Abstract
Introduction
Lung cancer is the leading cause of cancer-related death worldwide.1,2 In recent years, strategies such as early screening, surgical resection, targeted therapy, radiotherapy, chemotherapy, and immunotherapy have been used to prevent and treat lung cancer. However, the five-year overall survival (OS) and quality of life of patients with lung cancer remain below 30%.3,4 Therefore, new strategies with less toxicity and fewer side effects must be developed.
Cyclin-dependent kinases (CDKs) and CDK inhibitors (CDKIs) co-regulate the progression of the cell cycle and cell proliferation.5–7 CDKs promote cell-cycle progression by phosphorylating their target proteins, whereas CDKIs bind to and inactivate the function of CDKs.8–10 During the G1 phase, cyclin D-CDK4/6 complex phosphorylates and inactivates the retinoblastoma (Rb), thereby releasing E2F transcription factors that promote the expression of genes required for the transition from G1 to the S phase and DNA synthesis. Cyclin E-CDK2 complex reinforces pRb and initiates the gene expression required for the progression of the S phase.11–15 As CDKIs of the G1 phase, cyclin-dependent kinase inhibitors, 1A (p21) and 1B (p27), inhibit the activity of CDK2/4/6 and interrupt the transition of the cell cycle from G1 to the S phase.16–18 CDKs are over-activated in various cancer types. Some small molecular inhibitors of CDK, including CDK4/6 inhibitors such as palbociclib, ribociclib, and abemaciclib, and CDK2 inhibitors such as dinaciclib and seliciclib, have been applied clinically. 19
Numerous traditional Chinese herbs exert anti-tumor effects and have been promoted for application in lung cancer treatment in the clinical setting.20–22 Notably, the medicine Langdu, which was originally recorded in the
Therefore, we aimed to evaluate the efficacy of EA in lung cancer and elucidate the underlying molecular mechanisms.
Materials and Methods
Reagents
EA was obtained from Nature Standard (Shanghai Standard Technology Co., Ltd, Shanghai), with a purity ≥ 98.0%. Before use, the EA was stored at −80 °C in dimethyl sulfoxide (DMSO, Sigma-Aldrich, Darmstadt).
Cell Culture
The American Type Culture Collection (Manassas, VA) provided the human lung cancer cell lines A549 and H2030. A549 was developed in Dulbecco's Modified Eagle's Medium (DMEM, BasalMedia, Shanghai), and H2030 was cultivated in Roswell Park Memorial Institute-1640 (RPMI-1640, BasalMedia, Shanghai) with 10% fetal bovine serum (FBS, Biochrom AG, Berlin) and 1% penicillin–streptomycin solution (BasalMedia, Shanghai) at 37 °C with 5% CO2.
Cell Viability Assay
Lung cancer cells were planted at a density of 1200 cells per well and grown overnight in black 96-well optical-bottom plates. After 24, 48, and 72 h of treatment with either DMSO or EA at the indicated doses, an ATPlite luminescence assay kit was used according to the manufacturer's instructions. The ATPlite Luminescence Assay kit (BD Pharmingen, Franklin Lake, New Jersey) was used to determine cell viability.
Clonogenic Survival Assay
For the clonogenic survival assay, cells were seeded into 6-cm Petri dishes (300 cells) in triplicate overnight, and then treated with either DMSO or EA for approximately 7 to 10 days. Colonies were fixed with 4% paraformaldehyde, stained with crystal violet, and photographed. Colonies containing more than 50 cells were counted.
Cell-Cycle Assay
After 24 h of treatment with either DMSO or EA, the cells were collected for cell-cycle analysis and frozen in 70% ethanol at −20 °C overnight. After 30 min of treatment with propidium iodide (PI, 36 μg/mL, Sigma, St. Louis, Missouri) at 37 °C in the dark, the affected cells were identified using flow cytometry (BD Pharmingen, Franklin Lake, New Jersey). Statistical analyses were performed using the program FlowJo10.4.
Western Blotting
Radio Immunoprecipitation Assay (RIPA) lysis buffer was used to extract the total protein from the cells; the extracted proteins were resolved using SDS-PAGE and transferred to an Immobilon-PVDF membrane (Merck Millipore Ltd, Tulla Green). The membranes were incubated overnight with primary antibodies (p21, CST, #2947; p27, CST, #3686, Danvers, MA; CDK2, Santa Cruz Biotechnology, #1621; CDK4, Santa Cruz Biotechnology, #23896, CDK6, Santa Cruz Biotechnology, #7961, Santa Cruz, CA) following a 2 h blocking period in 5% non-fat milk in Tris-buffered saline with Tween 20 (TBST). The membranes were then incubated with a secondary antibody solution for 2 h. The images were captured using a Tanon 5200 imager (Shanghai). Immune complexes were identified using an ECL kit (Share Bio, Shanghai).
RNA Silencing
Gene-specific small interfering RNA (siRNA) oligonucleotides were transfected into cells using the Lipofectamine RNAi-MAX Transfection Reagent (Invitrogen) according to the manufacturer's instructions. All siRNAs were synthesized by GenePharma (Shanghai). The sequences of the siRNAs were as follows:
- siControl: 5′-UUCUCCGAACGUGUCACGUTT-3′; - sip21-1: 5′-AAUGGCGGGCUGCAUCCAGGA-3′; - sip21-2: 5′-GACCAUGUGGACCUGUCAC-3′; - sip27-1: 5′-CCGACGATTCTTCTACTCA-3′; - sip27-2: 5′-CCGACGAUUCUUCUACUCA-3′.
Tumor Model
Female BALB/c nude mice were provided by Lingchang Biological Technology Co., Ltd (Shanghai). Every mouse was maintained in a pathogen-free environment at the animal facility of Longhua Hospital. With permission from the Institutional Animal Care and Use Committee of Longhua Hospital, Shanghai University of Traditional Chinese Medicine, animal research was conducted in compliance with the National Guidelines for Experimental Animal Welfare. Briefly, 2 × 106 A549 cells were subcutaneously and bilaterally injected into the dorsal side of each mouse. Tumor-bearing mice were randomly divided into control and EA treatment groups (five mice per group) and intraperitoneally administered EA at concentrations of 0 and 36 mg/kg, respectively. EA (36 mg/kg) dissolved in 5% DMSO + 47.5% normal saline + 47.5% PEG300 was administered intraperitoneally every 2 days for 30 days as part of the treatment. Tumor volumes were determined using the ellipsoid volume formula (Length × Width2)/2, and tumor sizes were quantified using calipers. Electronic scale weights were obtained every 3 days from each mouse. At the end of the experiment, tumor tissue was collected, photographed, and weighed.
Immunohistochemical Staining
For immunohistochemistry, sections of mouse tumor sites were stained with Ki-67 antibody (IHC). After dehydrating the tissue pieces and blocking them with peroxidase, the sections were incubated with primary antibodies at 37 °C for 1 h. Subsequently, a biotinylated secondary antibody was added and incubated for 30 min, 3′-diaminobenzidine (DAB) was applied as a chemiluminescent substrate, and Harris's hematoxylin was used as a counterstain. Images were captured using a digital slice scanner (Konfoong Biotech International, Ningbo).
Statistical Analysis
GraphPad Prism 8 (GraphPad Software, Inc., San Diego, CA) was used to evaluate the significance of the differences between the groups. All data are presented as the mean ± standard error of the mean. Two groups’ parameters were compared using the Student's
Results
EA Suppresses the Proliferation of Lung Cancer Cells
Figure 1A depicts the chemical structure of EA. The half-maximal inhibitory concentration (IC50) values of EA in human lung cancer cell lines A549 and H2030 were 17.26 and 13.17 μM, respectively (Figure 1B). Cell viability assessed using the ATPlite cell proliferation assay revealed a time- and dose-dependent growth inhibition in both cell lines (Figure 1C,
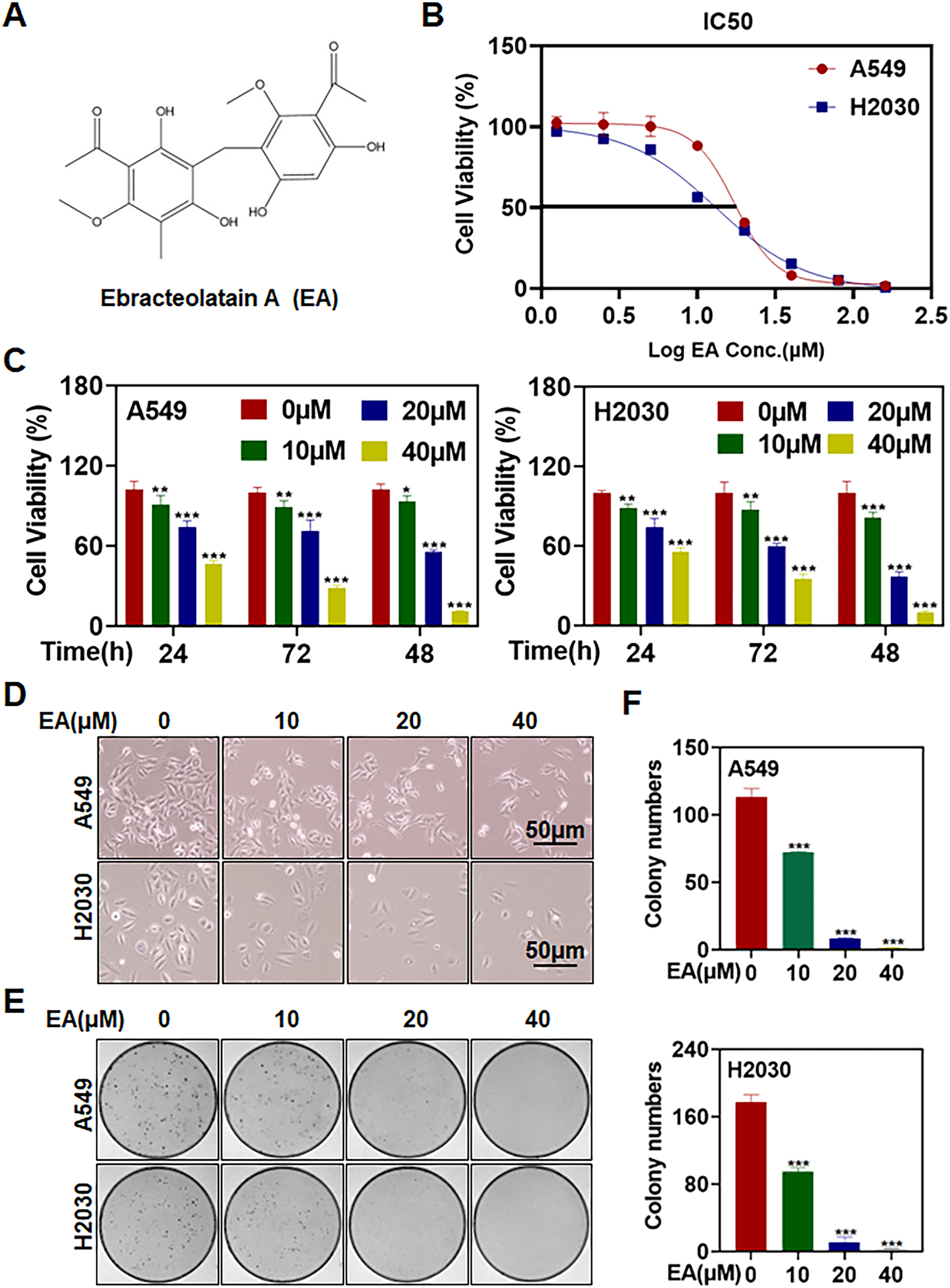
EA suppresses the proliferation of lung cancer cells. (A) Chemical structure of EA. (B) Cells were seeded into 96-well black plates, cultured overnight, treated with EA at the indicated concentrations for 72 h, and analyzed using an ATPlite luminescence assay kit. (C) Cells were seeded into 96-well black plates, cultured overnight, and treated with either 1‰ DMSO or EA (0, 10, 20, and 40 μM) for 24, 48, and 72 h, detected by ATPlite luminescence assay kit (n = 3). (D) Cells were treated with EA for 24 h, and cell morphologies were photographed using an inverted microscope. (E and F) Cells were seeded into 6-cm dishes and incubated with EA for 7 to 10 days. The cells were fixed and stained with crystal violet, and colonies with over 50 cells were counted (n = 3) (*
EA Arrests the Cell Cycle at the G1 Phase in Lung Cancer Cells
PI staining and fluorescence-activated cell sorting (FACS) analysis revealed that EA triggered dose-dependent G1 phase cell-cycle arrest (Figure 2A and B,

EA arrests the cell cycle at the G1 phase in lung cancer cells. (A and B) Cells were incubated with EA for 24 h, followed by PI staining and FACS analysis for cell-cycle profiling. (C) After 24 h of EA treatment, the cells were subjected to western blotting using antibodies against CDK2, CDK4, CDK6, p21, and p27, with β-actin as a loading control (***
EA-Induced p21 and p27 Accumulation Regulate G1 Phase Cell-Cycle Arrest
As CDK inhibitors, p21, and p27, can potentially impede cancer cell-cycle progression by compelling cancer cells to permanently quit the cell cycle and inhibit cancer cell proliferation.26,27 To clarify further the role of p21 and p27 in EA-induced G1 phase cell-cycle arrest in lung cancer cells, we used siRNA to reduce p21 and p27 expression. FACS (Figure 3A and C,

EA-mediated p21 and p27 accumulation regulates cell-cycle arrest in the G1 phase. (A-D) Cells were transfected with siRNA to knockdown p21 or p27 for 72 h and treated with EA (40 μM) for 24 h. Cells were fixed, stained with PI, and analyzed using FACS. Western blotting was performed to determine the expression of either p21 or p27, with β-actin as the loading control (**
EA Inhibits Lung Cancer Growth In Vivo in Mice
Gravimetric analysis revealed that the tumor-bearing mice in the EA-treated group had considerably smaller tumors than those in the control group (Figure 4A and B,

Ebracteolatain A (EA) inhibits lung cancer growth
Discussion
There has been increasing interest in the potential anti-cancer activity of Langdu as it can reportedly inhibit the proliferation, migration, and invasion of various tumor cells
Chinese medicine is regarded as an unexplored treasure trove of possible alternative treatment strategies. EA, which primarily exerts anti-inflammatory effects, also exhibits potential anti-tumor effects. 35 Despite promising results in the preliminary studies, our study has a few limitations. We have not yet identified the upstream regulatory molecular mechanism underlying the EA-induced accumulation of p21 and p27; therefore, further research is required to comprehensively understand the therapeutic potential and underlying mechanisms of EA in cancer treatment. Clinical trials are required to determine the safety and efficacy of EA in humans and identify any potential side effects or interactions with other medications (Figure 5).

Mechanism of action of ebracteolatain A (EA) inhibition of lung cancer.
Conclusion
In conclusion, our findings demonstrated the vital role of EA in inhibiting lung cancer tumor development
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231180083 - Supplemental material for Ebracteolatain A Inhibits Lung Cancer Growth via p21 and p27 Mediated Cell-Cycle Arrest
Supplemental material, sj-docx-1-npx-10.1177_1934578X231180083 for Ebracteolatain A Inhibits Lung Cancer Growth via p21 and p27 Mediated Cell-Cycle Arrest by Weili Zhao, Li Zhang, Meng Li, Shenghao Sun and Lihui Li in Natural Product Communications
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This research was approved by the Cancer Institute at Longhua Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai, China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Natural Science Foundation of China number needs to be changed to 82273028. National Natural Science Foundation of China (82273028), Scientific Research Project of Shanghai Science and Technology Commission (21ZR1482200), Shanghai Rising-Star Program (21QA140890).
Statement of Animal Rights
We confirm that the guidelines on animal rights and treatment have been met, and all details of the approval obtained are indicated within the text of the submitted manuscript.
Statement of Informed Consent Statements
No human participants were included in this study, and informed consent was not required.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
