Abstract
Neurodegenerative diseases (NDs) are neurological disorders characterized by pathological changes in neuronal structure and function, clinically manifesting as varying degrees of cognitive decline and motor impairment. They include conditions such as Alzheimer's disease (AD) and Parkinson's disease (PD). With the accelerating pace of global population aging, the incidence of NDs continues to rise annually, and no effective treatments currently exist, imposing a heavy societal burden.
Background
With the rapid progression of population aging, the incidence of common neurodegenerative diseases (NDs) such as Alzheimer's disease (AD) and Parkinson's disease (PD) continues to rise. 1 Globally, over 50 million individuals aged 65 and older are bearing the health and economic burdens imposed by NDs, with the number of affected individuals increasing by approximately 10 million annually.It is even projected to exceed 100 million people by 2050. 2 The pathogenesis of neurodegenerative diseases is complex and closely associated with oxidative stress, 3 neuroinflammation, 4 mitochondrial dysfunction, 5 and abnormal protein aggregation. 6 Currently, NDs treatment remains primarily symptomatic, relying on cholinesterase inhibitors and dopamine receptor agonists or agonists.7,8 These approaches cannot effectively delay or halt neuronal degeneration and disease progression, demonstrating limited control over disease progression. 9 Traditional Chinese medicines, characterized by their multi-component nature, multi-pathway mechanisms, multi-target effects, and low toxicity, demonstrate significant advantages in the prevention and treatment of NDs. 10
Accumulating evidence supporting the efficacy of
Primary Active Components and Pharmacological Effects of Polygonatum
Multiple bioactive compounds have been identified in

The pharmacological action mechanism of Polygonatum sibiricum(created by the author usingMicrosoft powerpoint).
Current reports have documented aspects of the pharmacokinetics and bioavailability of PS-containing compounds.
33
Fluorescein isothiocyanate (FITC) labeling showed low oral bioavailability of
Polygonati sibiricum Polysaccharide
Polygonati Rhizoma Saponin
Polygonatum Flavonoid
Flavonoids are one of the key bioactive components responsible for the pharmacological effects of plants. Over 50 flavonoids have been identified in PS.
56
primarily comprising isoflavones, flavones, chalcones, dihydroflavones, and palmatins.
Research on the Role of PS and Its Active Components in AD
Pathological Features of AD
AD is a central nervous system disorder primarily characterized by amnestic cognitive impairment. 64 The pathological hallmarks of AD are the deposition of Aβ plaques and the formation of neurofibrillary tangles composed of microtubule-associated protein tau (Tau).65,66 Tau is one of the most abundant proteins in neurons, maintaining the assembly and stability of microtubules within them. Tau accumulation occurs as oligomers forming neurofibrillary tangles, which enhance its neurotoxicity to neurons. 67
Pathogenesis of AD
Research indicates that glial cell dysfunction, mitochondrial dysfunction, oxidative stress, and inflammatory responses play significant roles in the onset and progression of AD. (Figure 2)68,69 Poor dietary habits and other factors can disrupt the gut microbiota, leading to excessive activation of microglia and the production of large amounts of inflammatory cytokines. 70 The PINK1/Parkin pathway initiates the mitochondrial autophagy program, 71 promoting mitochondrial damage and accumulation, accelerating neuronal death and oxidative stress, which can lead to the development of AD and other neurodegenerative diseases. Apolipoprotein E enhances tau phosphorylation, thereby promoting neurodegenerative disease. Furthermore, when apolipoprotein E4 (ApoE4) binds to Aβ, it slows down Aβ clearance, leading to its accumulation. 72 ApoE4 influences microglia regulation of brain development and aging. Hypercholesterolemia increases oxidative stress and inflammatory responses in the body, elevating blood-brain barrier permeability.73,74 Intracerebral cholesterol participates in axon and myelin formation, while cholesterol metabolites such as 24-hydroxycholesterol (24-OHC) and 27-hydroxycholesterol (27-OHC) can cross the blood-brain barrier. It has been demonstrated that 24-OHC from peripheral blood entering the brain increases Aβ deposition and tau phosphorylation, while 27-OHC activates the renin-angiotensin system, triggering a series of adverse reactions in the brain that lead to neuronal damage and loss.75,76 Impaired mitochondrial function leading to increased energy metabolism and oxidative stress is also one of the mechanisms underlying the development of Alzheimer's disease. 77 Mitophagy reduces ROS, alleviates neuroinflammation, and promotes microglial phagocytosis, thereby decreasing Aβ deposition and impeding AD progressio. 78
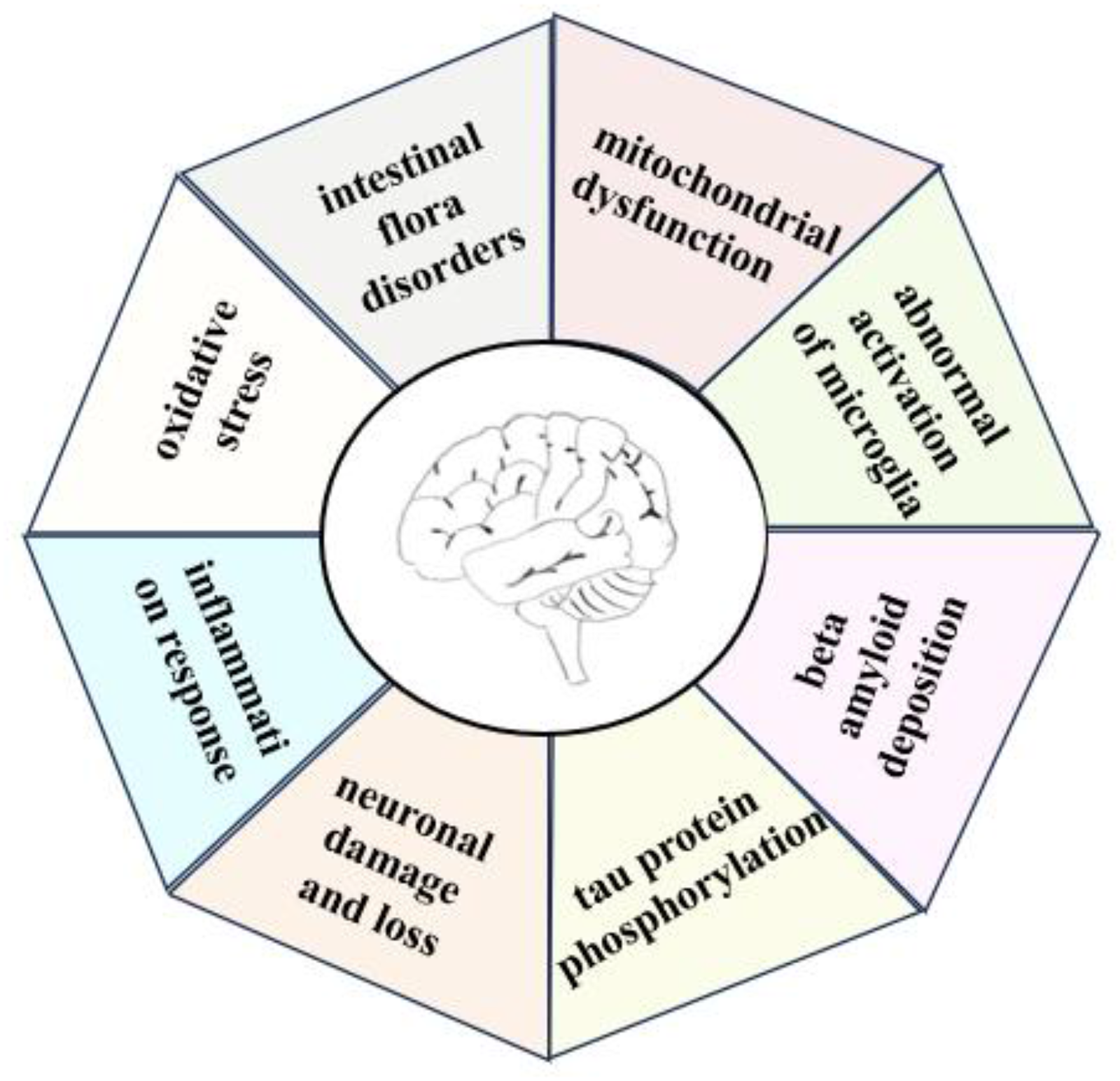
Pathogenesis of AD(Created by the author using Microsoft powerpoint).
Mechanism of Action of PS and Its Active Components on AD
Scholars have conducted network pharmacology and molecular docking analyses on PS, revealing that PS exerts its effects on AD through multiple components, multiple targets, and multiple pathways. 79 Multiple active components in PS, primarily PSP, exert effects on the onset and progression of AD. Their primary mechanisms are closely associated with inhibiting Aβ accumulation through antioxidant stress and mitochondrial autophagy. 80
Antioxidant Stress
A key driver of neurodegenerative diseases is aging. 81 The primary cause of aging is oxidative stress, resulting from extensive cellular damage caused by reactive oxygen species generated in mitochondria and the endoplasmic reticulum. 82 PSP can upregulate the protective proteins Nrf2 and HO-1 to enhance antioxidant defense capabilities and alleviate memory impairment in D-galactose-induced aged mice. 23 The Klotho gene and fibroblast growth factor 23 (FGF-23) share a common receptor that is closely associated with aging. PSP may exert its effects by regulating the Klotho-FGF23 endocrine axis, alleviating oxidative stress, and balancing calcium-phosphorus metabolism. 83 PSP alleviates neurotoxicity in Alzheimer's disease model mice by suppressing endoplasmic reticulum stress and lipogenesis through the AMPK/GSK3β/Nrf2 pathway. 31 A saponin component identified in Polygonatum cyrtonema Hua demonstrated protective effects against Aβ25-35-induced cytotoxicity and oxidative stress damage in PC12 cells, 84 exhibiting potential for therapeutic development in Alzheimer's disease.
Regulation of Neurotransmitters
Glutamate is the primary excitatory neurotransmitter in the central nervous system, participating in synaptic excitation transmission and neurotransmitter release. It is crucial for maintaining sensory cognition, and research indicates glutamate plays a significant role in synaptic plasticity. However, high concentrations of glutamate in the extracellular matrix cause neurotoxic damage to neurons. Both neurons and glial cells possess multiple excitatory amino acid transporters on their membranes, among which the L-glutamate-L-aspartate transporter (GLAST) and glutamate transporter-1 (GLT-1) have been demonstrated to be most closely associated with neurodegenerative diseases. Research indicates that GLT-1 possesses neuroprotective functions. AD mice lacking the GLT-1 allele exhibit rapid cognitive decline, whereas overexpression of GLT-1 alleviates cognitive impairment in these mice.
GLAST plays a crucial role in preventing excitotoxic neuronal damage. PSP suppresses excessive glutamate release and regulates the expression of its associated receptors. 28 Alleviate excitotoxic damage to neurons and provide comprehensive neural protection.Research indicates thatPR increases serum glutamate levels in AD mice, thereby inhibiting NLRP3/calcium pathways to alleviate neuroinflammation. 79 PRP suppresses oxidative stress and neuronal apoptosis by activating the protein kinase B/mechanistic target of rapamycin (Akt/mTOR) signaling pathway. Both mechanisms demonstrate no toxicity in C57BJ/6 mice, suggesting significant long-term therapeutic potential for AD. Akt/mTOR) pathway to suppress oxidative stress and neuronal apoptosis. Both approaches demonstrate no toxicity in C57BJ/6 mice, indicating long-term therapeutic potential for AD. Hippocampal neuroinflammation is an early manifestation of neurodegenerative diseases. 85 Research indicates thatadministering PSP via fecal microbiota transplantation to mice significantly elevates serotonin and norepinephrine levels in depressed mice while reducing pro-inflammatory cytokine levels in the hippocampus. 86 This suggests PSP possesses the capacity to mitigate neuroinflammation in the hippocampus.
Other Mechanisms
PSP can also significantly alleviate cognitive deficits in 5xFAD mice by modulating the gut microbiota, repairing the intestinal barrier, reducing synaptic damage, and enhancing the phagocytic capacity of microglia toward Aβ plaques. 87 Relevant studies indicate that PSP utilizes the gut-brain axis as a regulatory pathway. 88 By reducing intestinal inflammatory responses, protecting the integrity of the intestinal barrier, and increasing short-chain fatty acid production, it maintains normal physiological states. This mechanism reduces the accumulation of amyloid-beta (Aβ) and alleviates the progression of neurodegenerative diseases.Research has found that 89 PSP can regulate the composition and abundance of gut microbiota, increase the levels of butyrate—a metabolite produced by gut bacteria—and mitigate the severity of spinal cord injury in mice.Polysaccharide JP3 extracted from alcohol-processed Polygonatum sibiricum increases the content of probiotics such as Lactobacillus rhamnosus, improves hippocampal lesions and oxidative stress levels, and promotes the proliferation of Lactobacillus rhamnosus as well as the production of butyric acid and isobutyric acid more effectively than inulin. 90 Based on the above research findings, PSP plays a crucial role in neurodevelopment, synaptic plasticity, and neuroprotection by reducing inflammatory responses, regulating gut microbiota, and counteracting oxidative stress. The protective effect against Aβ(25-35)-induced apoptosis in PC12 cells is associated with enhanced PI3 K/Akt signaling. PRP modulates gene expression in critical processes, including stress resistance, aging, autophagy, and mitochondrial function. It reduces Aβ protein deposition in the AD model Caenorhabditis elegans and improves neuromuscular dysfunction. 91 Saponin of Rhizoma Polygonati can inhibit neuronal apoptosis and improve cognitive impairment in Alzheimer's disease mice by modulating the c-Abl/MST1 pathway. 92 The mechanism by which PS extract exerts its therapeutic effects on AD may be related to the following pathways (see Table 1).
Mechanism of PS Treatment for AD.

Research on the Role of PS and Its Active Components in PD
Pathological Features of PD
PD is a neurodegenerative disorder primarily characterized by tremor and motor paralysis, accompanied by autonomic dysfunction leading to multiple non-motor manifestations such as constipation, urinary incontinence, and dysphagia. Recently, its neuropsychiatric signs and symptoms—including depression, anxiety, and emotional apathy—have also drawn significant attention. 96 The pathological manifestations of Parkinson's disease primarily involve the degeneration and loss of dopaminergic neurons in the substantia nigra pars compacta (SNc) and striatum, along with the formation of Lewy bodies. 97
Pathogenesis of PD
Lewy bodies are protein inclusions abnormally aggregated within neurons, primarily composed of α-synuclein. Factors such as iron-induced oxidative stress and DNA damage can exacerbate their deposition. 98 Additionally, the lipid membrane promotes the aggregation of α-synuclein by binding to it. (Figure 3) 99 In PD patients, activated microglia within the substantia nigra produce pro-inflammatory factors such as TNF-α, IL-1, and IL-1β, which promote neuroinflammation. This neuroinflammation disrupts the integrity of the blood-brain barrier, allowing peripheral immune cells and toxins to cross into the brain.Recent research suggests that 100 white matter pathology in PD patients may precede that in the substantia nigra.

Pathogenesis of PD(Created by the author using Microsoft powerpoint).
Patients with PD commonly exhibit dysbiosis of the gut microbiota, specifically characterized by a decline in the family Bacteroidetes and an increase in the family Enterobacteriaceae. 101 This pattern correlates with motor dysfunction and disease progression in PD patients. Gut microbiota are closely linked to NDs. The gut microbiome is dominated by the phyla Firmicutes and Bacteroidetes, while Prevotellales, Bacteroidetes, and Ruminococcales constitute the gut-type composition.102,103 Abnormal shifts in this composition are primarily diet-related, and diet-induced dysbiosis compromises intestinal barrier integrity and normal mucosal immune function. The interaction between microbiota and the brain is bidirectional. The “gut-brain axis” mediates disease alleviation through mutual regulation between the gut and brain via the neuroendocrine-immune system. 104
Mechanism of Action of PS and Its Active Components on PD
Antioxidant Stress
PSP reduces the loss of dopaminergic neurons in the substantia nigra of Parkinson's disease mice and alleviates motor deficits, which is closely associated with its ability to mitigate oxidative stress and endoplasmic reticulum stress. 105 PSP reduces the expression levels of Bax and cleaved-aspartate 3 in Parkinson's disease mice while increasing Bcl-2 expression. It enhances the activity of antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, and catalase in the SNc of PD mice, thereby decreasing malondialdehyde levels. 106 PSP reduces malondialdehyde levels, reverses the expression of oxidative stress-related proteins Nrf2 and Keap1, and diminishes MPTP-induced increases in endoplasmic reticulum stress-related proteins. Furthermore, in vitro and in vivo studies indicate that PSP exhibits no chronic toxicity in C57BJ/6 mice. PSP also activates p70S6 K and 4E-BP1 signaling pathways via Akt/mTOR-mediated mechanisms and stimulates NADH quinone oxidoreductase 1 through NF-κB-related factor 2. This activation modulates heme oxygenase-1, glutathione S-transferase catalytic subunit, and glutathione S-transferase regulatory subunit, thereby exerting anti-apoptotic and antioxidant effects. 107 This demonstrates significant potential for neuroprotective applications in Parkinson's disease.
Regulation of Monoamine Neurotransmitters
PSP modulates monoamine neurotransmitters involved in PD, primarily including dopamine (DA), norepinephrine (NE), and serotonin (5-HT). These three neurotransmitters play crucial roles in regulating mood, cognition, and behavior.PSP improves cognitive impairment by alleviating neuroinflammation and synaptic damage. 108 It effectively suppresses lipid peroxidation injury, enhances serum superoxide dismutase activity in exercise-induced fatigue rats, reduces malondialdehyde levels, decreases 5-HT production in brain tissue, and increases DA content. Major depressive disorder frequently accompanies PD, with etiology intimately associated with neurotransmitter dysregulation. PSP significantly upregulates 5-HT and NE concentrations in murine hippocampal regions, consequently attenuating depressive-like phenotypes. 109 Cortisol can influence motor and cognitive behaviors in PD patients through the hypothalamic–pituitary–adrenal axis (HPA axis). 110 Research indicates that 30 PSP can reduce levels of IL-6 and IL-1β, increase acetylcholine (ACh)—crucial for learning and memory—and decrease gamma-aminobutyric acid (GABA) to regulate neurotransmitter balance.
Alleviating Neuroinflammation
Peroxisome proliferator-activated receptor gamma (PPAR-γ) may participate in the initiation of anti-inflammatory signaling in M2 macrophages. 111 Xia et al demonstrated that PSP can promote dopamine neuron regeneration by suppressing inflammatory responses and apoptosis through upregulating PPAR-γ expression.112,113 The HPA axis regulates central inflammation by modulating the expression of inflammatory cytokines and the activation of microglia. PSP enhanced the antioxidant capacity of mice in a chronic stress model 114 manifested by increased superoxide dismutase and catalase activity and reduced malondialdehyde concentration, thereby protecting neurons from oxidative damage. Additionally, PSP lowers circulating corticosterone levels, indicative of HPA axis regulation, and mitigates glucocorticoid-induced hippocampal injury commonly associated with chronic stress. 115 PSP can inhibit the TLR4/NF-κB-related pathway from exerting its effects. 116 Fecal microbiota transplantation suppresses the TLR4/MyD88/NF-κB signaling pathway and its downstream proinflammatory mediators in the substantia nigra and colon. 117 Suppression of neuroinflammation in the substantia nigra (SN) further mitigates damage to dopaminergic neurons. PSP may prevent SPS-induced PTSD-like behaviors and synaptic injury by modulating oxidative stress and NLRP3-mediated inflammatory responses, potentially through Nrf2/HO-1 signaling pathways. 108 The PSP regulates the PD through specific pathways. (See Table 2).
Mechanism of PS Treatment for PD.

Research on the Role of PS and Its Active Components in Age-Related Neurodegenerative Diseases
Aging accelerates neurodegenerative disease progression through multiple mechanisms. Brain cell senescence is accompanied by a high-secretory phenotype; upon entering cell cycle arrest, senescent neurons secrete diverse inflammatory cytokines, chemokines, and matrix metalloproteinases, thereby fostering neuroinflammation. 121 Reduced autophagy efficiency and lysosomal dysfunction caused by neuronal aging promote intracellular protein aggregation in the brain. 122 These processes further promote the occurrence of NDs. Brain-derived neurotrophic factor (BDNF) is a key neurotrophic protein that binds to tropomyosin receptor kinase B (TrkB), thereby upregulating downstream protein expression and enhancing cognitive function. 123 Research has demonstrated that PS can improve cognitive impairment in naturally aged rats by activating the BDNF-TrkB signaling pathway. 124
Additional studies indicate that advanced glycation end products (AGEs) trigger the accumulation of modified proteins leading to neurodegenerative diseases (NDs), ultimately promoting their pathological progression.125,126 Notably, significant gender differences exist in late-stage AGEs and oxidative damage levels among Alzheimer's disease (AD) patients, whereas such disparities are less pronounced in Parkinson's disease (PD) patients. 127 This suggests differing glycation-oxidation mechanisms between the two diseases, with specific distinctions requiring further investigation. Overall, PS and its active components can impede the progression of NDs through multiple pathways.
Conclusion
The primary distinction between AD and PD lies in their early clinical manifestations and affected neuroanatomical regions. AD predominantly presents as an amnestic dementia, characterized by hippocampal and medial temporal lobe atrophy alongside Aβ/tau pathology. In contrast, PD typically manifests initially with motor symptoms—specifically tremor and rigidity—reflecting core pathology involving substantia nigra dopaminergic neuronal degeneration and α-synuclein aggregation; cognitive decline usually emerges only in later stages. Regarding immune dysregulation, AD is distinguished by senescent microglia exhibiting impaired phagocytic capacity and oligoclonal expansion of CD8⁺ T cells, reflecting an immunosuppressive or senescent phenotype. Conversely, PD features persistently activated microglia coupled with CD4⁺ T-cell infiltration, indicative of a state of immune hyperactivation.
PS can influence the onset and progression of NDs through multiple pathways, including alleviating neuroinflammation, reducing oxidative stress, regulating neurotransmitters, and modulating gut microbiota. This provides a solid foundation for the clinical application of PS and its active component PSP. Early intervention with PSP may help slow the progression of neurodegenerative diseases and improve patients’ quality of life, offering new options and directions for the treatment of neurodegenerative disorders.
Despite its therapeutic potential, the clinical translation of PS faces significant hurdles. First, existing evidence derives predominantly from animal models and cellular assays, with a paucity of standardized preclinical or clinical investigations specifically targeting NDs. Second, extraction methodologies for Polygonatum and its bioactive constituents remain poorly standardized; variations in geographical origin, processing techniques, and formulation parameters compromise product consistency and efficacy, thereby constraining industrial scalability and clinical reproducibility. Nevertheless, PS and its principal constituent PSP demonstrate considerable promise in ameliorating NDs through attenuation of neuroinflammation and oxidative stress, modulation of gut microbiota and associated metabolites, and regulation of neurotransmitter systems. However, critical gaps persist in clinical validation, regulatory frameworks, manufacturing standardization, and neuropharmacological characterization. Future studies must elucidate the metabolic fate of PSP in animal and human subjects, determine whether microbiota-derived metabolites penetrate the blood-brain barrier to exert direct neurological effects, and generate comprehensive neuropharmacological data to enable successful clinical translation.
Footnotes
Author Contributions
Huiyang Tang: Conceptualization, Methodology, Formaoriginal draft. Xinlu Chen: Conceptualization, Methodology, Formal Analysis. Shanshan Wang: Conceptualization, Formal Analysis, Investigation. Yueying Wu: Methodology, Formal Analysis, Investigation. Xiaoya Li: Methodology, Formal Analysis, Investigation. Jiali Yuan: Conceptualization, Formal Analysis, Investigation. Bo Qiao: Conceptualization, Methodology, Formal Analysis, Writing-review & editing.
Funding
Yunnan Provincial Department of Education Science Research Fund (2025J0450). Yunnan Provincial Key Laboratory of Integration of Traditional Chinese and Western Medicine for Chronic Disease Prevention and Treatment Open Project (YPKLG2024-010).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
