukemia effect related to the attenuated Akt-E2F1 signaling pathway. Convallatoxin has potential anti-leukemic activity and can be further developed for clinical treatment, and the Akt-E2F1 signaling pathway may serve as an effective drug target.
Materials and Methods
Reagents
Leukemia cell line K562 cell was purchased from the American Type Culture Collection. IMDM medium, trypsin, penicillin, and streptomycin were purchased from Gibco| Life Technologies, fetal bovine serum (FBS) from Sijiqing Biological Engineering Materials Co. Ltd, dimethyl sulfoxide (DMSO), and enhanced chemiluminescence substrate ECL from Thermo Fisher Scientific, MMP detection kit (JC-1 method), lipofectamine 2000 kit, CCK-8 solution, PBS buffer, 4% tissue cell fixative, 1 mol/L Tris-HCl buffer (pH = 6.8), and 10% SDS from Beyotime Biotechnology, primary antibodies against cleaved caspase-9, cleaved caspase-3, Bcl-2, Bax, p-Akt (Ser 308), t-Akt, E2F1, and GAPDH from Cell Signaling Technologies, Annexin V-FITC apoptosis assay kit and cell cycle analysis kit from MultiSciences (Lianke) Biotech Co., Ltd, E2F1 overexpression lentiviral plasmid and empty vector lentiviral plasmid from Jikai Gene Chemical Technology Co., Ltd, and convallatoxin (purity: analyzed by SDS-PAGE gel and HPLC, greater than 98%) from Sigma-Aldrich.
Drug Configuration
Convallatoxin powder was weighed and placed in a 1.5 mL EP tube, and then a certain volume of DMSO, calculated according to the molecular weight, was added to obtain a 50 mM storage solution. An aliquot of this solution was stored at −80 °C, and different working concentrations were diluted according to the experimental requirements.
Cell Culture
K562 cell culture was performed using IMDM medium containing 10% FBS and 1% penicillin-streptomycin. The cells were cultured in an incubator at 37 °C and 5% CO2. The cells were passaged in a timely manner and cryopreserved when grown to the logarithmic growth phase.
Cell Proliferation Measurement
For the CCK-8 assay, K562 cells in the logarithmic growth phase were collected, and then 5 × 103 cells per well were seeded in a 96-well plate. Each group contained five replicates. After the cell density reached 80%, the cells were stimulated with 0, 3, 10, and 30 μM convallatoxin for 24 h. Then, 10 μL of CCK-8 solution was added to each well, and cells were cultured in an incubator at 37 °C and 5% CO2 for another 2 h. The optical density value of each well at 450 nm was measured using a microplate reader, and the cell growth curve was plotted to obtain the IC50 value.
Cell Cycle Measurement
K562 cells in the logarithmic growth phase were collected, and 4 × 105 cells per well were seeded in a 6-well plate. After the cell density reached 80%, the cells were stimulated with convallatoxin for 24 h. Then, the supernatant and cells were washed twice with PBS. The cells were digested, collected by centrifugation (4 °C, 1000 r/min, 4 min), resuspended in 1 mL pre-cooled 70% ethanol, and fixed at 4 °C for 10 h. The supernatant was discarded after centrifugation (4 °C, 1000 r/min, 4 min) and washed with pre-cooled PBS. The cell pellets were resuspended and incubated with propylene glycol iodide (PI) staining solution (0.5 mL) at 37 °C for 30 min in the dark. Finally, the cells were analyzed using a Beckman CytoFLEX flow cytometer (Beckman Coulter Biotechnology Co., Ltd). The proportion of cells at each phase (phase G0/G1, S, and G2/M) was determined, and a fluorescence density distribution graph was used to represent the results.
Cell Apoptosis Measurement
K562 cells in the logarithmic growth phase were collected, and 4 × 105 cells per well were seeded in a 6-well plate. After the cell density reached 80%, the cells were stimulated with convallatoxin for 24 h. Then, the supernatant and cells were washed twice with PBS. The cells were digested and collected by centrifugation (4 °C, 1000 r/min, 4 min), resuspended in 195 μL Annexin V-FITC binding solution and incubated with 5 μL Annexin V-FITC for 10 min at room temperature in the dark. The supernatant was discarded after centrifugation (4°C, 1000 r/min, 4 min), and the cells were resuspended in 190 μL Annexin V-FITC binding solution and incubated with 10 μL PI for 10 min at room temperature in the dark. The cells were detected using a Beckman CytoFLEX flow cytometer (Beckman Coulter Biotechnology Co., Ltd). PI-negative and FITC-positive cells represent early apoptotic cells (lower right quadrant), and double-negative cells represent normal cells (lower left quadrant).
Cell Transfection
A total of 4 × 105 K562 cells per well were seeded into a 6-well plate. After the cell density reached 80%, E2F1 overexpression lentiviral particles were transfected into K562 cells using Lipofectamine 2000 reagent, following the instructions of the manufacturer. The cells were cultured with 10 μM convallatoxin for 24 h to detect related indicators.
Western Blot
K562 cells in the logarithmic growth phase were collected, and 4 × 105 cells per well were seeded in a 6-well plate. After the cell density reached 80%, the cells were stimulated with 0, 3, 10, and 30 μM convallatoxin for 24 h, washed gently with pre-cooled PBS, and lysed with 1 × loading buffer. SDS-PAGE gels were then prepared as previously described.
Equal amounts of protein samples were added to the wells, and the voltage was set to 80 V. After 30 min, the voltage was set to 110 V. When the strips reached three-fourth of the way up the gel, the protein was transferred onto a nitrocellulose (NC) membrane at a voltage of 110 V for 90 min in an ice water bath. Then, the membranes were carefully blocked with 5% milk on a shaker at room temperature for 1 to 2 h. Membranes were incubated with the corresponding primary antibodies overnight at 4 °C and washed with 1 × TNET buffer. After incubation with horseradish peroxidase (HRP)-linked secondary antibody for 2 h at room temperature, the membranes were washed with 1 × TNET buffer and protein bands were analyzed using a Tanon-5200 system (Shanghai Tianneng Technology Co., Ltd). ImageJ software was used to analyze the gray values of each protein band.
MMP Measurement
After treatment with 0, 3, 10, and 30 μM convallatoxin for 24 h, K562 cells were collected and washed twice with PBS, resuspended in 0.5 mL culture medium, and incubated with 0.5 mL JC-1 staining working solution at 37 °C for 20 min. The supernatant was discarded, and the cells were washed twice and resuspended in staining buffer. Fluorescence intensity was detected using flow cytometry. Red and green fluorescence indicates normal and reduced MMP, respectively. The ratio of red to green fluorescence was used to reflect the change in cell MMP.
Molecular Docking Assay
Convallatoxin was constructed using Chemdraw software, and the three-dimensional (3D) conformation of convallatoxin was used for docking. The crystal structure of Akt1 was obtained from the Protein Database RCSB (https://www.pdb.org/) and used for docking. This structure was prepared using the Protein Preparation Wizard as follows: Autodock Vina software was used as the molecular docking program of this study to run a program with a semi-flexible docking method; Pymol software was used to separate the original ligands and protein structures, dehydrate them, and remove organic matter; and Autodock tools was used to hydrogenate, check the charge, specify the atomic type as the Ad4 type, calculate the Gasteiger charge, and construct the docking grid box of the protein structure. In addition, convallatoxin was used to determine the root and select the reversible bond of the ligand in AutoDock-tools. After docking with Vina, the scores of each pair of protein molecule combinations were calculated and the analysis results were analyzed and visualized using PyMOL software.
Statistical Analysis
Experiments were independently performed at least three times in this study, and the results are presented as the mean ± SD. One-way ANOVA and Tukey's test were used to compare the statistical significance of differences between groups using Prism software (ver. 8; GraphPad, San Diego, CA). Statistical significance is displayed as *
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Key Research and Development Projects in Anhui Province for A and Key Program of Natural Science Research of Anhui Provincial Education Department (grant numbers 202104j07020021 and KJ2020A0217).
Author’s Notes
Man Li and Wangwang Xu are equal contributors.
