Abstract
Oxyresveratrol, a polyphenolic compound, has been reported as having antioxidant and anti-inflammatory effects. This study determined the neuroprotective effects of oxyresveratrol, extracted from the heartwood of Artocarpus lakoocha Roxburgh (Moraceae), on parkinsonism induced by rotenone. Male Wistar rats were divided into control, rotenone (PD), and rotenone plus oxyresveratrol (OXY) groups. The OXY rats received oxyresveratrol (300 mg/kg orally) on days 1-20. Rotenone (3 mg/kg subcutaneously) was given to PD and OXY rats on days 15, 16, 18, and 20. Motor function was determined by the rotarod test. Brains were collected to analyze dopaminergic neurons, malondialdehyde (MDA) levels, and superoxide dismutase (SOD) and catalase activities. OXY rats exhibited a longer latency to fall than PD rats in the rotarod test (P < 0.01) on day 16. The number of dopaminergic neurons in PD rats was lower than that in controls (P < 0.01), while that of OXY rats was not different from controls. OXY rats showed a reduction in MDA levels (P < 0.01) and increased catalase activity (P < 0.05), while SOD activity was unaltered. The results suggest that oxyresveratrol pretreatment ameliorates motor impairment induced by rotenone and preserves dopaminergic neurons. The neuroprotective mechanism of oxyresveratrol is involved with its antioxidant properties.
Parkinson’s disease is a neurodegenerative disease that causes motor impairments. The main pathology of this disease is the progressive loss of dopaminergic neurons in the substantia nigra. 1 Recently, neuroprotective approaches, for example, the use of antioxidant, anti-inflammatory, and antiapoptotic compounds, have attracted more interest in delaying disease progression. 2 Natural neuroprotectants have attracted attention.
Oxyresveratrol (trans-2,4,3′,5-tetrahydroxystilbene) is a major constituent of the heartwood of Artocarpus lakoocha Roxburgh (Moraceae). 3 The antioxidant and anti-inflammatory effects of this compound have been reported in vitro, 4,5 and its protective effects have been previously demonstrated in in vitro and in vivo models of Alzheimer’s disease induced by β-amyloid, suggesting its promising neuroprotective effects. 6,7 Furthermore, oxyresveratrol has also been shown to protect against 6-hydroxydopamine induced cell death in SH-SY5Y cells, 8 suggesting its potential neuroprotective activity against Parkinson’s disease.
As mitochondrial dysfunction is an important pathogenic mechanism in Parkinson’s disease, 9 rotenone, a mitochondrial complex I inhibitor, has been widely employed to generate a parkinsonism rat model due to its pro-oxidative activity. 10 The molecular mechanisms of rotenone-induced parkinsonian behaviors are associated with the disruption of the mitochondrial electron transport chain, which leads to excessive reactive oxygen species (ROS) production, adenosine triphosphate depletion, oxidative stress, neuronal apoptosis, and motor deficits. 11,12 Therefore, the objective of this study was to determine the neuroprotective effect and mechanisms of oxyresveratrol in a rat model of Parkinson’s disease induced by rotenone.
Results
Effect of Oxyresveratrol on Motor Coordination
The motor coordination of the rats was measured using the rotarod test. Rats were divided into control, rotenone (PD), and rotenone plus pretreatment with oxyresveratrol (OXY) groups. Pretreatment with oxyresveratrol for 14 days had no effect on the motor coordination of the rats, as the latency to fall exhibited by OXY rats was not different from that exhibited by the control rats (110.9 ± 12.5 s vs 111.7 ± 10.0 s, P = 0.89). On day 16, rotenone impaired motor coordination, as PD rats showed a significantly decreased latency to fall compared with that of controls (51.1 ± 32.6 s vs 118.5 ± 3.7 s, P < 0.001). This result indicated that rotenone treatment can induce motor deficit, which represented parkinsonism behavior. Although OXY rats exhibited a significantly lower latency to fall than controls (86.7 ± 21.8 s vs 118.5 ± 3.7 s, P < 0.05), the latency to fall exhibited by OXY rats was significantly higher than that exhibited by PD rats (86.7 ± 21.8 s vs 51.1 ± 32.6 s, P < 0.01). However, the latency to fall exhibited by PD and OXY rats was lower than that exhibited by control rats on day 20 (66.4 ± 30.3 s and 63.6 ± 48.0 s vs 110.2 ± 13.2 s, P < 0.05; Figure 1).

Effects of rotenone and oxyresveratrol on motor coordination in the rotarod test on experimental days 1, 14, 16, and 20. *P < 0.05, ***P < 0.001 compared with control; $$ P < 0.01 compared with PD. PD, rotenone group; OXY, rotenone plus pretreatment with oxyresveratrol group.
Effect of Oxyresveratrol on Dopaminergic Neurons in the Substantia Nigra
Figure 2(A) shows tyrosine hydroxylase (TH)-positive neurons in the substantia nigra, which represent dopaminergic neurons. Rotenone treatment significantly decreased the number of TH-positive neurons in the substantia nigra of PD rats (201 ± 91 cells vs 366 ± 48 cells, P < 0.01 vs control). This result indicated that rotenone caused the dopaminergic neuronal loss, which resembles the pathology of Parkinson’s disease in the brain. In contrast, the number of TH-positive neurons in the substantia nigra of OXY rats (288 ± 77 cells) was not significantly lower than that in the substantia nigra of controls, suggesting that oxyresveratrol pretreatment had protective effects (Figure 2(B)).

(A) Immunohistological staining of dopaminergic neurons in the substantia nigra. (B) Tyrosine hydroxylase-positive cells were counted and presented as the mean ± SEM. **P < 0.01 compared with control. PD, rotenone group; OXY, rotenone plus pretreatment with oxyresveratrol group.
Effect of Oxyresveratrol on Lipid Peroxidation
The levels of malondialdehyde (MDA), a product of lipid peroxidation, were used to evaluate oxidative stress in rat brain tissue. MDA levels in the brains of PD rats appeared to be higher than those in the brains of control rats, but this effect was not significant (1.64 ± 0.46 nmol/mg protein vs 1.22 ± 0.41 nmol/mg protein, P = 0.0514). However, pretreatment with oxyresveratrol significantly decreased MDA levels in the brains of OXY rats compared with PD rats (1.04 ± 0.21 nmol/mg protein vs 1.64 ± 0.46 nmol/mg protein P < 0.01; Figure 3).
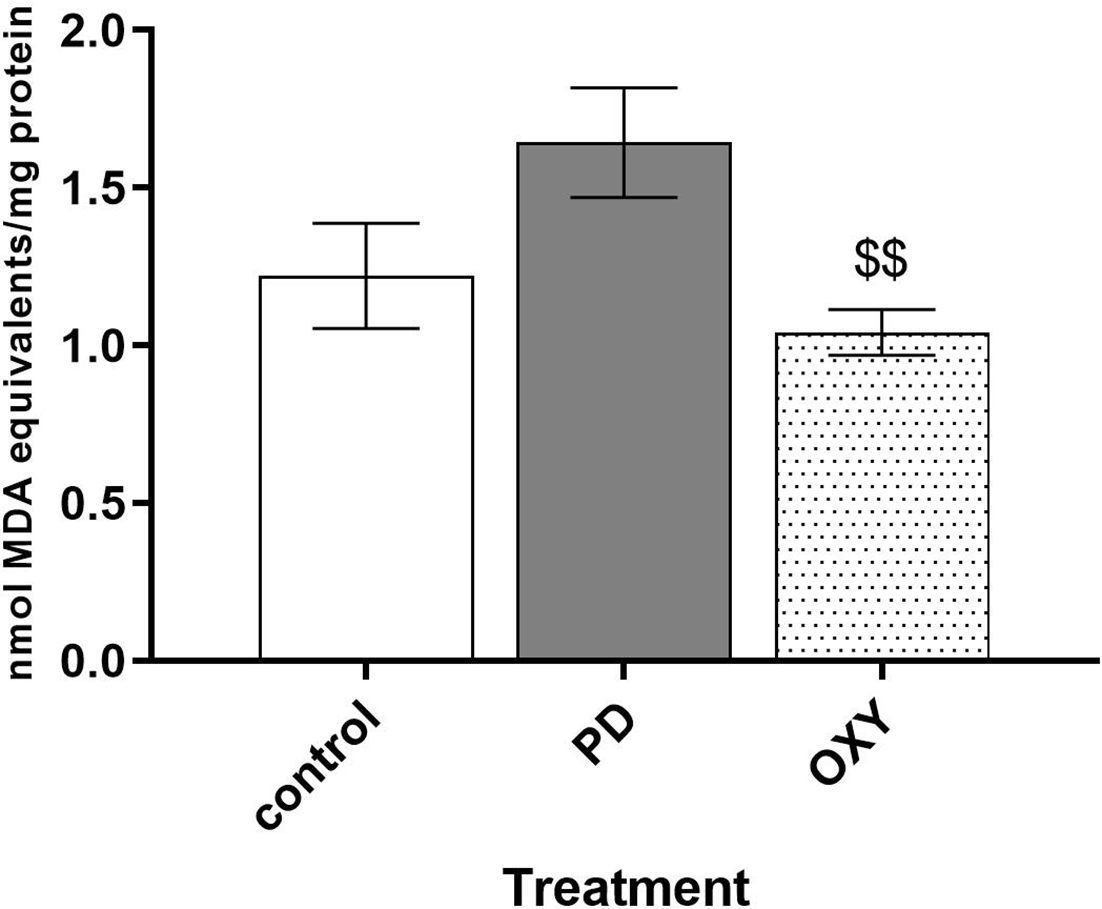
Effect of rotenone and oxyresveratrol on lipid peroxidation. MDA levels in rat brains are presented as the mean ± SEM. $$ P < 0.01 compared with PD. MDA, malondialdehyde; PD, rotenone group; OXY, rotenone plus pretreatment with oxyresveratrol group.
Effects of Oxyresveratrol on Catalase and SOD Activities
The activities of the antioxidant enzymes, catalase and superoxide dismutase (SOD), in rat brain tissue were examined to elucidate the antioxidant mechanisms of oxyresveratrol. Although catalase activity was not significantly changed in PD rats, oxyresveratrol pretreatment significantly increased catalase activity in OXY rat brains compared with control rat brains (0.60 ± 0.34 vs 0.19 ± 0.14, P < 0.05; Figure 4(A)). SOD activity was not altered in either PD or OXY rats (Figure 4(B)).
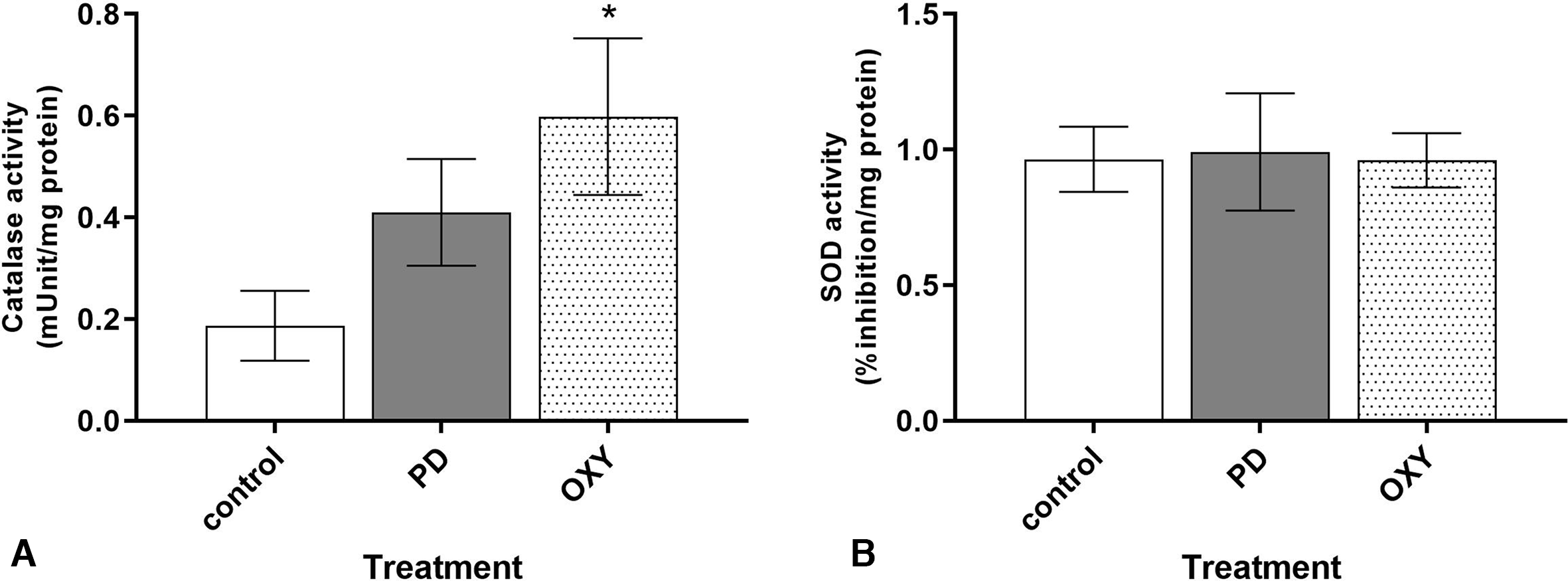
Effects of rotenone and oxyresveratrol on (A) catalase and (B) SOD activities in rat brains. *P < 0.05, compared with control. PD, rotenone group; OXY, rotenone plus pretreatment with oxyresveratrol group; SOD, superoxide dismutase.
Discussion
Neuroprotective approaches for the treatment of neurodegenerative diseases, including Parkinson’s disease, have been increasingly investigated. These therapeutic approaches aim to protect against the progressive loss of dopaminergic neurons and thus subsequently delay disease progression. The protective effects of oxyresveratrol in a Parkinson’s disease model were previously investigated in vitro. 8 The present study demonstrated the neuroprotective effects of oxyresveratrol in vivo.
Oxyresveratrol pretreatment alleviated motor symptoms and preserved dopaminergic neurons in the substantia nigra of rotenone-treated rats. Because oxyresveratrol had no motor-stimulating effect after 14 days of treatment, the preserved motor coordination in the oxyresveratrol pretreatment group after rotenone administration was indicative of its neuroprotective effects. The motor function of oxyresveratrol-treated rats was gradually decreased from day 16 to 20. This could be due to the systemic effect of complex I inhibition of rotenone in other organ systems. 13 Previous studies also reported neuronal damage in the myenteric plexus leading to gastrointestinal dysfunction in rotenone-induced parkinsonism models. 10 Although the motor performance of OXY rats was reduced, the dopaminergic neurons in the substantia nigra were preserved, indicating the neuroprotective effect of oxyresveratrol pretreatment. Previous studies have shown that oxyresveratrol can protect cortical and hippocampal neurons in animal models of stroke 12 and Alzheimer’s disease, 7 respectively. The present study provides evidence of its neuroprotective effect on dopaminergic neurons, which consequently contributed to the improvement of rat motor coordination, as discussed earlier.
Oxidative stress extensively contributes to neuronal death in neurodegenerative diseases. 9,14 The free radical scavenging properties of oxyresveratrol were previously reported in vitro. 5 In this study, a reduction in ROS was indirectly detected by measuring MDA, a product of lipid peroxidation. Pretreatment with oxyresveratrol significantly decreased MDA levels in the rat brain, indicating an enhancement of the capacity of brain tissue to alleviate lipid peroxidation. Under normal conditions, ROS homeostasis is regulated by pro-oxidant enzymes (such as xanthine oxidase and NADPH oxidase), and antioxidants and antioxidant enzymes (such as glutathione, SOD, and catalase). 15 Increasing antioxidant defenses is a promising neuroprotective strategy. 2 The present study revealed that oxyresveratrol pretreatment significantly increased catalase activity. On the other hand, SOD activity was unaltered in both rotenone-treated and oxyresveratrol plus rotenone-treated rats. This result can be explained by the different ROS and reactive nitrogen species (RNS) generated by rotenone treatment. Rotenone-treated rats show reduced levels of thiol, which catalyzes hydrogen peroxide (H2O2), and increased nitric oxide levels and nitric oxide synthase activity, while the levels of superoxide radicals and SOD activity are not changed in these rats. 16 Moreover, oxyresveratrol also directly scavenges superoxide radicals in vitro. 17 These factors might cause the lack of response of SOD to rotenone and oxyresveratrol treatment. Taken together, this study revealed that oxyresveratrol decreases lipid peroxidation marker and increases brain catalase activity. To understand the effect of oxyresveratrol on redox homeostasis in PD brain pro-oxidant enzyme activity, the activities of the glutathione system, as well as markers of protein and DNA damage, should be further investigated.
In conclusion, oxyresveratrol exhibited a neuroprotective effect in a rotenone model of Parkinson’s disease. The antioxidant property of oxyresveratrol is one of the neuroprotective mechanisms against neurodegenerative diseases. This study provides scientific evidence supporting the use of oxyresveratrol for the prevention of progressive neuronal loss in neurodegenerative disease.
Materials and Methods
Animals
Male Wistar rats (National Laboratory Animal Center, Mahidol University, Thailand), aged 8 weeks, were kept under controlled temperature (24 ± 2 °C) and relative humidity (60 ± 10%) on a 12-hour light/dark cycle. Food and water were provided ad libitum. The experiment was performed under Use of Animal for Scientific Purpose Act (2015) and approved by the Institutional Animal Care and Use Committee, Faculty of Pharmaceutical Sciences, Chulalongkorn University, Thailand (approval number 15-33-007).
Chemicals and Reagents
Oxyresveratrol (off-white powder >99% purity), extracted from the heartwood of Artocarpus lakoocha, 3 was suspended in 1% carboxymethylcellulose (CMC) at a concentration of 300 mg/mL. Rotenone (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in 100% dimethyl sulfoxide (DMSO) at a concentration of 150 mg/mL. A working solution of 3 mg/mL rotenone was freshly prepared in sunflower oil.
Experimental Design
Rats were divided into 3 groups (n = 6-8/group): the control, PD, and OXY groups. On days 1-20, control and PD rats were orally given 1% CMC (1 mL/kg), while OXY rats received oxyresveratrol (300 mg/kg, orally [p.o.]). Rotenone (3 mg/kg, subcutaneously [s.c.]) was given to rats in the PD and OXY groups on days 15, 16, 18, and 20, while control rats received 2% DMSO in sunflower oil (1 mL/kg, s.c.). The rotarod test was performed on days 1, 14, 16, and 20. On day 21, the rats were injected with pentobarbital sodium (100 mg/kg, intraperitoneally) and perfused transcardially with phosphate-buffered saline. The brain was quickly removed and cut along the midsagittal plane. The left hemisphere was snap-frozen in liquid nitrogen and kept at −80 °C for further analysis of lipid peroxidation and antioxidant activity. The right hemisphere was kept in 4% paraformaldehyde (PFA) for 48 hours and further analyzed for dopaminergic neurons in the substantia nigra using immunohistochemical techniques.
Rotarod
Motor performance was assessed by the rotarod test, as described, 18 with modification. Rats were trained on the rotarod (Ugo Basile, VA, Italy) at speeds of 10, 15, and 20 rpm 3 days before experimental day 1. On the testing day, each rat was placed on the rotarod, which was set to accelerate from 5 to 40 rpm over 2 minutes. The latency to fall was recorded 3 times per experiment. The longest latency of each rat was reported.
Immunohistochemical Analysis
Dopaminergic neurons in the substantia nigra were stained with an anti-TH antibody (Millipore, Billerica, MA, USA), as described previously. 19 Briefly, the right brain hemispheres in 4% PFA were transferred and kept in 30% sucrose for 48 hours. Brains were then serially cut (40 µm), and 3 sections of the substantia nigra (−5.04, 5.20, and −5.28 from bregma) were selected. The selected brain slices were incubated in 0.1% H2O2 for 30 minutes followed by skim milk for 1 hour. The sections were then incubated with primary anti-TH antibody (1:1000) at 4 °C overnight followed by a 2-hour incubation with secondary antigoat biotin-conjugated antibody (1:500, Millipore, Billerica, MA, USA) at room temperature. The antibody-antigen complexes were trapped with Avidin-Biotin Complex (Vector, CA, USA) followed by reacting with 3,3′-diaminobenzidine (Wako, Japan). The substantia nigra pars compacta areas were identified by comparison with the rat brain atlas. Then TH-positive cells were counted by an experimenter blinded to the treatment group. The total number of TH-positive cells in the 3 brain sections was reported.
Lipid Peroxidation
The thiobarbituric acid reactive species method was used to determine the level of MDA in brain tissues. This method was previously described by Teerapattarakan et al. 19 1,1,3,3- Tetraethoxypropane was used to produce a standard curve.
Catalase Activity
Brain catalase activity was measured using a catalase activity assay kit (Abcam ab83464, Cambridge, UK), and the experiment was performed according to the manufacturer’s protocol. Briefly, 200 µL of catalase buffer was added to 100 mg of each sample. The samples were homogenized and then centrifuged at 10 000×g for 15 minutes at 4 °C. The supernatant, catalase assay buffer, and 1 mM H2O2 were loaded onto a 96-well plate and incubated at 25 °C for 30 minutes. Stop solution (10 µL) was added followed by the developer mix (50 µL). The absorbance was measured at optical density (OD) 570 nm using a microplate reader.
SOD Activity
A SOD activity assay kit (Abcam ab65354, Cambridge, UK) was used to evaluate SOD activity in the brain. Analyses were performed according to the manufacturer’s protocol. Briefly, 10 mg of brain tissue was homogenized in ice-cold 0.1 M Tris/hydrochloric acid containing 5% Triton X, 5 mM β-mercaptoethanol, and 0.1 mg/mL phenylmethylsulfonyl fluoride, and then centrifuged for 5 minutes at 14 000×g at 4 °C. Each supernatant (20 µL) was collected and transferred to a 96-well plate, and then 200 µL of WST solution and 20 µL of enzyme solution were added. The mixtures were incubated at 37 °C for 20 minutes and later measured at OD 450 nm using a microplate reader.
The MDA levels and catalase and SOD activities of each sample were corrected per mg of protein (Bradford Assay kit, Abcam, Cambridge, UK).
Statistical Analysis
The data, presented as the mean ± SEM, were statistically analyzed by one-way analysis of variance, followed by Fisher’s least significant difference post hoc test (GraphPad Prism 7.0, GraphPad Software, Inc., USA). Differences were considered significant at P < 0.05.
Footnotes
Acknowledgments
We are grateful for research assistant funding from the Graduate School, Chulalongkorn University to Weerawan Boonlert.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were performed under the Use of Animal for Scientific Purpose Act (2015) and in accordance with the Ethical Principles and Guidelines for the Use of Animal for Scientific Purposes of the National Research Council of Thailand.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research was financially supported by National Research Council of Thailand (grant number GB-A_60_018_33_03). We are grateful for research assistant funding from the Graduate School, Chulalongkorn University to Weerawan Boonlert.
