Abstract
Phytochemical investigation of the 70% ethanolic extract of the roots of Rubia oncotricha Hand.-Mazz. led to the isolation of 2 new anthraquinone glucosides named 1-hydroxy-2-hydroxymethyl-9,10-anthraquinone-11-O-β-D-glucopyranoside
Introduction
Rubia oncotricha Hand.-Mazz., belonging to the plant family of Rubiaceae, is widely distributed in China as an endemic species. The dry roots and rhizomes of the plant have been used as Chinese folk herb for the treatment of cough, expectorant, jaundice, bronchitis, injuries, and so on by the local minorities in Guizhou province.1, 2 However, there have been few reports on the chemical constituents of this plant.
Previous phytochemical investigations of R oncotricha showed the isolation of anthraquinones, naphthoquinones, naphthohydroquinones, and triterpenoids,3–5 some of which exhibited biological activities, such as antitumor and antinematodal activities.6, 7 During the course of searching for bioactive compounds, we performed a phytochemical study on the root and rhizome of R oncotricha. Previous studies mainly focused on the isolation and identification of low polar compounds, and there have been few reports on the highly polar chemical constituents of R oncotricha. Therefore, an n-butanol fraction of 70% EtOH extract of this species was investigated, which led to the isolation of 2 new anthraquinone glucosides (
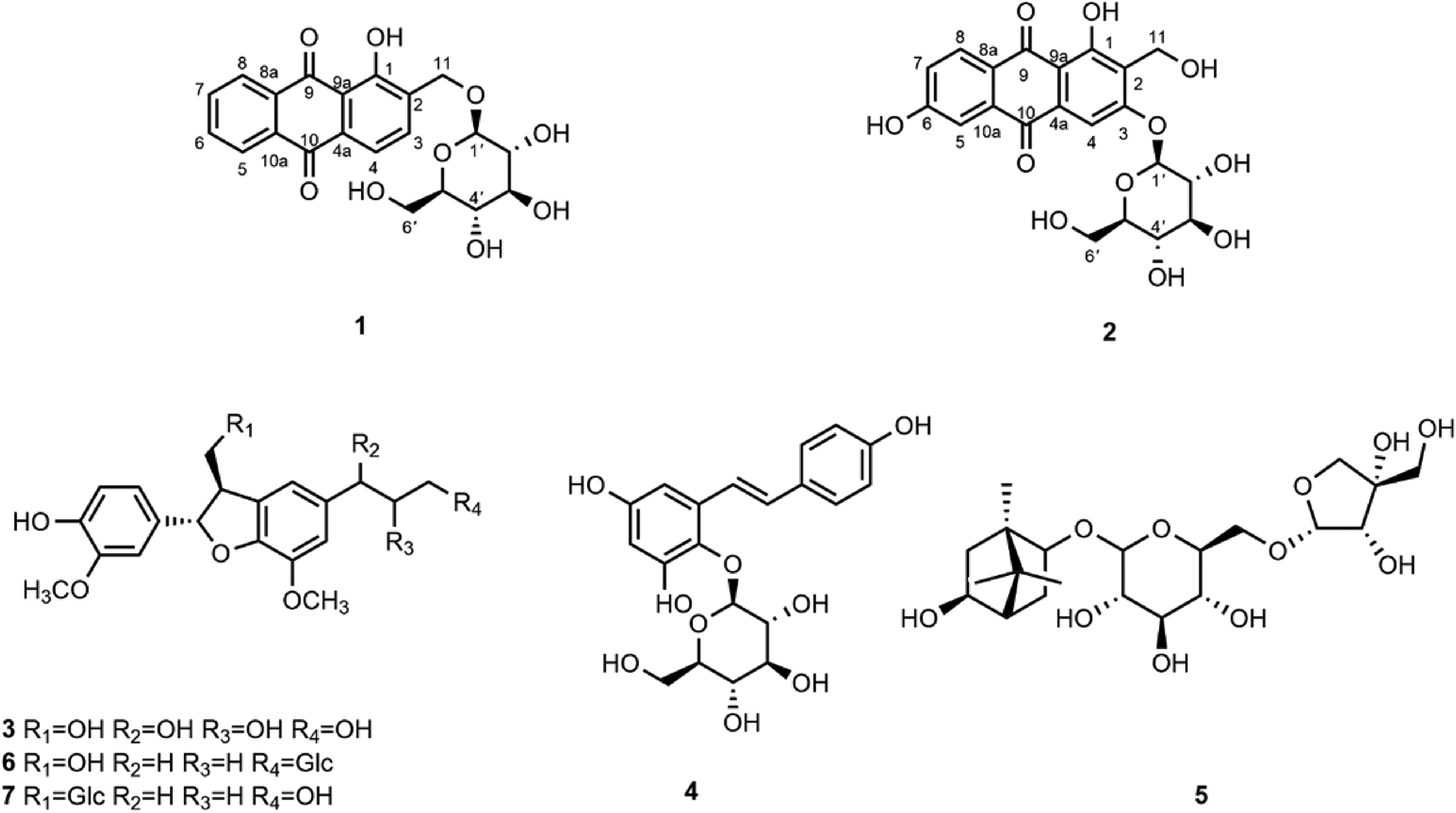
Chemical structures of compounds
Results and Discussion
Compound

Key HMBC correlations of
1H and 13C NMR Spectroscopic Data of Compounds

Abbreviations: NMR, nuclear magnetic resonance; DMSO-d6, dimethylsulfoxide-d6
1H at 400 MHz and 13C at 100 MHz in DMSO-d6.
Compound
In addition, 5 known compounds (
Conclusions
In the present work, 2 new anthraquinone glucosides (
Experimental
General
HR-ESI-MS was measured on a Bruker Daltonics micro TOF-Q II mass spectrometer (Bruker Daltoniks GmbH). 1D and 2D NMR were recorded on a JEOL ECS 400 NMR spectrometer (Jeol). Ultraviolet (UV) spectra were acquired on a UV-2700 spectrometer (Shimadzu Corporation). Optical rotation values were measured on an AUTOPOLⅥ spectrometer (Rudolph Co., Ltd). IR spectra were recorded on an IR Tracer-100 spectrometer (Shimadzu Corporation). Column chromatography was performed with macroporous resin (D101, Tianjin Haiguang Chemical Co., Ltd), silica gel (200-300 mesh and 300-400 mesh, Qingdao Haiyang Chemical Co., Ltd), Toyopearl HW-40C (Tocho Corporation), Toyopearl HW-40F (Tocho Corporation) and Sephadex LH-20 (Pharmacia Biotech). Semipreparative high-performance liquid chromatography (HPLC) was performed on Shimadzu LC-20AP with ACE C18-PFP columns (10 × 250 mm, 5 μm) (Phenomenex). Thin layer chromatography was performed on silica gel GF 254 plates (Qingdao Haiyang Chemical Co. Ltd). GC analysis was carried out on a Shimadzu-2010 plus gas chromatograph (Shimadzu Corporation) using a ZB-5 capillary column (30 m × 0.25 mm i.d. × 0.25 μm).
Plant Material
The roots and rhizomes of R oncotricha were purchased in September 2018 in Guiyang, Guizhou Province, China, and were identified by Associate Professor Qingde Long from the Department of Medicinal Botany and Pharmacognosy (School of Pharmacy, Guizhou Medical University). The specimen (No. 20180227) was stored at the Guizhou Provincial Key Laboratory of Pharmaceutical Preparations at Guizhou Medical University.
Extraction and Isolation
The dried root and rhizome of R oncotricha (5.0 kg) were extracted with 70% (v/v) EtOH 3 times. After evaporation of the solvent under reduced pressure, the crude extract (592.0 g) was dissolved in water and then partitioned in turn with petroleum ether, ethyl acetate, and n-butanol. The n-butanol was evaporated under reduced pressure to yield 215.0 g. The n-butanol extract (215.0 g) was chromatographed on silica gel column chromatography eluted gradient with a gradient of CHCl2-MeOH (30:1, 20:1, 15:1, 10:1, 7:1, 4:1, 1:1, v/v) to give 11 fractions (Fr.1-11).
Fraction Fr.2 (44.1 g) was subjected to silica gel column chromatography eluting with EtOAc-MeOH (15:1 to 1:1, v/v) afforded 9 subfractions (Fr.2.1-2.9), Fr.2.2 (2.7 g) was fractionated by silica gel column chromatography eluting with EtOAc and Toyopearl HW-40F column chromatography (MeOH) to yield compound
Fr.3 (2.5 g) was subjected to silica gel column chromatography eluted with EtOAc-MeOH (8:1-1:1, v/v) to obtain 7 subfractions (Fr.3.1-3.7). Fr.3.5 (334.8 mg) was further subjected to repeated Sephadex LH-20 column chromatography (MeOH), and Toyopearl HW-40F column chromatography (MeOH) to obtain Fr.3.5.1(18.0 mg). Further purification of Fr.3.5.1 with preparative HPLC (1.5 mL/min, MeOH/H2O 56:44, v/v) yielded compounds
Fr.4 (10.72 g) purified by Sephadex LH-20 column chromatography (MeOH) afforded 6 subfractions (Fr.4.1-4.6). Fr.4.3 (2.4 g) was applied to MCI column chromatography (50%-100% MeOH/H2O, v/v) and Toyopearl HW-40F column chromatography (MeOH) to get compound
Fr.6 (14.0 g) was subjected to silica gel column chromatography eluted with EtOAc-MeOH (15:1-1:1, v/v) to obtain 5 subfractions (Fr.6.1-6.5). Fr.6.3 was separated by repeated Sephadex LH-20 column chromatography (CHCl3/MeOH 1:1) and Sephadex LH-20 column chromatography (MeOH) to Fr.6.3.1. Fr.6.3.1 (95.0 mg) was subjected to silica gel column chromatography eluted with EtOAc-MeOH (15:1-1:1, v/v) and Toyopearl HW-40F column chromatography (MeOH) to obtain compound
1-Hydroxy-2-hydroxymethyl-9,10-anthraquinone-11-O-β-D-glucopyranoside (
1,3,6-Trihydroxy-2-hydroxymethyl-9,10-anthraquinone-3-O-β-D-glucopyranoside (
Compound
Acid Hydrolysis and Determination of Absolute Sugar Configuration
Each compound (0.5 mg) was hydrolyzed with 2 M HCl (2.0 ml) at 95 °C for 3 h. After cooling, the reaction mixture was partitioned between water and ethyl acetate 3 times. The aqueous layer was repeatedly evaporated to dryness with methanol, until neutral. The residue was dissolved in pyridine (0.4 mL), then L-cysteine methyl ester hydrochloride (1.0 mg) was added. The mixture was reacted at 60 °C for 1 h, and then trimethylsilyl imidazole (0.15 mL) was added to the reaction mixture at 60 °C for another 1 h, after the reaction was completed, the reaction solution was blown dry with nitrogen. The reaction residue was dissolved in water (1.0 mL), and then n-hexane (0.5 mL) was added for extraction 3 times, and the n-hexane layer was analyzed by GC. The absolute configurations of the monosaccharides were confirmed to be D-glucose by comparison of the retention times of the authentic sample [tR (D-glucose) 29.196 min, tR(L-glucose) 29.558 min)] (Supplemental material).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221117311 - Supplemental material for Chemical Constituents From the Roots of Rubia oncotricha
Supplemental material, sj-docx-1-npx-10.1177_1934578X221117311 for Chemical Constituents From the Roots of Rubia oncotricha by Li Jiang, Zhilong He, Yushan Nie, Yang Wang, Xue Ma, Ting Liu, Yuan Lu, Yonglin Wang and Yongjun Li in Natural Product Communications
Footnotes
Acknowledgements
We are grateful to Associate Professor Qingde Long from the Department of Medicinal Botany and Pharmacognosy (School of Pharmacy, Guizhou Medical University) for identifying plant species.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (U1812403), Central Government Guides Local Science and Technology Special Projects (20184006), and Innovation Talent Team Project of Guizhou Province ( 20165677).
Ethical Approval
Ethical Approval is not applicable for this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
