Abstract
Previous pharmacological research has demonstrated that Terminalia bellerica (Gaertn.) Roxb. (TB) extract possesses several pharmacological activities. However, there is scant evidence documenting the therapeutic activities of TB extract on inflammation, pain, and cancers. Our study examined the in vivo anti-inflammation, antinociception, and antitumorigenesis effects of TB extract and investigated possible mechanisms for those effects. Anti-inflammation activities of TB extract were evaluated using ethyl phenylpropiolate (EPP)- and arachidonic acid (AA)-induced ear edema models, a cotton pellet-induced granulation formation model, and a carrageenan-induced hind paw edema model. An antinociceptive property of TB extract was assessed using a formalin-induced nociception test. An anticarcinogenesis effect was investigated using a 7,12-dimethylbenz(a) anthracene (DMBA) and 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced tumorigenesis model. In the study, TB extract exhibited significant anti-inflammatory effects against EPP-induced ear edema and carrageenan-induced hind paw edema in rats. However, the TB extract showed insignificant inhibitory activity against AA-induced ear edema and cotton pellet-induced granuloma. A dose-dependent decrease in analgesic activity was observed with TB extract evidenced by decreased licking time in formalin-induced pain in mice in both the early and late phases. TB extract also significantly inhibited DMBA/TPA-induced mouse skin tumorigenesis. In conclusion, TB extract possesses anti-inflammatory, analgesic, and anticarcinogenesis properties which act, at least in part, through inhibitory effects of inflammatory mediator production.
Introduction
Inflammatory processes are complex immunological responses to noxious stimulants, including microorganisms, toxic compounds, injured cells, and irradiation. This response is intended to reduce damaging stimuli and to initiate the healing activities in the affected tissues.1,2 Clinically, inflammation is indicated by cardinal signs consisting of erythematous, edema, heat, pain, and loss of tissue function. 3 During the inflammatory process, an inflamed area increases vascular permeability, recruits and accumulates additional leukocytes, and releases inflammatory mediators, resulting in an exudation of plasma-rich fluid containing additional proteins into the damaged sites and subsequently presenting as an edematous lesion.1,4 The detection of the tissue damage involves not only the tissue-resident macrophages which lead to inflammation and repair but also with nociceptors (pain receptors) that stimulate pain sensation in the injured area. 3 There is also a functional association between inflammation and nociception that is responsible for inflammatory pain, including exudate formation, tissue edema, and inflammatory mediators, while nociception also monitors tissue homeostasis by means of complement inflammatory sensors.3,5 Many studies have demonstrated that chronic inflammation is one of the hallmarks of chemical, radiation, and other environmentally trigger-mediated skin carcinogenesis. 6 A well-known model employed to induce skin tumorigenesis involves initial topical application of 7,12-dimethylbenz(a) anthracene (DMBA), a polycyclic aromatic hydrocarbon that induces DNA alteration, followed by 12-O-tetradecanoylphorbol-13-acetate (TPA), which induces skin inflammation and proliferation, as well as promotion of tumors by inducing 2-stage skin cancer. 7
Effective treatments for inflammation, autoimmune diseases, and cancers remain an unmet medical need. Steroidal and nonsteroidal anti-inflammation medicines and chemotherapy are still the mainstay for cancers in conventional practice8,10; however, they can be accompanied by various adverse events, and the safety of their long-term use must also be considered. 11 For those reasons, there is great interest in identifying more viable therapeutic alternatives with better safety profiles that are also reliable, efficacious, and cost-effective, for example, natural compounds that have been used for a long time to alleviate inflammation and cancers. 12
Terminalia bellerica (Gaertn.) Roxb. (TB), family Combretaceae, is a traditional medicinal herb found in Southeast Asia, including Thailand.13,14 TB is a rich source of bioactive constituents, including glucosides, gallic acid, ellagic acid, ethyl gallate, gallyl glucose, tannins, and chebulinic acid. 13 The fruit of TB has been used extensively to treat constipation, fever, and as an anthelmintic agent13,15 and has also been used traditionally for the pharmacotherapy of eye disorders, scorpion stings, dyspepsia, hemorrhoids, diarrhea, hepatitis, coughs, bronchitis, and asthma. 15 Previous evidence has revealed that lignans isolated from TB extract exhibit activities against HIV-1, malaria, and fungi. 16 TB is also used as a free radical scavenger, and for treatment of various diseases, including infection, 17 cancers, 18 diarrhea, 19 diabetes, 20 hypertension, 21 and hepatic diseases. 22 Previous research by the authors found that acute and subacute toxicity of the ethanolic extract from TB produced no identifiable toxic effects in either mice 23 or rats. 24 Similarly, assessment of the acute and chronic toxicity of TB water extract found no toxic effects in rats. 25 However, there is only limited experimental evidence regarding the inhibitory activities on inflammation, analgesic activities, and antitumorigenesis of this plant in animal models. In an effort to fill this gap, the present study was conducted to determine the pharmacotherapeutic efficacy of the water extract of TB in the management of inflammation, pain, and tumors in rodent models.
Materials and Methods
Chemicals and Reagents
All experimental reagents and materials used in this study were obtained from Merck KGaA unless otherwise specified.
Plant Material and Standardization of Plant Extract
The TB plant was cultivated in Wangnumyen District, Sakaew Province, Thailand. When the TB fruits were mature, between September and November, they were harvested. Pharmacognostical analyses of herbal materials were carried out according to the Thai Herbal Preparation Pharmacopoeia and World Health Organization guidelines26,27 by Assoc. Professor Dr Noppamas Soonthornchareonnon, Mahidol University, Bangkok, Thailand. The voucher specimen of TB (PBM 02678) was deposited at the Faculty of Pharmacy, Mahidol University, Bangkok, Thailand.
To prepare the TB extract, fresh fruits of TB were exposed to sunlight for 15 days after which the dried fruits (104 kg) were boiled in water for 60 min then filtered. The boiling and filtering process was repeated a second and third time before drying using a spray-drying technique to evaporate the remaining traces of water. The yield of TB extract was 9.62% of the fresh weight. Quality control of the TB extract included evaluation of pharmacognostical, physicochemical, and microbiological parameters, together with the determination of the physical characteristics, weight loss (%) after drying, total ash and total acid-insoluble ash content, microbiological analysis, aflatoxin testing, as well as determination of the heavy metal contents and the quantity of chemical substances according to the Thai Herbal Pharmacopoeia. 27
High-Performance Liquid Chromatography (HPLC)
An HPLC technique was used to identify potential bioactive components of the TB extract. Test samples were separated using a Brownlee Spheri-5 RP-18 column (220 mm × 4.6 mm, 5 μm particle size) (PerkinElmer Inc). Isocratic elution was carried out using acetonitrile and 1 mM phosphoric acid (H3PO4) (8:92), with a flow rate of 0.7 mL/min. The absorption wavelength was selected at 216 nm and the injection volume of TB extract was 20 μL. To identify pharmacologically active components, peak area, and retention time were compared with standard curves of various concentrations of gallic acid and protocatechuic acid standards (Sigma-Aldrich Co).
Experimental Animals
Sprague-Dawley rats, weighing 40 to 60, 100 to 120, and 200 to 250 g, were used in the ear edema, paw edema, and granuloma models. Male MLAC: ICR mice (Mus musculus), weighing 25 to 40 g, were used in the formalin tests and skin tumorigenesis models. All animals were obtained from National Laboratory Animal Center (NLAC), Nakhon Phathom, Thailand. The rodents were housed in polypropylene cages provided with sterilized corncob bedding material with appropriate environmental enrichment and standard animal housing at 22 ± 1 °C with 50% to 70% humidity and a time-controlled 12/12 h light and dark cycle. An acclimatization period of at least 7 days allowed all animals to stabilize in their new surroundings before being used in the experiments. Commercial laboratory rodent diets and pathogen-free water were provided ad libitum. Prior to receiving drugs, the animals were fasted overnight, but clean water was freely available. Intraperitoneal administration of sodium pentobarbital was used to euthanize the animals. 28 All experiments adhered to the standard guidelines for the ethical use of laboratory animals in research.29,32 The protocol described was approved by the Animal Ethics Committee of the Faculty of Medicine, Thammasat University (Protocol approval number: 0001/2006).
Ethyl Phenylpropiolate (EPP)-Induced Ear Edema in Rats
To induce ear swelling, EPP dissolved in acetone was used according to a previously published protocol. 33 Thirty-six rats, weighing 45 to 65 g, were randomized into 6 groups of 6 animals each: a negative control group, a positive control group, and 3 TB extract groups. Edema induction of each rat was made by applying 1 mg of EPP (20 μL/ear) to the entire surface area of the pinna of one ear. Just prior to EPP application, the rats were treated topically (20 μL/ear) as follows: the negative control received the vehicle alone (1:1 combination of dimethyl sulfoxide and acetone), the positive control received 1 mg of phenylbutazone dissolved in the vehicle, and TB extract group received 1, 2, and 4 mg of TB extract dissolved in the vehicle. The outcome was evaluated by measuring the thickness of each rat's ear using a digital Vernier caliper at initiation and at 15, 30, 60, and 120 min after EPP induction. A graph was created plotting the thickness of the ears during the 2 h following EPP induction. The areas under the time-course curves were calculated from the overall swelling response of the individual experimental groups.
Arachidonic Acid (AA)-Induced Ear Edema in Rats
This test was performed following a previously published procedure. 34 Each rat received a topical application of 2 mg of AA dissolved in acetone (20 μL) on one ear to induce edema. Each group of rats then received a topical administration of one of the following agents (20 μL/ear): 1 mg of phenylbutazone, 2 mg of prednisolone, or 1, 2, or 4 mg of TB extract. One hour after AA stimulation, each rat was gently handled while the ear thickness was measured using a digital Vernier caliper.
Carrageenan-Induced Hind Paw Edema in Rats
This animal model study was performed following a previously published study.
35
Thirty-six male Sprague-Dawley rats, weighing 105 to 125 g, were allocated into 6 groups of 7 rats each. To induce swelling of the footpad, the subplantar surface of the rats’ right-hind foot was intradermally injected with 50 μL of newly prepared carrageenan (1%) dissolved in sodium chloride solution (0.9%). Before the inflammation-inducing injection, the paw volume was determined to ascertain the baseline value using a plethysmometer (Ugo Basile 7140). One hour prior to carrageenan injection, the rats were orally administered either TB extract at one of 3 doses (150, 300, or 600 mg/kg), aspirin (300 mg/kg), or 0.9% sodium chloride (10 mg/kg). The effect of the carrageenan on the edema volume of the paw was assessed again at 1, 3 and 5 h after carrageenan injection. A graph of the edema volume over that 5-h period was plotted. The areas under the time courses curves were determined by calculating the overall swelling response of each of the experimental groups. The inhibitory effect of TB extract was assessed as the percentage inhibition of paw swelling calculated using the following formula:
Cotton Pellet-Induced Granulation Formation in Rats
The effect of TB extract on chronic anti-inflammatory activity was evaluated according to a previously described method
36
using a cotton pellet-induced granulation formation model. Twenty-four rats, weighing 210 to 255 g, were randomized into 4 groups of 7 rats each. To prevent contamination, an autoclave was used to sterilize the cotton pellets under a pressure of approximately 15 pounds and at 120 οC for 30 min. One of 2 different standard drugs, either prednisolone (5 mg/kg) or aspirin (300 mg/kg), were administered to the reference groups, while TB extract (600 mg/kg) was given by oral gavage an hour before subcutaneous implantation of the sterile cotton pellets (21 ± 1 mg) in both the axillary regions of each rat under pentobarbital anesthesia. Test samples were administered by oral gavage each morning for one week. Changes in body weight were recorded throughout the experimental week. On day 8, the rats were again anesthetized. Wet weight was determined by surgically dissecting out the cotton pellets covered with granuloma tissue and the thymus and then weighing immediately. The cotton pellets covered with granuloma tissue and the thymus were surgically dissected out and their wet weight was determined immediately. The dry weight was determined by drying the dissected cotton pellets, granuloma tissue, and the thymus in an oven at 60 °C for 18 h to achieve consistent dry sample weight. To determine the transudative weight (mg), the mean difference between the wet weight and the constant dry weight of the cotton pellet was computed. To calculate the granuloma weight (mg/mg cotton), the mean of the cotton pellet weight was deducted from the mean of the granuloma dry weight and then divided by the mean weight of the cotton pellet. The percentage of granuloma inhibition (% GI) of exudates and granulomatous tissue was were calculated using the following formula:
Formalin Test
The chemical sensitivity test using formalin to test for acute pain was performed as mentioned in a previously published protocol. 37 Briefly, 36 ICR mice, weighing 40 to 60 g, were randomized into 6 groups of 6 mice. To assess the early phase of formalin-induced licking activity, each group of mice received either distilled water, morphine (10 mg/kg), aspirin (300 mg/kg), or TB extract (150, 300, or 600 mg/kg) by oral gavage 1 h before intraplantar injection into their right hind paws of 1% formalin (20 μL) dissolved in normal saline solution. Licking time, defined as the period that the mouse spent licking its formalin-administered footpad, was monitored and documented for 5 min following injection. To evaluate the late phase of licking activity in the formalin test, the right hind paws of another set of mice were injected with 1% of formalin 40 min after administering the test drugs. After this formalin administration, the licking time of the injected footpad was recorded for between 20 and 30 min. The licking times in both the early phase and the late phase were recorded and plotted.
DMBA/TPA-Induced Skin Tumorigenesis
A skin tumorigenesis model was used to assess the pharmacological activity of TB extract on DMBA/TPA-induced carcinogenesis using previously described methods.
38
Briefly, 3 days prior to the commencement of an experiment, hair was removed from a 2 × 4 cm area of the dorsal side of mouse's skin using surgical clippers. Mice were randomly segregated into 5 groups (8 animals each), as shown in Figure 7A. Group 1 mice, the vehicle control group, were treated with 100 μL of acetone (Merck KGaA) twice weekly using a micropipette. Mice in groups 2 to 5 were given a single topical application of DMBA (Sigma-Aldrich Co) at a concentration of 390 nmol dissolved in 100 μL of acetone as induction using a micropipette. One week after induction with DMBA, mice in groups 3 to 8 were treated twice weekly with TB extract at concentrations of 1, 2, or 4 mg dissolved in 100 μL. An hour after TB extract treatment, the mice in groups 2 to 5 were treated with TPA (4 nmol/100 μL acetone) using a micropipette. This treatment was repeated twice weekly for a period of 20 weeks. The size of nodules on the backs of the mice were carefully measured using a digital vernier caliper, and the tumor incidence and number of nodules were documented weekly. At the end of the 20 weeks, all the animals were euthanized under deep anesthesia and the skin with tumors was dissected and then fixed for 48 h in 10% neutral buffered formalin. The investigational dorsal skin and skin tumors were excised and stained with hematoxylin and eosin for microscopic investigation of histopathology. The suppressor effects of TB extract on DMBA/TPA-induced tumorigenesis were analyzed for (1) percentage of tumor-bearing mice (the number of mice showing tumor response divided by the total number of mice in the group × 100), (2) the average number of tumors per mouse, and (3) the volume of tumor mass determined using the following formula
39
:
Statistical Analysis
Outcome data are presented as mean ± standard deviation of the mean (SD). Differences of data in the normal, standard control, and TB-treated groups were analyzed with 1- or 2-way analysis of variance (ANOVA) using IBM SPSS Statistics (version 22.0). Further comparison by post hoc analysis was performed using the Least Significant Difference (LSD) test. P values of less than .05 were regarded as statistically significant.
Results
HPLC Chemical Profile of Gallic Acid and Protocatechuic Acid in Terminalia Bellerica (Gaertn.) Roxb Extract
It was observed that both the raw TB material and the TB water extract contained 34.5% tannins, 8% gallic acid, 5.3% uronic acid by weight, and 20% total carbohydrates. The monosaccharide components consisted of arabinose, galactose, glucuronic acid, and galacturonic acid. The TB extract had no contamination with aflatoxin, heavy metals, pesticides, or microbials. The major chemical composition of the water extract from TB was investigated by HPLC. The chromatographic fingerprint of the TB extract showed that the major peaks of both gallic acid and protocatechuic acid occurred at the retention times (RT) of 6.1893 and 12.5327, respectively. The detected retention times in the extract were highly compatible with those of standard gallic acid and protocatechuic acid (RT = 6.035 and 11.961, respectively). In the comparison calibration curve, the amount of gallic acid in TB extract was found to be 7.98%, w/w. These results clearly confirmed that gallic acid and protocatechuic acid were the major compounds, and probably were the active constituents of the TB extract.
Anti-Inflammatory Activity of TB Extract on Ethyl Phenylpropiolate (EPP)- or Arachidonic Acid (AA)-Induced Ear Edema in Rats
In the EPP-induced control group, pronounced inflammatory symptoms, including erythematous and edema, were observed on both the inner and outer sides of the ear pinna, as well as an almost 2-fold increase in the thickness of the ears (approximately 180 μm) at 60 min. Treatment with phenylbutazone (1 mg/ear), a reference anti-inflammatory drug, significantly decreased EPP-induced ear swelling to a thickness of around 60 μm (P < .05) at 60 min (Figures 1A and B) . Similarly, an induction with AA produced ear inflammation thickness of up to approximately 135 μm, whereas the treatment with phenylbutazone (1 mg/ear) and prednisolone (2 mg/ear) resulted in a significant decrease in ear thickness to approximately 70 and 60 μm, respectively (Figure 2). Interestingly, treatment with TB extract at doses of 1, 2, and 4 mg/ear resulted in a significant decrease in EPP-induced ear edema to 215, 205, and 195 μm, respectively, in a dose-dependent manner compared to the EPP-induced untreated group at all evaluation time points (Figure 1). Although TB extract at doses of 1, 2, and 4 mg/ear decreased ear thickness to 108.3, 91.70, and 98.30 μm, respectively, with AA-induced ear edema, no statistically significant reduction in ear thickness was found in the groups treated with TB extract compared with the control group (Figure 2).

Effects of TB extract treatment (1, 2, and 4 mg/ear) on ethyl phenylpropiolate (EPP)-induced ear edema formation in rats. (A) The thickness of ear swelling in the normal, control, phenylbutazone (PBZ), and TB-treated groups measured at different time points (15, 30, 60, and 120 min) after EPP induction. (B) Total change in ear edema was calculated as the area under the curve (AUC) in EPP-induced ear edema rats. Data are plotted as mean ± SD (n = 6). *P < .05 indicates a statistically significant difference between the drug treatment groups and the untreated group (one-way ANOVA followed by LSD post hoc test).
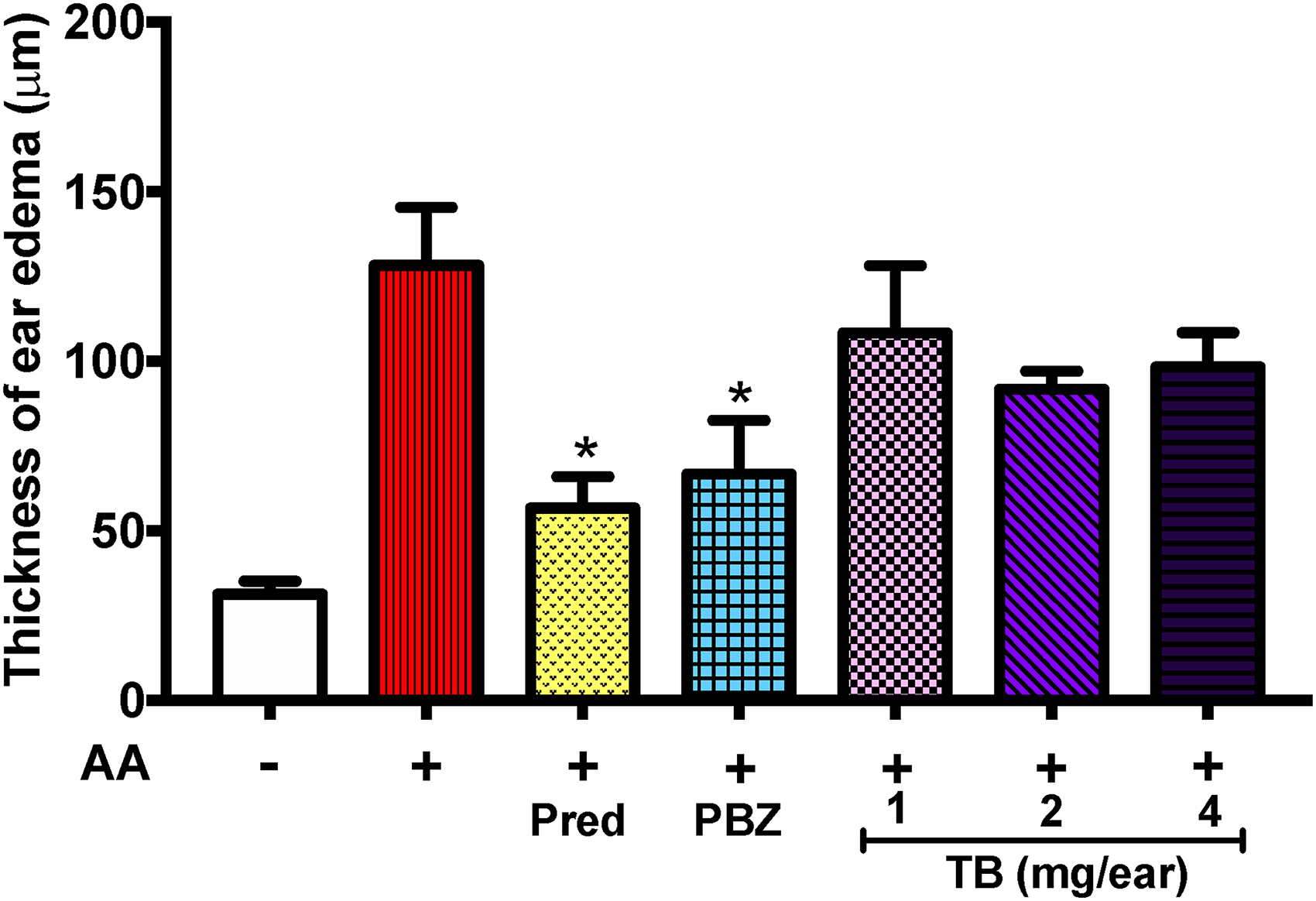
Effects of TB extract (1, 2, and 4 mg/ear) on arachidonic acid (AA)-induced ear edema formation in rats. The bar graph shows the thickness of ear edema in the normal, control, prednisolone (Pred), phenylbutazone (PBZ), and TB-treated groups at 60 min after AA induction. Data are shown as mean ± SD (n = 6). *P < .05 indicates a statistically significant difference between the drug treatment groups and the untreated group (one-way ANOVA followed by LSD post hoc test).
Anti-Inflammatory Activity of TB Extract on Carrageenan-Induced Paw Edema in Rats
During the 3 h following the 1% carrageenan induction in the control group rats’ paws, a marked time-related, progressive increase in edema volume (0.8 mL) was observed. A reference drug, aspirin (300 mg/kg), demonstrated a significant reduction in carrageenan-induced footpad swelling volume (by 57%) 3 h after carrageenan injection. Treatment with TB extract at various concentrations (150, 300, and 600 mg/kg) also significantly decreased total edematous paw edema volume by 40%, 44%, and 54%, respectively (P < .05) at 1, 3, and 5 h after injecting footpads with carrageenan (Figure 3).

Effects of TB extract (150, 300, and 600 mg/kg) on carrageenan-induced paw edema in rats. (A) Paw edema volume (mL) in normal, control, aspirin, and TB treated groups at 1, 3, and 5 h after 1% carrageenan injection in the right hind paw. (B) Total percentage edema calculated as the area under the curve (AUC) in carrageenan-induced paw edema. Data are plotted as mean ± SD (n = 6). *P < .05 indicates a statistically significant difference between the drug treatment groups and the untreated group (one-way ANOVA followed by LSD post hoc test).

Effects of TB extract (600 mg/kg) treatment on body weight gain and dry thymus weight of rats in the cotton pellet-induced granulation formation model. (A) Body weight gain (g) and (b) shows dry thymus weight (mg/100 g body weight) in the control, prednisolone, aspirin, and TB-treated groups. Data are plotted as mean ± SD (n = 6). *P < .05 indicates statistically significant difference between the drug treatments and the control group (one-way ANOVA followed by LSD post hoc test).
Anti-Inflammatory Activity of TB Extract on Cotton Pellet-Induced Granuloma Formation in Rats
On day 8 after sterile cotton pellets had been implanted in the abdomen of rats, the accumulation of granuloma tissue was observed in all experimental groups. Treatment with 5 mg/kg of prednisolone, a standard reference, showed a marked potential for reducing transudative weight, size, and weight of the granulomas with a granuloma inhibition (GI) of 16% (Table 1). In addition, body weight gain and dry thymus weight of the rats were also significantly reduced, as shown in Figure 4A and B. However, no statistically significant reduction in transudative weight, accumulated granuloma tissue (Table 1), body weight gain, or thymus weight in the rats treated with 600 mg/kg of TB extract and 300 mg/kg of aspirin was observed when compared with the untreated group.
Effect of TB Extract on Transudative Weight and Granuloma Formation of Cotton Pellet-Induced Granulomas in Rats. a

Data are shown as mean ± SD (n = 6). *P < .05 indicates a statistically significant difference between the drug treatments and the control group (one-way ANOVA followed by LSD post hoc test). %GI = % granuloma inhibition.
Antinociceptive Activity of TB Extract on Formalin-Induced Pain Reaction in Mice
Subplantar injection of 1% formalin (20 μL) induced a biphasic pain reaction. The early phase (neurogenic phase) is associated with direct chemical activation of nociceptors and is indicated by an immediate, intense pain response lasting up to 10 min post-injection, whereas the late phase (inflammatory phase) results from peripheral inflammation and alterations in central pain mechanisms and is marked by a suspended onset of pain beginning 10 to 60 min after injection due to the release of a variety of cytokines associated with the inflammatory process. The licking times decreased in a concentration-dependent manner in the morphine-, aspirin-, and TB extract-treated groups. An hour after formalin injection, a significant (P < .05) antinociceptive activity in a concentration-dependent manner was found in the group of mice that had been orally administered TB extract. In the early phase, TB extract at doses of 150, 300, and 600 mg/kg revealed a significant maximum suppression of the formalin-induced licking response of 24, 29, and 44%, respectively (Figure 5A and Table 2). In the late phase, the TB extract at low, medium, and high doses showed inhibition rates of 78, 78, and 89%, respectively (Figure 5B and Table 2). A reference drug, morphine (10 mg/kg), significantly (P < .05) suppressed both the early and late phase formalin-induced licking response with the maximum inhibition (100%), whereas the inhibitory effect of aspirin (300 mg/kg) was significant only in the late phase with inhibition of 100%.

Effects of TB extract (150, 300, and 600 mg/kg) treatment in the early phase (A) and late phase (B) of the formalin test in mice. The effect of morphine (10 mg/kg) and aspirin (300 mg/kg) on licking times were used as positive controls during the early phase (0-5 min) and the late phase (20-30 min). Data are plotted as mean ± SD (n = 6). *P < .05 indicates a statistically significant difference between the drug treatments and the control group (one-way ANOVA followed by LSD post hoc test).
Effect of TB Extract on Inhibition of Nociceptive Response in the Early and Late Phases of the Formalin Test in Mice.

Antitumor Activity on DMBA/TPA-Induced Tumorigenesis in Mice
Suppression activity of TB extract on tumorigenesis was examined in DMBA/TPA exposed mice. No nodules were observed in mice that were not exposed to DMBA/TPA, specifically, those treated with acetone (control group 1) and TB extract (groups 6-8). Compared to the control group, DMBA/TPA-induced mice showed a markedly higher incidence of skin papillomas, which was decreased by TB extract (Figure 6B). The onset of papillomatosis was observed at week 4 in the DMBA/TPA-treated mice (group 2), followed subsequently by a 5% to 10% increase in the tumor size each week. It was observed that TB extract at concentrations of 1, 2, and 4 mg significantly suppressed the number of tumors per mouse at week 20, indicating a reduced multiplicity of skin papilloma formation of 37%, 12%, and 18%, respectively, compared with the DMBA/TPA-induced group (Figure 6C). The tumor mass volume was consistently elevated by DMBA/TPA stimulation (100%) but was significantly reduced with 1, 2, and 4 mg of TB extract by 60%, 32%, and 27%, respectively, at week 20 (Figure 6D). Additionally, histopathological examination found normal skin with the well-defined epidermis, dermis, and subcutaneous tissue in the control group (Figure 7A). At week 20 of the study, the mouse skin in the DMBA/TPA group had formed nodules that were histologically characterized as squamous cell carcinoma (Figure 7B). The application of DMBA/TPA on mouse skin clearly induced atypically thickened keratinocytes and over proliferative epidermis, also known as hyperkeratosis. The DMBA/TPA-treated mice also showed the histological characteristics of islands of dysplastic squamous keratin cells in the dermis layer, indicating the invasive or malignant nature of the carcinoma. TB extract markedly ameliorated the increase in epidermal proliferation in mice with DMBA/TPA stimulation (Figure 7C). Representative pictures of mouse dorsal skin of each group are shown in Figure 7D to F. These findings strongly suggested that TB extract effectively suppressed skin tumorigenesis induced by DMBA/TPA in mice.

Inhibitory effect of TB extract on DMBA/TPA-induced mouse skin tumorigenesis. (A) Detailed study plan and treatment schedule for each group to evaluate the effect of TB extract on DMBA/TPA-induced skin tumorigenesis. (B) Tumor-bearing mice (percentage of mice with tumors). (C) The average numbers of tumors in each mouse. (D) Tumor volume (mm3). Data are plotted as mean ± SD (n = 8). *P < .05 indicates a statistically significant difference between the drug treatments and the DMBA/TPA group (one-way ANOVA followed by LSD post hoc test). (DMBA, 7,12-dimethylbenz[a]anthracene; TB, Terminalia bellerica; TPA, 12-O-tetradecanoylphorbol-13-acetate).

Representative images of epidermal hyperproliferation in DMBA/TPA-induced mice treated with 4 mg of TB extract analyzed by H&E staining. (A) Histopathology of the normal skin histoarchitecture of the control group at 40×. (B) Histopathology of the DMBA/TPA-induced tumorigenesis in a mouse at 40× showing characteristic squamous keratin pearls in the DMBA/TPA-stimulated mouse skin tumors at 200× (bottom right corner of (b) image). (C) Histopathology of TB extract-treated mouse skin (4 mg) at 40×. (D) Dorsal skin picture of a control mouse. (E) Dorsal skin picture of a DMBA/TPA-induced tumorigenesis mouse. (F) Dorsal skin picture of a TB extract-treated mouse (4 mg). (DMBA, 7,12-dimethylbenz[a]anthracene; TB, Terminalia bellerica; TPA, 12-O-tetradecanoylphorbol-13-acetate).
Discussion
Our study was performed to assess the pharmacotherapeutic use of Terminalia bellerica water extract derived from its dried fruit in treating swelling and pain caused by acute and chronic inflammation in animal models. Ethyl phenylpropiolate (EPP), arachidonic acid (AA), and carrageenan were applied to induce experimental edema, whereas cotton pellets were used to induce chronic inflammatory activity. In addition, a formalin-induced nociceptive model was used to evaluate pain and to investigate possible mechanisms of action of the TB extract. The anti-skin cancer effect of TB extract was also evaluated using a DMBA/TPA-induced skin tumorigenesis model.
EPP and AA were topically applied to the ear pinnae of rats leading to induction of ear edema 34 because different kinds of chemical stimulants produced skin irritation by multiple mechanisms. 40 The secretion of many mediators of inflammation, including histamine, serotonin, kinin, and prostaglandins, is stimulated by an EPP-induced acute inflammatory model, 41 whereas an AA-induced edema model activates the lipoxygenase pathway. 34 Our results are in line with Patrick et al (1985), demonstrating that the rat's ears treated with EPP show vasodilation and become engorged, while edema and infiltration of neutrophils are observed in blood vessels. 40 Prostaglandins have the ability to synergistically increase the permeability of blood vessel walls and formation of edema either directly by promoting vasodilation or indirectly by releasing histamine from mast cells.42,43 Phenylbutazone, a nonsteroidal anti-inflammatory drug (NSAID), elicits an inhibitory effect on edema through prostaglandin inhibition and attenuates the sensitivity of blood vessels to histamine and bradykinin. 44 In the current study, TB extract exhibited an anti-inflammatory activity against ear edema formation induced by EPP but failed to afford protection against an AA-induced ear edema response. These outcomes suggest that TB extract may predispose inflammatory mediator synthesis and release leading to edema, although this might not involve the lipoxygenase pathway.
To ascertain better the inhibitory properties of TB extract on inflammation, acute inflammation of the hind foot pad was stimulated by carrageenan. When carrageenan is intradermally administered into a rat's paw, it causes inflammation in 3 phases. The initial phase (hours 0 to 2 after injection) is characterized by the secretion of serotonin and histamine from mast cells, while the second phase, hours 2 to 4, involves the secretion of kinins, and finally, in the third phase, hour 4 to 6,45,46 prostaglandins, COXs, and cytokines are released and neutrophil infiltration can be observed.47,48 Aspirin is the standard reference drug used in this type of experimental model for the therapeutic treatment of inflammation and is part of many direct nonselective NSAIDs that inhibits the COX enzyme. 49 The present study found that at 1, 3, and 5 h after administration of carrageenan, TB extract significantly inhibited edema formation of the hind paw in a dose-dependent manner. This protective property began in the first hour and was sustained throughout the 3 phases of inflammation, indicating that TB extract exhibits its anti-inflammatory action, at least in part, through blocking prostaglandin synthesis and interfering with the cyclooxygenase-mediated pathway. These findings are in line with previous evidence showing that the aerial part of Terminalia bellerica can reduce cyclooxygenase-2 (COX-2), nuclear factor-kappaB (NF-κB), and tumor necrosis factor-alpha (TNF-α) overexpression in damaged livers of carbon tetrachloride (CCl4)-intoxicated mice, suggesting amelioration of the inflammatory response. 50
Cotton pellet-induced granuloma formation has been commonly used in in vivo tests to evaluate the extent of chronic inflammation related to the transudative, exudative, and proliferative components. 36 This model involves 3 phases of inflammatory responses following the implantation of a cotton pellet. The first phase is the transudative phase (0-3 h), which is characterized by an increase in the wet weight of the pellet due to increased vascular permeability.51,52 The second phase is the exudative phase (3-72 h), which is defined by the permeability of blood vessels around the granuloma that allows proteins to leak and is categorized by a delayed and prolonged change in vascular permeability resulting from inflammation. The third phase is the proliferative phase (3-6 days) that is marked by an increase in the dry weight of the granuloma which presents with a collagen appearance in granulation tissue. 36 Specifically, a transudate, defined as a fluid that is low in protein and noninflammatory in origin, 53 develops from an imbalance of hydrostatic and oncotic pressure. 54 Our study showed prednisolone significantly inhibited the increase in transudative weight, granuloma formation, body weight, and dry thymus weight; however, neither TB extract nor aspirin reduced any of those parameters. The insignificant decrease of transudative and exudative weight with TB extract and aspirin in this model does not indicate an inhibitory effect on the capacity of the vascular endothelium to allow for the flow of small molecules. Our findings are consistent with a report by Swingle et al (1972) indicating that the exudative phase is refractory to inhibition by most NSAIDs, with the exception of indomethacin. 36 The granulomatous inflammatory response is a distinctive type of chronic inflammatory manifestation. 52 The effectiveness of anti-inflammatory drugs in inhibiting granuloma formation is possibly a result of their inhibitory effects on the proliferative phase of inflammation. 36 The thymus of rodents is considered as the primary lymphoid organ where immature CD4− and CD8− T-cell precursors develop into mature T cells. 55 The administration of corticosteroids causes not only a rapid and profound lymphopenia 56 but also a dramatic reduction in levels of circulating thymus-derived lymphocytes. Additionally, corticosteroids also have a catabolic effect on lymphatic and connective systems, and consequently the animals’ body weight gain and dry thymus weight decreased.57,58 The present study found that TB extract neither inhibits the proliferative phase of chronic inflammation nor does it possesses steroid-like properties, as it did not change either body weight gain or dry thymus weight.
Formalin-induced acute inflammatory pain, characterized by paw licking, is a model used to mimic acute and clinical pain.59,60 The intraplantar injections of formalin stimulate the C fibers by direct activation of the peripheral nociceptors, leading to a biphasic behavioral reaction. The early phase (neurogenic pain), which initiates promptly after injecting a paw with formalin and lasts for 3 to 5 min, produces various substances, including bradykinin and substance P. Then, after a quiescent period, the late phase (moderate inflammatory pain) occurs and lasts for between 20 and 30 min. During this phase, many mediators of inflammation, for example, histamine, prostaglandins, bradykinin, interleukin, TNF-α, and adrenergic amines, are activated, occurring with functional modifications of the spinal cord, particularly in the dorsal horn.59,61,64 The intensity response of the nociceptive behaviors response is determined by the concentration of formalin that is administered. 62 Our study used an antinociceptive drug, morphine, as a standard reference, which was effective against pain in both phases. In contrast, aspirin seemed to markedly suppress pain only during the late phase.37,65 Thus, TB extract is more promising than the standard reference drug, aspirin, in the early phase. Interestingly, TB extract produced a significant analgesic effect in a dose-dependent pattern in both the neurogenic and inflammatory phases, a result of the release of the aforementioned inflammatory mediators. This finding agrees with a recent study showing that the ethanolic extract of T. bellerica exhibits analgesic activity in a rodent tail immersion model. 66 Hence, the TB extract is a potential agent against nociception, operating in part through inhibiting the production and secretion of inflammatory pain mediators, for example, prostaglandins, at peripheral nociception sites.
The antitumorigenesis effect of TB extract was evaluated using DMBA/TPA-induced skin carcinogenesis in a mouse model. Our study showed that TB extract significantly decreased tumor incidence, the mean number of tumors per mouse, and tumor volume in mice exposed to DMBA/TPA. Our HPLC results revealed that the major active constituents of TB extracts are gallic acid and protocatechuic acid, which are consistent with previously published evidence.67,69 Protocatechuic acid has been reported to be associated with potent antioxidant effects and anticarcinogenic effects by influencing the activity of enzymes involved in oncogenesis, for example, cyclooxygenase, nitric oxide synthase, and inflammatory mediators.70,71 Gallic acid exerts antitumorigenesis effects via the regulation of genes which modulate the cell-division cycle, angiogenesis, programmed cell death processes, cell migration, and metastasis.72,73 The inhibitory effect of gallic acid also mediates through inhibition of NF-κB and PI3 K/Akt signaling transduction pathways, as well as through regulation of cyclooxygenase, ribonucleotide reductase, and glutathione. 72 Evidence is accumulating which supports the inhibitory effects of TB extract on the growth of various tumors. 74 A recent study reported suppression activity of T bellirica seed extract and pure gallic acid against cancer cell lines, including HaCaT keratinocytes, tongue (Cal33, SCC-4, and SCC-25), and pharynx (FaDu) squamous cell carcinoma. 75 The gallic acid in the TB extract possessed potent antioxidant and antiproliferative activities which resulted in programmed cell death, suggesting promising pharmacological effects against oral squamous cell carcinoma. 75
Conclusions
Our current study suggests that TB extract exhibits marked anti-inflammatory activity, causing significant reductions in ear edema and paw edema. This indicates the probable mechanism of action may be inhibition of acute phases of inflammation. In addition, TB has significant inhibitory effects on pain response induced by formalin stimulation of nociceptors, reflecting its analgesic activity. TB extract also dose-dependently ameliorates DMBA/TPA-induced skin tumorigenesis. Further investigation of the specific cellular targets and biochemical pathways underlying these anti-inflammatory, analgesic, and antitumor functions in specific disease models is needed to elucidate further the active compounds involved, as well as the precise mechanisms of action of TB extract. Target-specific compounds such as TB extract offer promise for the treatment of diseases related to inflammation, pain, and tumors.
Footnotes
Acknowledgments
The authors would like to thank the Faculty of Medicine, Chiang Mai University, and the National Research Council of Thailand for their financial support. The authors also would like to thank Dr Lamar Robert for English proofreading.
Authors’ Note
The data that support the findings of this study are available from the corresponding author, [SS], upon reasonable request. All experiments were conducted according to the International and Thai National Guidelines for Ethical Conduct in the Care and Use of Animals and were approved by the Animal Ethics Committee of the Faculty of Medicine, Thammasat University (Protocol approval number: 0001/2006). This study was conceptualization and conducted by MN, KJ, NS, and SS. The analysis was made by MN, KJ, and SS, data curation and visualization by MN and SS, and the manuscript was written by MN and SS with contributions from all authors. All authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The National Research Council of Thailand, Faculty of Medicine, Chiang Mai University,



