Abstract
Cell suspension culture is an attractive alternative source to wild plant for the production of novel biological metabolites. Hence, in this study, chemical investigation of cell suspension cultivated Marchantia polymorpha L. was performed, and led to the isolation of one new bis-bibenzyl (
Introduction
The bryophytes are found all over the world, it was considered as the ancestor of land plants. There are around 23 000 species were reported and divided into 3 phyla including Bryophyta, Marchantiophyta, and Anthoceratophyta. 1 Among those bryophytes, lipophilic metabolites such as terpenoids, polyketides, and aromatic components are the major ones, which exhibited interesting biological activities.2‐9
Marchantia polymorpha, known as a traditional Chinese herbal medicine used in the treatment of cuts, fractures, snake bites, burns, scalds, and open wounds. Phytochemical research declared that the major metabolites in M. polymorpha are polyphenols (bis-bibenzyls and flavonoids), long-chain polyunsaturated fatty acids (AA, EPA), and terpenoids.10‐15 Among which especially the bis-bibenzyls exhibited significant antibacterial, antifungal, anti-inflammatory, and anti-oxidant properties16‐18; AA and EPA are also commonly known for their beneficial roles in human health. 15
Cell suspension culture is an attractive alternative source to wild plants for the production of secondary metabolites,19‐23 it offers a clear production system to make continuous supply, even quality, and stable rates; meanwhile, it is possible to obtain novel components to that are not found in parent plants. In this study, chemical composition, total phenols and total flavonoids contents, and biological activity were investigated. The cell cultivated M. polymorpha was found to produce superior phenol yields and preferable biological activities, which can be explained by the better production of some effective constituents in artificial conditions, it was also supported by the HPLC fingerprint analyses. Furthermore, guided by antioxidant and antibacterial activity, phytochemical research of methanol-soluble fraction of cultivated M. polymorpha was investigated, a totally of 10 bis-bibenzyls derivatives were isolated, and structure elucidated (Figure 1). Herein, details of the cell cultivation, contents determination, chemical isolation, structural identification, and the results of the biological activities screening are described.

Bis-bibenzyl derivatives (
Results and Discussion
Total Phenol and Flavonoid Contents Determination
Phenolics and flavonoids are important metabolites that exhibited antioxidant activity. Consequently, the total phenol contents (TPC) and total flavonoid contents (TFC) values were evaluated and presented. Clearly, a significant difference with respect to their TPC values has been found in methanol and petroleum extracts. As presented in Table 1, the higher TPC value was found in methanol extracts of cell cultivated M. polymorpha (ME, 271.23 ± 2.07 mg GAE/g extract), Followed by the methanol extracts of wild M. polymorpha (WM, 225.45 ± 1.48 mg GAE/g extract). On the other hand, the lowest TPC value was recorded for the petroleum extract cell cultivated M. polymorpha (CP, 11.15 ± 1.13 mg GAE/g extract).
The Content of Total Phenols and Flavonoids in Different Extracts.

Note: Each value is the mean ± SD of 3 independent measurements and 95% confidence intervals; TPC, total phenols content; TFC, total flavonoids contents; GAE, gallic acid equivalents; QE, quercetin equivalents; WP, petroleum extract of wild M. polymorpha; WM, methanol extract of wild M. polymorpha; CP, petroleum extract of cultivated M. polymorpha; CM, methanol extract of cultivated M. polymorpha; ND, not detected.
Furthermore, the total flavonoid contents of crude extracts were also evaluated. However, none of the samples investigated exhibited a high TFC value, with the highest value found in methanol extracts of wild M. polymorpha was around 0.31 ± 0.07 mg QE/g extract. Moreover, no flavonoid metabolites were detected from the crude extract of cultivated M. polymorpha.
Structure Identification
Compound
Four benzylic methylenes, 24 benzene ring carbon atoms, and 1 methyl group were determined based on the 1H and 13C NMR spectrum, indicating that compound
The linkage of rings A and B, rings C and D were determined by the HMBC experiment (Figure 2). Meanwhile, the biphenyl linkage between C-14 and C-14′ was also confirmed based on the HMBC correlations from H-13′ to C-14 (Figure 2). Thus, compound
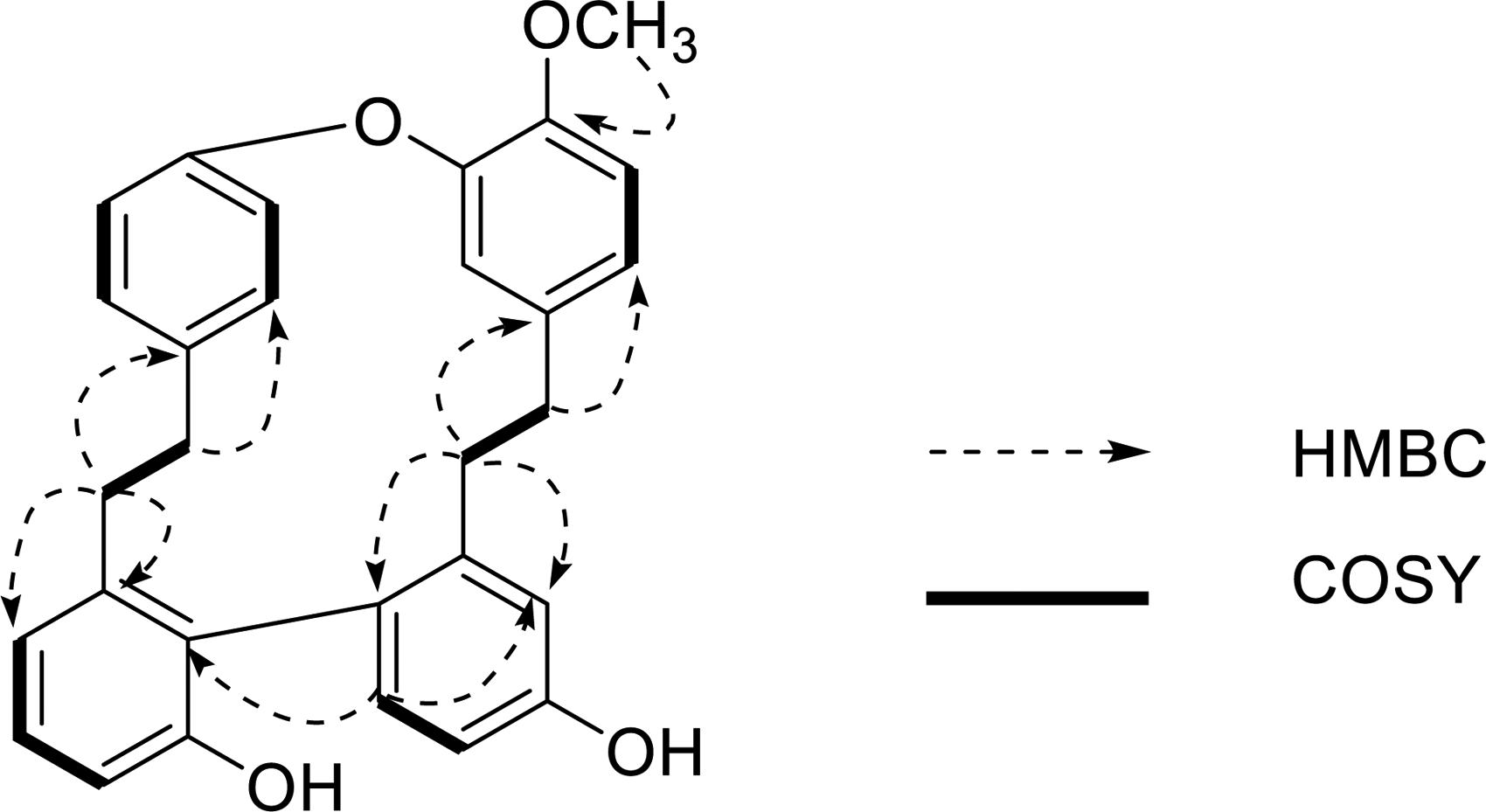
Key HMBC and COSY connections of compound
In addition to the aforementioned new compound, 9 known components including plagiochin G (
Compound
HPLC Fingerprint Analyses of Phenolics in Methanol Extracts between Wild and Cell Cultivated M. polymorpha
To determine the chemical propriety differences between wild and cell cultivated M. polymorpha, methanol extracted was prepared for HPLC analysis, using isolated compounds as reference standards. In HPLC analysis, compounds
Antioxidant Activities
The DPPH and reducing power assays are easy and sensitive methods used in the evaluation of the antioxidative activity. Among which the DPPH assay has been commonly used as a tool for evaluating free radical scavenging activities of antioxidants, the characteristic absorption possessed and measured at 520 nm; while the reductant conversed the iron ion/ferricyanide complex to ferrous ion, and the remained iron ion can be monitored at 700 nm in reducing power assay.
Those results obtained from the DPPH assay are presented in Figure 3, the radical scavenging effects of isolated compounds on DPPH radicals were significant and concentration-dependent. Meanwhile, the reducing power results of tested samples are in agreement with the DPPH assay (Figure 4), all the tested samples exhibited moderate to significant activity. While methanol extracts were the most active ones, which showed comparable antioxidant effects to that of vitamin E in both assays.

Radical scavenging activity (RSA) of tested samples (

Reducing power of tested samples (
Antimicrobial Activities
To screen the antimicrobial activities of crude extract and isolated components, the in vitro antibacterial effect of crude extracts and pure components against human pathogenic bacteria is evaluated. The minimum inhibition concentration (MIC) values were presented in Tables 2 and 3. The methanol extracts exhibited interesting antibacterial activity against Staphylococcus aureus, Bacillus subtilis, and Listeria monocytogenes, as well as weak antibacterial activity against Escherichia coli. It declared that S. aureus and L. monocytogenes were the most sensitive bacterial strains to methanol extract. Alternatively, none of them exhibited an obviously antibacterial effect against Salmonella typhimurium.
Minimal Inhibitory Concentration (MIC, µg/mL) Values of Tested Samples Against Selected Microbial Strains.

Note: Bacterial strains: Staphylococcus aureus; Bacillus subtilis; Listeria monocytogenes; Salmonella typhimurium; and Escherichia coli, Fungal strains: Aspergillusflavus; Aspergillus niger; Candida albicans; Trichophyton mentagrophytes; PE, petroleum ether extract of cultivated M. polymorpha; ME, Methanol extract of cultivated M. polymorpha; “–”: No activity.
Minimal Inhibitory Concentration (MIC, µg/mL) Values for Crude Extract of Marchantia polymorpha on Selected Microbial Strains.

Note: Bacterial strains: Staphylococcus aureus; Bacillus subtilis; Listeria monocytogenes; Salmonella typhimurium; and Escherichia coli, Fungal strains: Aspergillus flavus; Aspergillus niger; Candadi albicans; Trichophyton mentagrophytes.
Furthermore, the antifungal activity is also reported in Tables 2 and 3. Obviously, both extracts exhibited antifungal activity against selected fungi strains. The most effective antifungal activity was found for methanol extracts, which showed the best antifungal effect against Candida albicans and Trichophyton mentagrophytes, with MIC values at 64 and 128 µg/mL; respectively. Additionally, all the bis-bibenzyls (
Materials and Methods
Plant Material
The fresh plant of M. polymorpha (100 g) was collected in the suburb of Chongqing province by one of the authors (T. Chen). Following a rinse with sterile distilled water, a small piece of M. polymorpha callus tissue was transferred into freshly prepared Murashige-Skoog's medium (MSK-2 medium), 28 which contained 2,4-dichlorophenoxyacetic acid (1 mg/L) 28 as phytohormone for cell growth, and then cultivated with continuous shaking (100 rev/min) in light at 25 °C (2000 Lux). Five days later, the cells were sub-cultured into a 1-L conical flask containing 500 mL of MSK-2 medium and then grown under the same condition for 4 weeks.
Extraction, Isolation, and Structure Elucidation
The cultured medium (15 000 mL) was filtered, and the cultivated plant cells were dried in vacuo and extracted by EtOH at room temperature for 3 days (1 L × 3 times), the crude extracts were further partitioned between petroleum ether and 90% methanol–water system (v/v) to give petroleum ether extract (CP, 4.3 g) and methanol extract (CM, 11.2 g), respectively. Meanwhile, about 80 g of fresh plants of M. polymorpha were extracted with petroleum at room temperature for 24 h (100 mL × 3) and filtered, the filtrate was further extracted with methanol under the same condition to give petroleum (WP, 0.4 g) and methanol extract (WM, 0.9 g).
Guided by biological activities, the methanol extract (11.2 g) was subjected to an MCI gel column and eluted with a gradient methanol–water system (from 50% to 100% methanol) to give 4 fractions. Fraction 2 was subjected to RP-HPLC and eluted with 42% MeCN (2.0 mL/min) to yield compounds
HPLC Fingerprint Analysis for Methanol Extracts of Wild and Cultivated M. polymorpha
Methanol extracts of the wild and cultivated M. polymorpha were dissolved in methanol to prepare sample solutions for HPLC analysis. Previously yield compounds in methanol were used as reference standards. The HPLC analysis was carried out on a YMC packed C18 column (5 µm, 4.6 mm × 250 mm, 12 nm), eluted with MeOH–H2O system in linear-gradient (from 30% MeOH at the initial time to 90% MeOH at 90 min; flow rate, 0.6 mL/min) and detected at 254 nm.
Determination of Total Phenols and Total Flavonoids Contents
The total amount of phenols (TPC) in extracts was investigated as previously described. 16 To 50 µL of prepared samples, 2.5 mL diluted Folin-Giocalteau's reagent, and 2 mL of sodium carbonate (7.5% v/v) were added and kept in a water bath (45 °C) for 15 min. The data was measured spectrophotometrically at 760 nm (Bio-Rad 680 microplate reader). The calibration curve was prepared by using a standard gallic acid solution from 20 to 1000 mg/L (r2 = 0.998). The TPC values were presented as mg gallic acid equivalents per gram of extract (mg GAE/g extract).
The TFC values were determined as previously described. 29 In brief, 250 µL of extracts, 1.0 mL of distilled water, and 75 µL of 5% sodium nitrite were mixed and kept for 6 min, and then, 150 µL of aluminum trichloride (10% v/v) was added and further cultivated for another 5 min, and then, 0.5 mL of sodium hydroxide (1 M) was added. Finally, distilled water was added to make up to 2.5 mL and kept for another 15 min, the data was recorded immediately at 510 nm. The TFC was calculated using an equation obtained from the calibration curve of quercetin (r2 = 0.999) and recorded as milligrams of quercetin equivalents per gram of extract (mg QE/g extract).
Antioxidant Activity Evaluation
DPPH assay: The DPPH (1,1-diphenyl-2-picrylhydrazyl) assay is commonly used to evaluate potential antioxidants to determine the antioxidant activity.
30
In brief, 0.1 mL of DPPH (150 µM) methanol solution was added to freshly prepared tested samples (0.1 mL), vigorously shaken and kept in darkness, 30 min later. The absorbance was recorded on a Bio-Rad 680 microplate reader at 520 nm. In this text, 0.1 mL of methanol was used as the blank control, while vitamin E was used as the positive standard. The RSA values were recorded:
Reducing power: The reducing power of each fraction was evaluated as previously described. 30 Briefly, tested samples (0.5 mL) of different concentrations were prepared and mixed with 0.5 mL of phosphate buffer (0.2 M, pH 6.6), 0.5 mL of potassium ferricyanide (1%), this mixture was initially cultivated at 50 °C for 20 min, and 0.5 mL of trichloroacetic acid (10%) were added. And then, the mixture was centrifugated at 3000 rev/min to remove the residue. Finally, 0.5 mL of the top layer, 0.5 mL distilled water, and 0.1 mL ferric chloride (1%) were mixed. The uniform mixture was kept at room temperature for 5 min and the data was determined at 700 nm (Shimadzu UV-2401 PC). In this study, antioxidant vitamin E was used as the positive control.
Antibiotic Susceptibility Tests
The antibacterial effect was simply evaluated by agar-well diffusion assay. 31 Selected strains were cultivated in LB media overnight (37 °C) and diluted (1/100). Then, diluted bacterial culture (0.15 mL) was transferred to 100 mL autoclaved LB media (2.5 g powdered LB medium, and 1.5 g agar) to make plates (90 × 15 mm). Once the plates had been aseptically solidified, 10 μL of the test sample (10 mg/mL) were loaded, and further incubated at 37 °C for 18 h. Streptomycin (≥ 98%, 30 μg/disk) was used as a positive control. The inhibition zones surrounding the disks were recorded. Based on the aforementioned screening results, the MIC values were further evaluated. 32 In brief, 20 μL selected bacterial cultures (105 CFU/mL) were added to a 96-well plate and mixed with 180 µL of prepared diluted samples. After 24 h incubation, the lowest concentration in which the bacterial strains do not visible growth was recorded and defined as the MIC values.
The antifungal activity was also investigated via a modified microdilution technique. 33 The fungal spores were harvested and diluted (1.0 × 106 CFU/mL). The MIC values were determined by a serial dilution assay using 96-well microtiter plates. The crude extracts in broth medium and 1/10 diluted fungal inoculum were added to achieve different concentrations (0.1-10 mg/mL) and incubated at 28 °C. After 5 days, the lowest concentrations in which the fungal spores do not visible growth were defined as the MIC values.
Chemicals
All chemicals used in this study were purchased from Sigma.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221096172 - Supplemental material for Antimicrobial and Antioxidant Metabolites From the Cultured Suspension Cells of Marchantia polymorpha L.
Supplemental material, sj-docx-1-npx-10.1177_1934578X221096172 for Antimicrobial and Antioxidant Metabolites From the Cultured Suspension Cells of Marchantia polymorpha L. by Ya-yun Cai, Ting Chen and Jia-fu Cao in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Nantong City (grant no. JC 2020002).
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supporting Information
NMR and MS data were available as Supporting Information.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
