Abstract
The present review is focussed on the structural elucidation of the bicyclic and tricyclic diterpenoid title compounds isolated from Pseudopterogorgia species, displaying a direct structural relationship with the biosynthetic precursor GGPP (regular isoprenoid skeletons). A compilation of 1H and 13C NMR spectroscopic data is presented grouped by similar spin systems. Apparent inconsistencies or potential missassignments are discussed, pointing out convenient revisions of data assignment to improve structure correlations. Some hemisynthetic intermediates in the structural elucidation process are included, as well as data of representative synthetic compounds.
Pseudopterosins A-D (PsA-PsD), tricyclic diterpene D-xylosides, were reported as new naturally occurring metabolites isolated from the gorgonian octocoral Pseudopterogorgia (now Antillogorgia) elisabethae with promising antiiinflammatory and analgesic activities.1,2 seco-Pseudopterosins A-D (sPsA-sPsD), a related set of bicyclic arabinosides, were reported shortly after. 3 The structures of the isolated compounds are shown in Figure 1.

Structures of pseudopterosins (Ps) and seco-pseudopterosins (sPs).
In the second report on pseudopterosin derivatives (PsE-PsL), 4 two compounds (PsE-PsF) displayed the same PsA aglycone (PsAa) part, but with 4-O (instead of 5-O) glycosides, one four-member set (PsG-PsJ) with different aglycones and two (PsK-PsL) with enantiomeric PsAa. However, the aglycone structure of PsG was revised as the 13-epimer of PsAa. 5
Later on, PsM-PsO and sPsE-sPsG were reported,6,7 but the structure drawings of the first group displayed xyloside sugar moieties (with revised drawings in Marine Natural Products annual review) 8 , while the text suggested D-arabinose (revised in MNP annual review). 9
Duplication in structures and names (PsP-PsQ, 10 PsP-PsV,11,12 PsP-PsZ 13 ) resulted, unfortunately, from three almost simultaneous publications in 2004 (the case being revised in MNP annual reviews.9,14 Many of the trivial names were clarified in the 2007 review 14 [according to communication with the authors]
Finally, the hemisynthetic iso-PsE 15 and a reduced seco-pseudopterosins group (sPsH-sPsI, 13 sPsJ16,17 and sPsK 17 ) were added to the list. The diterpenes from gorgonian octocorals (including an array of nonglycosylated: diterpenes amphilectanes, serrulatanes, elisabethanes, elisapteranes, colombianes, cumbianes, seco-cumbianes, elisabanes, sandresanes, caribanes and related skeletal systems) were reviewed. 18 Occasionally, complementary spectroscopic data were provided21,22 of known (newly isolated) metabolites for activity testing.23,24
In Figure 1, a phytane numbering system is used (owing to the direct structural relationship of the structures with the biosynthetic precursor GGPP)25,26 and the preferred sugar drawings (with standard axial and equatorial substituent orientations) 27 and numbering of R subscripts (the same number of ring attachment, thus, R1 = aglycone) for unequivocal assignments are shown in Figure 2.

Preferred sugar drawings and numbering.
Along with the absolute configuration of sugars determination, controversial data, revised names, and convenient revisions of NMR data in compilation tables will be discussed next.
Actually, while this manuscript was in preparation, a partial phytane numbering was specified for some structures, including one reported as “iso-PsA” prepared from the dimethoxy PsA-PsF aglycone. 28
The Phytane Naming and Numbering System: Introductory Notes and Benefits
In this review, the parent phytane numbering system, already reported for the monocyclic “prenylbisabolanes” 25 , will be applied in the further cyclised compounds discussed, considered a more convenient atom numbering and naming for “regular” skeletons, such as Ps and sPs. The recommended numbering of side-chains for prenols 26 is a convenient addition for a straightforward locant assignment of methyl groups as 31, 71, 111, 151, 16 (avoiding the inconsistent exchange of positions 16/17 or 19/20).
The locant direct relationships of the parent skeletons are considered the most remarkable of benefits: position of O-derivatives are always C-4 and C-5 (only OH and OR positions are exchanged). Thus, names for PsC and sPsC as examples are:
3-O-acetyl-α-D-xyloside 3-O-acetyl-α-arabinoside
Other compounds may be named simply by changing to the new stereochemistry, glycoside name and position, if different (-5-hydroxy-4-yl).
The Identification and Chirality of the Sugar Components
The reported Ps and sPs glycosides are β-D-xylo, α-L-fuco, β-D-arabino and β-L-xylopyranosides. The anomers are designated α or β, depending on the configurational relationship between the anomeric center and the anomeric reference atom. In L-fucopyranoses, 29 the 1,5- substituents are on opposite sides (1C4 conformation), the axial anomer is α. Vicinal proton-proton coupling constants for pyranose rings were predicted by a simple additivity rule. 30
The 1,4- in β-xylosides are trans-diequatorial,1,2 and the absolute configuration of xylose in PsA-PsD was established by acidic hydrolysis of PsA and treatment in the same manner as D- and L-xylose. D-xylose afforded an [α]20D + 23° (c 1.64, H2O), L-xylose [α]20D = −26° (c 1.54, H2O) and the sugar component from PsA [α]20D = + 29° (c 0.95, H2O). On the other hand, the later reported β-L-xylosides
Rotations obtained from pure D-arabinose and PsF were negative {[α]D −104° (c = 3.0, H2O) and [α]D −17.5° (c = 1.7, H2O) respectively}, pointing out a very strong decomposition (less reliable method) and the axial anomer was β. Although a similar decomposition result was reported in PsG and PsK, L-fucose was assigned on the basis of the negative rotations observed, in spite of the low values: PsG {[α]D = −20.5° (c = 0.7, H2O)} and PsK {[α]D −11.5° (c = 1.0, H2O)}, whereas from pure L-fucose the result was {[α]D −75° (c = 10, H2O), and the axial anomer was α
Attempts to determine the absolute stereochemistry of arabinose in sPsA-sPsD 3 failed, “presumably due to decomposition of the sugar under conditions required for glycoside cleavage” (but the L structure was shown). Unfortunately, the same result was reported for sPsH-sPsI (the L structure was also shown). However, the hydrolysates of PsU and PsZ under standard conditions afforded strong negative optical rotations (pointing out D-arabinose resistance in pseudopterosins). 13 In sPsK, 17 chirality as α-L-fucopyranoside was assumed (signals essentially identical to PsP, unambiguously established previously).
The sugar chirality of aldose enantiomers was established also by a GLC protocol.
31
Retention times reported when applied to fucose, arabinose and xylose,
12
were:
D/L-fucose derivatives rt: Found 13.4 and 15.2 min; the sample from PsP 15.2 min. D/L-arabinose derivatives rt: Found 11.5 and 10.4 min; the sample from PsT 11.5 min D/L-xylose derivatives rt was also reported: 11.05/12.01 min.
Controversial Data Reported of PsM-PsO and SPsE-SPsG Structures
In spite of the poor elucidation data (lacking important coupling constants to assess sugar structure) reported for PsM-PsO and sPsE-sPsG and “non-standard” structures drawing, the paper went through the reviewing process (quite a surprise!). Fortunately, the MNP annual reviews pointed out some inconsistencies (as already mentioned). A diaxial relationship according the J1,2 = 9.0 Hz of H-C1′,H-C2′ may fit either β-ara or β-xyl glycosides.
30
Furthermore, the pseudopterosins were elucidated as D-arabinopyranosides (
Again, in sPsE a “non-standard” sugar drawing was depicted (only one out of five substituent bond orientations was acceptable) and 1H and 13C NMR data assigned to the aglycone components of sPsE-sPsG (no correspondence with those values reported for sPsA aglycone portion nor the sPsH and sPsI glycosides). The large (8.9 Hz) diaxial J1,2 coupling indicated the equatorial (β) linkage with the aglycone (instead of α), while the L configuration of the fucose was assigned on the basis of a strong negative optical rotation of the sugar obtained by hydrolysis.
The Revised Names Agreement
Introduction
Up to three structures were given the same trivial name (PsP, PsQ), while two different names were given to some compounds. 10 The revision of some trivial names without a joint communication of the authors may be found since 2008 32 (revised names are shown in Figure 1) pointing to a proposal by Rodríguez or by Blunt after communication with Rodríguez14,33
One Structure and two Trivial Names
The identity of L-fucopyranosides PsP (
Other Renamed Glycosides
Diacetylated glycosides pseudopterosins R, S, W, × and Z (
NMR Data Compilation Tables and Structure Elucidation Comments
Introductory Note
The report of leubethanol, 34 a serrulatane closely related to erogorgiane, included 1D and 2D NOE experiments and 1H-NMR full spin analysis. The results may be a useful model for sPs and Ps aglycones. The small calculated J values are a good rationale for broad multiplicities, as well as for prediction of arabinosides or fucosides [equatorial H-C(4′) small axial-equatorial J3,4 and J4,5a, and very small eq-eq J4,5e. 30 ]
Proton Multiplicities
Multiplicities (real) are quoted with the common s (singlet, no J quoted), d (doublet omitted in the tables as implied in every J), t (triplet), q (quartet) or combinations such as dd, td, qd, ddd, tdd, qdd, etc When J values with several nonequivalent systems are very close (or equal), multiplicities may appear as simple “multiplete-like” and are indicated as m* (t* = dd, q* = td/ddd, etc); b is quoted for broad signals (unresolved small couplings) and m to mean “complex or overlapping multiplets” (implied but not quoted if a chemical shift interval is reported).
Data Table Arrangements
Data are presented in separated groups according to multiplicity (m). Groups of m = 0,1, and 2 (aglycone) are in decreasing chemical shifts, while m = 3 and sugar are in locant order. Revised locant assignments show the original position as superscripts. Data of some synthetic compounds are included5,35,36 (to provide better spectroscopic characterization).
Data of seco-pseudopterosins, aglycones and some methyl ether derivatives are compiled as Table 1 (1H NMR arranged as reported for sPsK) and Table 2 (13C NMR). Tables 1 to 10. 1H NMR and 13C NMR Data of seco-Pseudopterosins and Pseudopterosins.
1H NMR Data of Seco-Pseudopterosins, Aglycones and Methyl Ether Derivatives.

13C NMR Data of Seco-Pseudopterosins and Aglycone Methyl Ether Derivatives.
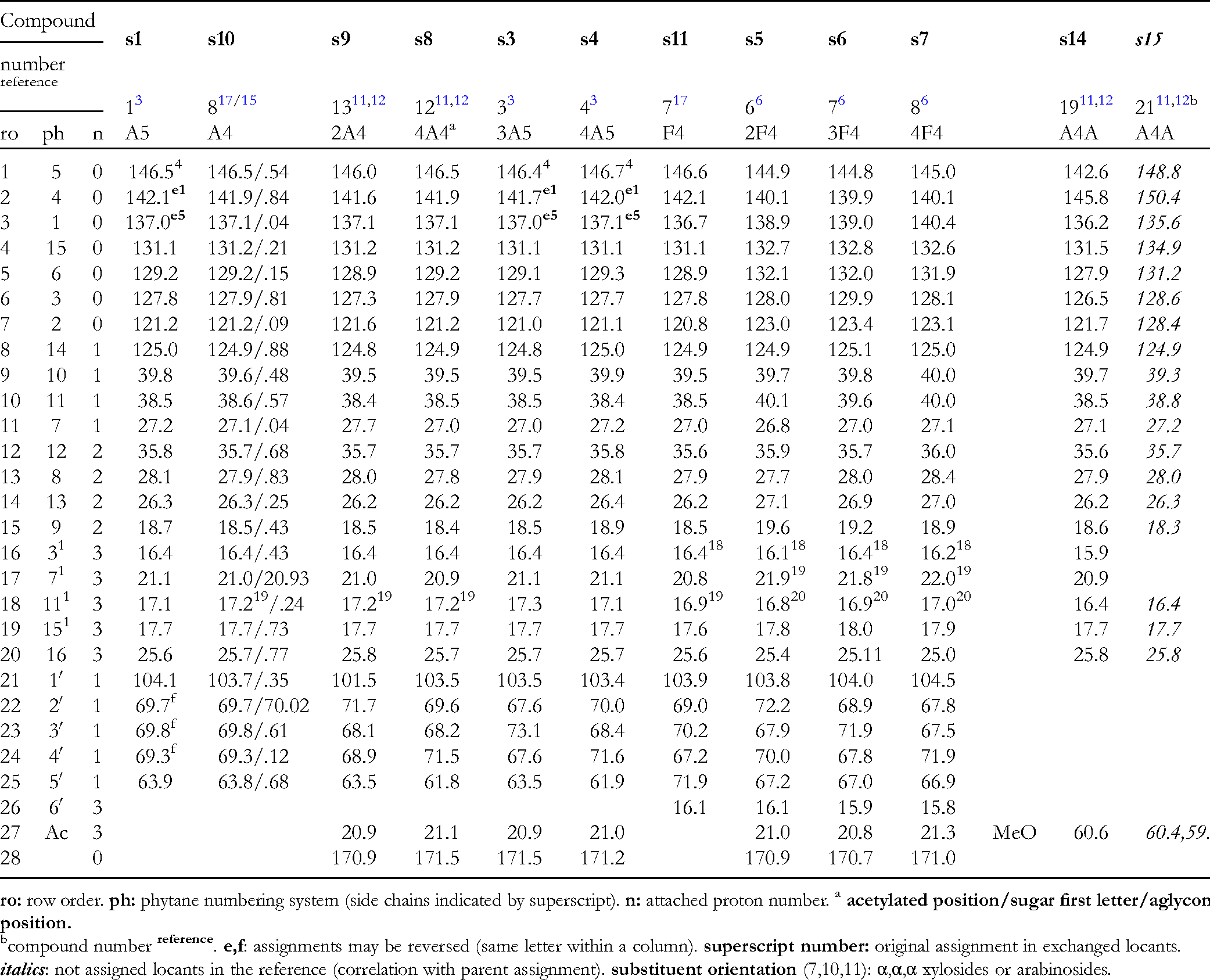
compound number
1H NMR Data of Pseudopterosin Xylosides.

13C NMR Data of Pseudopterosin Xylosides.

1H NMR Data of Pseudopterosin 4-O-Fucosides.

OH signals not included (D2O exchg.).
1H NMR Data of Pseudopterosin 5-O-Fucosides.

13C NMR Data of Fucose Glycosides of Pseudopterosins.
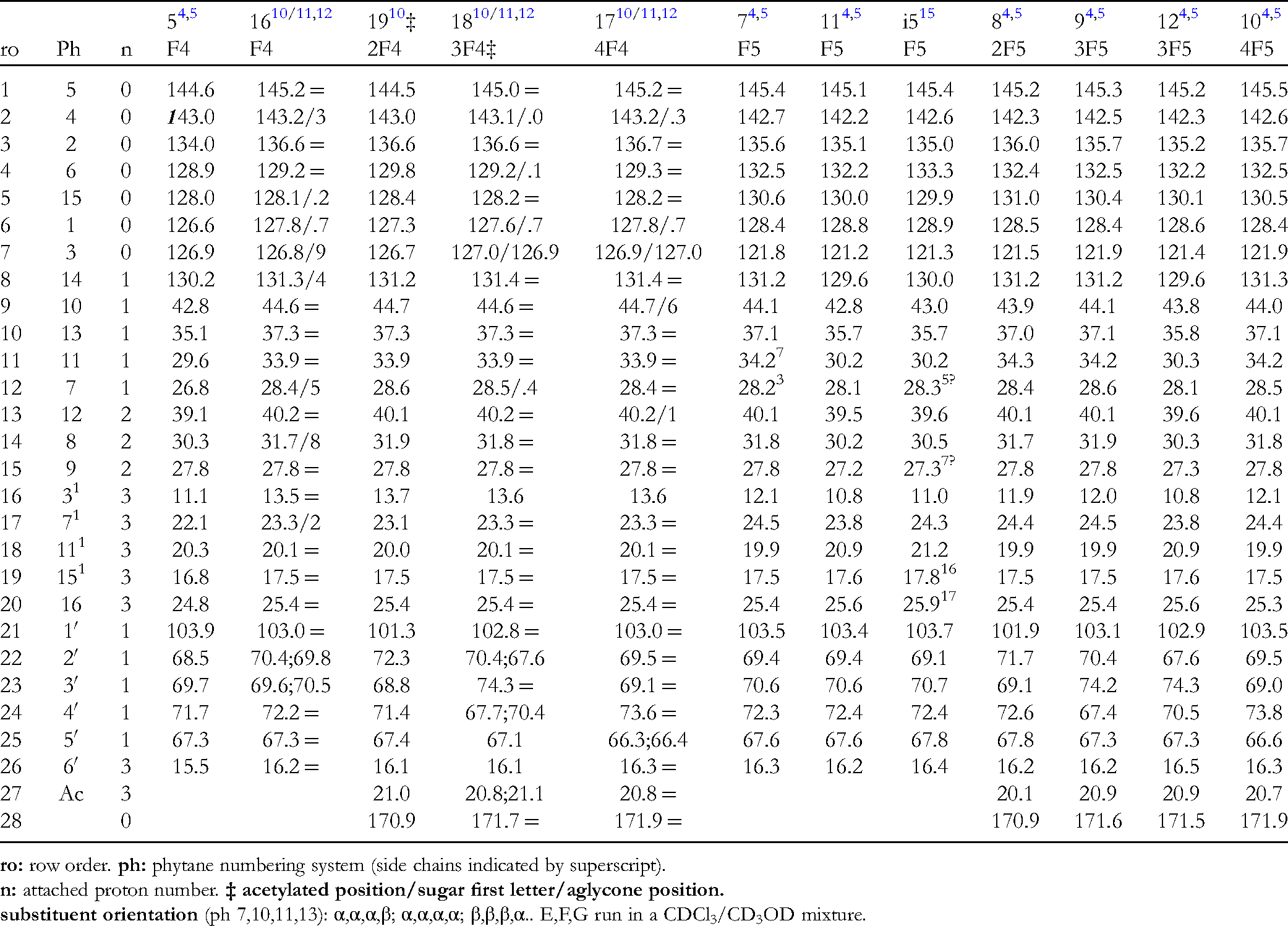
1H NMR Data of Pseudopterosins Arabinose Glycosides.

13C NMR Data of Arabinose Glycosides of Pseudopterosins.

same signals within a column may be reversed.
1H NMR and 13C NMR Data of Pseudopterosin Dicetylated Glycosides (Revised Names)7.

Revised Assignments According to the Logical Sequence of Coupling Constants
Before 2D-COSY spectra were developed, the proton-proton vicinal relationship was established through the logical sequence of coupling constants. Signal reassignments have been proposed for sPsJ 4-(β-D-arabinopyranoside), PsD [5-(4-O-acetyl-β-D-xylopyranoside)], PsE 4-(α-L-fucopyranoside) and PsL [5-(3-O-acetyl-α-L-fucopyranoside)], and are summarized in Table 11.
Revised Assignments According to the Logical Sequence of Coupling Constants.

Sugar Acetylation Effects
Downfield chemical shifts were recognised as showing the location of sugar acetylation. A related effect in the vicinal 13C chemical shifts has been observed. The chemical shift effects on 2′-O-acetylation
Chemical Shifts Effects in Pseudopterosins and Seco-Pseudopterosins Acetylated Glycosides.
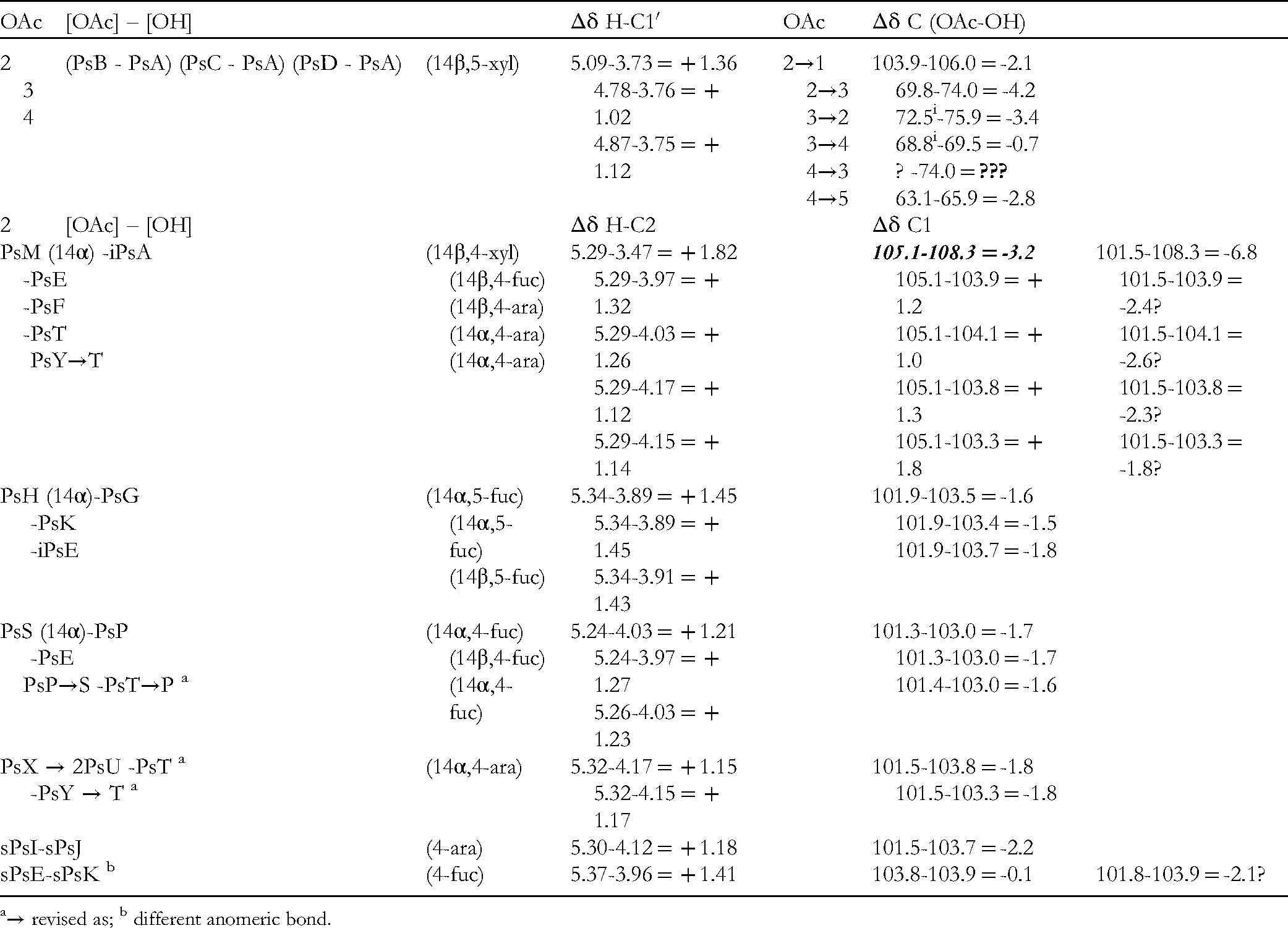
→ revised as; b different anomeric bond.
The controversial data of PsM are out of range. Thus, the chemical shift is either “true” or a simple typing error (101.5 may fit quite well). The extremely large downfield shift for iPsA data may be due (in part) to a solvent effect (MeOH-d4: ca. 4.0 ppm).
There is also a remarkable apparent lack of effect in sPs-4-fucosides (103.8/103.9), but the sPsI/sPsJ 4-arabinoside pair displayed the expected shift (101.5/103.7: Δδ −2.2 ppm). In Ps pairs, only PsM, as mentioned, appears again as a real effect or another simple typing error (101.8 may fit well: Δδ −2.1 ppm).
Small shift differences (ca.< 0.2 ppm) may not be significant as recorded for the anomeric proton (4.74- 4.49 = + 0.25) with 2′-OAc. Overlapping multiplets in PsA (2′, 3′, 4′/14,7) were partially separated in monoacetates. Curiously, a real typing error occurred with the 5′a PsB proton: it appears at ca. 3.25 ppm in the figure, but was printed as 3.52! (Table 3).
The 13C Chemical Shifs of 4 - or 5 -O-Glycosyl sPs Derivatives
As pointed out, 16 the 13C data of the aglycone moieties for 4-O-glycosylated seco-pseudopterosins (eg, sPsK and sPsJ) 17 and 5-O-glycosylated congeners (seco-PsA–seco-PsD) are coincidentally closely similar to each other.
Actually, the average shielding effect for sPsA-sPsD observed (

Revised possible sugar structures of PsM and sPsE.
13C NMR Data of C-OR sPs and Ps Aglycones.
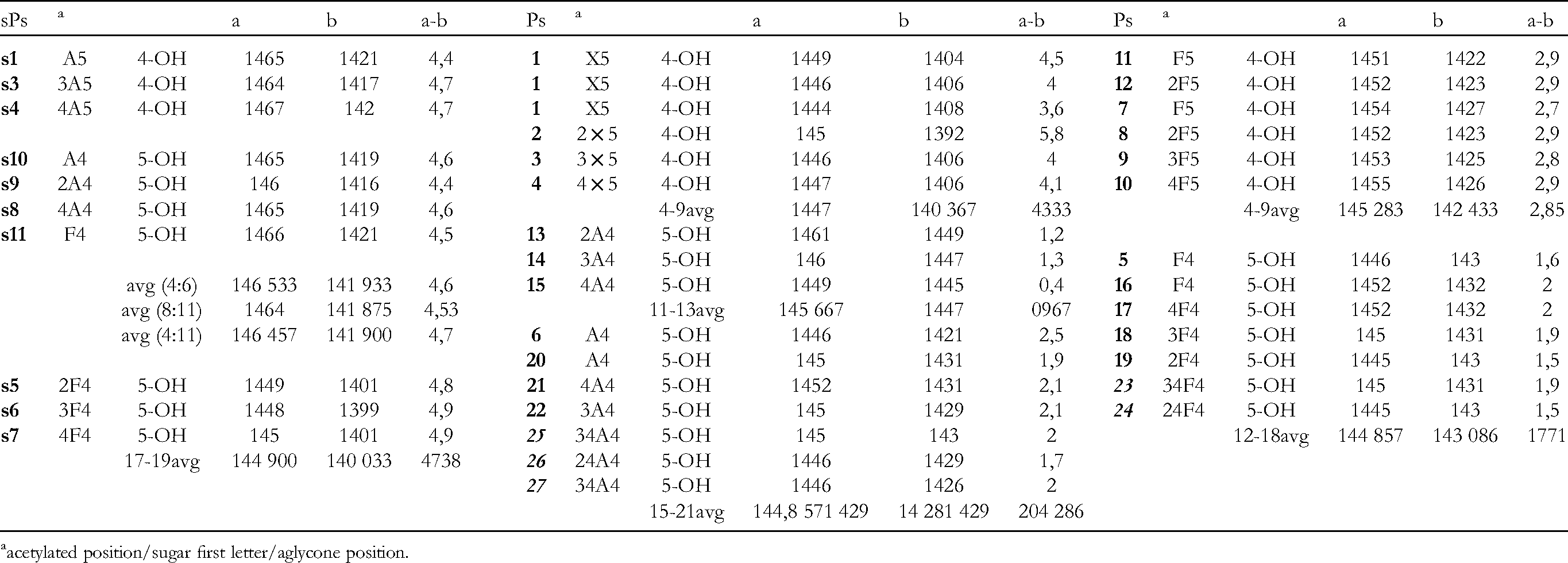
acetylated position/sugar first letter/aglycone position.
Surprisingly, the averages of Ps 5-O-xylosides are 144.7 and 140.4 (Δ 4.3), of 4-O-arabinosides 144.71 and 142.81 (Δ 1.9) ppm, 4-O-fucosides 144.86 and 143.09 (Δ 1.8), and of 5-O-fucosides 145.28 and 142.43 (diacetylated glycosides included) (Δ 2.85). Thus, large differences in glycosyloxy substitution compared with that of the vicinal free hydroxyl substituted position are observed in sPs aglycone moieties and also for Ps 5-O-xylosides. Other Ps display a smaller range, from 1.6 to 2.8 ppm. The PsM-PsO group averages are 145.7 and 144.7 (Δ = 0.97).
Further Comments on Seco-Pseudopterosins Data
Some multiplicities are not rigorous. H-14 is reported as br t (actually t*), but no J is quoted. Whereas a t* may be due to coupling with the vicinal H2-13, signal broadening may result from long-range allylic (4J) couplings with (one or both) terminal methyl groups. Actually, for the sPsH aglycone 5-methoxy and 4,5-dimethoxy derivatives they have been reported as tdd (7.1, 2.5, 1.3 Hz and 7.1, 1.3, 1.2 Hz).11,12 A t* (J = 7.1 Hz) is reasonabe, but couplings with terminal methyl groups may be expected as q or qq.
In sPsE-sPsG the separation of geminal protons (Δδ) for H2-8, H2-9, H2-12 and H2-13 are quite different from those of sPsK and also the methines H-7, H-10 and H-11. These results support a certain difference in the sugar attached, but structural elucidation could not be concluded, as mentioned already.
Assignments of the HC(14) and HC(1′) Methine Doublets
The doublets are reported with close chemical shifts (occasionally overlapping), but are easily distinguished because small HC(1′) equatorial-axial couplings in fucosides (Ps, Table 5; sPs, Tables 1,6) and arabinosides (Ps, Table 8; sPs, Table 1). However, large HC(1′) axial-axial couplings in xylosides (Table 3) are close to 9.0 Hz, as found for HC(14). Exchange of assignments may be carefully excluded.
The Dimethyl Vinyl Unit (HC-14, C-15, H3C-16 and H3C-151) NMR Data
In the early reports, the assignments of the methyl groups were not disclosed (“may be reversed: same letter within a column”). Assignments in the tables were revised according to the results of the stereochemical analysis of leubethanol, 34 showing the H-14 signal enhancement on H3-16 irradiation (as a figure, although not quantified, as was for β,β-dimethylacrylic acid). 37 Thus, locants assignment were fixed in the HMQC: H3C-151 (δ 1.582/17.49) and H3C-16 (δ 1.683/25.59).
Furthermore, H-14 was reported at δ 4.97 ppm when the 2-methyl-1-propenyl side chain has an α-orientation” (β H-13), while with a β-oriented side chain exhibits a downfield shift to δ 5.11 ppm. 5
Concentration and Temperature Effects
1H NMR and 13C NMR spectra may vary with concentration and temperature. 36 1H NMR spectra of the peracetylated derivatives of PsP-S and PsT-V were recorded at different temperatures (20, 40, 60°). 10 When recorded at 20 °C, the spectrum showed two sets (ca. 45:55 ratio) of signals for some peaks, which might correspond to two conformers of the tetraacetate, or broad signals (the interchange of the two conformers at this temperature was not rapid enough for sharp peaks), pointing out a “higher energy barrier for the rotation of the fucose derivative, compared to that of the arabinose derivative” caused by acetylation of the C-5 hydroxyl, as well as the hydroxyl groups of the sugar moiety.
Concluding Remarks
The improved consistency of reported chemical shifts and coupling constants revision (through the proposed reassignments) may provide some answers, but some questions remain to be properly addressed, while pointing out presumed typing errors or misassigned structures. Hopefully, data revised and compiled may be useful for further studies, axial and equatorial bonds will show standard orientations in sugar chair cyclohexane drawings, NMR multiplicity will be treated rigorously, and the phytane naming/numbering proposal may proof useful in structure relationships.
Footnotes
Acknowledgments
The sending of requested copies of papers is gratefully acknowledged to Professors Athar Ata, Yoshinori Fujimoto and Dan Little, and useful answers and comments to Professor Carmenza Duque and Hebelin Correa.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
