Abstract
The absolute configuration of diaporthe B, a pimarane diterpene isolated from the mangrove derived endophytic fungus
Introduction
Prostate cancer is one of the common malignant tumors of the male urinary system. It is the second leading cause of cancer-related death in men in Western countries. 1 Risk factors, including advancing age, race, geographical distribution, diet, and family history, are contributing to the incidence of this disease.2,3 The common treatment for prostate cancer is hormone deprivation. However, prostate cancer cells lose their hormone dependence and therapeutic responsiveness during disease progression.4,5 The resulting androgen-independent prostate cancer (also referred to as castration-resistant or hormone-refractory) progresses and metastasizes, with poor prognosis.6,7 Chemotherapy is usually applied for the treatment of the androgen-independent prostate cancer. 8
Docetaxel (Figure 1) belongs to the chemotherapy drug class taxane, and is currently the first-line chemotherapy drug for androgen-independent prostate cancer. Docetaxel treatment combined with either prednisone or estramustine increased survival, reduced pain, and serum prostate specific antigen (PSA) level, as well as increased the quality of life.9,10 However, prostate cancer patients receiving docetaxel usually develop chemo-resistance. Toxic side effects and the development of chemo-resistance limit the clinical success of docetaxel therapy. The cancer stem cell (CSC) model provides insights into the mechanisms behind tumor initiation, progression, and chemo-resistance.11,12 It has been reported that docetaxel-resistant prostate cancer cells exhibited CSC features.13,14 Therefore, CSCs could be used as critical targets for a more effective treatment of prostate cancer.

Structures of docetaxel and diaporthe B.
Mangrove derived fungi are an important source of secondary metabolites useful for drug discovery purposes. Although mangrove derived fungi are less explored in comparison to their terrestrial counterparts, a number of useful hits have been obtained from the drug discovery perspective adding to their importance in natural product discovery.15–17 Diaporthe B (Figure 1) is a pimarane diterpene isolated from the mangrove endophytic fungus
This study aimed to explore the inhibitory effects and mechanisms of diaporthe B on prostate cancer cells resistant to docetaxel. In our study, we determined sphere formation and expression levels of CSC-related markers in prostate cancer PC-3 cells that were resistant to docetaxel. The effects of diaporthe B on the stemness of docetaxel-resistant PC-3 cells were also examined, as was the effect of diaporthe B in combination with docetaxel on growth and apoptosis in docetaxel-resistant PC-3 cells. The results of the present study demonstrated that diaporthe B in combination with docetaxel synergistically inhibited the growth and induced apoptosis in docetaxel-resistant PC-3 cells. The effects of diaporthe B on docetaxel-resistant PC-3 cells were associated with inhibition of the stemness of the cells.
Results and Discussion
Isolation of Diaporthe B
Diaporthe B (Figure 1) was isolated as colorless black crystals from the mangrove derived fungus
Docetaxel-Resistant PC-3 Cells Show Features of CSC
In initial experiments, the effects of docetaxel on cell viability and apoptosis were determined in parental PC-3 and docetaxel-resistant PC-3 cells. The docetaxel-resistant cells were generated by continuous exposure of the PC-3 cells to increasing concentrations of docetaxel as described previously 21 . As shown in Figure 2A, treatment of PC-3 cells with docetaxel resulted in a dose-dependent decrease in the number of viable cells, while docetaxel only had a small effect on cell viability in the resistant cells. Docetaxel also dose-dependently induced apoptosis in PC-3 cells, but only a small increase in apoptosis was seen in the resistant PC-3 cells treated with docetaxel (Figure 2B). Docetaxel is an antimitotic agent that is able to restrain microtubule disassembly, and docetaxel-based chemotherapy is the standard care for later stage metastatic prostate cancer22,23. However, prostate cancer cells can develop resistance to the drug, which is the main limitation of its use24,25. The molecular mechanisms of resistance are not fully understood. The expansion of CSCs after chemotherapy has been identified as an important factor responsible for acquired therapeutic drug resistance.13,14,26

PC-3 and docetaxel-resistant PC-3 cells. (A) PC-3 cells and the docetaxel-resistant PC-3 (PC-3/DTX) cells were treated with various concentrations of docetaxel (DTX) for 72 h. The number of viable cells was determined by the trypan blue exclusion assay. (B) PC-3 cells and PC-3/DTX cells were treated with various concentrations of docetaxel for 72 h, and apoptosis was determined by propidium iodide staining. (C) Morphology of PC-3 cells cultured in medium containing FBS. (D) PC-3 spheres and (E) PC-3/DTX spheres cultured in ultralow attachment plate with serum-free medium for 12 days. (F) The number of PC-3 and PC-3/DTX spheres formed after 12 days in serum-free medium. (G) PC-3 and PC-3/DTX cells were seeded in collagen-coated dishes for 5 min. Unattached cells were removed and the attached cells were collected and counted. (H) The levels of CD44, CD133, and ALDH1A1 in PC-3 and PC-3/DTX cells were determined by Western blot analysis. Superscript
To investigate whether CSCs are involved in the resistance of PC-3 cells to docetaxel, we determined tumor sphere formation, which is one of the characteristics of CSC 27 in PC-3 and docetaxel-resistant PC-3 cells. In contrast to a monolayer of cells grown in adherent cultures (Figure 2C), in serum containing medium, PC-3 (Figure 2D) and the docetaxel-resistant PC-3 (Figure 2E) cells formed nonadherent spheroid cell clusters when they were cultured with the serum-free medium in ultralow attachment plates. The size of spheres after 12-day cultures was around 50-200 μm. The morphology of the spheres was similar to those derived from other tumors such as lung and renal cancers28,29. As shown in Figure 2F, the resistant cells had much higher sphere-forming efficiency than the parental PC-3 cells. Another feature of prostate CSCs is their ability to quickly adhere to collagen-coated culture plates30,31. Therefore, the assay for quick cell adherence was performed in the cells. As shown in Figure 2G, a strong increase in adherent cells was observed in the docetaxel-resistant cells. In additional experiments, the levels of CSC-related markers CD44, CD133, and ALDH1A1 (aldehyde dehydrogenase) were PC-3 determined in PC-3 and docetaxel-resistant PC-3 cells by Western blotting using corresponding antibodies. As shown in Figure 2H, the abundance of CD44 was higher in the resistant cells than that in the parental PC-3 cells. CD44 is a transmembrane glycoprotein which possesses a particular cell adhesion function and consequently assists the matrix adhesion and migration of CSCs 32 . Prostate cancer patients with high expression of either CD44 or β-catenin are associated with more aggressive clinicopathological features and poor prognosis 33 . CD44 contributes to the activation of stem cell regulatory genes and can be a target of these genes 34 . Elevation of CD44 has been observed in some drug-resistant cancer cell lines35,36. Our present study showed that the levels of CD44 increased in the docetaxel-resistant cells implying that these cells have a higher CSC population. This result is in line with a recent study showing that docetaxel-resistant prostate cancer cells contained a higher CD44+ population 14 .
The Western blot analysis results also showed that the level of CD133 was higher in the docetaxel-resistant cells than in the parental PC-3 cells (Figure 2E). CD133 (also called Prominin-1 or AC133) was the first member verified in the prominin family of pentaspan transmembrane glycoproteins, and is now widely used as a CSC-related marker in various types of cancer, including prostate cancer37,38. Overexpression of CD133 in prostate cancer cells promotes bone marrow metastasis 39 . The CD133+ cells isolated from human prostate cancer cell lines are resistant to radiation and docetaxel 40 . The result of our present study suggests that the docetaxel-resistant cells contained a higher CSC population.
As shown in Figure 2E, the level of ALDH1A1 was higher in the resistant cells than that in parental PC-3 cells. ALDH1A1 is a cytosolic enzyme belonging to the ALDH family of enzymes that catalyze the intracellular oxidation of different aldehydes 41 . It plays an important role in retinoic acid signaling and is used as a marker to identify various types of CSCs41–44. A high expression of ALDH1A1 in prostate cancer has been shown to be positively correlated with tumor aggressiveness and inversely correlated with patient survival45,46. It has been shown that CSC related markers, as well as markers associated with tumor pathology and progression such as CD34, CD117, CD133, ALDH1A1, and MMP-13, may have prognostic value for prostate cancer patients46–48. These markers may also be promising candidates for targeted therapy of prostate cancer. Taken together, our studies on the expression levels of CD44, CD133, and ALDH1A1 indicate that docetaxel-resistant prostate cancer cells contain a higher population of prostate CSCs. Since CSCs are associated with chemotherapeutic drug resistance, and tumors with a higher population of CSCs have poor clinical outcomes, these CSC-related markers may have potential value as prognostic markers for prostate cancer patients.
Effects of Diaporthe B on Cell Growth and Apoptosis in Docetaxel-Resistant PC-3 Cells
The growth of docetaxel-resistant PC-3 cells treated with different concentrations of diaporthe B was determined. As shown in Figure 3A, diaporthe B inhibited the growth of docetaxel-resistant PC-3 cells in a time- and dose-dependent manners. Diaporthe B also dose-dependently decreased the cell viability (Figure 3B). The effect of diaporthe B on apoptosis of the resistant cells was determined using propidium iodide staining and caspase-3 activity assays. As shown in Figure 3C, treatment of the resistant cells with diaporthe B resulted in a dose-dependent increase in the number of apoptotic cells. The result of the caspase-3 activity assay was similar to that of the propidium iodide staining assay, confirming the stimulatory effect of diaporthe B on apoptosis in docetaxel-resistant cells (Figure 3D). Our results demonstrated for the first time that diaporthe B effectively inhibits the growth and induces apoptosis in docetaxel-resistant PC-3 cells. Diaporthe B may represent a promising candidate for the future development of drugs for the management of chemoresistant prostate cancer.
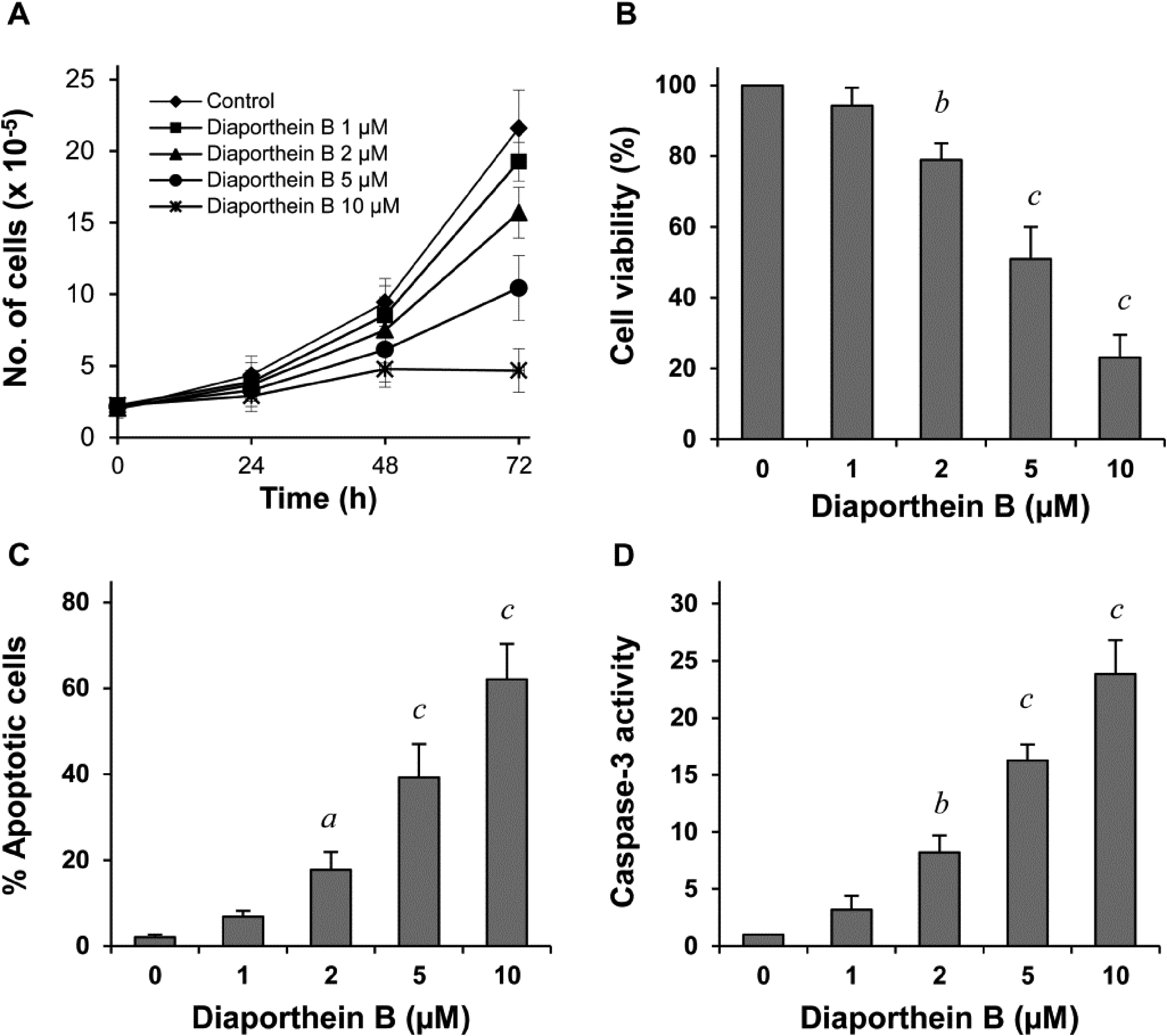
Effects of diaporthe B on growth and apoptosis in docetaxel-resistant PC-3 cells. The cells were treated with diaporthe B (1-10 µM) for 72 h. (A) The number of viable cells was determined by the trypan blue exclusion assay. (B) Cell viability was determined by the trypan blue exclusion assay and expressed as a percentage of control. (C) Apoptosis was determined by propidium iodide staining and expressed as a percentage of control. (D) Caspase-3 activity was examined by the caspase-3 assay kit and expressed as an arbitrary unit. Each value represents mean ± S.D from three separate experiments. Superscripts
Effects of Diaporthe B on the Stemness of Docetaxel-Resistant PC-3 Cells
To select a nontoxic dose of diaporthe B, we determined its toxicity on normal prostate epithelial RWPE-1 cells by MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) assay. As shown in Figure 4A, treatment of RWPE-1 cells with 1 and 2 µM diaporthe B had no significant effect on the viability of the cells. Higher concentrations of diaporthe B (5 and 10 µM) significantly decreased the viability of RWPE-1 cells (Figure 4A).

Effects of diaporthe B on normal prostate epithelial cells and docetaxel-resistant PC-3 cells. (A) Human prostate epithelial cells (RWPE-1) and docetaxel-resistant PC-3 cells (PC-3/DTX) were treated with different concentrations of diaporthe B for 72 h. The number of viable cells was expressed as percentage of control. (B) PC-3/DTX cells were cultured in an ultralow attachment plate with serum-free medium and treated with diaporthe B for 12 days. The number of spheres was counted at the end of the experiment and expressed as a percentage of control. (C) PC-3/DTX cells were treated with diaporthe B for 24 h. The cells were harvested and seeded in collagen-coated culture dishes. After 5 min, cells adhered to the dishes were counted and expressed as a percentage of control. (D) PC-3/DTX cells were treated with diaporthe B for 24 h and the levels of CD44, CD133, and ALDH1A1 were determined using Western blot analysis. Superscripts
Since diaporthe B at 2 µM significantly inhibited the docetaxel-resistant PC-3 cells, but had no toxicity towards RWPE-1 cells, this concentration was used in the subsequent experiments. The inhibitory effect of diaporthe B on sphere formation in the resistant cells was determined and the result is shown in Figure 4B. Diaporthe B (2 µM) significantly decreased the sphere-forming efficiency of the cells. As shown in Figure 4C, diaporthe B (2 µM) also significantly decreased the adherent cells on the collagen-coated surface. We further examined the levels of CSC-related markers CD44, CD133, and ALDH1A1 in the docetaxel-resistant PC-3 cells treated with diaporthe B. Western blot analysis showed that the levels of CD44, CD133, and ALDH1A1 were decreased in the resistant cells treated with diaporthe B (Figure 4D). Results of the above experiments indicated the diaporthe B inhibited the stemness of docetaxel-resistant PC-3 cells. It is possible that the inhibitory effect of diaporthe B on the docetaxel-resistant cells was mediated by the suppression of CSCs.
Effects of Diaporthe B and Docetaxel in Combination on Growth and Apoptosis in Docetaxel-Resistant PC-3 Cells
We further investigated the effects of diaporthe B in combination with docetaxel on the growth and apoptosis of docetaxel-resistant PC-3 cells. In these experiments, diaporthe B at a nontoxic dose of 2 µM was used to combine with different concentrations of docetaxel. As shown in Figure 5A, diaporthe B in combination with docetaxel had stronger effects on decreasing the number of viable cells than each agent alone. The results of our study demonstrated for the first time that diaporthe B increased the effectiveness of docetaxel on growth inhibition and apoptosis in the resistant cells. Results of our mechanistic studies described above showed that the effects of diaporthe B on docetaxel-resistant cells were associated with inhibition of CSCs including reduced sphere-formation efficiency, reduced adherence to a collagen-coated surface, and decreased levels of CSC-related markers. Further studies using suitable animal models to examine the in vivo effect of diaporthe B on docetaxel-resistant prostate cancer cells are warranted.

Effects of diaporthe B in combination with docetaxel on growth and apoptosis in docetaxel-resistant PC-3 cells. The cells were treated with docetaxel (DTX; 1-20 nM) either alone or in combination with diaporthe B (2 µM) for 72 h. The number of viable cells was determined by the trypan blue exclusion assay and apoptosis was determined by propidium iodide staining and caspase-3 assay. (A) Percentage of viable cells. (B) Percentage of apoptotic cells. (C) Caspase-3 activity.
Materials and Methods
Chemical Experimental Procedures
Diaporthe B was isolated from the mangrove derived fungus
Fungal Materials
The mangrove derived fungus
Extraction and Isolation of Diaporthe B
The fermentation mixture was broken up with a spatula and extracted three times with methanol. The methanol layer was filtered and evaporated. The dry residue was partitioned with ethyl acetate to yield crude extract (28.0 g). The EtOAc extract was chromatographed on a Si gel column (Qingdao silica gel 200-300 mesh, 600 g), eluting with a step gradient of light petroleum—ethyl acetate (9:1, 3.5 L; 8:2, 4 L; 7:3, 4L; 6:4, 3L; 1:1, 7.5 L; 4:6, 6L; 3:7, 2.5 L; 2:8, 2 L; 7:3, 4L; 6:4, 3L, 1:1, 7.5 L, 4:6; v/v), and MeOH (100%, 5L) to yield eight fractions (Fr.1-Fr.12). Fr. 3 was eluted with a gradient of light petroleum/ethyl acetate from 80:20 to 20:80 to give 6 fractions (Fr.4.1-Fr.4.6); diaporthe B was obtained (64.8 mg). Its purity (>98%) was confirmed by HPLC (Supplemental Material).
Cells Culture and Reagents
Human prostate cancer cell line PC-3 and human prostate epithelial cell line (RWPE-1) were obtained from the American Type Culture Collection (ATCC), and docetaxel-resistant PC-3 cells by continuous exposure of the cells to increasing concentrations of docetaxel 21 . The cells sustained exponential growth by culturing in Roswell Park Memorial Institute Medium (RPMI) 1640 tissue culture medium supplemented with 10% fetal bovine serum (FBS) (Gibco), penicillin (100 units/mL), streptomycin (100 mg/mL), and L-glutamine (300 µg/mL). Docetaxel was purchased from Sigma-Aldrich (St. Luis). Cultured cells were placed into tissue culture dishes and grown at 37°C in a humidified atmosphere of 5% CO2. Diaporthe B and docetaxel were dissolved in dimethyl sulfoxide (DMSO), and the final concentration of DMSO in all experiments was 0.1%.
Determination of the Number of Viable Cells
The number of viable cells after each treatment was determined by using the trypan blue exclusion assay. After each experiment, a single cell suspension was made by trypsinized the cells. The trypan blue exclusion assay was performed by mixing 80 µL of cell suspension and 20 µL of 0.4% trypan blue solution for 2 min. Blue cells were marked dead and the cells that did not absorb dye were marked as alive. A hemacytometer was used to count the number of viable cells under a Nikon light microscope (Optiphot, Nikon).
Assessment of Apoptotic Cells
Apoptosis was determined by morphological assessment of cells stained with propidium iodide. After each experiment, cells were trypsinized and cytospin slides were prepared. The cells were fixed with acetone/methanol (1:1) for 10 min at room temperature, followed by 10 min with propidium iodide staining (1 µg/mL in PBS), and finally analyzed using a Nikon fluorescence microscope (Eclipse TE200, Nikon). Apoptotic cells were identified by their classical morphological features such as nuclear condensation, cell shrinkage, and formation of apoptotic bodies.
Caspase-3 Activity Assay
Caspase-3 activation was measured using an EnzoLyte 7-amido-4-methylcoumarin (AMC) Caspase-3 Assay Fluorimetric kit (AnaSpec), following the manufacturer instructions. Briefly, after drug treatment, caspase-3 substrate was added to each well. Plates were incubated for 30 mins at room temperature. Fluorescence intensity was measured in a Tecan Inifinite M200 plate reader (Tecan US Inc.).
Sphere Formation Assay
The sphere-formation assay was performed as described previously 49 . PC-3 and the docetaxel-resistant PC-3 cells (1000 cells/mL) were seeded in a 24-well ultralow attachment surface culture plate (Corning Co) and cultured in Dulbecco's Modified Eagle Medium (DMEM)/F12 serum-free medium supplemented with basic fibroblast growth factor, epidermal growth factor, insulin, selenium, transferrin, and bovine serum albumin for 12 days. The number of spheres was counted under a Nikon light microscope (Optiphot, Nikon).
Collagen Adherence Assay
The collagen assay was performed as described previously 30 . In brief, cell culture dishes were coated with 70 mg/mL of collagen I (Sigma-Aldrich) for 1 h at room temperature. The dishes were then washed with PBS, blocked with 0.3% bovine serum albumin (BSA) for 30 min and washed with phosphate buffered saline (PBS) again. The cells were plated on collagen I coated dishes for 5 min. The cells that did not adhere were removed and the adherent cells were counted.
Western Blotting
After treatment, the cell lysates were prepared. Proteins were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to nitrocellulose membranes. After blocking nonspecific binding sites with blocking buffer, the membranes were incubated overnight at 4°C with primary antibodies (#3570 for CD44, #5860 for CD133, #12035 for ALDH1A1, Cell Signaling Tech.). β-Actin (sc-47778, Santa Cruz Biotechnology Inc.) was used as a loading control. Following the removal of the primary antibodies, the membranes were then washed three times with TBS (tris buffered saline) phosphate buffered saline containing 0.05% Tween 20) buffer at room temperature and later incubated with fluorochrome-conjugated secondary antibody (925-32211, Li-Cor Biotechnology). The membrane was then washed with TBS three times. Final detection was made with an Odyssey infrared imaging system (Li-Cor Biotechnology).
Statistical Analyses
Statistical analyses were made by using the software InStat (GraphPad Software, Inc.). Comparisons of treatment outcome were analyzed for the statistical difference by analysis of variance. Statistical significance was assumed at a value of
Supplemental Material
sj-doc-1-npx-10.1177_1934578X211049688 - Supplemental material for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells
Supplemental material, sj-doc-1-npx-10.1177_1934578X211049688 for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells by Yao Xu, Zhiwei Zhong, Yiwen Gao, Yuhui Wang, Lanyue Zhang, Huarong Huang, Junxia Zheng, Kun Zhang, Xi Zheng and Susan Goodin in Natural Product Communications
Supplemental Material
sj-jpg-2-npx-10.1177_1934578X211049688 - Supplemental material for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells
Supplemental material, sj-jpg-2-npx-10.1177_1934578X211049688 for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells by Yao Xu, Zhiwei Zhong, Yiwen Gao, Yuhui Wang, Lanyue Zhang, Huarong Huang, Junxia Zheng, Kun Zhang, Xi Zheng and Susan Goodin in Natural Product Communications
Supplemental Material
sj-jpg-3-npx-10.1177_1934578X211049688 - Supplemental material for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells
Supplemental material, sj-jpg-3-npx-10.1177_1934578X211049688 for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells by Yao Xu, Zhiwei Zhong, Yiwen Gao, Yuhui Wang, Lanyue Zhang, Huarong Huang, Junxia Zheng, Kun Zhang, Xi Zheng and Susan Goodin in Natural Product Communications
Supplemental Material
sj-jpg-4-npx-10.1177_1934578X211049688 - Supplemental material for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells
Supplemental material, sj-jpg-4-npx-10.1177_1934578X211049688 for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells by Yao Xu, Zhiwei Zhong, Yiwen Gao, Yuhui Wang, Lanyue Zhang, Huarong Huang, Junxia Zheng, Kun Zhang, Xi Zheng and Susan Goodin in Natural Product Communications
Supplemental Material
sj-jpg-5-npx-10.1177_1934578X211049688 - Supplemental material for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells
Supplemental material, sj-jpg-5-npx-10.1177_1934578X211049688 for The Mangrove-Derived Diterpenoid Diaporthe B Inhibits the Stemness and Increases the Efficacy of Docetaxel in Prostate Cancer PC-3 Cells by Yao Xu, Zhiwei Zhong, Yiwen Gao, Yuhui Wang, Lanyue Zhang, Huarong Huang, Junxia Zheng, Kun Zhang, Xi Zheng and Susan Goodin in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81773593, 21102020) the Department of Education of Guangdong Province (2017KZDXM084, 2017KSYS010), and the Jiangmen Program for Innovative Research Team (2017TD02). Dr Xi Zheng is the Unilever Chair in Nutrition and Disease Prevention Research at Rutgers University.
Supplemental Material
NMR spectra, ESI-MS data, x-ray diffraction data of diaporthe B, and purity analysis of diaporthe B by HPLC.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
