Abstract
Objectives
Diabetic nephropathy (DN), a severe microvascular complication of diabetes mellitus, is a leading cause of end-stage renal disease. Crocin (CRO), an active ingredient extracted from Crocus sativus and Gardenia jasminoides, has multiple bioactivities such as anti-oxidative, anti-inflammatory, anti-tumor, and anti-depressive activities. However, the potential effects and mechanisms of CRO in the treatment of DN are still unclear.
Methods
In this study, we aimed to assess the efficacy of CRO in treating DN using in vivo and in vitro experiments, and intensively investigate the potential therapeutic mechanisms of CRO against DN based on the inhibition of epithelial-mesenchymal transition (EMT) by inducing adenosine monophosphate-activated protein kinase (AMPK)/mammalian target of rapamycin (mTOR)-mediated autophagy.
Results
The results showed that CRO had a therapeutic effect and anti-EMT effect in kidney of DN mice. CRO also moderated AMPK/mTOR pathway and improved autophagy in kidney of DN mice. In high glucose (HG)-induced tubular epithelial cell EMT model, CRO inhibited EMT, moderated AMPK/mTOR pathway and improved autophagy. AMPK inhibitor abolished the above effects of CRO on tubular epithelial cells.
Conclusion
CRO exhibited considerably therapeutic and anti-EMT effects on DN both in vivo and in vitro, these may be associated with restoring autophagy through regulating AMPK/mTOR pathway.
Keywords
Introduction
Diabetic nephropathy (DN), a severe microvascular complication of diabetes mellitus, is a leading cause of chronic kidney disease and end-stage renal disease (ESRD), representing a major cause of death among patients with diabetes mellitus worldwide.1–3 To date, there is no consensus concerning the pathogenesis of DN. Generally, DN is associated with renal microangiopathy, and is influenced by inflammation, oxidative stress, renal hemodynamic changes, metabolic disorders, genetic factors, and aberrant autophagy. 4 Therefore, exploring the potential molecular mechanisms involved in DN and seeking novel therapeutic strategies are of far-reaching significance.
Glomerulosclerosis and renal tubulointerstitial fibrosis are critical features of DN development and progression.5–7 Specifically, renal tubulointerstitial fibrosis is a crucial pathological change observed in progressive DN, and the trans-differentiation of renal tubular epithelial cells into myofibroblasts, known as epithelial-mesenchymal transition (EMT), is a marker of renal tubulointerstitial fibrosis. 8 During EMT, epithelial cells lose their cell polarity and adhesion capacity, gain migratory and invasive properties to become mesenchymal cells, and secrete a substantial quantity of extracellular matrix, resulting in the down-regulation of epithelial surface markers like E-cadherin and the up-regulation of mesenchymal markers like vimentin and α-smooth muscle actin.9,10 Natural products such as curcumin, 11 astragaloside IV, 12 and berberine 13 have been proven to improve DN by inhibiting EMT.
Crocin (CRO), an active compound extracted from Crocus sativus and Gardenia jasminoides, 14 has multiple bioactivities such as anti-oxidative, anti-inflammatory, anti-tumor, and anti-depressive activities. Thus, it is extensively applied in the traditional medicine and food industries. 15 Studies suggest that CRO demonstrates potential in treating diabetes mellitus. 16 Experimental findings have revealed that CRO can improve mitochondrial dysfunction and alleviate endothelial dysfunction in patients with diabetes mellitus. 17 CRO can regulate a variety of biological pathways, including insulin signaling pathways, glucose metabolism and oxidative stress.18,19 Additionally, a controlled clinical research trial found that CRO supplementation is conducive to reducing oxidative stress and inflammatory markers in patients with type 2 diabetes mellitus (T2DM). 20 Although the above studies indicate that CRO can improve diabetes mellitus symptoms, further investigations should be performed to determine its potential effect and mechanism in the treatment of DN. In this study, we aimed to assess the efficacy of CRO in treating DN in vivo and in vitro experiments, and intensively investigate the potential therapeutic mechanisms of CRO against DN via the inhibition of EMT by autophagy activated by the 5′ adenosine monophosphate-activated protein kinase (AMPK)/mammalian target of rapamycin (mTOR) pathway.
Methods
Animals and Reagents
A total of 50 specific pathogen-free healthy male C57BL/6 mice weighing 20–22 g (laboratory animal production license number: SCXK [Beijing] 2019-0008) were purchased from HuaFuKang Bioscience Co. Ltd (Beijing, China). Each cage housed 5 mice and was maintained at a room temperature of 21 ± 2 °C with a relative humidity of 50%–60% and under a 12 h light/12 h dark cycle; the mice were given free access to food and water. See Supplemental materials for details of the materials and reagents employed in this study.
Animal Experiments
This study was approved by Ethics Committee of Hebei University of Chinese Medicine (Approval no. CZX2021-KY-026), and all animal experiments were conducted in compliance with “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health. Refer to previous studies to establish a DN mouse model. 21 Briefly, the mice underwent 1-week adaptive feeding. Afterwards, 40 mice were randomly selected to receive a high-sugar and high-fat diet (HFD) composed with 21% fat, 34% sucrose, 0.15% cholesterol, and 44.85% normal chow for 8 weeks, whereas the remaining 10 mice were assigned to a normal control (NC) group to receive a normal diet. At the end of week 8, all mice underwent fasting for 12 h with unrestricted access to water, and then were intraperitoneally injected with 30 mg/kg streptozotocin (STZ) except for mice in the NC group, who were intraperitoneally injected with an equivalent volume of 1% sodium citrate buffer. After 72 h, blood samples were collected from the caudal vein to determine random blood glucose levels, with a blood glucose level of ≥16.7 mmol/L considered as the modeling criterion of T2DM. Then, the mice continued being fed and were tested weekly for 24 h urinary protein content, with the random blood glucose level of ≥16.7 mmol/L and 24 h urinary protein content of ≥20 mg considered as the modeling criteria for DN.
After modeling, 40 DN mice were randomly divided into DN, irbesartan (IRB), low-dose CRO (CRO-L), and high-dose CRO (CRO-H) groups, with 10 mice assigned to each group. The mice in NC and DN groups received 0.2 mL of normal saline via oral gavage, mice in IRB group received IRB for 0.2 g/kg/d via gavage, and mice in CRO-L and CRO-H groups received CRO for 10 and 20 mg/kg/d via gavage, respectively. The duration of administration in each group was 4 consecutive weeks, and fasting blood glucose (FBG) levels and body weight of mice in each group were measured weekly. After 4 weeks of treatment, 24 h urinary samples of mice in each group were collected in metabolic cages. In addition, blood samples were collected via the inner canthus. After collecting the blood samples, the mice were euthanized, the abdominal cavity was opened; the left kidney was collected and immobilized in 4% polyoxymethylene, whereas the right kidney was frozen and stored.
Biochemical Analysis of Renal Function
The obtained 24 h urinary samples were centrifuged at 4 000 × g for 10 min, and then the supernatant was collected to detect the 24 h urinary total protein (UTP) content according to the kit instructions. The collected blood samples were centrifuged at 400 × g for 15 min to collect serum, and creatinine (Cr) and blood urea nitrogen (BUN) levels in mouse serum in each group were detected according to the kit instructions. After reaction with relevant reagents, the optical absorbance of each reaction well was determined using a microplate reader at the corresponding wavelength for quantitative analysis.
Pathological Staining Analysis
For histopathological assessment of renal function and fibrosis, the renal tissues that had been immobilized for 48 h were cut into 5 μm sections. After dewaxing and rehydration, hematoxylin and eosin (H&E) staining, periodic acid–Schiff (PAS) staining, Masson's trichrome staining, and Sirius Red staining were performed, and the stained renal slices were observed and analyzed using an optical microscope. Based on previous studies, we used kidney damage score to evaluate HE staining 22 and Glomerulosclerosis index to evaluate PAS staining. 23 We also employed Image Pro Plus to quantify the area of collagen fibers in MASSON staining and Sirius red staining.
Cell-Based Experiments
Human renal tubular epithelial cells (Human kidney 2, HK-2), purchased from iCell Bioscience Inc. (Shanghai, China), were cultured in Defined K-SFM supplemented with growth factors in an atmosphere of 5% CO2 at a temperature of 37 °C and a humidity level of 98%. The cells were subcultured in a 1:3 ratio when they reached 80% confluent.
For CRO cytotoxicity tests, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was performed. In brief, HK-2 cells were seeded into 96-well plates at a density of 2 × 104 cells/well, incubated for 24 h, and then treated with CRO at various concentrations (0, 1, 10, 50, and 100 μM) for 24 h. After the incubation, 10 μL of 5 mg/mL MTT solution was added to each well. After an additional 4-h incubation, the medium was gently removed, and 100 μL of dimethyl sulfoxide was added. The optical absorbance of 96-well plates was measured using a microplate reader at a wavelength of 550 nm.
To investigate the protective effect of CRO toward high glucose (HG)-challenged HK-2 cells, HK-2 cells were cultured in normal D-glucose medium (NC, 5.5 mM) and high D-glucose medium (HG, 30 mM) for 48 h.13,24 When HK-2 cells were challenged by HG, CRO was added to the medium at the concentrations of 1, 10, and 50 μM, respectively at the same time. To validate the efficacy of CRO in inhibiting EMT by regulating autophagy activated via the AMPK/mTOR pathway, CRO, 20 nM rapamycin (RAPA),25,26 or CRO + 5 μM dorsomorphin (CRO + DM)25,26 were added to the medium while HK-2 cells were challenged by HG. The NC group was treated with vehicle. After the 24-h incubation, the cells in each group underwent subsequent tests immediately after being collected using a cell scraper.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR)
The procedures were performed as previously described. 27 Total RNA was extracted from renal tissues using Trizol reagent, followed by reverse transcription into cDNA according to the kit instructions. Gene expression was analyzed using a real-time PCR detection system and SuperReal PreMix Plus kit. At the mRNA level, the expression quantity of target genes relative to the housekeeping gene Actb was calculated by the 2−ΔΔCT method. See Supplemental materials for primer sequences.
Western Blot
Total proteins in renal tissues or cells were extracted using radioimmunoprecipitation assay buffer. After protein concentrations were determined, the samples were homogenized and protein loading buffer was added and mixed well. Next, the proteins samples were denatured in a water bath at 95 °C for 5 min, and then underwent sodium dodecyl-sulfate polyacrylamide gel electrophoresis for further analysis. Proteins were transferred onto polyvinylidene fluoride membranes. After 1 h blocking in 10% skim milk at room temperature, the membrane was incubated with primary antibodies (VIM, 1:1000; CDH1, 1:1000; tight junction protein 1 (TJP1), 1:1000; α-SMA, 1:2000; AMPK, 1:2000; p-AMPK, 1:2000; mTOR, 1:5000; p-mTOR: 1:2000; LC3, 1:2000; P62, 1:20 000; Beclin-1, 1:2000; β-actin, 1:5000) overnight at 4 °C, washed in Tris-buffered saline with Tween 20 three times and incubated with secondary antibodies at room temperature for 1 h. Then, enhanced chemiluminescence developer was added, and the membrane was developed and exposed to a chemiluminescence detection system to reveal immunoreactive western blot bands. Relative protein levels were quantified using ImageJ software.
Statistical Methods
Statistical analyses were performed on data using SPSS Statistics 17.0. All data were expressed as means ± standard deviation, and inter-group differences were analyzed by one-way analysis of variance. Then, Tukey's honestly significant difference test was conducted, and P < .05 was considered statistically significant.
Results
Therapeutic Effect of CRO in DN Mice
Figure 1a and b showed that CRO markedly affected body weights and FBG levels of DN mice. The DN group exhibited an obviously decreased body weight compared with the NC group, and IRB, CRO-L, and CRO-H treatments notably delayed this weight loss. Additionally, hyperglycemia in DN group was effectively controlled after treatment with CRO. The renal function test results revealed that 24 h UTP levels and serum Cr and BUN levels in DN group were noticeably higher than those in the NC group, indicating continuous and severe renal damage (Figure 1c-e). Noteworthily, IRB or CRO exerted renal protective effects; specifically, CRO-H exerted better protective effect than CRO-L.

CRO had a therapeutic effect on DN mice.
H&E and PAS staining results indicated that kidneys in DN group mice underwent severe pathological changes, including deformed glomeruli, balloon cavities with varying widths, increased and unevenly distributed mesangial matrices, and thickened glomerular basement membranes (Figure 1f-i). Additionally, Masson's trichrome staining and Sirius Red staining results suggested that collagen were deposited in renal tissues, further confirming a severe increase in the number of matrices and severe fibrosis involving the kidneys (Figure 1j-m).
Effect of CRO on EMT in Kidney
The occurrence of EMT in renal tissues was evaluated by detecting the levels of epithelial cell markers, cadherin 1 (CDH1) and TJP1, and interstitial cell markers, vimentin (VIM) and alpha-smooth muscle actin (α-SMA), in kidney of mice. PCR results demonstrated that compared with the NC group, DN mice showed decreased mRNA levels of Cdh1 and Tjp1 and markedly elevated mRNA levels of Vim and alpha-smooth muscle actin (Acta2) in kidneys (Figure 2a-d). Additionally, western blot results revealed that CDH1 and TJP1 levels remarkably decreased, whereas VIM and α-SMA levels noticeably increased (Figure 2e-i). The above results confirmed that EMT occurred in the kidneys of DN mice. After treatment with IRB or CRO, the changed EMT markers in the kidney and their corresponding mRNA levels were moderated to varying degrees. Specifically, the CRO-H group exhibited better improvement effects than CRO-L, and no differences were identified between IRB and CRO-H groups.

CRO inhibited EMT in kidney of DN mice.
Effects of CRO on Autophagy and AMPK/mTOR Pathway in Kidney
Autophagy, a highly conserved self-protection mechanism responsible for removal of damaged proteins and organelles in cells to avoid cell apoptosis and necrosis, is vital for maintaining cellular homeostasis. 28 Studies have suggested that when the body functions normally, renal glomerular epithelial cells and proximal renal tubular epithelial cells are at a high level of autophagy to effectively remove damaged proteins and organelles from cells; however, a persistent HG state in DN mice leads to inhibition of autophagy and prevention of the timely removal of damaged proteins and cytotoxins in cells, thereby resulting in the occurrence of EMT. 29 On this basis, we detected levels of autophagy-related proteins LC3, p62, and Beclin-1 in renal tissues using western blot and found that levels of the autophagy-related proteins in the DN group were markedly lower than those in the NC group, confirming previous conjecture that autophagy was inhibited in DN mouse kidneys (Figure 3a-d). After treatment with IRB or CRO, LC3 and Beclin-1 levels increased significantly, whereas p62 levels decreased considerably, proving that autophagy was restored. Specifically, the CRO-H group was markedly superior to the CRO-L group with respect to the improvement effect, and no differences were present between the CRO-H and IRB groups.
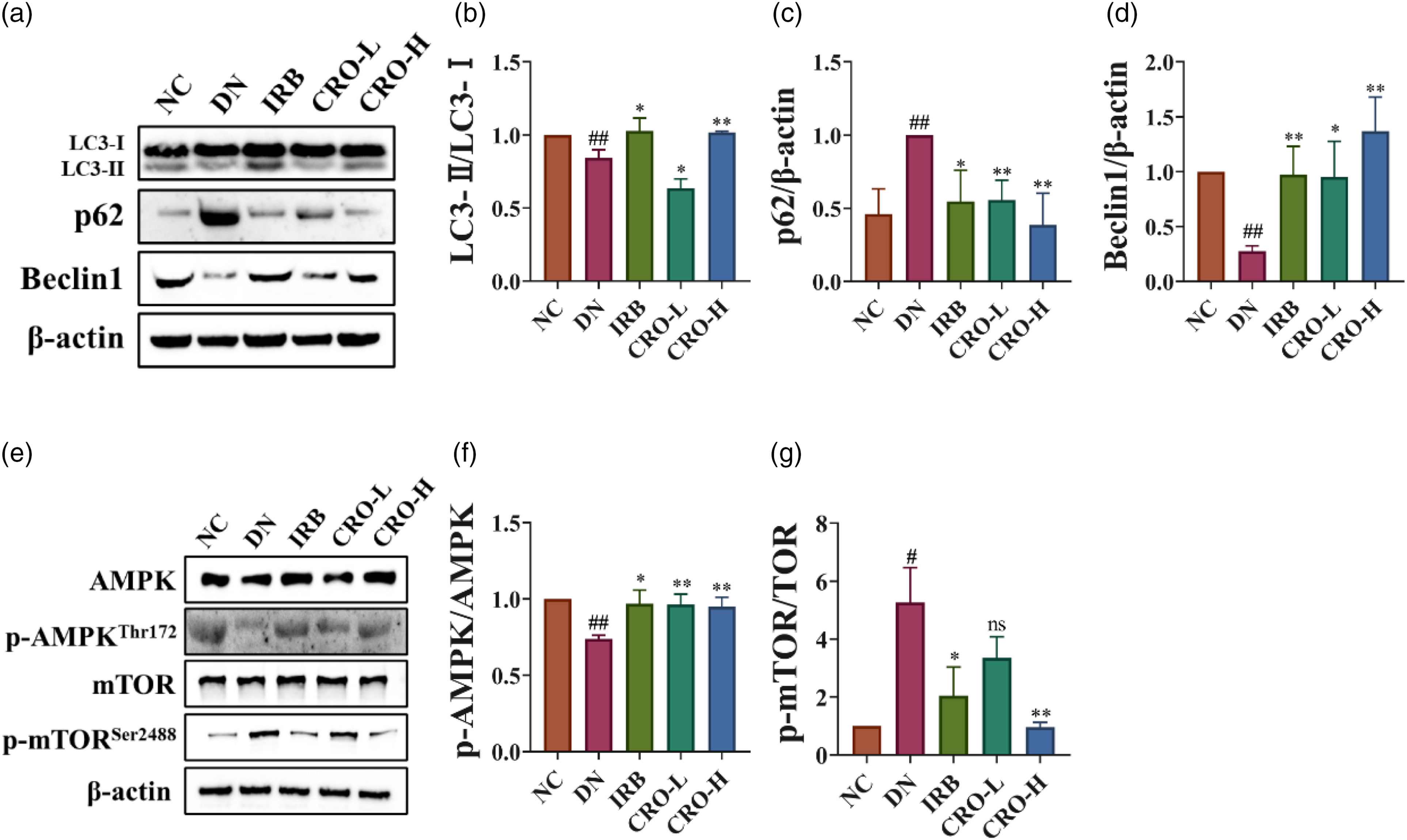
CRO improved autophagy and moderated AMPK/mTOR pathway in kidney of DN mice.
AMPK/mTOR pathway, the classical regulation of autophagy, can function to restore impaired autophagy, reduce cell injuries, and delay the progression of DN upon activation. 30 Studies revealed that AMPK can promote the expression of autophagy-related proteins, LC3-II and Beclin-1, and autophagy formation by inhibiting the activation of the mTOR pathway, to protect diabetes mellitus patients from renal impairment. 31 Western blot results revealed that compared with the NC group, DN mice showed decreased AMPK phosphorylation levels in renal tissues and elevated mTOR phosphorylation levels; after treatment with IRB or CRO, the phosphorylation levels of AMPK and mTOR were increased and decreased, respectively (Figure 3e-g).
Effects of CRO on EMT of HG-Challenged HK-2 Cells
The cytotoxicity of CRO against HK-2 cells was detected using the MTT assay, and the results indicated that CRO at concentrations of 1, 10, 50, and 100 μM did not obviously affect the activity of HK-2 cells (Figure 4a). Therefore, subsequent experiments were conducted using CRO at concentrations of 1, 10, and 50 μM to investigate the effects of CRO on EMT.

CRO inhibited EMT in HG challenged HK-2 cells.
A previous study indicated that HG can induce EMT in HK-2 cells, and this model is extensively applied to investigate EMT in patients with DN. 13 Therefore, we employed HG to induce EMT in HK-2 cells while performing interventions using CRO at varying concentrations and assessed the effects of CRO on EMT by detecting levels of CDH1 and TJP1 as well as VIM and α-SMA in each group. PCR results demonstrated that compared to those in the NC group, the mRNA levels of CDH1 and TJP1 decreased, while the mRNA levels of VIM and ACTA2 increased significantly in HG-challenged HK-2 cells (Figure 4b-e). Additionally, western blot results revealed that CDH1 and TJP1 levels were remarkably decreased, while VIM and α-SMA levels noticeably increased (Figure 4f-j). The above results confirmed that EMT occurred in HG-challenged HK-2 cells. After treatment with CRO, the changed EMT markers and their corresponding mRNA levels improved to varying degrees. Specifically, the 50 μM concentration group showed the most significant improvement effect.
Effects of CRO on Autophagy and AMPK/mTOR Pathway in HG-Challenged HK-2 Cells
Phosphorylation levels of AMPK and mTOR in each group were detected by western blot and the results showed that compared to those in the NC group, LC3 and Beclin-1 levels decreased remarkably and p62 levels increased in HG-challenged HK-2 cells, suggesting that HG inhibited autophagy of HK-2 cells (Figure 5a-d). Additionally, phosphorylation levels of AMPK and mTOR decreased and increased in HG-challenged HK-2 cells, respectively (Figure 5e-g). The above results were identical to the in vivo study results. After treatment with CRO, LC3 and Beclin-1 levels increased, while p62 levels decreased in HK-2 cells, proving that autophagy was restored. Furthermore, phosphorylation levels of AMPK and mTOR were corrected. Specifically, the 50 μM concentration group showed the most significant improvement effect. Therefore, we selected this concentration as the validation test concentration.

CRO moderated AMPK/mTOR pathway and improved autophagy in HG challenged HK-2 cells.
DM Eliminated the Protective Effect of CRO in HG-Challenged HK-2 Cells
To validate the mechanism of CRO whereby CRO inhibits EMT by regulating autophagy activated via the AMPK/mTOR pathway, CRO, RAPA (an mTOR inhibitor that can activate autophagy), and CRO + DM (DM is an AMPK inhibitor) were administered when HK-2 cells were challenged by HG. The results indicated that CRO and RAPA were not significantly different in terms of their effects on EMT, and DM eliminated the inhibitory effect of CRO on EMT in HG-challenged HK-2 cells. Compared with CRO groups, the CRO + DM group showed decreased CDH1 and TJP1 mRNA levels and notably elevated mRNA levels of VIM and ACTA2 (Figure 6). The detection results of AMPK/mTOR pathway- and autophagy-related proteins also proved that CRO and RAPA were not significantly different with respect to their effects on AMPK/mTOR and autophagy (Figure 7). Furthermore, DM eliminated the effect of CRO on restoring autophagy of HG-challenged HK-2 cells. Compared with CRO groups, the CRO + DM group exhibited decreased AMPK phosphorylation levels, elevated mTOR phosphorylation levels, significantly decreased LC3 and Beclin-1 levels, and increased p62 levels.

Dm abolished the effects of CRO on inhibiting EMT in HG challenged HK-2 cells.

Dm abolished the effects of CRO on moderating AMPK/mTOR pathway and improved autophagy in HG challenged HK-2 cells.
Discussion
The classical mouse model of DN induced by HFD combined with STZ, which is highly reliable, can effectively reproduce renal injuries observed in patients with DN. 32 The 24 h UTP levels and serum Cr and BUN levels, which are critical parameters in evaluating renal function, are sensitive biochemical parameters. Damaged renal tissue leads to changes in filtration function, resulting in failure to prevent proteins from entering urine. Therefore, renal damage can be rapidly judged by 24 h UTP levels at an early stage, and serum Cr and BUN levels increase as DN progresses, indicating the degree of renal hypofunction. 33 The progression of DN to ESRD is characterized by progressive scarring of glomeruli and, subsequently, similar fibrosis in the tubulointerstitial region. These can be clearly observed on histopathological staining of renal tissues. 34 Our study findings showed that CRO could remarkably improve impaired renal function and reduce fiber deposition in the kidneys of DN mice, which may help to delay the process of fibrosis. IRB, commonly used to treat DN in clinical practice, can protect the kidneys of patients with diabetes mellitus, independently of its antihypertensive effect. 35 Additionally, the study found no difference between CRO-H and IRB in terms of their efficacy, indicating that CRO is a potential candidate drug to treat patients with DN. In addition, while CRO has demonstrated hypoglycemic effects, numerous studies have shown that, CRO can directly act on cells to improve pathological damage, including its protective effects on HK-2 cells 36 and neurosynaptic protection. 37
The occurrence and progression of DN involve multiple factors like hyperglycemia, inflammation, and oxidative stress. These factors can induce EMT wherein renal tubular and glomerular epithelial cells are transformed into interstitial cells. 38 During EMT, epithelial cells loose certain markers, CDH1 and TJP1, to gain interstitial cell markers, VIM and α-SMA. Such transition can lead to changes to renal structures and function, and promote progression of DN. 39 Interstitial cells produced during EMT can secrete massive amounts of collagen proteins and fibronectins to facilitate the deposition of glomerular basement membranes and increase the degree of renal tubulointerstitial fibrosis.40,41 This eventually results in further deterioration of the renal function and in the occurrence of ESRD in patients with DN. Our study findings demonstrated that CDH1 and TJP1 levels were notably decreased, while VIM and α-SMA levels were markedly increased in HG-challenged HK-2 cells of DN mice. These changes showed improvement to varying degrees after treatment with CRO, suggesting that CRO noticeably improved EMT in kidneys of DN mice. Additionally, IRB has been proven to effectively improve EMT. 42 Our study findings showed that CRO-H and IRB were no different in terms of the improvement effect on EMT, further proving that CRO exhibits high potential in treating patients with DN. In the future, we plan to utilize immunohistochemistry to evaluate the aforementioned indicators, which will help us gain a deeper understanding of the impact of CRO on renal tissue fibrosis.
AMPK, a nutrient-sensing kinase, is activated when energy reserves are depleted, and is a critical positive regulator of autophagy. 43 The phosphorylation and activity of AMPK were inhibited in glomeruli and renal tubules of DN mice, resulting in the inhibition of autophagy and, thus, the occurrence of DN.44,45 mTOR, a nutrient-sensing kinase too, can directly inhibit the activity of the Ulk1 complex after being formed through phosphorylation. Contrary to AMPK, mTOR is a negative regulator of autophagy. 46 It is noteworthy that crosstalk occurs between AMPK and mTORC signals, and AMPK inhibits the activity of mTORC by regulating the levels of other related proteins.47,48 Therefore, the induction of autophagy is subject to an equilibrium between AMPK and mTOR pathways. Our study findings suggested that, in DN mice and HG-challenged HK-2 cells, the phosphorylation level of AMPK decreased whereas that of mTOR increased, and the levels of LC3, p62, and Beclin-1 were decreased, indicating that autophagy was inhibited. After treatment with CRO, phosphorylation levels of AMPK and mTOR improved, and the levels of LC3 and Beclin-1 increased significantly, while p62 levels decreased, signaling that autophagy was restored. Additionally, IRB has been proven to restore autophagy by regulating the AMPK pathway. 49 In this study, IRB and CRO-H had no significant difference with respect to their efficacy in improving AMPK/mTOR pathway activity and levels of autophagy-related proteins, demonstrating that CRO can effectively improve autophagy. In future studies, we plan to observe the changes in autophagosomes through transmission electron microscopy to further evaluate the effect of CRO intervention on cellular autophagy.
To further validate the mechanism of CRO whereby CRO inhibits EMT by promoting autophagy through the AMPK/mTOR pathway, RAPA was selected as a positive control, as it can bind to mTOR to inhibit its activation to promote cell autophagy. 50 The study findings indicated that CRO and RAPA were not different in terms of their effects on improving EMT and regulating the AMPK/mTOR pathway and autophagy in HG-challenged HK-2 cells. In addition, we compared the effects of CRO on autophagy in HG-challenged HK-2 cells and on EMT before and after inhibition of AMPK activity. DM, an AMPK inhibitor, is commonly employed to investigate the AMPK pathway. 51 Our study results revealed that the protective effects of CRO were eliminated after inhibition of AMPK activity, suggesting that the regulatory effects of CRO on autophagy and EMT may be mediated by regulating the AMPK pathway.
The above analysis indicates that crocin could inhibit EMT in renal tubular epithelial cells to treat diabetic nephropathy through improving AMPK/mTOR-mediated autophagy (Figure 8).

Graphical abstract.
Although this study highlights the pivotal roles of AMPK and mTOR pathways in DN, it is crucial to note that these pathways do not operate in isolation within the disease pathology. In fact, they likely interact with other signaling pathways, including those involved in inflammatory responses, oxidative stress, and endoplasmic reticulum stress.52–54 The research in this domain is not fully explored, thus potentially limiting our comprehensive understanding of the complex pathophysiological mechanisms of DN. To overcome this limitation, future research will adopt an integrative and systematic approach to meticulously investigate the interactions between these signaling pathways and their collective impact on the progression of DN. Specifically, given the complexity of intracellular signaling networks, we plan to employ strategies in systems biology to deeply analyze the dynamics and interrelationships among various signaling pathways. We will utilize high-throughput genomic, proteomic, and metabolomic techniques to explore the interactions of CRO with the AMPK and mTOR pathways and other related pathways involved in inflammation, oxidative stress, and cellular stress responses.55,56 This multidimensional analysis will provide a holistic perspective, enhancing our understanding of the roles and complex interactions of these pathways in the pathology of DN. Through this approach, we aim to elucidate the molecular mechanisms and pathway interplays that are crucial for the pathogenesis and therapeutic strategies of DN, thereby laying a more robust scientific foundation for the development of new treatment modalities.
Conclusion
In summary, CRO exhibited considerably therapeutic and anti-EMT effects on DN both in vivo and in vitro, these may be associated with restoring autophagy through regulating AMPK/mTOR pathway.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241286968 - Supplemental material for Crocin Inhibited Epithelial-Mesenchymal Transition in Renal Tubular Epithelial Cells to Treat Diabetic Nephropathy Through Improving AMPK/mTOR-Mediated Autophagy
Supplemental material, sj-docx-1-npx-10.1177_1934578X241286968 for Crocin Inhibited Epithelial-Mesenchymal Transition in Renal Tubular Epithelial Cells to Treat Diabetic Nephropathy Through Improving AMPK/mTOR-Mediated Autophagy by Jinhao Su, Wei Chen, Hui Zhang, Hanzhou Li, Baochao Pan, Ziang Ma, Yuansong Wang, Huantian Cui and Shuquan Lv in Natural Product Communications
Supplemental Material
sj-doc-2-npx-10.1177_1934578X241286968 - Supplemental material for Crocin Inhibited Epithelial-Mesenchymal Transition in Renal Tubular Epithelial Cells to Treat Diabetic Nephropathy Through Improving AMPK/mTOR-Mediated Autophagy
Supplemental material, sj-doc-2-npx-10.1177_1934578X241286968 for Crocin Inhibited Epithelial-Mesenchymal Transition in Renal Tubular Epithelial Cells to Treat Diabetic Nephropathy Through Improving AMPK/mTOR-Mediated Autophagy by Jinhao Su, Wei Chen, Hui Zhang, Hanzhou Li, Baochao Pan, Ziang Ma, Yuansong Wang, Huantian Cui and Shuquan Lv in Natural Product Communications
Footnotes
Author Contributions
Jinhao Su carried out the experiments and manuscript writing. Wei Chen, Hui Zhang, Hanzhou Li, Baochao Pan, and Ziang Ma provided experimental help and performed data analysis and result interpretation. Yuansong Wang provided experimental guidance. Huantian Cui and Shuquan Lv provided ideas and technical guidance for the whole work.
Data Availability
All datasets generated and analysed during this study are included in this published article and its Supplementary Information files. Additional data are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by Ethics Committee of Hebei University of Chinese Medicine (Approval no. CZX2021-KY-026).
Funding
This research was funded by Construction Project of Workshop of Prestigious Chinese Physician in Cangzhou Hospital of Integrated Traditional Chinese Medicine and Western Medicine, grant number 2022-75, 2024 Scientific Research Project Plan of the Hebei Provincial Administration of Traditional Chinese Medicine, project number: 2024458. Wang Yuansong National Renowned Traditional Chinese Medicine Expert Inheritance Studio.
Statement of Human and Animal Rights
All procedures conducted in this study were in accordance with the Institutional Animal Care guidelines of Hebei University of Chinese Medicine, China and approved by Ethics Committee of Hebei University of Chinese Medicine (Approval no. CZX2021-KY-026).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
