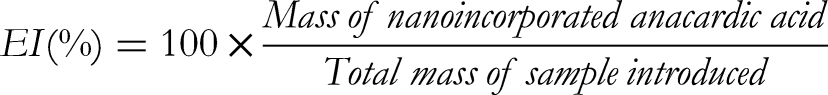Abstract
Anacardic acid (AA) is a natural active ingredient that accounts for 65% of the liquid extract from the shell of the cashew nut. Due to the stronger cytotoxic activity of hydrogenated AA (HAA) against NTERA-2 cancer stem cells (CSCs) than AA itself, HAA was co-conjugated with CD133 monoclonal antibody (mAb^CD133) into nanoliposomal particles (AMC). This nanoliposomal complex is expected to improved HAA activities against CSCs based on the targeting capacity of mAb^CD133 toward CD133, a typical CSCs’ surface marker. AMC was manufactured with a mean size of 100.9 nm, a zeta potential of −40.7 mV, and a PDI of 0.283. We report a 100% encapsulation efficiency of HAA into liposomes and a 90.7% conjugation efficiency with mAb^CD133. The penetration of AMC into NTERA-2 CSCs after 2 h was 83.7%. The AMC complex inhibited NTERA-2 growth with an IC50 (inhibition concentration at 50%) value of 75.83 ± 6.70 µM, showing the targeting ability and lower toxicity (IC50 > 100 µM) on healthy cells. The AMC nanoparticles also demonstrated significant potential apoptotic induction by activating caspase 3 activity by up to 2.57 and 2.06 folds compared to that of the negative control at 20 and 4 µM, respectively. This induction was significant improvement in comparison with that of unconjugated HAA (P < .05). AMC presented a clear effect on the solid structure of NTERA-2 spheroids and significantly suppressed the proliferation of CSCs in the 3D tumorspheres with an IC50 = 64.25 ± 3.15 µM, compared to the free form with an IC50 = 82.22 ± 0.65 µM (P < .05). Therefore, this nanoliposomal complex exhibits promising capacities as an effective material against NTERA-2 CSCs.
Keywords
Introduction
Despite numerous efforts, cancer remains a life-threatening disease that is a burden for society and the healthcare system. According to the international cancer organizations, in 2020, 19.3 million new cancer cases were diagnosed, resulting in 9.96 million deaths; compared to 2018, the incidence had increased by 2 million cases and 360,000 deaths. 1 Cancer is a chronic disease associated with exposure to environmental risks (smoking, radiation exposure, pollution), prolonged unhealthy lifestyle (sedentary habits, mental distress, unbalanced diets, and nutritional deficit diets), hereditary syndromes, chronic inflammations, diabetes, obesity, viral infections, and many other factors. 2 The first-line cancer regimens include surgical removal, radiation therapy, and chemotherapy. However, systemic therapeutics are gradually being substituted by new remedial options due to clinical failures in cancer treatment and the limited drug concentration delivered to the tumor site. Also, active pharmaceutical ingredients currently used in chemotherapy have high cytotoxicity against both cancer and normal cells. For that reason, the liposomal system of loading anti-cancer drugs provides safer and more effective delivery platforms for targeting antineoplastics to specific locations, as well as helping to reduce the cytotoxic side effects of anticancer drugs on normal cells. The same may be feasible for anacardic acid (AA), a natural active ingredient that accounts for about 65% of the liquid extract from the shell of cashew nuts. Cashew is the main crop in the Midland Highlands in Vietnam. AA was described as a cytotoxic agent, 2 possessing anti-tumor and antioxidant 3 abilities against prostate cancer and breast cancer. 4 However, AA is a mixture composed of 4 different compounds with hydrophobic unsaturated chains that block cell absorption. Therefore, to be effective, high doses need to be used, but may cause acute side effects. Also, the hydrogenated form of AA (HAA) could be an alternative to improve the effectiveness.
Nanotechnology can create many new materials, 5 and is one of the methods to overcome the limitations of poorly absorbed biological substances, such as AA. In recent decades, nanoliposomes have been used as an anti-tumor drug carrier 6 and offer many advantages over other drug delivery systems. Therefore, to improve the bioavailability of AA, nanotechnology is a viable and effective solution. For that reason, AA isolated from cashew nut shells was hydrogenated to form HAA before undergoing conjugation into a nanoliposomal complex. The nanoliposomes obtained were expected to be small enough for easy absorption, helping to reduce the dosage, lessen toxicity, and improve the bioavailability of AA. Several studies have successfully encapsulated AA within liposomes and encapsulated AA in combination with other compounds to exert an inhibitory effect on cancer cells. Filipczak et al. 7 reported that liposomal formulation of mitoxantrone, ammonium ascorbate, and AA induced high levels of reactive oxygen species in skin cancer cells—with no toxic propensity toward normal cells. Legut et al. 8 entrapped a combination of AA, vitamin C, and mitoxantrone in liposomes and showed that the combination exhibited cytotoxicity to cancer cells and protection to normal cells. However, cancer treatment remains challenging, primarily due to cancer stem cells (CSCs). CSCs directly participate in drug resistance, metastasis, cancer recurrence, and reduce the effectiveness of cancer treatment. 9 Further, targeting CSCs can lead to tumor regression. 10 Therefore, CSCs are the target of the search for more effective cancer prevention and therapies. The properties of CSCs are expressed by many cellular surface markers, including CD133, a pentaspan transmembrane glycoprotein. CD133 is proposed to be a CSC marker present on many tumor cell types and a specific surface antigen for the isolation and detection of CSCs. 11 In this study, instead of combining hydrolyzed AA with other active ingredients, such as ammonium ascorbate or mitoxantrone, to help increase tumor suppression effectiveness, as in previous studies, we used a monoclonal antibody (mAb) as an indicator to bind specifically to the CSC marker 12 and conjugate with the HAA-loaded liposome, generating a targeting nanocomplex that inhibits CSCs.
Materials and Methods
Materials
Cancer stem cell line (NTERA-2, human Caucasian pluripotent embryonic carcinoma), human continuous health cell line (HEK-293A), and human lung cancer cell line (SK-LU-1) were kind gifts from Prof. Chi-Ying Huang, National Yang Ming Chiao Tung University, Taiwan; and from Dr P. Wongtrakoongate, Mahidol University, Thailand.
DMEM medium supplemented with 10% fetal bovine serum, mouse anti-human CD133 mAb conjugated with FITC (CD133-FITC), and mouse anti-human CD133 mAb were purchased from Miltenyi (Germany), mouse-IgG-ELISA kit from Abcam (UK), and other chemicals from Sigma-Aldrich (St. Louis, MO, USA).
AA was extracted from cashew nut shell and purified to more than 99% purity. HAA was obtained by hydrogenation of AA. The chemical structures of AA and HAA were identified from ESIMS and NMR spectra 13 (Refer to Supplemental Material, Figures S1-S8, Tables S1 and S2).
Fabrication of mAb^CD133 Conjugated HAA Nanoliposomes (AMC)
Producing CD133-Conjugated HAA Nanoliposomes
After hydrogenation and purification, HAA was used to produce AMC. Incorporation of HAA to nanoliposome subunits was predicated on the modified method of Bangham et al. (1965). 14 Accordingly, a mixture of miphosphatidylcholine (PS), cholesterol, and DSPE-PEG2000-maleimide were dissolved in dichloromethane, and supplemented with HAA in a molar ratio of 9:1:0.1:2. Then the solvent was vacuum-evaporated to create a thin-film lipid layer. The mixture was then thoroughly hydrated, for 10 min by a PBS buffer that was previously supplemented with mAb^CD133 (0.5 µg/mL) in a suitable volume. Sonication was applied to the solution at 2 rpm for 20 s with a resting time of 10 s and the process was repeated 10 times. After that, the solution was centrifuged at 12,000 rpm for 90 min to obtain a nanoliposome precipitate. The supernatant was collected to assess the incorporation efficiency. The liposomes obtained varied in size, and to acquire harmonized nanoliposomes, large particles were filtered from the suspension through a 13 mm PVDF membrane with a pore size of 0.22 µm. The physical characteristics of the obtained nanocomplex were evaluated using a Zetasizer Nano ZS (Malvern Instruments, Malvern, UK).
Determination of Encapsulation Efficiency (EE)
After AMC fabrication, unconjugated HAA was separated from the HAA-encapsulated liposomes in Sephadex G-50 Fine minicolumns (5.5 × 70 mm) that were balanced previously with 150 mM NaCl solution. Fifty microliter of liposome samples were put on a column and free AA was isolated from the liposome containing HAA. The supernatant, after centrifugation, was used to quantify the residual HAA content by HPLC, while the concentration of unconjugated anti-CD133 monoclonal antibodies was determined using the Mouse-IgG-ELISA kit provided by Abcam (UK) by strictly following the instruction of the kit manufacturer.
The incorporation efficiency of HAA was calculated using the following formula:
Evaluation of Biological Activities of AMC Against NTERA-2 Cells
Evaluation of the Penetration Capability of AMC to the Cell
NTERA-2 cells were pre-seeded in 6-well culture plates for 24 h in an incubator maintained at 37 °C, with 5% CO2. Next, samples were added to the wells and incubated for 3 h. Then, samples were removed from the medium. The cells were washed with PBS and removed from the bottom of the well with 1 mM EDTA. Novocyte flow cytometry and relevant software (ACEA Bioscience Inc.) were used to determine AMC penetration.
MTT Anti-Proliferation Assay
The survival rate of cells under the treatment of the samples was assessed by using the (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, as previously described. 15
Cell Cycle Analysis
To analyze the cell cycle, treated cells were collected, washed with cold PBS 1X, and fixed with 70% ethanol at 4 °C for 2 h. Then, they were rinsed twice with cold PBS 1X, incubated with RNase A for 15 min (1 mg/mL) in a 37 °C water bath, and stained with propidium iodide (1 mg/mL) for 1 h. Cell cycle control was determined by examining 10,000 cells/sample using a Novocyte flow cytometer and NovoExpress software (ACEA Bioscience Inc.).
Caspase 3 Inducible Assay
The caspase 3 Colorimetric Assay Kit (Biovision Inc.) was employed to measure the AMC apoptotic inductive activities, as previously described. 16
Effects of AMC on the Growth of 3D Tumor Spheroids
The hanging drop method was used to create 3D tumor spheroids, as previously detailed. 17 After 3 days of culture, the cancer cells aggregate, with a formed single spherical cell mass. The viability of these 3D tumor spheroids was evaluated using the MTT assay and cell mass imaging through the Zeiss Vert A1 inverted microscope.
Statistical Analysis
Results were analyzed using Graphpad Prism7.0 and displayed as mean ± standard deviation (SD). The hypothesis of the mean of the 2 samples was examined by unpaired Student's t-test. The difference was considered to be statistically significant when P ≤ .05.
Results and Discussion
Fabrication of AMC (AA-mAb^CD133 Conjugated Nanoliposomes)
In primary screening for cytotoxic activity against NTERA-2 cells, AA presented an IC50 (concentration to inhibit 50% cell growth) value of 68.80 ± 0.95 µg/mL, while the value for HAA was 18.02 ± 0.60 µg/mL. Therefore, it is obvious that the hydrogenated form exhibits a much stronger effect in inhibition of cell growth and was selected to conjugate into nanoliposomal particles. As detailed in the experiment, the AMC nanoliposomes were produced. The characteristics of HAA-mAb^CD133 conjugated nanoliposomes, including mean size, particle size distribution, and zeta potential are presented in Table 1.
Effect of the Ratio of Active Ingredients on Liposome Formation.

Additionally, mean size, zeta potential, and PDI index of AMC were quantified by DLS measurements using the Zetasizer Nano-Z instrument. The results showed that the AMC were approximately 100.9 nm in size and had a PDI index of about 0.28, indicating that the liposome complex was relatively uniform. Furthermore, the zeta potential of AMC reached −40.7 ± 3.2 mV, while that of liposome-HAA was −35.5 ± 1.9 mV. Those zeta potentials were preferably higher than that of another reported liposome-AA, which was − 4.5 ± 0.47 mV. 8 The zeta potential is a particle surface charge that acts as an energy barrier that controls the stability of dispersion and counteracts the proximity and aggregation of particles, where higher values indicate lower aggregation. 18 Therefore, the high zeta potential of this liposome complex may prevent the liposome particles from aggregating and aid in maintaining the stability of the AMC.
The PDI reflects the formation of narrowly dispersed nanostructures without any agglomeration in water, where PDI ≤ 0.3 represents uniform density. 19 According to Do et al., 7 TMC (the nanocomplex TbPO4·H2O@silica-NH2 + mAb^CD133 + mAb^PD-L1) was a rare earth nanomaterial that carried mAb^CD133, with a size of 50 to 80 nm, showing good efficiency in detecting and inhibiting colon cancer cells. 20 Additionally, PMCs (piperine and anti-CD133 monoclonal antibodies containing nanoliposomal complexes) were a nano-piperine material that conjugated mAb^CD133 with a size of 100 to 200 nm, which targeted CSCs effectively. 17 As such, the properties of AMC are consistent with those of a biomedical nanoparticle. However, as shown in Figure 1, SEM revealed AMC as small unilamellar vesicles, rod-shaped and slightly different from PMC, which were spherical in shape. 16

Scanning electron microscopy (SEM) images of HAA-mAb^CD133 conjugated nanoliposomes (HITACHI 8100, Japan).
The encapsulation of HAA in liposomes showed high efficiency (100%). This result is consistent with the study by Legut where the MIT liposomes (formulation of mitoxantrone, AA, and vitamin C) achieved 95% efficiency. 8 The amount of mAb^CD133 was evaluated in the supernatant after centrifugation using the Mouse-IgG-ELISA kit. The results showed that the conjugation efficiency of mAb^CD133 with HAA-encapsulated liposomes reached 90.7 ± 2.7%.
Evaluation of Infiltration Ability of AMC
After HAA and mAb^CD133 were combined into liposomes, the penetration of the active substance into cells was assessed. AMC showed the highest penetration of 76.61% after 1 h of incubation, while at the same time, only 67.29% and 1.78% mAb^CD133 nanoliposomes and blank nanoliposomes penetrated the cell, respectively. Similarly, AMC exhibited the best penetration rate of 83.71% after 2 h of incubation, while at this time, mAb^CD133 nanoliposomes at 2 h were 73.52%, and blank nanoliposomes were 2.69%. As such, when the incubation time was increased, the penetration level of AMC increased substantially and was higher than that of the other samples.
Determination of the Effect of Samples on Cell Growth
The effects of the studied sample on the NTERA-2, SK-LU-1 and HEK-293A cell lines are shown in Table 2.
The Ability to Inhibit Cell Growth.

Nanoliposome-HAA-mAb^CD133 exhibited strong targeting capability against CSCs (NTERA-2 cell line) with an IC50 of 75.83 ± 6.70 µM as compared to cancer cells (SK-LU-1 cell line) which did not present the CD133 marker, with an IC50 of 94.51 ± 3.58 µM, and showed little or much less toxicity to healthy cells (HEK-293A cell line) at the same concentration (IC50 > 100 µM). AMC also presented improved anticancer activity compared to the unconjugated form with lower IC50 values for both cancer cells and CSCs, and with remarkedly reduced cytotoxicity to healthy cells (HEK-293A cells).
It was reported that AA has potential anti-cancer activity that could induce caspase-independent apoptosis 21 inhibition of the NF-κB pathway 22 and impede histone acetyltransferase (HAT) activities that are involved in the regulation of gene expression. One of the HAT family members, Tip60, is modulated in cellular responses to genotoxic events. 23 Inhibition of this enzyme diminishes the DNA repair system and AA can sensitize cancer cells to genotoxicity by Tip60 inhibition. 24 AA also represses the CSCs phenotypes by decreasing CD44+/hi/ALDH+/hi markers in IOWA-1T cells. 25 In this case, for the first time, HAA-containing nanoliposomes were fabricated and studied for anti-CSCs activities on NTERA-2 cells. The obtained nanocomplex presented potential targeting effects against NTERA-2 CSCs with an IC50 value of 75.83 ± 6.70 µM, while the value for non-CD133 marker cells (SK-LU-1 cell line) was 94.51 ± 3.58 µM. The stronger inhibition activities of AMCs of which mAb^CD133 was conjugated in the complex could attribute to this action of HAA. Still, it could also be a synergistic effect between HAA and mAb^CD133. However, this needs to be clarified in future studies.
Cell Cycle Arrest Effect of AMC
The effects of the samples studied at different concentrations on the NTERA-2 cell cycle are displayed in Figure 2.

NTERA-2 cell cycle control under the influence of samples at different concentrations. (a) Negative control; (b), (c), and (d) represent data for treatment with AMC at 100, 20, and 4 µM, respectively; (e), (f), and (g) represent data for treatment with HAA at 100, 20, and 4 µM, respectively.
The above results demonstrated that AMCs noticeably affected the NTERA-2 cell cycles and were different from HAA in free form. The nanoliposomal complex arrested the NTERA-2 cell cycle in the sub-G1 and G2/M phases, and reduced cell number in the G1 phase in a concentration-dependent manner. While HAA noticeably acted in the sub-G1 phase, it had no effect on other phases of the NTERA-2 cell cycle. Through the targeting capability of mAb^CD133, AMCs seemed to remarkably modify HAA anti-CSCs capacity, leading to cell cycle arrest in the G2/M phase (Figure 2). As reported, increased mAb^133 activity to a glycosylated form of CD133 in human embryonic stem cells is correlated with increased DNA content and mutually leading to the least reactive cells in the G0/G1 portion of the cell cycle. 26 Thus, in this study, AMCs which have mAb^CD133 and HAA in complexes might be the reason for their strong effect on the sub-G1 and G2/M phases of the NTERA-2 cell cycles.
Effect of AMC on the Induction of Caspase 3
To evaluate the caspase 3 induction of the examined materials, their activities were determined and presented as fold change in comparison with that of the negative control (Table 3).
Caspase 3 Induction Activity of the Samples.
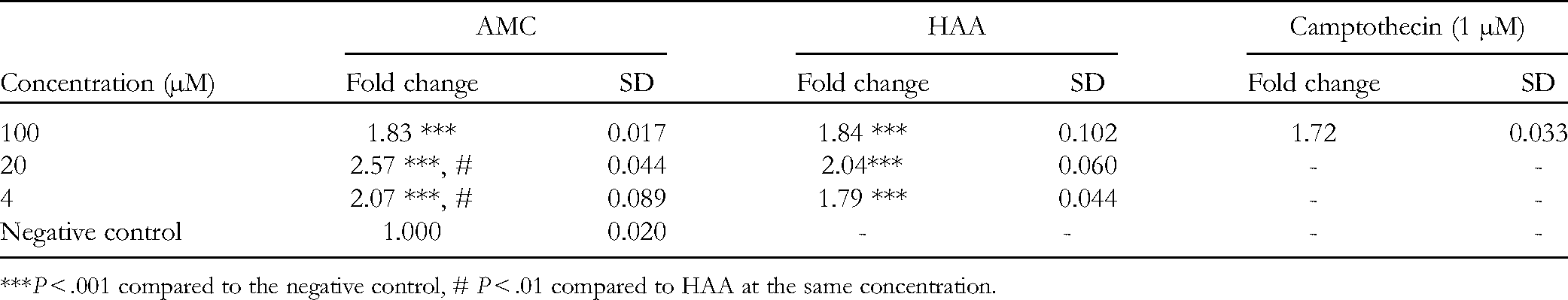
***P < .001 compared to the negative control, # P < .01 compared to HAA at the same concentration.
The above results show that AMC clearly induced caspase 3 activity at all 3 concentrations (P < .001), which differed significantly compared to the free HAA at 20 and 4 µM. The obtained results were similar to those in other studies which reported AA apoptotic inducible activities. According to Seong et al., 21 the 3 typical executioners of the intrinsic pathway, caspase 3, cleaved caspase 7, and cytochrome c, were gradually increased under the treatment of AA in a time-dependent manner. From our study, HAA was, for the first time, reported for its caspase 3 apoptotic induction on NTERA-2 embryonic carcinomas. Furthermore, HAA in AMC nanoliposomal complex presented significant improvement on caspase 3 inducible activities compared to that of the unconjugated form (P < .01). Due to the fact that caspase 3 moderates either mitochondria-mediated or extrinsic apoptotic inductive pathways, increased activity of this enzyme definitely presents the potential apoptotic inducible capacity in caspase-dependent manner in CSCs of AMC.
Inhibitory Effect of AMC on the Growth of Tumor Spheroids In Vitro
The MTT results showed that AMC had the ability to inhibit CSCs, and had a lower cytotoxic effect on healthy cells (Table 2). However, in vitro studies have limitations. During cell line establishment, it was easy for cancer cells to access nutrients and oxygen, creating populations expressing homogenous genotypes and phenotypes. 27 Furthermore, monolayer cultured cells lack the complexity of the tumor structure. 28 Therefore, to assess further the possible anti-tumor potency of AMC, we employed a 3D tumor spheroidal culture model. The effectiveness of AMC on the growth of NTERA-2 cells in 3D tumorspheres is shown in Figure 3 and Table 4.
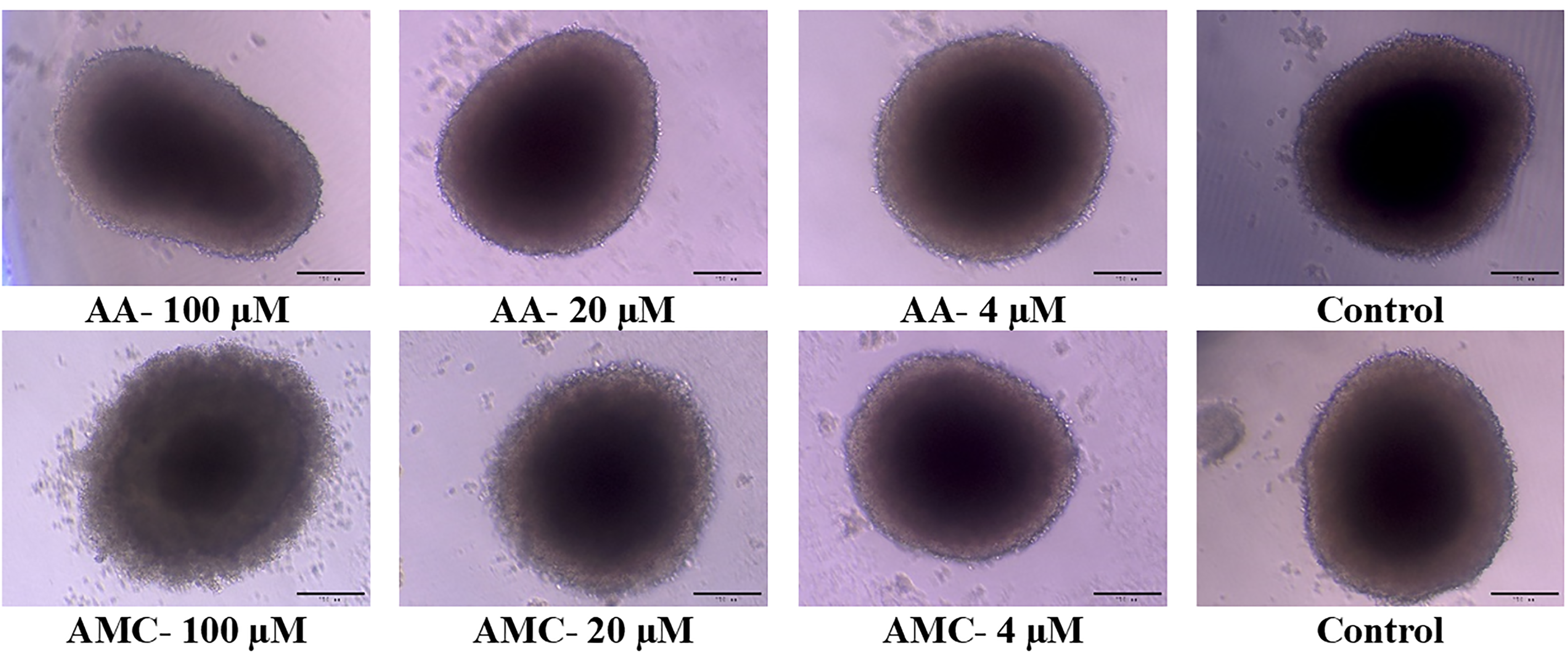
Effect of the samples studied on the 3D tumor spheroid of NTERA-2 cells observed under a Zeiss Vert A1 inverted microscope; scale bar 200 µm.
Inhibition of NTERA-2 Spheroid Development at Different Concentrations of Samples.

Under AMC exposure at 100 µM, the cell mass observed showed more significant shrinkage and disintegration relative to the effect of free HAA. Compared to the control, this result demonstrated the destruction of the 3D spheroids by AMC. By examining cell viability by MTT assay, AMC presented the stronger anti-proliferative activities with an IC50 = 64.25 ± 3.15 µM, compared to that of the unconjugated form (IC50 = 82.22 ± 3.15 µM). Thus, AMC significantly improved the inhibitory activity against the NTERA-2 cells in 3D spheroids in comparison to that of free HAA. Here, we proved the potential anti-tumor inhibitory effect of the AMC nanocomplex in a 3D tumor spheroidal platform which reflected the typical characteristics of cancer stemness of NTERA-2 cells. The tumor spheroid that exhibited characteristics in the early stages of tumor growth in vivo was likely to be in the intermediate state between monolayer cultures and spontaneously growing tumors. 29 Therefore, these results, again, demonstrated the promising anticancer inhibitory effect of AMC on CSCs.
As such, the outstanding question is whether the increased effectiveness of AMC is due to either the synergistic effect of mAb^CD133 and HAA or the targeting of mAb^CD133 enhancing the inhibitory effect of AA in AMC compared to free HAA. Zhao et al. 30 elucidated that activated T cells with a bispecific antibody, which was an asymmetric BiAb consisting of a monomer of chimeric AC133 (mouse anti-human CD133 mAb) and a single chain of humanized OKT3, could generate anti-tumor effects in vitro. Do et al. 20 reported that the targeting capacity of mAb^CD133 in conjunction with mAb^PD-L1 increased cancer tumor inhibition. With the results described earlier, changing to the nanoliposomal type and replacing the inhibitory active ingredient have much improved the anti-tumor ability of the material. The remaining problem of the synergistic effect of active ingredients in AMC has revealed new research directions for the future.
Conclusions
Due to hydrogenated modification, AA was saturated, crystallized, and presented a stronger cytotoxic effect against NTERA-2 CSCs. Thus, HAA was co-conjugated into nanoliposomes (AMC nanocomplex) together with CD133 mAb. The nanocomplex was manufactured with a mean size of 100.9 nm, a Zeta potential of −40.7 mV, and a PDI of 0.283. The EE of AA in liposomes reached 100%, and the conjugation efficiency with mAb^CD133 was 90.7%. The AMC nanocomplex presented targeting ability against NTERA-2 cells with an IC50 of 75.83 ± 6.70 µM, noticeably arresting the cells in the subG1 and G2/M phases, and a strong caspase 3 inducible activity. The complex also suppressed the proliferation of NTERA-2 cells in tumor spheroids by 72.02 ± 4.59% and with an IC50 = 64.25 ± 3.15 µM, which were significantly improved compared to those of free HAA.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221105696 - Supplemental material for A New Liposomal Formulation of Hydrogenated Anacardic Acid to Improve Activities Against Cancer Stem Cells
Supplemental material, sj-docx-1-npx-10.1177_1934578X221105696 for A New Liposomal Formulation of Hydrogenated Anacardic Acid to Improve Activities Against Cancer Stem Cells by Le Tri Vien, Nguyen Thi Nga, Phung Thi Kim Hue, Tran Hoang Bao Kha, Nguyen Hong Hoang, Pham Thi Hue, Pham Ngoc Thien, Chi-Ying F Huang, Phan Van Kiem and Do Thi Thao in Natural Product Communications
Footnotes
Acknowledgments
The authors are grateful to Prof. Dr Tran Van Loc, Institute of Chemistry, Vietnam Academy of Science and Technology for the extraction of AA from cashew nut shell and preparation of its hydrogenated production, HAA.
Author Contribution
Research idea: DT Thao and LT Vien. Biological experiment: LT Vien, NT Nga, PTK Hue, THB Kha, NH Hong, PT Hue, Thi Nga Nguyen, Thi Hue Phung, Hoang Bao Kha Tran, Hoang Hong Nguyen, and Thi Hue Pham. Methodology: CYF Huang. Writing—original draft: LT Vien, NT Nga. Structure elucidation and writing: PV Kiem. Review & editing: D T Thao and PV Kiem.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Institute of Biotechnology, Vietnam Academy of Science and Technology (grant number CSCL08.01/22-22).
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.