Abstract
Introduction
Parkinson's disease (PD) is an age-related, progressive neurological condition clinically characterized by involuntary muscle-related movement due to the slow and steady degradation of nigral dopaminergic neurons, followed by striatal dopamine (DA) depletion.
1
The prevalence of PD in Chinese patients is ∼3.9% of those ≥50 years of age, and by 2030, it is estimated to rise to 4.94 million, thus accounting for almost 50% of PD patients worldwide.
2
Yang and Chen reported that in China, PD is associated with an average annual cost of $3226 per patient, within which the majority proportion is related to
The efficacy of
Recently, DA receptor agonists, including pergolide, pramipexole, and ropinirole, have been reported as an effective therapeutic regimen for managing PD, besides
Fisetin is a natural flavonoid that offers neuroprotection against cognitive/synaptic dysfunction induced in mice by amyloid-beta via inhibition of apoptotic protein (caspase-9), and activation of phosphoinositide 3-kinases (PI3Ks) and Akt (Protein Kinase B [PKB]). 9 Prakash and Sudhandiran reported that fisetin ameliorated aluminum chloride-induced neuronal toxicity via inhibition of c-Jun N-terminal Kinase (p-JNK), apoptosis stimulating kinase (ASK-1), cytochrome c, caspases-3 and 9, and modulation of the Bax/Bcl-2 ratio. 10 In addition, fisetin exerts its antidepressant-like effect via inhibition of MAO-A/B activities and modulation of monoamines (5-HT, 5-hydroxy indole acetic acid [5-HIAA], 5-HIAA/5-HT, noradrenaline, DA, and 4-dihydroxyphenylacetic acid [DOPAC]). 11 In the PC12 cell line, fisetin produces a neuroprotective effect in models of Huntington's disease, mainly through the extracellular signal-regulated kinase (ERK) cascade pathway. 12 Although the efficacy of fisetin has not been evaluated against LID, in light of existing evidence, fisetin may exhibit remarkable protective action against neurodegenerative disorders such as PD. Thus, the current study aimed to determine the neuroprotective efficacy of fisetin against LID in 6-hydroxydopamine (6-OHDA)-lesioned experimental rats.
Results and Discussion
LID-Induced Alteration in AIMs Scores and Attenuation by Fisetin
A notable elevation (P < .05) was observed in the axial, forelimb, orolingual, and total AIMs of the LID control group compared to the sham control group on respective days. Fisetin (10 and 25 mg/kg) administration failed to show any noticeable reduction in any AIMs compared to the LID control group on day 7. Fisetin (10 and 25 mg/kg) administration effectively decreased (P < .05) LID-induced increased axial, forelimb, orolingual, and total AIMs compared to the LID control group from day 14 days onward. When compared with the LID control group, amantadine (40 mg/kg) showed markedly lessened (P < .05) AIMs scores on various days. These scores did not differ significantly in sham-

Effect of fisetin on LID-induced behavior alterations of axial (A), limb (B), orolingual (C), and total (D) AIM scores of rats. Data are expressed as mean ± SEM (n = 6) and analyzed by nonparametric 2-way ANOVA followed by Mann-Whitney posthoc test. #P < .05 and &P < .05 in contrast to sham control rats, *P < .05 in contrast to the LID control rats, and $P < .05 in contrast to each other (fisetin and amantadine). AIMs, Abnormal Involuntary Movement Scale;
LID-Induced Alteration in Fall-off Time in Rota Rod Test and Attenuation by Fisetin
Fall-off time was strikingly repressed (P < .05) in the LID control in contrast to sham control rats on respective days. Fisetin (10 and 25 mg/kg) treatment effectively augmented (P < .05) fall-off time from day 14 onward in contrast to the LID control group. Amantadine (40 mg/kg) prominently increased (P < .05) the fall-off time from day 14 to day 42 compared to the LID control group. The fall-off time did not differ significantly in sham-

Effect of fisetin on LID-induced alterations in fall-off time in rota rod test (A), time spent on catalepsy bar (B), numbers of steps in FAS (C), and percent intact stepping in FAS (D) of rats. Data are expressed as mean ± SEM (n = 6) and analyzed by 2-way ANOVA followed by Tukey's multiple range test (for fall-off time in rota rod test and time spent on catalepsy bar) and nonparametric 2-way ANOVA followed by Mann-Whitney posthoc test (for FAS). #P < .05 and &P < .05 in contrast to sham control rats, *P < .05 in contrast to the LID control rats, and $P < .05 in contrast to each other (fisetin and amantadine). FAS, Forelimb Adjustment Stepping;
LID-Induced Alteration in Time Spent on Catalepsy Bar and Attenuation by Fisetin
The time spent on the catalepsy bar was effectively elevated (P < .05) over 42 days in the LID control in contrast to sham control rats. Fisetin (10 and 25 mg/kg) strikingly attenuated (P < .05) time spent on the catalepsy bar from day 7 onward compared to the LID control group. However, fisetin treatment (5 mg/kg) did not significantly reduce the time spent on the catalepsy bar compared to the LID control rats on respective days. Amantadine (40 mg/kg) treatment significantly decreased (P < .05) the time spent on the catalepsy bar from day 7 onward in contrast to the LID control group. Time spent on the catalepsy bar did not alter significantly in sham-
LID-Induced Alteration in Numbers of Steps in FAS and Attenuation by Fisetin
The LID control group showed fewer (P < .05) steps during the FAS test than a sham control group from 7 days onward. Fisetin (5 mg/kg) treatment showed no effective increase in the steps compared to a LID control group on various days. However, treatment with fisetin (10 and 25 mg/kg) markedly elevated (P < .05) the number of steps during the FAS test contrasted to the LID control group from 14 days onward. When contrasted with the LID control group, amantadine (40 mg/kg) showed a significant increase (P < .05) in the number of steps from 7 days onward. The number of steps did not vary in sham-
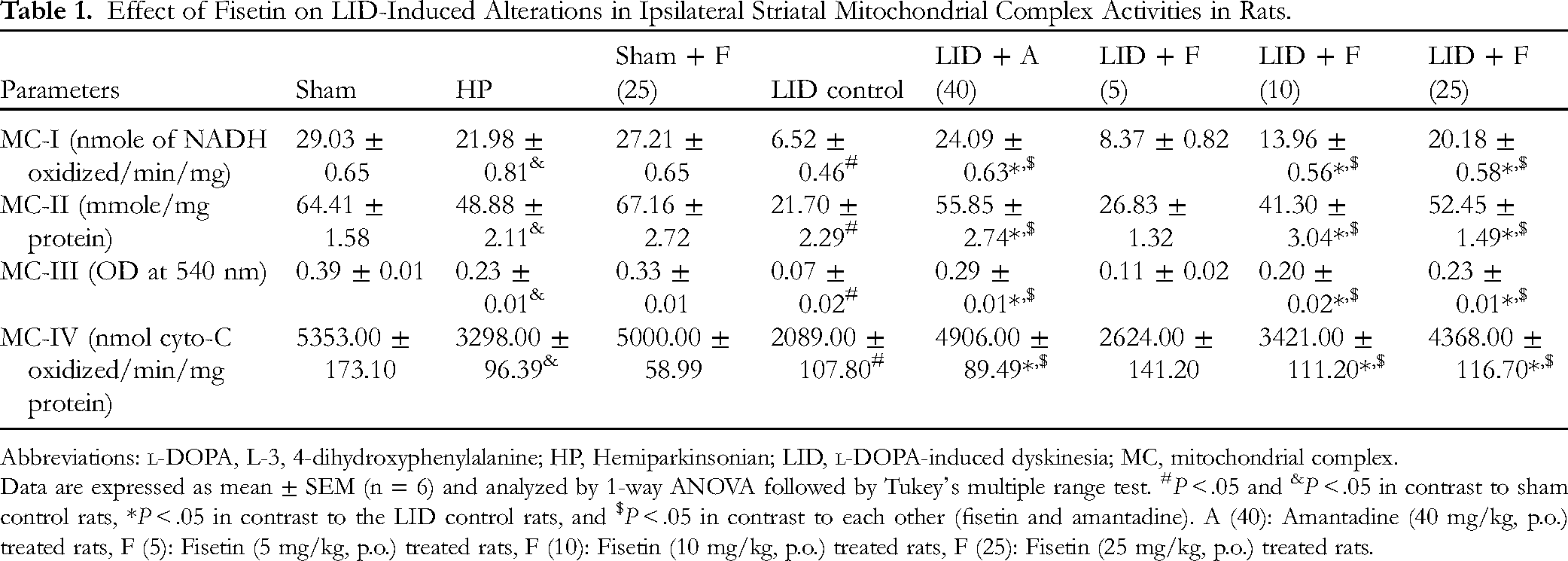
LID-Induced Alteration in Striatal Mitochondrial Complex Activities and Attenuation by Fisetin
The ipsilateral striatal mitochondrial complex (I-IV) activities were strikingly suppressed (P < .05) in LID control rats in contrast to the sham control rats. Moreover, hemiparkinsonian rats also showed a noticeable decrease (P < .05) in the ipsilateral striatal mitochondrial complex (I-IV) activities when compared with sham control rats. Fisetin (10 and 25 mg/kg) prominently augmented (P < .05) ipsilateral striatal mitochondrial complex (I-IV) activities in contrast to the LID control group. However, treatment with fisetin (5 mg/kg) did not show any significant increase in the ipsilateral striatal mitochondrial complex (I-IV) activities in contrast to LID control rats. Treatment with amantadine (40 mg/kg) also noticeably ameliorated (P < .05) the LID-induced diminished ipsilateral striatal mitochondrial complex (I-IV) activities in contrast to the LID control group (Table 1). When compared with hemiparkinsonian rats, fisetin (25 mg/kg) and amantadine (40 mg/kg) treatment in hemiparkinsonian rats showed a significant increase (P < .05) in mitochondrial complex (I-IV) activities (Supplementary File 2). The ipsilateral striatal mitochondrial complex (I-IV) activities did not alter between sham and per se treated rats (Table 1).
Effect of Fisetin on LID-Induced Alterations in Ipsilateral Striatal Mitochondrial Complex Activities in Rats.
Abbreviations
Data are expressed as mean ± SEM (n = 6) and analyzed by 1-way ANOVA followed by Tukey's multiple range test. #P < .05 and &P < .05 in contrast to sham control rats, *P < .05 in contrast to the LID control rats, and $P < .05 in contrast to each other (fisetin and amantadine). A (40): Amantadine (40 mg/kg, p.o.) treated rats, F (5): Fisetin (5 mg/kg, p.o.) treated rats, F (10): Fisetin (10 mg/kg, p.o.) treated rats, F (25): Fisetin (25 mg/kg, p.o.) treated rats.
The contralateral striatal mitochondrial complex levels of sham, hemiparkinsonian, LID control, and treatment groups (amantadine [40 mg/kg] and fisetin (5, 10, and 25 mg/kg)) did not differ significantly compared to each other (Supplementary File 2).
Cumulative evidence suggests that defective mitochondrial respiration is associated with the progression of PD. Induction of cytotoxicity in nigrostriatal dopaminergic neurons via elevated inflammatory responses and neuronal apoptosis results in mitochondrial protein misfolding and aggregation. 13 6-OHDA is one of the mitochondrial neurotoxins responsible for programmed cell death.13,14 The present study observed a decrease in ipsilateral striatal mitochondrial complex activity in the 6-OHDA lesioned rats. Previous investigators also showed that flavonoids, such as quercetin, and resveratrol exert their neuroprotective efficacy against LID.15,16 In the current investigation, the fisetin treatment effectively ameliorated 6-OHDA-induced alteration in mitochondrial complex activities to produce its neuroprotective effect. Recently, Alikatte et al (2021) also reported that fisetin protects against rotenone-induced neuronal damage via the prevention of mitochondrial damage, 17 and our study results are in accordance with this finding.
LID-Induced Alteration in Brain Monoamine Levels in the Striatum and Attenuation by Fisetin
There was a noticeable decrease (P < .05) in the ipsilateral striatal 5-HT and DA levels, and a noticeable increase (P < .05) in ipsilateral striatal 5-HIAA and DOPAC levels of LID control rats in contrast to the sham control rats. Moreover, hemiparkinsonian rats also showed an effective decrease (P < .05) in the ipsilateral striatal 5-HT and DA levels, and a marked increase (P < .05) in ipsilateral striatal 5-HIAA and DOPAC levels when compared with sham control rats (Table 2). When compared with hemiparkinsonian rats, fisetin (25 mg/kg) and amantadine (40 mg/kg) treatment in hemiparkinsonian rats showed significant amelioration (P < .05) in brain monoamine levels (Supplementary File 3). Treatment with fisetin (10 and 25 mg/kg) prominently increased (P < .05) ipsilateral striatal 5-HT and DA level, whereas prominently lessened (P < .05) ipsilateral striatal 5-HIAA and DOPAC levels in contrast to the LID control group. However, there were no noticeable changes in ipsilateral striatal monoamine levels in fisetin (5 mg/kg) treated rats compared to LID control rats. Amantadine (40 mg/kg) prominently elevated (P < .01) ipsilateral striatal 5-HT and DA levels and significantly diminished (P < .01) ipsilateral striatal 5-HIAA and DOPAC levels in contrast with the LID control group. The ipsilateral striatal brain monoamine levels did not alter between sham and per se treated rats (Table 2).
Effect of Fisetin on LID-Induced Alterations in Levels of Ipsilateral Striatal 5-HT, 5-HIAA, DA, DOPAC, and Their Turnover in Rats.

Abbreviations:
Data are expressed as mean ± SEM (n = 6) and analyzed by 1-way ANOVA followed by Tukey's multiple range test. #P < .05 and &P < .05 in contrast to sham control rats, *P < .05 in contrast to the LID control rats, and $P < .05 in contrast to each other (fisetin and amantadine). A (40): Amantadine (40 mg/kg, p.o.) treated rats, F (5): Fisetin (5 mg/kg, p.o.) treated rats, F (10): Fisetin (10 mg/kg, p.o.) treated rats, F (25): Fisetin (25 mg/kg, p.o.) treated rats.
The contralateral striatal brain monoamine levels of sham, hemiparkinsonian, LID control, and treatment groups (amantadine [40 mg/kg]and fisetin [5, 10, and 25 mg/kg]) did not differ significantly compared to each other (Supplementary File 3).
DA degeneration is the hallmark of PD pathophysiology; however, daily administration of
LID-Induced Alteration in Brain 5-HT and DA Turnover in the Striatum and Attenuation by Fisetin
The ipsilateral striatal 5-HT and DA turnover (5-HT: 5-HIAA and DA: DOPAC) were noticeably increased (P < .05) in LID control rats in comparison with the sham control rats. Moreover, hemiparkinsonian rats also showed an effective increase (P < .05) in the ipsilateral striatal 5-HT and DA turnover when compared with sham control rats (Table 2). When compared with hemiparkinsonian rats, fisetin (25 mg/kg) and amantadine (40 mg/kg) treatment in hemiparkinsonian rats showed a significant decrease (P < .05) in brain ipsilateral striatal 5-HT and DA turnover (Supplementary File 3). The turnover of 5-HT and DA in ipsilateral striata was effectively decreased (P < .05) in fisetin (10 and 25 mg/kg) treated rats in contrast with the LID control group. Treatment with amantadine (40 mg/kg) also significantly decreased (P < .05) ipsilateral striatal 5-HT and DA turnover in contrast with the LID control group (Table 2).
The turnover of 5-HT and DA in contralateral striatal of sham, hemiparkinsonian, LID control, and treatment groups (amantadine [40 mg/kg] and fisetin [5, 10, and 25 mg/kg]) did not differ significantly compared to each other (Supplementary File 3).
The role of DA and 5-HT in the pathogenesis of PD has been well-established.21–24 The ratio of DOPAC: DA is considered a hallmark for DA turnover and its utilization, and an increase in this ratio indicates locomotor impairment.21,22 Direct and indirect degeneration of dopaminergic neurons is attributed to PD-associated motor deficits. Administration of serotonergic antagonists caused blockade of 5-HT reuptake inhibiting the rapid increase in synaptic 5-HT levels extracellularly.
25
This 5-HT reuptake blockade has been documented as an important pathway in the antidyskinetic effects of LID.
26
Uncontrolled DA release from the 5-HT terminals is one of the important causes of dyskinesia during PD.
27
In an experimental model, serotonin afferents removal showed a complete blockage of
LID-Induced Alteration in Striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA Expressions, and Attenuation by Fisetin
There was prominent upregulation (P < .05) in the ipsilateral striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions of LID control rats in contrast to sham control rats. Moreover, hemiparkinsonian rats also showed noticeable upregulation (P < .05) in the ipsilateral striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions compared with sham control rats (Figure 3). When compared with hemiparkinsonian rats, fisetin (25 mg/kg) and amantadine (40 mg/kg) treatment in hemiparkinsonian rats showed significant downregulation (P < .05) in ipsilateral striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions (Supplementary File 4). Treatment with fisetin (10 and 25 mg/kg) effectively downregulated (P < .01) ipsilateral striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions in contrast with LID control rats. Amantadine (40 mg/kg) treatment also markedly down-regulated (P < .05) ipsilateral striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions in contrast to the LID control group. The ipsilateral striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions did not differ notably in sham and per se treated rats (Figure 3).
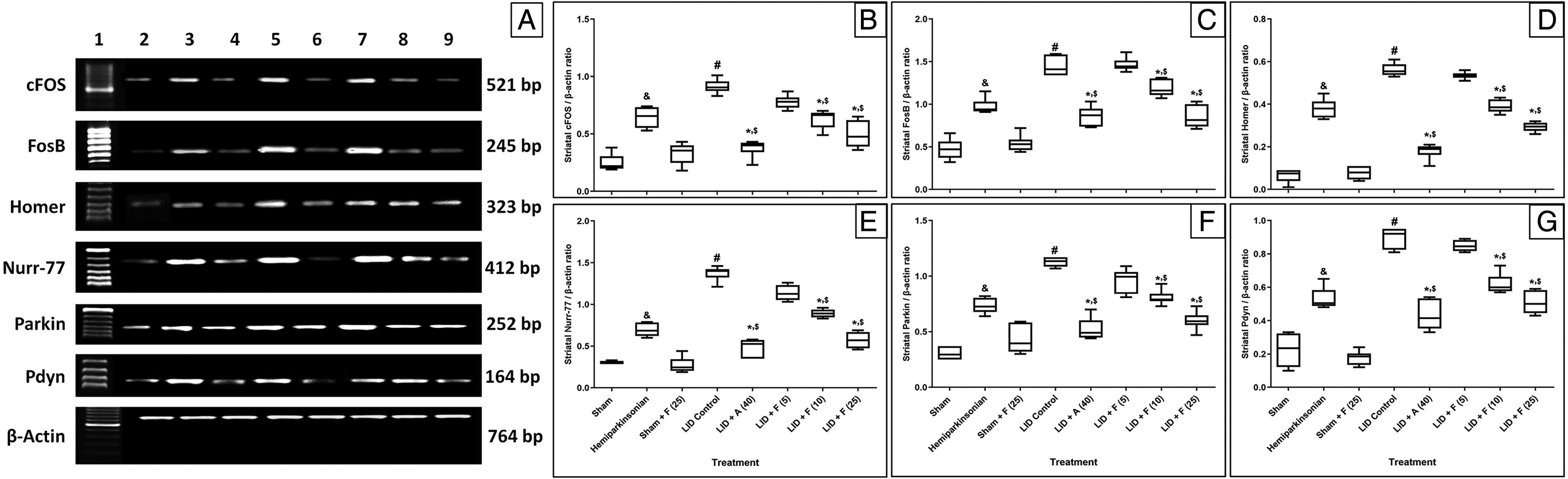
LID-induced alterations in brain ipsilateral striatal cFOS, FoB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions of rats (A), quantitative representation of the mRNA expression of cFOS (B), FoB (C), Homer (D) Nurr-77 (E), Parkin (F), and Pdyn (G). Data are expressed as mean ± SEM (n = 6) and analyzed by 1-way ANOVA followed by Tukey's multiple range test. #P < .05 and &P < .05 in contrast to sham control rats, *P < .05 in contrast to the LID control rats, and $P < .05 in contrast to each other (fisetin and amantadine). F (25): Fisetin (25 mg/kg, p.o.) treated rats. Lane 1: Ladder 1000 bp; Lane 2: mRNA expression of sham rats; Lane 3: mRNA expression of hemiparkinsonian rats; Lane 4: mRNA expression of sham + F (25, Fisetin [25 mg/kg, p.o.]) treated rats; Lane 5: mRNA expression of LID control rats; Lane 6: mRNA expression of LID + A (40, Amantadine [40 mg/kg, p.o.]) treated rats; Lane 7: mRNA expression of LID + F (5, Fisetin [5 mg/kg, p.o.]) treated rats; Lane 8: mRNA expression of LID + F (10, Fisetin [10 mg/kg, p.o.]) treated rats; Lane 9: mRNA expression of LID + F (25, Fisetin [25 mg/kg, p.o.]).
The contralateral striatal cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expressions of sham, hemiparkinsonian, LID control, and treatment groups (amantadine [40 mg/kg] and fisetin [5, 10 and 25 mg/kg]) did not differ significantly compared to each other (Supplementary File 4).
There was a positive correlation between ipsilateral striatal cFOS (R2 = 0.6347, Figure 4A), FosB (R2 = 0.7587, Figure 4B), Homer (R2 = 0.7215, Figure 4C), Nurr-77 (R2 = 0.8231, Figure 4D), Parkin (R2 = 0.7315, Figure 4E), and Pdyn (R2 = 0.7678, Figure 4F) mRNA expression, and total AIMs score. Upregulation in ipsilateral striatal FosB, Homer, Nurr-77, Parkin, and Pdyn mRNA expression was significantly (P < .05) associated with total AIMs score.

A simple regression of ipsilateral striatal cFOS (A), FoB (B), Homer (C), Nurr-77 (D), Parkin (E), and Pdyn (F) on the cumulative AIMs score in
The present investigation demonstrated the anti-LID potential of the dietary flavonoid fisetin during
Multiple lines of evidence indicated that cFos and FosB are vital transcription factors from the Fos family involved in the mechanisms of dyskinesia.28,29 During post DA denervation, the levels of FosB explicitly increased in the striatal region of chronic
A systematic review and meta-analysis support the use of herbal medicine therapy to manage PD. 39 Senolytic therapy, which contains quercetin as an important flavonoid, has been recently evaluated against neurodegenerative diseases in a pilot clinical study. 40 Furthermore, several clinical investigations have been initiated to estimate the potential of fisetin against various disorders. Senolytic formulation, which contains fisetin as an essential agent, has been evaluated against osteoarthritis (NCT04815902) and related articular cartilage degeneration (NCT04210986). Another pilot study assessed the efficacy of fisetin against SARS-CoV-2-associated complications, including inflammation (NCT04476953). In addition, a randomized, double-blind, placebo-controlled study determined the anti-inflammatory effect of fisetin against frailty and inflammation in older adults (NCT03675724) and older women (NCT03430037). Furthermore, another randomized trial recruited patients with advanced chronic kidney disease and diabetic nephropathy to determine the effect of fisetin (NCT03325322). This clinical profile and the existing pharmacological evidence of fisetin's properties support its implication as an adjuvant for managing PD-associated dyskinesia.
Conclusion
The current investigation demonstrated that fisetin has synergistic potential with
Materials and Methods
Animals
To conduct this experiment, 120 healthy rats (male, Sprague-Dawley strain, 220-250 g) were obtained from Mudanjiang Medical University. Upon their arrival, the rats were housed in a clean cage at the animal care facility of Mudanjiang Medical University until the beginning of experiments. They were provided with food and water ad libitum under a normal light/dark cycle. Relative humidity and temperature were maintained at 50 ± 10% and 23 ± 2°C. This study was approved by the Committee for the welfare and ethics of experimental animals of Mudanjiang Medical University (approval no. 20210225-68) and followed approved guidelines and regulations of the National Institute of Health Guide for Care and Use of Laboratory Animals.
Induction of Unilateral 6-OHDA Lesion
Unilateral 6-OHDA-lesioned PD rats were induced by a previously described method. 41 Briefly, desipramine HCl (25 mg/kg, intraperitoneal [i.p.]; Sigma-Aldrich) was administered 30 min before injection of 6-OHDA (to protect degeneration of NE neurons). All 6-OHDA injections were conducted in anesthetized rats using thiopental sodium (45 mg/kg, i.p.) under stereotaxic apparatus (coordinates: anterior-posterior (AP), −2.5 mm; Medial-Lateral (ML), + 2.0 mm; dorsal-ventral (DV), −9.0 mm relative to bregma). The 6-OHDA (12 µg; Sigma-Aldrich) was dissolved in 0.9% NaCl and 0.1% L-ascorbic acid and administered with a Hamilton syringe (10 µL using a 26-gauge needle) at a rate of 2 µL/min (total volume: 4 µL). The needle was equilibrated for 3 min, then withdrawn later.
Animal Screening Using Apomorphine-Induced Rotational Response
Assessment of sensorimotor movement of unilateral 6-OHDS-lesioned rats was performed as described elsewhere. 42 Four weeks following surgery, lesioned rats were selected for a subsequent experiment based on stable apomorphine (0.5 mg/kg, i.p.)-induced asymmetry rotations (at least 7 full-body turns/min) away from the lesioned side. Rats showing below 7 full-body turns/min were selected for the hemiparkinsonian group.
l -DOPA-Induced Dyskinesia
To develop an animal model of dyskinesia, previously selected rats were administered daily with a combination of
Measurements of AIMs Rating
Dyskinesia behavioral changes were assessed for 1 min (every 30 min interval) over a 120 min period. AIMs were divided into 3 subtypes, viz., axial, limb, and orolingual, and scored from 0 to 4 (absent [0], intermittently present at half of the observation period [1], intermittently present more than half of the observation period [2], interruptible and present through the entire rating period [3], uninterruptable and through the entire rating period [4]).16,19 At the end of the experiment, each AIMs subtype score was summarized to obtain the total AIM scores.
Treatment
Rats were arbitrarily divided into the following treatment groups (n = 14):
Rota Rod Test for Motor Skill Learning
Muscular coordination and neurological impairments were evaluated using a rota rod test, according to a method reported elsewhere. 13 Rats were trained for 2 days on rods before experimenting. A rotation speed of 20 rpm was used with a cutoff time of 180 s. The total duration of time spent on a rod by the rats was recorded.
Catalepsy Bar Test
The catalepsy test was conducted based on a previously reported method. 44 A horizontal bar (9 cm above ground level) was used on which both front paws of rats were kept in a half-rearing position. The time taken for removing one foot by animals from the bar was noted. A fixed time of 180 s was considered the maximum cutoff for the catalepsy bar test.
Forepaw Adjusting Steps (FAS) Test
This is implicated in determining akinesia, that is, stepping deficits produced post unilateral depletion of DA. Therefore, FAS was performed based on a previously described method. 18
Tissue Isolation and Estimation of Mitochondrial Function
The rats were sacrificed by decapitation at the end of the experiment to isolate the brain striatum (n = 4-5). The left and right striata were micropunched separately, and the striatum of the rat in the lesioned hemisphere injected was processed for homogenate preparation. Then the supernatant of homogenates (in 0.1 M ice-cold lysis buffer, Tris 10 mM pH 7.5) was used to determine mitochondrial complex (I-IV) activities based on a previously reported method. 45
Estimation of Contents of Biogenic Amines and Metabolites
The levels of biogenic amines (DA, NE, and 5-HT) and metabolites (3,4-dihydroxyphenylacetic acid and 5-hydroxy indole acetic acid) were estimated in the brain striatum (n = 6) according to the method reported by Blasco-Serra et al. 46
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
The mRNA levels of cFOS, FosB, Homer, Nurr-77, Parkin, and Pdyn were estimated in brain striatum tissue (n = 6) using RT-PCR according to a method reported previously.47‐49 The primer sequence for cFOS, FosB, Homer, Nurr-77, Parkin, Pdyn, and β-actin is presented in Supplementary File 1.
Statistical Analysis
Data were expressed as mean ± standard error of the mean (SEM). Statistical significance was analyzed by either 1- or 2-way analysis of variance (ANOVA) using GraphPad Prism software (GraphPad Software Inc). Scores assigned for AIM were analyzed by non-parametric Mann-Whitney's multiple comparisons ANOVA, whereas remaining data were analyzed by either 1-way ANOVA or 2-way ANOVA followed by Tukey's multiple range test. A value of P < .05 was considered to be statistically significant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221136674 - Supplemental material for Fisetin Ameliorates Levodopa-Induced Dyskinesia in Experimental Model Parkinson's Disease: Role of Mitochondrial Activities and Monoamines Turnover
Supplemental material, sj-docx-1-npx-10.1177_1934578X221136674 for Fisetin Ameliorates Levodopa-Induced Dyskinesia in Experimental Model Parkinson's Disease: Role of Mitochondrial Activities and Monoamines Turnover by Wenhui Cao, Shaodong Liang, Yindong Yang, Chuanzhen Zhu, Li Sun and Liming Zhang in Natural Product Communications
Footnotes
Acknowledgment
This project was funded by Mudanjiang Applied Technology Research and Development plan (contract number HT2020NS057).
Authors’ Note
This study was approved by the Committee for the welfare and ethics of experimental animals of Mudanjiang Medical University (approval no. 20210225-68) and followed approved guidelines and regulations of the National Institute of Health Guide for Care and Use of Laboratory Animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mudanjiang applied technology research and development plan (grant number HT2020NS057) and Science and technology project of “Hongqi Scientific Research Fund”, No.: 2020HQ-11.
Supplemental Material
Supplemental material for this article is available online.
Correction (December 2022):
Funding statement is updated with Science and technology project of “Hongqi Scientific Research Fund”, No.: 2020HQ-11.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
