Abstract
Diterpenes are a class of critical taxonomic markers of the Euphorbiaceae family, representing small compounds (eg, molecules) with a wide range of biological activities and multi-target therapeutic potential. Diterpenes can exert different activities, including antitumor and multi-drug resistance-reversing activities, and antiviral, immunomodulatory, and anti-inflammatory effects, mainly due to their great structural diversity. In particular, one polycyclic skeleton has been highlighted: ingenane. Besides this natural diterpene, promising polycyclic skeletons may be submitted to chemical modification—by in silico approaches, chemical reactions, or biotransformation—putatively providing more active analogs (eg, ingenol derivatives), which are currently under pre-clinical investigation. This review outlines the current mechanisms of action and potential therapeutic implications of ingenol diterpenes as small cancer molecules.
Introduction
Folk or traditional medicine is considered a summation of health practices and approaches, popular knowledge, and beliefs which comprise plants, fungi, minerals, and animal-based products employed either singly or in combination in the maintenance of health, as well as for diagnosis, treatment, or prevention of illnesses, and may be found in diverse regions of the world.1,2 Medicinal plants and other natural products have been used to treat various ailments in folk medicine systems. 3
Importantly, natural products, particularly derived from plants (ie, “secondary” metabolites), are the primary sources of chemical diversity and, as such, play essential roles in the field of cancer pharmaceutical discovery.4-6 These natural compounds are classified according to their biological function, biosynthetic origin, or main structural characteristics. Secondary metabolites have four main classes: terpenoids, polyketides, phenylpropanoids, and alkaloids. 7
Over 60% of the antineoplastic drugs are directly or indirectly related to natural products. 8 Vinca alkaloids, such as vincristine and vinblastine, isolated from leaves of the periwinkle plant (Catharanthus roseus L.) are the best-known examples. 9 Teniposide, etoposide, and etoposide phosphate (Etopophos®), which are semi-synthetic derivatives of epipodophyllotoxin, have also been used as clinically active agents. 10 Topotecan, belotecan, and irinotecan (CPT-11) are semi-synthetic camptothecin derivatives, isolated from the Chinese ornamental tree Camptotheca acuminata Decne. and used in clinical practice. 11 Furthermore, paclitaxel (Taxol®), a plant-derived anticancer drug, was found in Taxus species. 12 Taxol is a natural diterpene alkaloid, a family of small molecules with a wide range of pharmacological activities. Nevertheless, the vast majority of the more than 350,000 known plant species remain uninvestigated.5,13
Ingenol 3-angelate (Picato®), a diterpenoid structure isolated from Euphorbia peplus L., was approved for the treatment of actinic keratosis (a pre-cancerous skin condition) by the U.S. Food and Drug Administration (FDA) in 2012, and the European Medicines Agency (EMA) in 2013. However, it was withdrawn from the EU in 2020, due to the observed development of skin cancer in ∼6% of patients using Picato®.14-19 Several studies regarding the isolation, synthesis, and anticancer effects of diterpenoids from Euphorbia species have been published. These diterpenoids were reported to have antiproliferative, 20 cytotoxic, 21 antimicrobial, 22 and antipyretic–analgesic activities. 23
Other species have also been studied for their antitumor effect. The ethanolic and methanolic crude extracts of Boerhavia diffusa L., belonging to the Nyctaginaceae family, showed anticancer activity against cervical, prostate, and breast cancers, and inhibited metastases in vitro and in vivo, 24 as well as the extract of Tabebuia pallida Lindl. leaves, which showed inhibition of the growth of tumor cells through induction of apoptosis. 25
Furthermore, a literature review highlighted flavonoids that have very interesting palliative effects on clinical symptoms, such as diarrhea, mucositis, neuropathic pain, and others, often associated with chemotherapy treatment of colorectal cancer. 26
Emodin (13,8-trihydroxy-6-methyl-anthraquinone), a natural anthraquinone derivative found in species of Rhamnaceae, Polygonaceae, Rubiaceae, and Fabaceae, has been suggested to be effective in pancreatic cancer by modulating tumor growth, angiogenesis, invasion, and metastasis. 27 Other biological studies performed on furanocoumarins and coumarin derivatives revealed that these compounds have antitumor effects.28,29
However, the biological activities of natural compounds are not limited only to antitumor activity. The ethanol extract of Ophiorrhiza rugosa L. leaves demonstrated analgesic and anti-inflammatory potential in different animal and computational models. 30 Prominent antidiabetic properties have been reported for Gynura species through in vitro and in vivo studies. 31 Clinical trials have evaluated the anti-infection activity of Andrographis paniculata Burm. F. against URTIs, influenza, and HIV, as well as its effectiveness in treating osteoarthritis and multiple sclerosis. 32 Phytocannabinoids exert potent actions throughout the body. Data report their high therapeutic potential to treat the diseased gastrointestinal tract, from functional to organic pathologies. 33
In summary, compounds of plant origin have been extensively researched to find a better and safer alternative treatment, mainly for neurological diseases. Experimental studies have shown that phytochemicals such as alkaloids, terpenes, flavonoids, and phenolic acids, as well as lipids, have significant potential in in vitro and in vivo models of psychiatric disorders.34-37 This review addresses, in the cancer context, the mechanisms of action of macrocyclic diterpenes, such as ingenane and their synthetic/semi-synthetic derivatives, from Euphorbiaceae, plants with wide application in traditional medicine. We highlight the in vitro and in vivo anticancer effects of natural, semi-synthetic, and synthetic derivatives, as well as the in silico approaches involved in developing anticancer agents from ingenane. Additionally, we summarize the available information concerning these ingenane small molecules to support future antitumor drug discovery.
Euphorbiaceae
The Euphorbiaceae family (also known as spurges) is considered one of the largest plant families, having about 300 genera. Within this family, the Euphorbia genus is one of the richest, exhibiting a great chemical diversity among circa 2000 species. Plants of this genus have a cosmopolitan distribution, are found in all tropical and temperate regions,38,39 and have been widely used in traditional medicine.40-42 To date, 33 species have been reported in folk medicine. 40 An array of applications can be found, such as the treatment of gonorrhea, migraine, intestinal parasites, purgative agents, digestive and respiratory disorders, skin diseases,40-43 inflammation, disorders of the respiratory system, and wart cures.42,44 Moreover, several studies using extracts derived from Euphorbia species have also demonstrated their anti-inflammatory, immunomodulatory, and antiproliferative action in tumor cell cultures.4,45-49
Although the milky latex is the most relevant and useful product obtained from Euphorbia species, 50 roots, seeds, wood, barks, and leaves have been used for the applications and properties mentioned above,40,42,43 as well as the essential oils (EOs). 43
Six major classes form the chemical structures of the Euphorbia genus; namely, sesquiterpenoids, flavonoids, triterpenoids (including steroids), phenolics, cerebrosides, and a wide diversity of macro- and poly-cyclic diterpenoids.44,47,51 Diterpenoids constitute an extensive class of isoprenoid compounds, one of the widest families of natural molecules. These compounds are classified according to their biogenesis, reaching up to 126 distinct carbon skeletons. 42
Traditional Uses of Euphorbiaceae Plants
Over the decades, the literature has indicated several traditional uses of Euphorbiaceae plants. The roots of Euphorbia fischeriana Steud., a perennial herbaceous plant known as “lang-du,” have been commonly used in traditional Chinese medicine (TCM) for thousands of years to cure immune diseases. 52 It is also used to treat cancer, ascites, edema, indigestion, skin ulceration, asthma, and tuberculosis.53-55 This species contains diterpenoids as its major chemical constituents, followed by triterpenoids, steroids, and phenolic acids.56,57
Lathyrane diterpenoids and some triterpenoids are present in the roots of Euphorbia kansuensis Prokh., which are used as a purgative and to treat apocenosis, cholagogue, and pyretolysis. 58 This same class of diterpenes, including ingenanes, are the main constituents of Euphorbia lathyris L. Its seeds possess biological activities to treat hydropsy, constipation, amenorrhea, ascites, migraine, and snakebites. 59
The Chinese herbal medicine, Euphorbia esula L., has been used to treat cancer, warts, edema, diarrhea, vomiting, and fever, and as a diuretic remedy.60,61 Previous phytochemical studies on this genus have led to the identification of numerous diterpenoid classes, such as jatrophane and lathyrane, but mainly ingenane. 62
Diterpenoids and terpenoids are also commonly found in species such as Euphorbia helioscopy L. and Euphorbia resinefera Berg., which are used in traditional medicine to treat malignant tumors and chronic obstructive pulmonary diseases, 63 and to suppress chronic pain and tuberculosis, respectively. 64 The roots of the perennial herb, Euphorbia stracheyi Boiss. are used to treat hemostasis, muscular regeneration, and analgesia. 65 Previous research reported diterpenoids, sesquiterpenes, flavonoids, steroids, and phenolic compounds as responsible for several of these bioactivities.65-67
E. peplus L. is commonly known as “petty spurge.” The sap from these plants has been used to treat asthma, as a topical treatment for warts and corns, tumors, and as a purgative. 68 An active ester diterpene extracted from E. peplus was clinically approved for treating actinic keratosis69,70; however, its use was suspended due to its risks outweighing its benefits. 19
The latex of Euphorbia caducifolia Haines., also known as “danda thor,” is used in popular medicine for treating bleeding and wounds. Furthermore, it has been used to cure cutaneous eruptions and earache. 71 Euphorbia canariensis L. was also described as having vascular properties. 72
The latex of Euphorbia antiquorum L. has been used for the treatment of skin sores, toothache, scabies, palsy, and bronchitis, 73,74 and the fresh stem is applied as a purgative, diuretic, and emetic. 75
The shrub of Euphorbia tirucalli L., named “aveloz” or “pencil-tree”' by Brazilian indigenous communities, has been used in traditional medicine as a remedy for rheumatism, asthma, and stomach ailments. 76 Depending on the region, E. tirucalli is reported to be used in the treatment of warts, toothache, hemorrhoids, snake and scorpion bites, epilepsy, cough, swellings, asthma, dropsy, colic, sexual impotence, and a remedy against syphilis, among others. 77
In traditional medicine, Euphorbia trigona Mill. has been used to heal constipation and epilepsy, 78 inflammation, and infections, mainly in the urinary tract, 79 while Euphorbia palustris L. is used to treat hydrophobia. 80 Other Euphorbia species, such as Euphorbia marginata Pursh. and Euphorbia hirta L., are used to treat inflammatory conditions, as well as respiratory system disorders and afflictions of the skin and mucous.43,81
Belonging to the Euphorbiaceae family, Croton tiglium L. is spread in tropical and subtropical regions and is one the most used plants in folk medicine. 82 Its seeds are popularly known as “badou.” In TCM, their fruits are used to treat gastrointestinal diseases (eg, stomachache, abdominal pain, abscesses, constipation, and ulcer), inflammation, malaria, and rheumatism.82,83 Homalanthus nutans is indicated to treat back pain, abdominal swelling, yellow fever (hepatitis), diarrhea, and gonorrhea.84,85
Euphorbia cornigera Boiss. is described as one of the most toxic species in this family. Nevertheless, its leaves, roots, shoots, and fruits are used in the treatment of various ailments. Esters of ingenol and phorbol have been reported in this species. 86 With regards to toxic properties, Euphorbia pekinensis Rupr. is known as a toxic Chinese medicinal herb, which is used for edema distention, hydrothorax, and ascites. 87 Previous reports assume that the toxicity may be related to many diterpenoids present in this species.88-90
Lastly, Euphorbia serrata L. has been used in Nigeria to treat skin diseases, migraines, intestinal parasites, gonorrhea, warts, asthma, bronchitis, and dysentery, 91,92 whereas Euphorbia granulate Forssk. is described with diuretic properties and is capable of purifying the blood. Besides, it has been used as an anthelmintic and purgative. 93
As evidenced, the literature has shown that the Euphorbiaceae is abundant in diterpenoids, which are believed to be responsible for several properties indicated in traditional medicine as described above. Many extensive and detailed studies of extracts and molecules derived from Euphorbiaceae plants are proving to be promising for the treatment of several diseases.
Diterpenes
Structural Diversity and Classification
Natural products are classified based on their biogenic origins (terpenoid/isoprenoid, phenylpropanoid, polyketides, and alkaloid). Within terpenoids, diterpenes and diterpenoids are of particular interest. Diterpenoids are compounds comprising 20 carbon atoms, formed by a head and tail (ie, isopropylidene and alcoholic ends, respectively) of four isoprene building block (C5H8) units. They form part of a large class of polycyclic compounds which can be widely found in plants, fungi, bacteria, and the animal kingdoms, in both terrestrial and marine environments.12,51,94,95 Numerous interactions with living organisms are created, due to the large variability of diterpene skeletons and acylation patterns, which have been potentially investigated for treating different diseases. 12 Briefly, the structure of this class originates from the isomeric five-carbon building blocks of isopentenyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP). Both are synthesized through the cytosolic mevalonate (MEV) and plastidial methyl-erythritol phosphate (MEP) pathways. 96
The known diterpenes constitute a variety of approximately 18,000 (or more) compounds derived from (E,E,E)-geranylgeranyl diphosphate (GGPP), 97 and their classification can be based on the biogenetic construction origin of the hydrocarbon ring. Therefore, they are classified and represented as acyclic, monocyclic, bicyclic, tricyclic, tetracyclic, and pentacyclic, in which the ingenanes can be found, and macrocyclic diterpenes (Table 1).
Summary of Diterpene Classes, Their Compounds, Derived species, and Properties.
Furthermore, diterpenoids can also be classified into higher and lower diterpenoids. The higher diterpenoids include ent-abietanes, ent-atisanes, ent-kauranes, ent-isopimaranes, and ent-pimaranes; while lower diterpenoids comprise casbanes, jatrophanes, lathyranes, myrsinanes, tiglianes, ingenanes, segetanes, paralianes, pepluanes, and euphoractine. Both include the most-studied diterpenoids isolated from Euphorbia species 44 (Figure 1).

Representation of diterpenoid classification: higher diterpenoids (abietane, atisane, kaurene, and rosane) and lower diterpenoids (ingenane, tigliane, lathyrane, casbane, daphnane, mirsinane, and jatrophane). The image was created using the biorender platform (https://biorender.com/).
Diterpene Biological Activity
The structural diversity of diterpenes enables a wide array of biological activities, including antioxidant, anti-inflammatory, antimicrobial, wound-healing, antihypertensive, analgesic, neuropharmacological, and antineoplastic activities.122-124
Diterpenoids are significant components of E. fischeriana Steud. and exhibit various pharmacological properties, such as antiviral, immune-enhancing, sedative, analgesic, and anticancer. 53 These diterpenoid-associated antineoplastic mechanisms include cell cycle arrest, modulation of apoptosis, and inhibition of tumor cell metastasis by interfering with multiple signaling pathways in several tumor types. 125 Moreover, studies have reported that diterpenoids from Euphorbia spp. also induce different immune responses, through the modulation of the protein kinase C (PKC) pathway, as well as the release of prostaglandins and proinflammatory cytokines such as TNF-a, interleukin-6 (IL-6), and interleukin-1 beta (IL-1 beta).46,53,126,127
Due to the extensive bioactivities of diterpenes, they have garnered attention in the drug discovery industry.13,128 Paclitaxel, also known under its commercialized name Taxol, is used for chemotherapy in ovarian, prostate, breast, esophageal, melanoma, lung, and other solid tumors. In the same manner, like Taxol, another compound used in cancer therapy is TPI287, a synthetic derivative of the taxane diterpenoid drugs (taxonomic), and which has been related to passage through the blood–brain barrier. 124 Other examples are ingenol mebutate, from E. peplus, which was used for the treatment of actinic keratosis, and forskolin, isolated from Coleus forskohlii roots, which is used as a cardioprotective compound.128,129 Other less-explored compounds include the ginkgolides derived from Ginkgo biloba L., resinous acids from Copaifera spp., and stevioside from Stevia rebaudiana Bertoni.130,131
Interestingly, it has been evidenced that a lathyrane diterpene, Euphorbia factor L1 (EFL1), derived from seeds of E. lathyris, reversed multi-drug resistance (MDR) by inhibiting the efflux function of ABCB1. 132 The most significant cause of MDR in cancer cells has been linked to the high expression and activity of ABCB1 in the efflux of chemotherapeutics. 133 In this study, the authors suggested that to overcome MDR, EFL1 might be used in combination with an ABCB1 substrate chemotherapeutic.132,133
Regarding key structures in ingenanes being mainly responsible for the reported activities, the literature demonstrates that there are limitations to drawing clear structure–activity relationships within this family of compounds. 134 Most studies covering the period 1998–2014 that report their chemical structures and biological activities have limited reference to the structure–activity relationship of these constituents.
Thus, what is currently known is that possibly the structure–activity relationship of these diterpenes revealed that acetylation and esterification of hydroxyl groups, particularly at C-3 and C-8, positively affect these activities. 135
Due to the various substitution patterns in isolated ingenol-type diterpenoids, structure–activity relationships remain under investigation. The rigid ingenol scaffold conveys several structural features, including the 5- and 20-hydroxyl groups, which may be essential for biological activity. 136 However, this information is still not understood.
Ingenane Diterpenoids
Natural Occurrence
Ingenols are diterpene compounds derived from Euphorbia members. 40 Ingenol was identified for the first time in 1968, in Euphorbia ingens E.Mey.137,138 Formerly, it had been isolated from the seed oil of Croton tiglium, belonging to the Euphorbiaceae family. 139 In plants, ingenol is commonly present in a conjugated form and can be altered by several carboxylic acids. The main types and their species are compiled in Table 2.
Main Ingenane-Type Diterpenoids Found and Their Respective species.

Another essential feature of ingenols is their intrinsic affinity with PKCs.51,140 The most descriptive molecular exploration of the interaction of ingenols with PKC was performed using ingenol 3-benzoate, which has been considered a potent PKC modulator and anticancer compound. 136 One ingenol derivative, ingenol 3,20-dibenzoate (IDB), has been shown to activate the classical isoforms of PKCs. 141
Semi-Synthetic Molecules
To improve the clinical efficacy and decrease the side effects, such as drug resistance and cytotoxicity to normal cells, semi-synthetic analogs can be designed from an original compound. 188 Semi-synthetic ingenols have been conceived to enhance their latent activity.186,189
The first complete ingenol synthesis, through the use of an intramolecular de Mayo reaction, was accomplished by Winkler. 190 Other successful synthetic approaches, such as those of Wood and Tanino/Kuwajima, have also been reported.191-193 One of the reasons ingenol has raised great interest is due to its atypical structure, holding an “inside − outside” bridged BC ring, and its broad spectrum of pharmacological activities. 193 However, the downsizing of ingenanes, such as ingenol mebutate, presents low quantities when obtained from plant sources, therefore making its production inefficient; thus, alternative synthesis sources are desirable for such compounds. 194
In order to overcome the barrier of the production limitations mentioned above, Nakamura et al, in 1997, carried out an efficient method for synthesizing ingenane in tandem with the complete C and D ring structure of ingenol. 195 Thereafter, using the same approach with alternative commercial compounds, the authors described another complete ingenol synthesis. 193 Rigby et al, in 2002, developed a competent form of access to the ingenane core through Lewis acid-catalyzed intramolecular [6 + 4] cycloaddition. 196 The major hurdles regarding the synthesis are the strained “in − out” bicycloundecane ring and a heavily oxygenated and functionalized molecule part. Picato®, also called PEP005 or ingenol mebutate, which can be obtained from the sap of E. peplus, was formed through a semi-synthetic method designed by Liang et al in 2012. 182 Although complete synthesis of the critical precursor—ingenol—has been reported, 194 its possible use in industrial applications remains unclear.
Other macrocyclic compounds include resiniferatoxin (RTX), a daphnane diterpene from the latex of E. resinefera, 197 for which the first total synthesis was reported by Wender et al in 1997. 198 To date, a few synthetic efforts towards the partial synthesis of RTX have also been documented. 199 In 2008, Wender et al created analogs of prostratin [13-O-acetyl-12-deoxifolia], a phorbol ester derived from Homalanthus nutans (G.Forst.) Guill., by applying a semi-synthetic approach utilizing C-13 modifications; so far, its complete synthesis has not yet been reported. 200
Recently, Amazônia Fitomedicamentos Ltda, a Brazilian enterprise, reported three novel ingenol semi-synthetic esters derived from the sap of E. tirucalli. 185 The ingenol core ring was modified biochemically by adding an ester chain at C-3, which led to three new molecules, named ingenol-3-trans-cinnamate (ingenol A), ingenol-3-hexanoate (ingenol B), and ingenol-3-dodecanoate ingenol C). The synthesis of ingenols originated at Kyolab Laboratories (Campinas, Brazil) and was distributed by Amazônia Fitomedicamentos Ltda, Brazil, through a patent, as previously described by Abreu et al.185,186
These results demonstrate the potential therapeutic application of ingenol; however, the findings and their implications should be discussed in the broadest context possible. Future research directions may also be highlighted.
Ingenane Biological Activity
Ingenane-type diterpenes share an ingenol core, showing antineoplastic or anti-HIV activities. 201 Their proinflammatory activities have been evaluated, indicating a moderate irritant activity of the compounds, 47 thus providing good pesticidal activity against brown planthopper, 146 as well as molluscicidal activities. 202
At present, ingenol 3-angelate (Picato®, PEP005, ingenol mebutate), isolated from E. peplus, 203 was the only ingenol diterpene which has been approved for clinical use by the FDA and the EMA. Despite, it having shown promising chemotherapeutic effects for skin cancer treatment, including topical therapy for actinic keratosis and superficial basal cell carcinoma,204,205 this drug had its use suspended in 2020 due to the occurrence of skin cancers in a circa 6% of patients. 19 Ingenol 3-angelate is considered a PKC activator and has shown antiproliferative activity against human melanoma cell lines, inducing apoptosis and cell cycle arrest, and suppressing the growth of PAM212 and B16 subcutaneous tumors in mice.69,206 Moreover, it also appears to have antineoplastic effects in leukemia cell lines and primary acute myeloid leukemia blasts. 205
20-O-Acetyl-ingenol-3-angelate (PEP008) is considered a novel ingenol ester, which is a structural analog of PEP005. Both are derived from E. peplus and are PKC activator diterpenes. This compound induced permanent growth arrest and senescence in a panel of breast, melanoma, and colon cancer cells, 167 and sensitized leukemia cell lines 166 in vitro. Nonetheless, mouse model studies are missing to demonstrate their in vivo antitumor activity.
According to Dang et al, the diterpenes EK-1A, EK-5A, and EK-15A, isolated from Euphorbia kansui, can reactivate latent HIV-1 infection in a primary CD4 + T-cell model and Jurkat cell lines at a nanomolar level of concentration. 141 Moreover, four diterpenes isolated from E. kansui, 13-undecanoyl-3-(2,4-dimethylbutanoyl) ingenol, 3-(2,4-decadineacyl) ingenol, kansuiphorin A, and kansuinin A increased the cytoimmunity of an organism resulting in declining virus levels. 207 These associations among bioactivities and structural features have been determined to be due to the ingenol derivatives holding acetyl groups on C-13 and C-20, which seems essential for their antiviral activity. 208
Ingenol-3-hexanoate (ingenol B), isolated from E. tirucalli, effectively promoted HIV LTR-induced gene expression through the activation of PKCs. 209 Generally, most ingenol derivatives function as PKC agonists. 185
Furthermore, the ingenane diterpenes 3-O-angeloyl-17-angeloyloxy-20-deoxyingenol and 20-O-acetyl-3-O-angeloyl-17-angeloyloxyingenol can be used as MDR modulators but have toxic effects. In another study, the ingenanes called 6 and 7 from Euphorbia taurinensis. All have demonstrated remarkable MDR modulating effects at 20 μM. 202
Major Ingenane Compounds and Anticancer Effect
The shift from cytotoxic chemotherapy-based treatment to the discovery and development of new antitumor drugs based on well-defined molecular targets has resulted in an intensifying number of successful therapies, which have a direct impact on the lives of cancer patients. 210 Many studies have reported the antitumor activities of Euphorbia spp. diterpenes in recent years, and studies have shown diterpenes to have excellent antitumor effects in both in vivo and in vitro models.43,53
Diterpenes isolated from E. kansui (ie, tigliane-, ingenane-, and daphane-type diterpenoids) have been shown to possess antiallergy, antiviral, and antitumor activities. 211 Some constituents have even shown selective cytotoxicity to melanoma (SK-MEL-5), lung (H-322), breast (MDA-MB-435), and colorectal (Colo205) cancer cells. 212 Most of the compounds evaluated showed inhibitory and antiproliferative activity against MDA-MB-435 and Colo205 cells.
Miyata et al explored the biological effects of ingenol diterpenes isolated from the roots of E. kansui on the proliferative capacity of Xenopus W.Roelofs’ embryo cells. 213 Eight of the analyzed diterpenes—that is, 20-O-(2′(E),4′(E)-decadienoyl) ingenol, 20-O-(2′(E),4′(Z)-decadienoyl) ingenol, 3-O-(2′(E),4′(Z))-decadienoyl) ingenol, 3-O-(2′(E),4′(E)-decadienoyl) ingenol, 20-O-decanoylingenol, 5-O-(2′(E),4′(E)-decadienoyl) ingenol, 5-O-benzoyl-20-deoxyingenol, and 3-O-(2′(E),4′(E)-decadienoyl)-20-deoxyingenol—significantly inhibited cellular proliferation at low concentrations. Furthermore, most of these diterpenes also inhibited topoisomerase II activity. 214 Previously, Yoshida et al reported that 20-O-(2′(E),4′(Z)-decadienoyl) ingenol inhibited topoisomerase II through cell cycle arrest in the G2/M phase without activating γ-H2AX, due to breaks in the DNA molecules in mouse mammary tumor cells. 215
Khiev et al reported that two ingenane-type diterpenoids, 20-O-acetylingenol-3-O-(2′E,4′Z) decadienoate and 3-O-(2,3-dimethylbutanoyl)-13-O-dodecanoylingenol, isolated from E. kansui, exhibited the most significant response in interferon-gamma (IFN-γ) production. 147 IFN-γ proteins are critical in the early innate defense against tumor control, viral, and bacterial infections, as they are responsible for the production of immune cells. 216 These findings indicated that ingenane-type diterpenoids might be promising anticancer compounds. 53 20-O-D-ingenol Z isolated from E. kansui Liou. inhibited cell entrance into the S- and M-phase sub-stages. On the other hand, 20-O-D-ingenol E induced a growth arrest of 75–80% in embryonic cells and suppressed mitosis progress. 217
Moreover, using in vitro models, 14 diterpenoids derived from E. fischeriana have been shown to inhibit cancer cell proliferation with promising IC50 values. Among them, 13 (including 3 ingenanes: ingenol-3-myristinate, ingenol-3-palmitate, and ingenol-20-myristinate) demonstrated inhibition of the formation of mammospheres using human breast cancer cells (MCF-7 cell line). 125 These results demonstrated the potential of these bioactive diterpenoids for the further investigation of cancer stem cells. Another study evaluated the cytotoxicity of 11 compounds extracted and isolated from the roots of E. fischeriana using the MTT method. Exposure to three diterpenes (ingenol 6,7-epoxy-3-tetradecanoate, ingenol 3-palmitate, and ingenol-3-myristinate) proved effective in the A549 human lung tumor cell line. 125 Moreover, studies have revealed that E. fischeriana has the potential to produce further diterpenoids, of which 24 have been identified as having antitumor activities. 53
Yan et al extracted and isolated the root constituents of E. kansuensis Prokh. in 2019, resulting in a highly modified compound named euphorkanlide A, 58 a diterpene of the ingenane type. This compound may induce cell-cycle interruption by generating ROS and apoptosis in cells of the HCT-15/5-fluoracil (5-FU)-resistant cell line. In the study by Fallahian et al in 2017, a tetrahydroingenol diterpene, identified as 7,13-diacetyl-5-angeloyl-20-nicotinyl-3-propionyl-1,2,6,7-tetrahydroingenol (DANPT), was extracted and isolated from the aerial part of Euphorbia erythradenia Bioss. and assessed for its anticancer potential and its mechanisms of action in human melanoma tumor lines (A375 and HMCB). 49 Two new ingenol derivatives from E. cornigera, called 214 and 215, have been isolated by Baloch et al. 218 In vitro, compound 214 showed cytotoxicity in mouse macrophage cells (RAW) and a colon cancer cell line (HT-29). In contrast, compound 215 was more effective than amrubicin hydrochloride (used as positive control), but demonstrated moderate cytotoxic activity against both cell lines. 218
Diterpenes derived from Euphorbia royleana Boiss. have also been evaluated, in which ingol-3,7,12-triacetate-8-benzoate stood out as a potent MDR modulator, acting as an inhibitor of P (gp-P) transport, thus influencing the efficacy of doxorubicin. 219
Three new diterpenoids have recently been isolated from the roots of E. stracheyi Boiss., while the cytotoxicity of 19 known diterpenoids was evaluated in human cancer cell lines in the study by Ye et al (2020). Of the new compounds evaluated, the ingenane-type diterpene 3β,20-diacetoxy-5β-deca-2′′E,4′′E,6′′E-trien-4β-hydroxyl-1-one and 20-O-acetyl-[3-O-(2′E, 4′Z)-decadienoyl]-ingenol both showed significant toxicity against the tumor cell line. 220
In animal models, IDB derived from E. esula attenuated the anemia induced by the cytotoxicity of the drug 5-FU, exhibiting a synergistic effect on erythropoietin (EPO) at low concentrations. However, IDB has also been used as a potential antitumor agent at higher concentrations, in which apoptosis was the endpoint in Jurkat cells, and an antagonistic effect was discovered. 221 Systematic fractionation of an extract of E. esula L. led to the characterization of diester IDB as a compound with significant inhibitory activity against P-388 lymphocytic leukemia in mice. 138 Several in vitro and in vivo pre-clinical studies have indicated the antitumor activity of ingenol 3-angelate through a double mechanism of action: the first is a cytotoxic effect (tumor cells death promoting necrosis), 222 while the second consists of the activation of the immune system, mediated by neutrophils causing antibody-dependent cellular cytotoxicity (ADCC). 223 Regarding antitumor activity, Serova et al have demonstrated that, in colon cancer cells (Colo205), both the AKT and Ras/Raf/MAPK pathways are modulated by ingenol 3-angelate; however, only the MAPK pathway is mediated by PKC signaling. 224 Thus, these findings suggest that ingenol 3-angelate has both antitumor and proinflammatory activities. 205
Recently, Silva et al (2019) addressed the antitumor potential of novel ingenol semi-synthetic esters derived from compounds extracted from the latex of E. tirucalli. Ingenols A, B, and C exhibited dose-dependent cytotoxic effects in 70 commercial human tumor cell lines comprising 15 different tumor types. Ingenol C was the most efficacious for esophageal and pancreatic cancer cell lines. Furthermore, ingenol A revealed effective activities in esophageal, lung, melanoma, and colon cancer cell lines. In contrast, ingenol B was less effective than the other semi-synthetic compounds and demonstrated no significant activity against any cell line. 185 Additionally, the authors described the cytotoxic activity of ingenol C in glioma cell lines. The semi-synthetic compound showed potential for inhibiting colony formation, promoting the overexpression of cell cycle regulatory proteins, and inhibiting PKC activity. 187
Ingenols Acting as Chemotherapeutic Drug Sensitizers
Ingenols can be applied uniquely or in combination with existing chemotherapeutic agents to achieve a synergistic effect. This effect can increase cytotoxicity in target cells, and can have additional effects on the tumor microenvironment and the immune response to tumors. 225 In vitro and in vivo studies have shown that ingenol derivatives can act efficiently, both in monotherapy and with other drugs (eg, JQ1 and vorinostat).70,186,226-228
Gillespie et al (2004) presented the results for treatment of melanoma with PEP005 (Picato
As drug-associated toxicity remains one of the main barriers to the action of chemotherapeutic and chemopreventive drugs at present, the use of natural compounds as adjuvant therapy may reduce such drug-associated toxicities. By sensitizing conventional therapies synergistically, it may be possible to observe better responses to the treatment. 233
Ingenane/Diterpenes Toxicities and Tumor Promotion Effects
Despite the fact that diterpenes have demonstrated several biological activities, some may present toxicity to humans, culminating in acute or chronic impacts in multiple tissues and organs, as well as immunostimulatory effects when combined with chemotherapeutic drugs. 234 These components have also been described as being responsible for caustic, mucosal irritant, proinflammatory, and carcinogenic characteristics, as well as milky latex activities. These severe irritant effects are due to the diterpene esters (mainly esters of phorbol), which activate PKC, an essential enzyme that is responsible for many cellular functions, including uncontrolled cancerous growth. 235 The development of peripheral neurotoxicity is a well-known side effect of paclitaxel chemotherapy, 236 which can be enhanced when administered with other drugs. 237
Regarding the irritations mentioned above, diterpenes from E. pekinensis can cause effects in the skin, oral, and gastrointestinal mucosa of humans, 238 as well as also nephrotoxicity when taken long-term or in great quantities. 239 Furthermore, these toxic diterpenes may act as tumor promoters, when consumed directly or indirectly from animal products. 238
In 1971, Adolf and Hecker showed that the carcinogenic activity in mice due to oil extracted from the seeds of E. lathyris L. resulted from the irritating activity of the esters of ingenol 7-hydroxy-lathyrol and a monoester of ingenol. 240 Previously, Upadhyay et al (1976) evaluated the latex extracted from E. serrata, from which ingenol-3-palmitate originated, which exhibited irritant and carcinogenic activities in an ear and dorsal skin test of NMRI mice. 241
Opferkuch and Hecker (1982) isolated compounds from E. ingens E. Mey. and displayed their potential to cause irritation and promote tumor formation on the skin of mice. 150 Ingenol-3-hexadecanoate was shown to be the compound with the most significant capacity to promote tumors 242 ; however, its activity was not considered reasonable when compared to the positive control (TPA). 16-Angeloyloxy-3-O-(2.4.6-decatrienoyl) ingenol was shown to be a potential irritant, but was less significant as a tumor promoter than ingenol-3-hexadecanoate. 150
Vogg et al (1999) reported potential tumor-promoting diterpene esters in the Euphorbiaceae. They described that the latex extracted from the leaves of the cultivar Euphorbia leuconeura Boiss. and Euphorbia lomii (hybrid E. lomii Gabi) showed high inducing activity, where only the fractions of the latex that contained ingenol showed tumor-promoting capacity. 243 Ingenol-3,20-dibenzoate extracted from E. leuconeura showed significant tumor-promoting activity in Raji cells. 244
Diterpenes extracted from the aerial part of Euphorbia nubica N.E.Br. and E. helioscopy showed irritation and promoting-tumor potential when evaluated in vivo by Zayed et al (2001). 162 The irritant activity promoted by tigliane-type diterpene esters derived from E. nubica presented moderate activity. Ingenol 3-O-(24,6-decatrienoyl) and 3-O-angeloyl-20-deoxyingenol derived from E. helioscopy extracts were the most active compounds that caused irritation and had the potential for tumor-promoting activity. 153
In the investigations by Baloch et al (2008), seven diterpene compounds from an Euphorbia cauducifolia extract were obtained and tested as possible tumor-promoters. These compounds were 17-[(2Z,4E,6Z)-deca-24,6-trienoyloxy] ingenol, 3-O-angeloyl-17-[(2Z,4E,6Z)-deca-24,6-trienoyloxy] ingenol, 3-O-acetyl-20-O-angeloyl-17-hydroxyingenol, 17-(acetyloxy)-3-O-angeloyl-ingenol, 20-O-acetyl-3-O-angeloyl-17-hydroxyingenol, 3-O-angeloyl-17-(benzoyloxy) ingenol, and 20-O-acetyl-3-O-angeloyl-17-(benzoyloxy) ingenol. When evaluated on the back skin of NMRI mice, these compounds all resulted in high toxic activity. All compounds were considered to be tumor promoters, although only with weak activity. 86
In the study conducted by Tsai et al (2016), the authors described that diterpenes derived from plants belonging to the Euphorbiaceae family, phorbol esters 12-myristate 13-acetate (TPA) and 16-angeloyloxy-13α-isobutanoyloxy-20-acetoxy-4β, and 9α-dihydroxytiglia-1,6-dien-3-one, are inducers of PKCs, showing tumor-promoting and platelet aggregation properties. 245
These reports of toxicity and tumor-promoting activities in natural products/derivatives showing potential for anticancer treatment suggest that precautions should be taken, including assessing the best fit between possible adverse effects and antitumor efficacy, in terms of clinical practice. 233
Clinical Trials in Diterpene-Based Therapy
The use of alternative medicines has increased in recent years. Besides being used as alternatives, the use of natural-origin medicines is also common as complementary medicine in cancer treatment, as well as to improve the side-effects of conventional therapies. They have also been used in patients with either progressive disease or those who have undergone several treatments without a positive response. Moreover, the use of natural compounds in clinical trials has also expanded.
According to the official clinical trials website [https://www.clinicaltrials.gov/], up to December 2019, about 540 natural compound studies have been presented. This ever-growing number is due to the fact that researchers have increasingly focused their efforts on the potential use of natural products in the treatment of cancer. However, in order to be used by patients, more clinical evidence of the pharmacokinetic parameters of these products is necessary.
At present, clinical trials related to cancer and diterpenes can be found, such as one assessing the treatment of acute myeloid leukemia using the Minnelide drug; however, this study was withdrawn. Minnelide (Minne = Minnesota and -lide = triptolide) has been shown to be exceptionally useful in pancreatic and liver cancer, 246 after synthesis (at the University of Minnesota) by adding a phosphate ester group, which generated a chemically unstable compound, due to its limited in vivo solubility.
Clinical studies have evaluated the effect of a supplement based on diterpenes from Rosmarinus officinalis L. and alkylglycerols targeting the immune system and related disorders.247,248 On the other hand, studies using bevacizumab, a humanized monoclonal antibody known as Avastin®, and TPI287, a synthetic derivative of the taxane diterpenoid (abeo-taxane) drugs, for glioblastoma treatment were performed. 249 However, these studies were also canceled.
Clinical trials from natural products can present several difficulties, mainly in relation to commercial availability, structural complexity of the compounds, and the fact that the potency, selectivity, and pharmacokinetic characteristics are not optimized for the clinic. 250
With regard to diterpenes, the limited number of clinical trials can also be related to previous studies that report the expressed toxicity of the compounds. Structural modifications through biotransformation using enzymes or by the chemical route can produce relevant compounds for clinical use. 238
In silico approaches and use of bioengineering to explore the potential of ingenane-based compounds
The characterization of ingenol-based compounds from synthetic or natural sources may boost the target identification, hit discovery, and lead optimization for clinical practice through in silico approaches. Computational methods have been applied extensively to accelerate drug discovery and to reduce drug development costs in both academia and industry, mainly due to the continuous increase in the amount of molecular data and public chemical databases, such as PubChem 251 and ChEMBL. 252
Target identification is an essential step in drug development, for which several computational target prediction strategies have been developed, using different methods, such as traditional ligand- and structure-based applications or machine learning models. 253 Moreover, virtual screening techniques allow for the identification of new hit compounds for a specified target using large chemical libraries. 254 However, classical structural-based virtual screening requires the identified target's three-dimensional structure, while ligand-based screening relies on significant sets of ligands–target interactions. Along with several chemoinformatic tools, computational information can lead to insights into the rational drug design of ingenol derivatives.
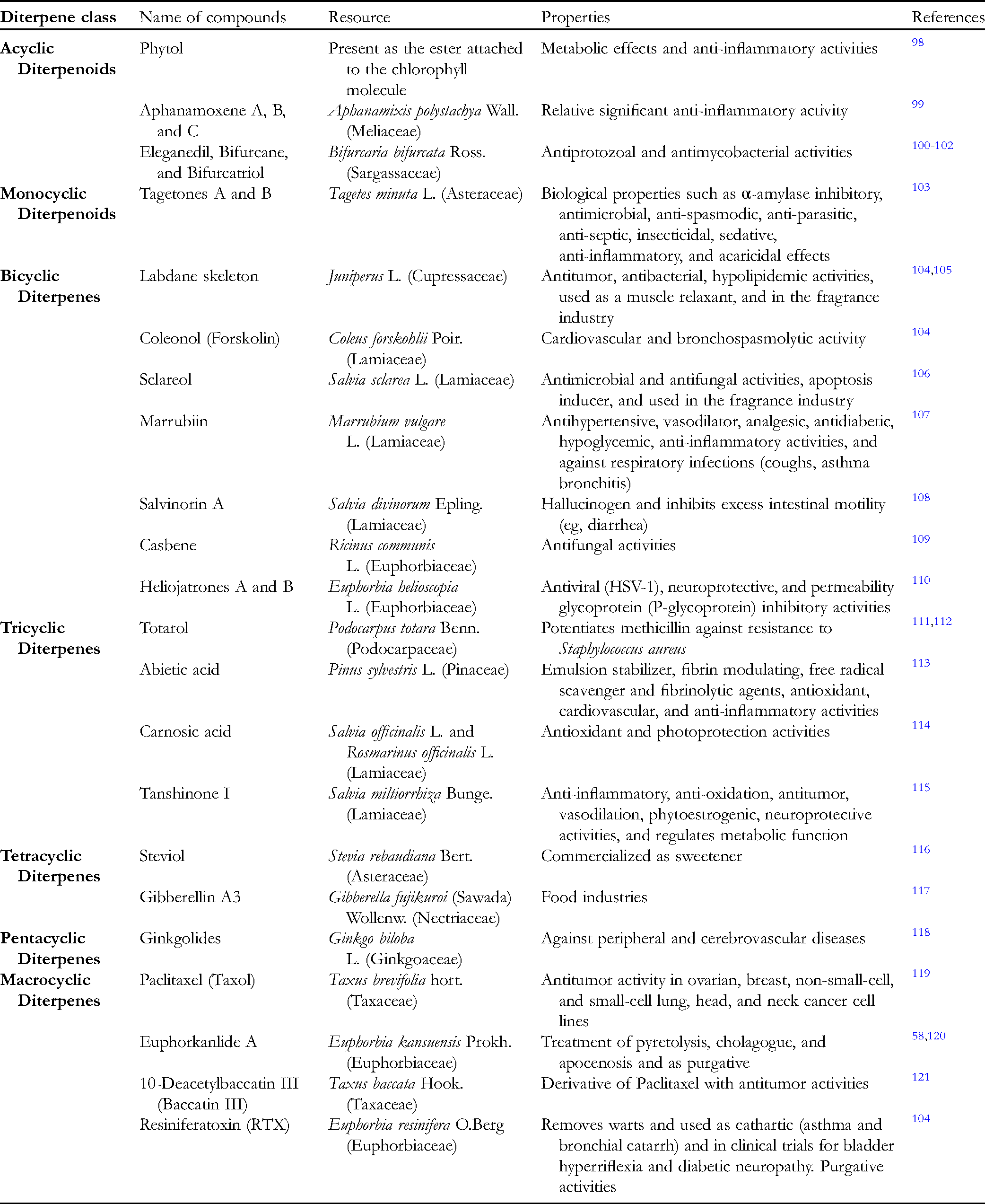
The ingenol structure (Figure 2A) was submitted to a sub-structure search in Clarivate Analytics Integrity (accession date: Jun 20, 2020), a commercial database for drug discovery that integrates biological, chemical, and pharmacological data. This search resulted in 57 small molecule compounds, as presented in Table 3 and detailed in Table S1. The selected compounds, of which 13 were indicated as natural products, have a molecular weight ranging from 430.5 to 726.9 Daltons, including one phase III compound (ingenol disoxate), and three at the pre-clinical stage (ingenol-3-hexanoate, GSK-445A, and ingenol 3,20-dibenzoate). Clarivate Analytics Integrity classified 13 compounds as protein kinase C activators, although 42 compounds were assigned with undesirable molecular mechanisms. Regarding the cellular mechanisms, 42 were described as not specified, 11 as signal transduction modulators, four as HIV latency-reversing agents, three as apoptosis inducers, two as angiogenesis inhibitors, and one as an antimitotic drug.
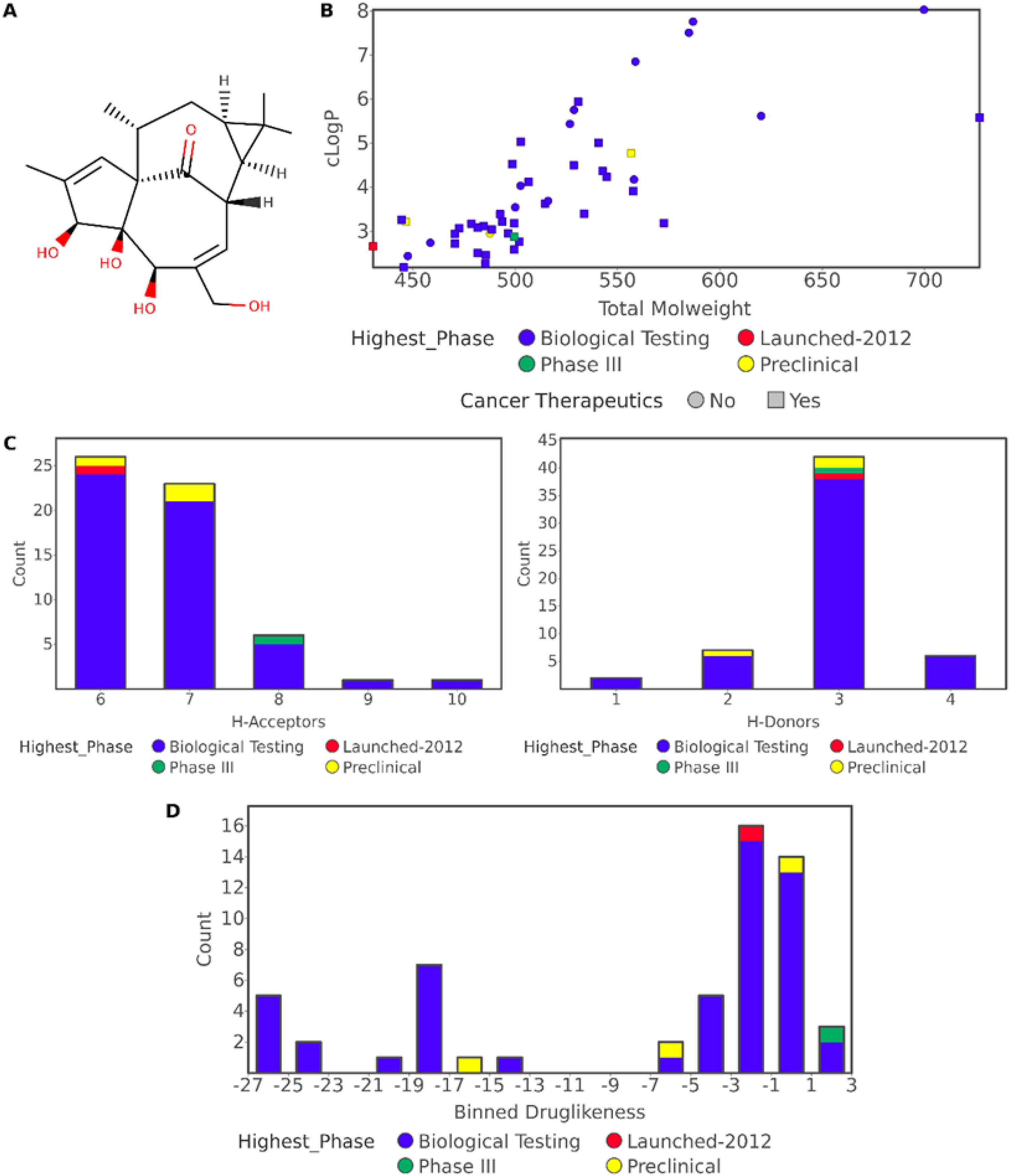
Molecular properties of compounds from ingenol sub-structure search in clarivate analytics integrity (accession date: Oct 20, 2020):
Summary of Small Molecule Compounds Retrieved from Substructure Search of Ingenol in Clarivate Analytics Integrity. The Structure of Current Drugs, Molecular, Cellular Mechanism, and Therapeutic Group of 57 Small Molecules are Reported.
Chemical structures were generated from Integrity sdf files with molconvert, version 20.10.0 (ChemAxon Ltd).
At least one drug name was retrieved from the Integrity database, if available.
The molecular properties of the 57 ingenol derivatives were calculated using DataWarrior, 255 a cheminformatics software for data visualization and analysis (Figure 2 and Table S2). According to Lipinski's rule of five, orally administered drugs are more likely to have low absorption or permeation if they present a molecular weight of more than 500 Daltons, a cLogP over 5, more than 10 hydrogen-bond acceptors, or more than five hydrogen-bond donors. 256 In practical applications for drug discovery, compounds usually should not violate more than one of Lipinski's rules of five. However, natural products may account for most of the favorable exceptions to this rule. 257 As shown in Figure 2B, most of the compounds have a cLogP below 5.0, while around 54% exhibit molecular weight below 500 Daltons. No violations of Lipinski's rules regarding the number of hydrogen donors or acceptors were identified for ingenol derivatives (Figure 2C).
Additionally, the fragment-based drug-likeness, for which traded drugs tend to have more positive values, was calculated, as shown in Figure 2D and Table S2. In this analysis, we observed a group of around 70% of compounds with higher drug-likeness, encompassing Picato®, ingenol disoxate, ingenol 3,20-dibenzoate, and GSK-445A. These molecular property data reinforce the potential of ingenol-based compounds for therapeutic applications.
The discovery of a novel and effective drug, once identified, has scientific and technical hurdles to overcome as establishing and isolating the best targets can be very challenging. 258 The association of the suggested target to the behavior of the clinical disease, and the consequences of this modulation, must be studied, to deliver an appropriate biological and therapeutic effect in applicable experimental models.
Hanson et al 238,259,260 described that biotransformation processes use biological systems to promote chemical transformations from unnatural substrates. In this sense, the biotransformation of diterpenoids through the metabolic engineering of relevant biosynthetic pathways plays a vital role in drug discovery and has attracted increased attention in recent years, becoming a viable alternative strategy. Inexpensive sequencing technologies, such as the available high-throughput transcriptomics and metabolomics technologies, have made this approach increasingly easy.261,262
The study of biotransformation processes applying in silico approaches is a potential way to analyze molecules and the metabolism of medicines in a computational manner. 263 These computational methods are favorable for the investigation of metabolic pathways, and for obtaining new chemical structures and active compounds.
The intensified availability of sequenced genomes in the Euphorbiaceae family, enhanced bioinformatics pipelines for genome assembly, and the evidently conserved clustering of macrocyclic diterpenoid pathways in these species is likely to facilitate novel pathway discoveries, making this platform a reality. 263 The combination of the analysis of metabolite profiling and transcriptome of E. lathyris seeds, which are rich in ingenanes, has been performed by Luo et al, in 2007, to investigate better the associated biosynthetic pathways. 264
Diterpenes are a potential class for biotransformation, due to their extensive biological activities, an important factor in the process of obtaining new derivatives. 238 As examples of biotransformation with ingenols, 16-hydroxy-ingenol-3-angelate originated through hydroxylation of the methyl group at C-16, a deacylated product was formed as a metabolite of ingenol, and an acyl rearrangement of C-17 led to the development of ingenol-5-angelate. 265
Conclusions and Future Research
The active bioconstituents derived from popular medicinal plants have been commonly exploited in the pharmaceutical industry, as active principles for the development of new medicines. Diterpenes are a class of taxonomic markers of the Euphorbiaceae family, representing small molecule compounds with multi-target therapeutic biological activities. Thereby, structural diversity investigations considering diterpenoids from Euphorbia species are considered very promising. At present, approximately 100 Euphorbia species have been explored so far, which is less than 5% of the species belonging to this genus. Moreover, this percentage is further lowered when the target is restricted to the ingenane-type diterpenoids.
The only clinical data on ingenol derivatives are those of I3A, introduced into the pharmaceutical market as a gel for managing actinic keratosis. Despite being withdrawn due to risks that outweigh benefits, it has been a considerable time since a natural product without structural modification has been introduced into clinical practice.
Generally, the active constituents of natural products, such as diterpenoids, are obtained mainly from plants. Although plant biomass recovery is limited due to taxonomic distribution and depressed availability, techniques that allow for the complete chemical synthesis of these molecules despite being laborious, due to the great structural complexity of the members of the diterpene class, are efficient. In this sense, the biomanufacturing of diterpenoids by metabolic engineering has been widely explored. 128
Virtual screening or in silico simulations have gained prominence in the development of new drugs for use in clinical practice. Using these computational approaches better allows for the determination of compounds that exhibit several physiological activities, thus reducing the associated time and cost. In silico approaches to study biotransformation makes it possible to investigate metabolic pathways and obtain new chemical structures and active compounds. 12
Once identified, these new bioactive molecules require extensive in vitro and in vivo studies to verify their effectiveness in biological models. These tests are essential to validate and provide basic knowledge about the pharmacodynamics of the newly discovered drug. In addition, they are significant in facilitating comprehension of the selectivity of the compound and its possible interactions with the therapeutic target.
Considering the results presented in this review, Euphorbia spp. plants have shown great potential as sources of bioactive extracts and compounds, such as ingenane-type diterpenes. Understanding both the biological activities and the roles of these compounds in nature and their optimization through computational methods may accelerate the development of safe and effective new drugs for clinical use.
Supplemental Material
sj-rar-1-npx-10.1177_1934578X221105691 - Supplemental material for The Role of Ingenane Diterpenes in Cancer Therapy: From Bioactive Secondary Compounds to Small Molecules
Supplemental material, sj-rar-1-npx-10.1177_1934578X221105691 for The Role of Ingenane Diterpenes in Cancer Therapy: From Bioactive Secondary Compounds to Small Molecules by Ana Laura V. Alves, Luciane S. da Silva, Camila A. Faleiros, Viviane A. O. Silva and Rui M. Reis in Natural Product Communications
Footnotes
Acknowledgments
This study was supported by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) (2019/05142-4—A.L.VA.) and (2019/23245-5—C.A.F.), Public Ministry of Labor Campinas (Research, Prevention and Education of Occupational Cancer)—L.S.S, CAPES and Barretos Cancer Hospital, all from Brazil. We thank Caio Fernando Oliveira for the assistance in using the Biorender platform and CAPES to access Integrity through the periodic CAPES portal.
Author Contributions
A.L.V.A.—conception or design of the work, writing—original draft preparation, review and editing, and final approval; L.S.S.—conception or design of the work, writing—original draft preparation, review and editing, and final approval; C.A.F.—conception or design of the work and final approval; V.A.O.S.—conception or design of the work, critical review, and final approval; Rui M. Reis (corresponding author)—conception or design of the work, critical review, and final approval.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hospital de Câncer de Barretos, Fundação de Amparo à Pesquisa do Estado de São Paulo, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, and Public Ministry of Labor Campinas (grant number 2019/05142-4, 2019/23245-5).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
