Abstract
Background:
Hepatocellular carcinoma stem cells (HCSCs) contribute to tumor recurrence and therapy resistance, necessitating novel therapeutic strategies. Artesunate (ART), an anti-malarial drug, has demonstrated anti-cancer effects, but its mechanism in HCSCs remains unclear.
Methods:
CD133+ Hep3B cells were treated with ART, and cell viability was assessed via CCK-8 assay. Ferroptosis-related pathways were analyzed using RNA sequencing and KEGG analysis. Key ferroptosis markers and oxidative stress indicators were tested. The ferroptosis inhibitor deferoxamine (DFO) was used to validate the mechanism. Stemness (spheroid formation), malignancy (Ki67, HSP90), and metastatic potential (migration, invasion) were examined
Results:
ART dose- and time-dependently inhibited HCSCs proliferation (IC50: 82.35 μM at 12 h, 34.97 μM at 24 h and 19.76 μM at 48 h, respectively). RNA sequencing revealed ferroptosis as the primary pathway. ART upregulated TFRC, FTL and FTH1 while increasing ferric ions, ROS and MDA effects reversed by DFO. ART suppressed stemness, malignancy and metastatic potential
Conclusion:
ART induced ferroptosis in HCSCs by disrupting iron metabolism, leading to oxidative stress and lipid peroxidation. This mechanism underlies its suppression of stemness, proliferation, and metastasis, highlighting ART's potential as a targeted therapy for hepatocellular carcinoma (HCC).
Introduction
Hepatocellular carcinoma (HCC) represents a significant global health burden, being the third leading cause of cancer-related deaths worldwide.
1
The complexity of HCC, characterized by heterogeneity and chemoresistance, often leads to poor prognosis and limited treatment options.
2
In this context, the role of cancer stem cells in HCC has garnered substantial interest.
3
Cancer stem cells, known for their self-renewal capacity and tumorigenic potential, contribute to the recurrence and metastasis of HCC, making them a critical target for therapy.
4
CD133 is a glycoprotein with five transmembrane domains and two large extracellular glycosylation chains, which can be used as one of the main markers of cancer stem cells for the isolation of hepatoma carcinoma stem cells (HCSCs).
5
CD133 positive (CD133+) HCSCs can be isolated from HCC cell line cultured
Recent paradigms in oncology have shifted towards inducing specific forms of regulated cell death in HCSCs to overcome the refractory HCC. 9 Ferroptosis 10 is an iron-dependent form of non-apoptotic cell death, characterized by the accumulation of lipid peroxides.11,12 Transferrin promotes ferroptosis by delivering extracellular iron into cells via the transferrin receptor (TFRC). This imported iron fuels the Fenton reaction, catalyzing the lipid peroxidation that drives ferroptotic cell death. 13 Notably, HCSCs demonstrate vulnerabilities in their redox homeostasis, making them potentially susceptible to ferroptosis induction, which presents a novel therapeutic avenue. 14
In addition to its traditional anti-malarial effect, several studies have shown that artesunate (ART), a derivative of artemisinin,
15
significantly inhibits the proliferation of tumor cells.16,17 Our previous studies have found that ART can induce the ferroptosis of HCC cells
18
and ovarian cancer cells,
19
ultimately exerts a good anti-cancer effect. It is still unknown whether ART can induce ferroptosis in HCSCs and the molecular mechanism underlying this induction. This study was therefore conducted to address these critical gaps in knowledge. Our research innovatively focused on the CD133+ HCSC subpopulation, and employed an integrated approach combining high-throughput transcriptomic sequencing to map ART-induced pathway alterations, with functional
Materials and Methods
Isolation and Culture of HCSCs
The human normal liver cell line (HL-7702, ATCC, PCS-450-012) and human HCC cell line (Hep3B, ATCC, HB-8064) was obtained from the Shanghai Institute of Cell Research, Chinese Academy of Sciences (Shanghai, China). HL-7702 cells and Hep3B cells were cultured in MEM (GIBCO, America) containing 10% fetal bovine serum (GIBCO, America). Subsequently, CD133 magnetic bead antibody, FCR blocking reagent and MACS separation buffer were used to isolate CD133+ HCSCs from Hep3B cells according to the manufacturer's instructions (Miltenyi, Germany). 20 CD133+ Hep3B cells were cultured in DMEM/F12 (GIBCO, America) containing special cell growth factors (2% B27, 1% ITS, 20 ng/ml EGF and 20 ng/ml FGF). All cells were supplemented with streptomycin and penicillin (GIBCO, America), and were cultured at 37°C in a humidified environment with 5% CO2. CD133− Hep3B cells were collected for the following experiments.
Reagent Interventions
1 × 106 CD133+ Hep3B cells/well were seeded into low-adsorption 6-well plates and treated with different concentration of ART for 12, 24 and 48 h. Additionally, 80 μM ART+10 μM Fer-1, 80 μM ART+5 μM DFO, 80 μM ART+20 μM Nec-1 and 80 μM ART+20 μM Z-VDA-FMK were added in 6-well plates for 48 h. 19 All cells were collected for subsequent experiments.
CCK-8 Assay
According to the experimental procedures of our previous research. 19 Briefly, 2 × 103 CD133+ Hep3B cells/well were seeded into 96-well plates overnight and treated with experimental agents. Following intervention, 10 μl CCK-8 reagent (Beyotime Biotech, China) was added to each well and incubated at 37˚C for more 30 min. Finally, the absorbance was measured using a microplate reader (BioTek, America) at a wavelength of 450 nm. The cell viability rate was calculated according to the formula: cell viability rate (%) = (experimental absorbance value-blank absorbance value) / (control absorbance value-blank absorbance value) × 100.
RNA Isolation and mRNA Sequencing
All samples total RNA were isolated using TRIzol reagent (Vazyme, China) according to the manufacturer's protocol. RNA degradation and DNA contamination were evaluated by agarose gel electrophoresis. In order to enrich pure RNAs, ribosomal RNA was removed from each total RNA sample after treatment with the kit (Thermo Fisher, America). Subsequently, RNA-seq library preparation was carried out using Illumina. Following cluster generation, the libraries were sequenced on an Illumina HiSeq platform, and paired-end reads were generated. The above steps were assisted by Meggie Bio (Shanghai, China). 19
Real-Time Quantitative Reverse-Transcription Polymerase Chain Reaction (RT-qPCR)
All samples total RNA were prepared as above. cDNA was synthesized and the expression of mRNAs was measured using a RT-qPCR kit (Vazyme, China) in a 20 μl reaction volume for RT-qPCR. All primers were described in Table.S1. The reaction was performed as follows: 10 min at 95 °C and 40 cycles of 5 s at 95 °C, 30 s at 60 °C, and 30 s at 72 °C. β-actin served as a control. Relative expression was calculated using the 2 −ΔΔCt method. 19
Western Blot (WB)
Cells lysates were prepared in RIPA buffer with proteinase inhibitors and phosphatase inhibitors (Beyotime, China). The equal amount of protein from each sample was added into the SDS-PAGE gel for electrophoresis. After electrophoresis, all protein were transferred from the gel to a PVDF membrane (Millipore, America). The PVDF membrane was then sealed and exposed to the corresponding antibody. All antibodies were described in Table.S2. The blots were visualized, and digital images and densitometry were determined by a chemiluminescent imaging system (Tanon, China). 19
Cell Apoptosis Assay
CD133+ Hep3B cells were inoculated in 6-well plates at 1 × 106 cells/well and treated with 0 and 80 μM ART for 48 h. The cells were then washed with PBS prior to resuspending in Annexin V binding buffer. Later, the cell suspension was introduced with Annexin V-FITC as well as propidium iodide (PI), then incubated in dark at ambient temperature for 15 min. The samples were analyzed by flow cytometer (Becton Dickinson, USA) with FlowJo software. 21 Apoptosis rate (%) = % (Annexin V+/PI-, early apoptotic cells) + % (Annexin V+/PI+, late apoptotic cells). 22
Mitochondrial Morphological Observation
CD133+ Hep3B cells were harvested in electron microscope fixative (Servicebio, China). Next, samples were dehydrated in ethanol (with 3% uranyl acetate) for 24 h. The mitochondrial morphology of sample was detected under a transmission electron microscope (JEM 1011, Japan). 19
Determination of Trivalent Iron Ions, Ferrous Iron Content, ROS Level, Malondialdehyde (MDA) Content, GSH Content and GPX4 Content
After various interventions, the total ROS level was detected in accordance with the instructions of the ROS assay kit (Beyotime Biotech, C0033S). The intracellular MDA content, GSH content and GPX4 content were measured according to the manufacturer's instructions provided by the kits (Beyotime Biotech, S0053S and S0131S, Abcam, ab304936), as well as the total iron content and the ferrous iron content were determined according to the kit instructions (Solarbio, BC5515 and Abcam, ab83366) using a microplate reader (BioTek, America). 23
Fluorescent Immunostaining
The cells received various interventions were inoculated in a petri dish containing the cell slides, and then were postfixed for 1 h in 4% paraformaldehyde at 4 °C. The cell slides were washed three times in PBS and then blocked in 1% BSA in TBST for 30 min. The slides were incubated overnight at 4 °C in the antibodies, and then washed in TBST and incubated with appropriate secondary antibodies. Sections were mounted with DAPI. The images were obtained from a fluorescence microscope (Leica, Germany). 23
Sphere Formation Assay
2 × 103 CD133+ Hep3B cells were planted in low-adsorption 6-well plates and exposed to different reagents for a week. Then the spheres were collected and photographed, and their size were measured by the Image pro plus software. 24
Transwell Invasion Assay
After reagent treatments, 1 × 105 cells were suspended in the upper compartment with 200 μL serum-free medium and 600 μL complete medium was added into the lower compartment. The matrigel (Becton Dickinson, America) existed between upper compartment and lower compartment or not. The invasive cells migrated to the lower compartment and were stained with crystal violet (Solarbio, China) after 24 h. Three visual fields were selected at random from each compartment and counted under the microscope (Leica, Germany). 24
From the first day of cell injection, the mice were randomly divided into 3 groups: control group, ART group and ART + DFO group with 4 mice in each group. The control group received intraperitoneal injection of saline daily, the ART group received intraperitoneal injection of ART (5 mg/kg) daily, and the ART + DFO group received intraperitoneal injection of ART (5 mg/kg) + DFO (0.5 mg/kg) daily for 15 days. Every 3 days, the diameter of the transplanted tumor was measured to calculate tumor volume using a Vernier caliper. Tumor volume was calculated according to the following formula: V = (a × b2)/2 (‘a’ is long axis, ‘b’ is short axis). 15 days later, the mice were sacrificed and the weight of transplanted tumor was tested at the same time. During this period, mice were monitored every 3 days for signs of pain, such as hunched posture and reduced mobility after tumor cell injection. Euthanasia was considered in case of severe signs of pain even before the experiment's endpoint. If euthanasia is needed, mice were subjected to 70% CO2, and complete euthanasia was confirmed by checking the tail pinch. All transplanted tumors, major organs, and blood of mice were harvested for subsequent experiments.
Immunohistochemistry (IHC)
Firstly, fixing tissues in 4% paraformaldehyde, followed by paraffin embedding and sectioning. After deparaffinization in xylene and ethanol rehydration, heat-induced antigen retrieval is performed in citrate/EDTA buffer. Slides are then blocked with 3% H₂O₂ and 5% BSA, incubated overnight with primary antibody at 4 °C, and treated with HRP-conjugated secondary antibody. Signal detection uses DAB or other chromogens, followed by hematoxylin counterstaining, dehydration, and mounting for microscopy. 19
Quantification and Statistical Analysis
All statistical analyses were conducted using GraphPad Prism 8.0. Differences between two groups were analyzed by two-tailed unpaired Student's
Results
ART Induced the Ferroptosis of HCSCs
Firstly, we verified the CD133 expression characteristics of HCSCs screened by the magnetic bead antibody by WB (Fig. S1A, B). To assess the cytotoxicity of ART for the CD133+ Hep3B cells, we treated them with different concentrations of ART at 0, 12, 24, and 48 h. CCK8 assays showed that ART reduced viability in a dose-dependent and time-dependent manner (Fig. 1A). After 12 h of interventions, 80 μM ART significantly suppressed proliferation of CD133+ Hep3B cells. 40 μM and 80 μM ART both had remarkable efficacy at 24 h. After 48 h, 20 μM ART was also beginning to have a significant effect. Collectively, the IC50 of ART for CD133+ Hep3B cells was approximately 82.35 μM at 12 h, 34.97 μM at 24 h and 19.76 μM at 48 h, respectively. We selected 80 μM ART intervention for 48 h in subsequent experiments.
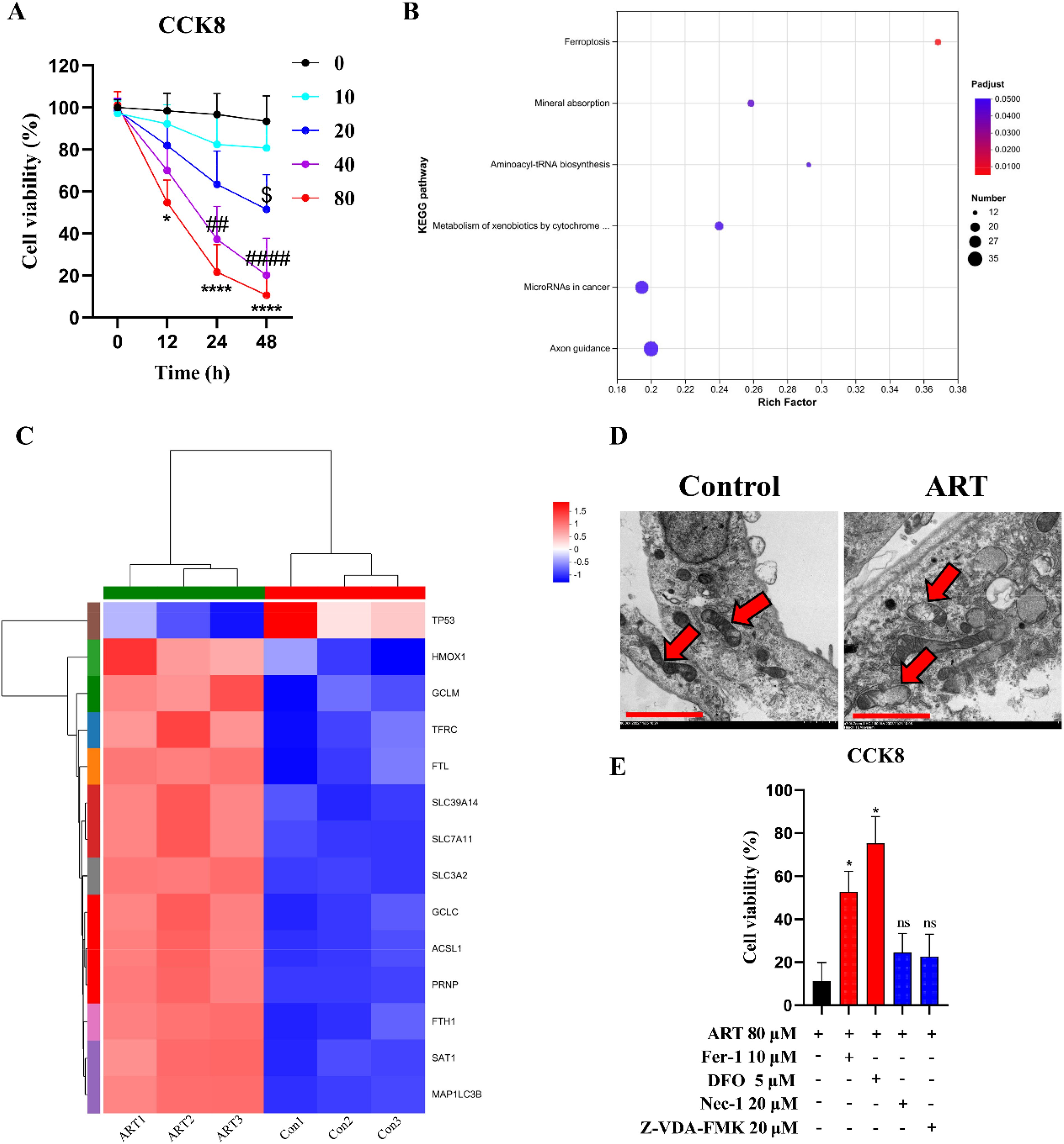
ART inhibited the proliferation of HCSCs via inducing ferroptosis. A Cell viability of CD133+ Hep3B cells exposed to 0, 10, 20, 40 and 80 μM ART for 0, 12, 24 and 48 h (n = 5 independent samples,
In order to reveal the reason why ART inhibited the proliferation of CD133+ Hep3B cells, we conducted mRNA sequencing and KEGG analysis indicated that the changes of gene profile induced by ART were mainly concentrated in ferroptosis related pathways (Fig. 1B). Data showed that ART reduced the expression of TP53 and improved the expression of HMOX1, GCLM, TFRC, FTL, SLC39A14, SLC7A11, SLC3A2, GCLC, ACSL1, PRNP, FTH1, SAT1, and MAP1LC3B (Fig. 1C). Moreover, we also observed the collapse of the mitochondrial membrane after the ART intervention (Fig. 1D). Deferoxamine (DFO, ferroptosis inhibitor to chelate iron ions) and ferroxstatin-1 (Fer-1, ferroptosis inhibitors that blocked lipid oxidation) remarkedly weakened the inhibition effect of ART on the proliferation of the CD133+ Hep3B cells, but DFO showed a stronger efficacy than Fer-1. Necrostatin-1 (Nec-1, necroptosis inhibitor) and Z-VAD-FMK (apoptosis inhibitor) had no ability to counter ART efficacy (Fig. 1E). More importantly, the apoptosis rate of CD133+ Hep3B cells did not change significantly after 80 μM ART intervention. Apart from this, there was no significant difference in the expression of serine/threonine protein kinase 3 (RIP3), mixed lineage kinase domain-like protein (MLKL) and the phosphorylation of MLKL in CD133+ Hep3B cells after 80 μM ART intervention, which were the key proteins of necroptosis. These results demonstrated that ART induced ferroptosis rather than apoptosis and necroptosis in the CD133+ Hep3B cells.
ART Induced Ferroptosis by Promoting the Iron Transport Mechanism in HCSCs
To further uncover the mechanism of ART-induced ferroptosis in the CD133+ Hep3B cells, we then assessed key regulatory factors of ferroptosis. As expect, ART certainly reduced the mRNA expression of TP53 and improved the mRNA expression of HMOX1, GCLM, TFRC, FTL, SLC39A14, SLC7A11, SLC3A2, GCLC, ACSL1, PRNP, FTH1, SAT1, and MAP1LC3B in the RT-qPCR experiments. Interestingly, ART + DFO intervention abrogated ART-induced increase of HMOX1, TFRC, FTL, SLC39A14, FTH1 mRNA levels, but had no significant effect on other genes (Fig. 2A). There was a consistent trend at the protein level (Fig. 2B, C). Additionally, ART increased the contents of trivalent iron ions (Fig. 2D), ferrous iron (Fig. 2E), ROS (Fig. 2F, G) and MDA (Fig. 2H), but made no difference in GSH content (Fig. S1G) and GPX4 content (Fig. S1H) in the CD133+ Hep3B cells. DFO could significantly reverse these changes caused by ART. Briefly, the iron transport mechanism might be more closely related to ART-induced ferroptosis in the CD133+ Hep3B cells.

ART induced ferroptosis by promoting the transport of trivalent iron ions in HCSCs. A Quantification of relative mRNA expression of TP53, HMOX1, GCLM, TFRC, FTL, SLC39A14, SLC7A11, SLC3A2, GCLC, ACSL1, PRNP, FTH1, SAT1, and MAP1LC3B in CD133+ Hep3B cells treated with saline, 80 μM ART and 80 μM ART+5 μM DFO (n = 3 independent samples). B, C Graphic representation and representative immunoblots of TP53, HMOX1, GCLM, TFRC, FTL, SLC39A14, SLC7A11, SLC3A2, GCLC, ACSL1, PRNP, FTH1, SAT1, and MAP1LC3B protein expression in CD133+ Hep3B cells treated with saline, 80 μM ART and 80 μM ART+5 μM DFO (n = 3 independent samples). D, E The trivalent iron ions and ferrous iron concentrations in CD133+ Hep3B cells treated with saline, 80 μM ART and 80 μM ART+5 μM DFO (n = 3 independent samples). F, G Representative immunofluorescence images and ROS levels (scale bar = 50 μm) in CD133+ Hep3B cells treated with saline, 80 μM ART and 80 μM ART+5 μM DFO (n = 3 independent samples). H MDA concentrations in CD133+ Hep3B cells treated with saline, 80 μM ART and 80 μM ART+5 μM DFO (n = 3 independent samples). All bars indicate mean ± SD (
ART Inhibited the Spherical Formation, Migration and Invasion Ability of HCSCs
We also observed the expression of Ki67 and HSP90 (markers of tumor malignancies) after ART intervention and ART + DFO intervention in the CD133+ Hep3B cells, and found that ART reduced the expression levels of Ki67 and HSP90, while the addition of DFO reduced the negative regulatory effects of ART on Ki67 and HSP90 (Fig. 3A, B). In addition, ART significantly decreased the cloning of stem cells into pellets, and ART + DFO intervention weakened this effect (Fig. 3A, C). With or without the matrigel, the number of cells that crossed the membrane was significantly reduced after ART intervention, meaning ART not only inhibited the migration ability, but also inhibits the invasion ability of the CD133+ Hep3B cells. DFO also had negative regulatory effects on the efficacy of ART (Fig. 4A, B). Collectively, these results suggested that the iron transport mechanism contributed significantly, though not exclusively, to ART's suppression of stemness, migration, and invasion in CD133+ Hep3B cells.

ART inhibited the spherical formation ability of HCSCs. A Representative immunofluorescence images of Ki67 and HSP90 and spherical images in CD133+ Hep3B cells treated with saline, 80 μM ART and 80 μM ART+5 μM DFO (scale bar = 50 μm). B Quantification of relative expression of Ki67 and HSP90 in A (n = 3 independent samples). C Graphic ratio of spherical forming cells in A (n = 3 independent samples). All bars indicate mean ± SD (
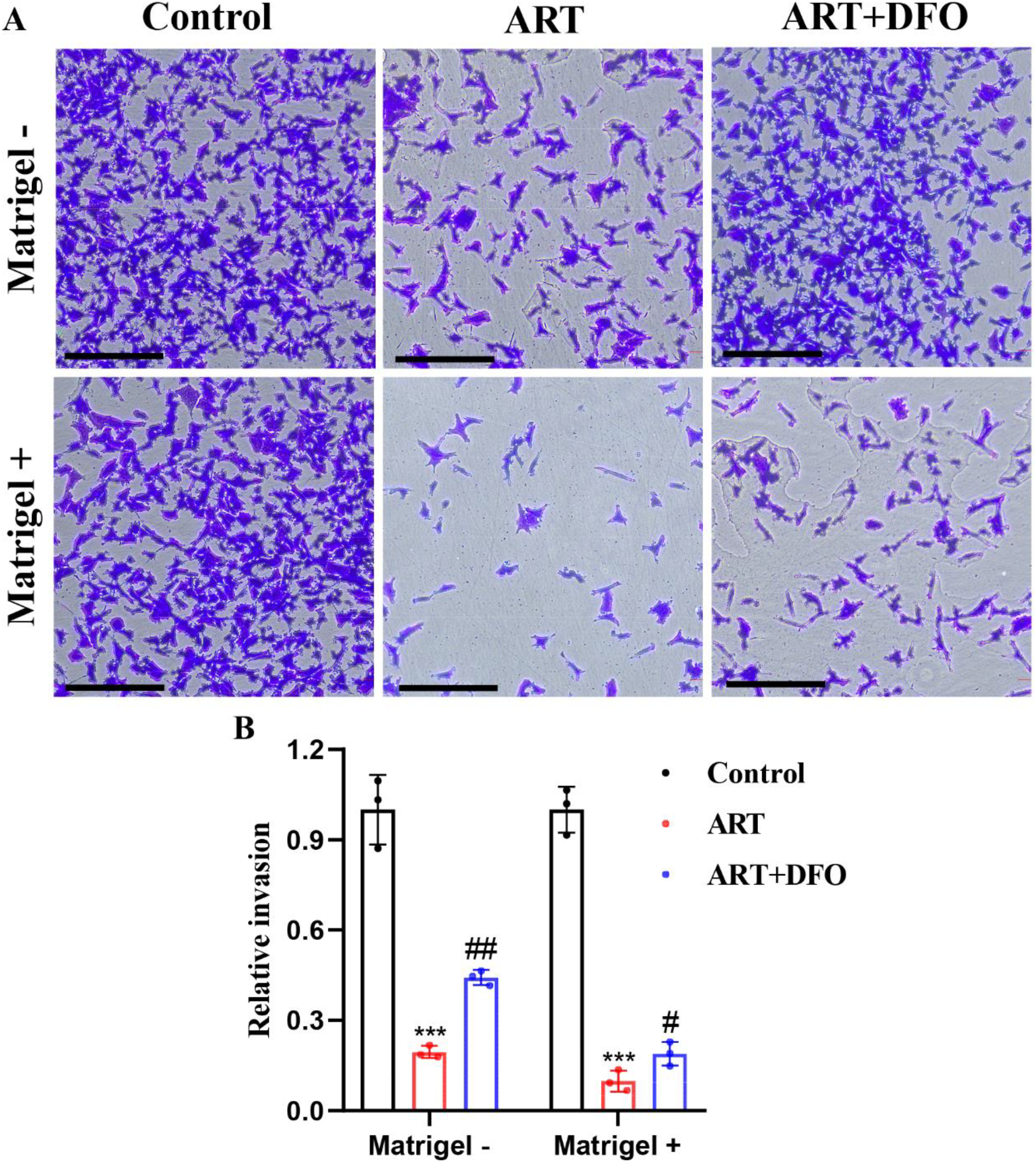
ART inhibited the ability of migration and invasion in HCSCs. A Representative image of transwell assay in CD133+ Hep3B cells treated with saline, 80 μM ART and 80 μM ART+5 μM DFO (scale bar = 100 μm). B Graphic representation of transwell assay in A (n = 3 independent samples,
ART Slowed Down the Tumorigenesis of HCSCs by Inducing Ferroptosis in Vivo
Given the ferroptosis-inducing effect of ART on the CD133+ Hep3B cells, we further explored the

ART slowed down the tumorigenesis of HCSCs
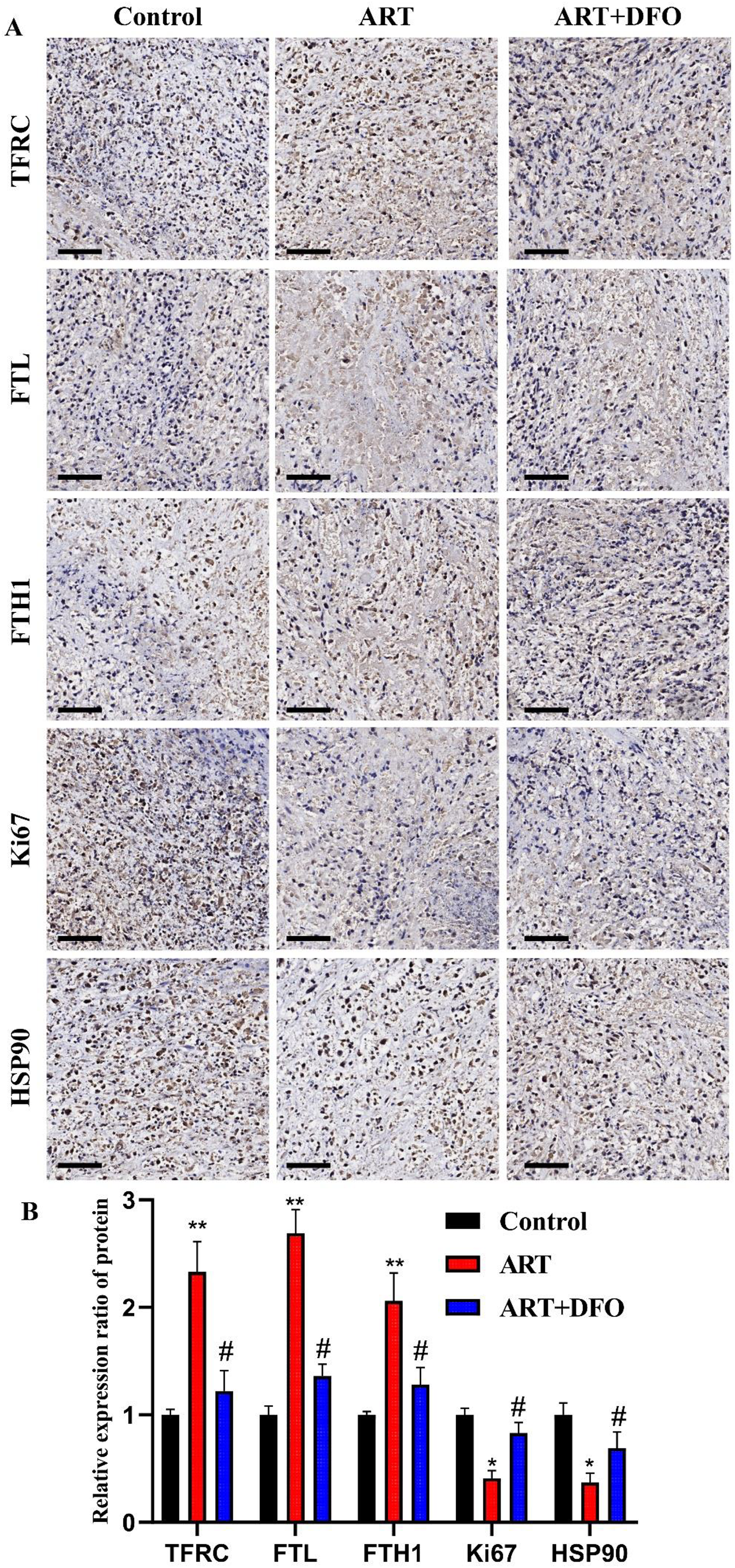
ART increased the expression of TFRC, FTL and FTH1, as well as decreased the expression of Ki67 and HSP90
Discussion
The isolation of HCSCs is the premise of this research. In clinical practice, flow cytometer and immunomagnetic bead are commonly applied for isolation of HCSCs. 26 In this study, we used immunomagnetic bead to obtain HCSCs for little damage to cell activity and the whole aseptic process, which was convenient for subsequent cell culture. This study has pivotal implications in the field of HCC treatment, particularly highlighting the potential of ART in targeting HCSCs via ferroptosis. The dose-dependent and time-dependent efficacy of ART in reducing the viability of HCSCs underscores its potential as a selective therapeutic agent. However, the investigation was primarily conducted using a single HCSC subpopulation (CD133+ Hep3B). Although Hep3B is a widely used HCC cell line, HCC is a highly heterogeneous disease. Therefore, the generalizability of these findings to other HCSC subpopulations (eg, EpCAM+, CD44+, CD13+) 27 or other HCC cell lines requires validation. Future studies should extend these observations to primary patient-derived HCSCs and HCC organoids 28 to confirm the robustness of the mechanism.
Ferroptosis, an iron-dependent form of regulated cell death, is characterized by the accumulation of lipid peroxides and ROS due to iron overload or impaired GPX4 activity. 29 We observed that ART intervention caused mitochondrial cristae collapse 30 and the increase in the iron ions, ROS and MDA, these as the marker of ferroptosis. Moreover, DFO and Fer-1 remarkedly weakened the ART-induced cytotoxicity, but Z-VAD-FMK and Nec-1 had no significant impact on the cytotoxicity of ART. These further supported the induction of ferroptosis by ART. The lack of change in GSH and GPX4 levels, which should have been reduced, might be due to a compensatory balance caused by the increase in SLC7A11 treated with ART.
Based on mRNA sequencing, 31 ART significantly upregulated key ferroptosis-related genes, including HMOX1, TFRC, FTL and FTH1, which were involved in iron uptake, storage, and metabolism. When trivalent iron ions were transported into cells via transferrin (TFRC), they could be reduced to divalent iron ions through the ferritin, which is composed of heavy chains (FTH1) and light chains (FTL), mediated iron autophagy pathway and then participated in lipid oxidation, ultimately leading to ferroptosis. The regulatory effect of ART on iron ions was mainly manifested as an increase in trivalent iron ions, confirming that iron chelation mitigated ART-induced cytotoxicity. DFO acts as a chelating agent for trivalent iron ions but has weak binding affinity for divalent iron ions. 32 Thus, DFO exhibited a stronger reverse ART effect than Fer-1, possibly because ART preferred to directly improve iron transport mechanism. Lipid peroxidation and ROS accumulation are the molecular mechanisms that intersect with iron metabolism to cause ferroptosis. Fer-1 as a synthetic antioxidant that inhibits cell ferroptosis by preventing membrane lipid damage through a reduction mechanism, 33 thus its effect against ART was not as significant as DFO.
Traditional Chinese medicine has been applied in prevention and treatment of tumor for over thousands of years owing to its characteristics of multiple targets. ART treatment led to decreased expression of Ki67 and HSP90, markers associated with tumor proliferation and malignancy, while also inhibiting spheroid formation, migration, and invasion. The reversal of these effects by DFO suggested that iron transport mechanism-mediated ferroptosis was central to ART's anti-HCSCs activity. Then, the
Our findings align with previous studies showing that ferroptosis inducers can selectively target HCSCs, which often exhibit heightened iron dependency and susceptibility to oxidative stress. However, the inability of DFO to completely reverse the effects of ART suggests that ART might induce ferroptosis through multiple pathways or mechanisms,34,35 which warrants further exploration. This multi-faceted approach could be key in overcoming the notorious drug resistance observed in HCC. Furthermore, the findings raise questions about the potential synergistic effects of combining ART with other treatments. Exploring combinations of ART with chemotherapy, targeted therapy, or immunotherapy could enhance efficacy and potentially overcome drug resistance. The first-line multikinase inhibitor for advanced HCC, sorafenib, has been reported to indirectly predispose cells to ferroptosis by downregulating SLC7A11 and depleting glutathione. A combination of ART and sorafenib could synergistically attack HCC by simultaneously targeting the bulk tumor cells (via sorafenib's anti-proliferative/anti-angiogenic effects) and the resilient HCSCs pool (via ART's iron metabolism-driven ferroptosis), potentially overcoming the therapeutic resistance commonly observed with sorafenib monotherapy. 36 ART also sensitized HCC to sorafenib via exacerbating AFAP1L2-SRC-FUNDC1 axis-dependent mitophagy. 37 Moreover, the combined therapeutic schedule of ART and selective EGFR-tyrosine kinase inhibitor erlotinib demonstrated excellent anti-cancer effects in the clinical trial. 38 However, further studies are needed to optimize dosing strategies and evaluate potential synergies with other ferroptosis inducers or conventional chemotherapies.
Conclusion
In summary, our findings establish that ART induces ferroptosis in CD133+ Hep3B cells by disrupting iron metabolism, leading to oxidative stress and lipid peroxidation. This mechanism underlies ART's ability to suppress stemness, proliferation, migration, and invasion
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261428190 - Supplemental material for Artesunate Induced Ferroptosis in Hepatocellular Carcinoma Stem Cells: Unveiling a new Therapeutic Approach
Supplemental material, sj-docx-1-npx-10.1177_1934578X261428190 for Artesunate Induced Ferroptosis in Hepatocellular Carcinoma Stem Cells: Unveiling a new Therapeutic Approach by Dengyun Nie, Lingjun Liu, Rong Jia, Qian Zhu, Xiaofang Yin and Han Zhang in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X261428190 - Supplemental material for Artesunate Induced Ferroptosis in Hepatocellular Carcinoma Stem Cells: Unveiling a new Therapeutic Approach
Supplemental material, sj-docx-2-npx-10.1177_1934578X261428190 for Artesunate Induced Ferroptosis in Hepatocellular Carcinoma Stem Cells: Unveiling a new Therapeutic Approach by Dengyun Nie, Lingjun Liu, Rong Jia, Qian Zhu, Xiaofang Yin and Han Zhang in Natural Product Communications
Footnotes
Acknowledgements
Special thanks to the Shanghai Meiji Biological Co. for their support of this study.
Consent for Publication
All authors approved the final manuscript and the submission to the Natural Product Communications.
Author Contributions
Methodology: D.N.; Acquisition of data: L.L.; Analysis of data: Q.Z. and R.J.; Conception and design: X.Y.; Writing-original draft: D.N.; Supervision: H.Z.
Ethics Considerations
The Institutional Animal Care and Use Committee of the Jiangsu Huachuang Xinuo Medical Technology Co., LTD approved all animal experiments performed in this study (Approval Number: IACUC-2022-0010).
Funding
This work was financially supported by the Nanjing Hospital of Chinese Medicine Feihong program (FHJH202404).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Statement of Human and Animal Rights
According to the International Society for the Study of Cancer guidelines, we attempted to use the minimum number of mice necessary to achieve statistical significance.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
