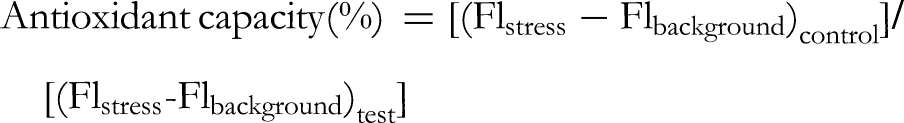Abstract
Background
Terminalia chebula Retz. var. chebula (T chebula) has been traditionally used as a crude drug for treating various diseases, including skin disorders. This study aimed to investigate the potential antioxidant effects and antitumorigenic activity of an aqueous extract from the fruit of T chebula.
Methods
The extract's total phenolic content and antioxidant activity were measured using Folin-Ciocalteu, 1,1-diphenyl-2-picrylhydrazyl (DPPH), and ferric reducing antioxidant power (FRAP) tests, respectively. The 2 ´,7 ´-dichlorodihydrofluorescin diacetate assay was employed to evaluate the extract's ability to reduce intracellular reactive oxygen species (ROS) in U-937 human monocytic cell lines which had been treated with hydrogen peroxide for 30 min. Male ICR mice (n = 30, 5 groups) were used to investigate the influence of T chebula extract on dimethylbenz[a]anthracene (DMBA)/12-O-tetradecanoylphorbol 13-acetate (TPA)-induced carcinogenesis. After 20 weeks of treatment with T chebula extract, tumor incidence, size, and number were assessed. The major phenolic compounds in the extract were determined using high-performance liquid chromatography. Statistical analysis was performed using ANOVA followed by post hoc least significant difference test.
Results
T chebula extract effectively reduced intracellular ROS and scavenged antioxidants in vitro. The IC50 values of the extract as measured by DPPH and FRAP assays were 109.0 ± 14.5 μg/mL and 2.39 ± 0.17 μg/mL, respectively. Moreover, 4 mg of T chebula extract significantly decreased the incidence, volume, and number of tumors in DMBA/TPA-induced skin tumorigenesis in mice.
Conclusion
T chebula extract effectively reduced intracellular ROS, and significantly decreased the incidence, volume, and number of tumors in DMBA/TPA-induced skin tumorigenesis in mice. These findings suggest that T chebula extract could be a promising therapeutic agent for skin tumors and other oxidative stress-related diseases.
Introduction
Skin cancer, including melanoma and keratinocyte carcinoma, has become a growing concern globally due to its increasing incidence and prevalence in recent decades. 1 Skin cancer accounts for one-third of all diagnosed cancers, with 132 000 new cases of melanoma occurring annually. 2 Melanoma rates have been rising by 4%-6% each year in light-skinned populations in North America, Northern Europe, Australia, and New Zealand. 2 During 2013-2015, the incidence rate of skin cancer in Thailand was 3.6 per 100 000 person-years for males and 3.8 per 100 000 person-years for females. 3 Although this is lower than the incidence rate in Western countries, skin cancer is still relatively common in Thailand and ranks among the top fifteen types of cancer diagnosed in the country. 3 Reactive oxygen species (ROS) have an intense oxidative power that is generated during metabolic processes and in response to various environmental stressors such as ultraviolet radiation. 4 At low concentrations, ROS are essential to cellular communication, but excessive ROS production can damage cellular components such as DNA, lipids, and proteins, leading to oxidative stress, inflammation, and ultimately, tumorigenesis.5,6 Skin cancer or tumorigenesis has been associated with an imbalance between ROS production and antioxidant defense mechanisms in the body. Given the association between ROS and tumorigenesis, antioxidants, which are molecules that can neutralize ROS and protect cells from oxidative damage, have come to the forefront as a promising alternative to conventional cancer treatments, and are often used in combination with chemotherapy drugs. 7 While conventional cancer therapies such as chemotherapy, radiotherapy, and chemically derived drugs are effective, 8 they often have adverse effects that can be physically and emotionally taxing for patients and significantly affect their quality of life. 9 In contrast, many plant-derived compounds have fewer adverse effects and are considered to have a more favorable impact on patients. 10 For many years, plants have been utilized in Eastern cultures as folk medicines. Examples of plant-derived compounds which have the capability to fight cancer include polyphenols, taxanes, and flavonoids. 11
Terminalia chebula Retz. var. chebula (T chebula), family Combretaceae, is a medicinal herb that is widely grown in northeast Thailand. 12 The fruit of T chebula has a long history of traditional usage as an astringent for treating various ailments including gastrointestinal disorders, tumors, and skin diseases, as well as issues related to cardiovascular disorders and diabetes.13‐15 The bioactive chemicals present in T chebula are phenolic compounds including phenolic acids, tannins, and flavonoids 16 eg, gallic acid, 17 ellagic acid, 18 and chebulinic acid. 19 T chebula has been found to be beneficial in treating various diseases, owing to its antioxidant activity, anti-inflammatory, 20 cardiac protective, 21 blood glucose-modulating, 22 and anticancer effects. 23 Additionally, T chebula has been demonstrated to possess chemotherapeutic properties against infectious diseases24‐26 and protective effects on various organs and systems, including neurons, 27 kidney, 28 liver, 29 and stomach, 30 as well as skin wound healing. 31 Many studies have demonstrated most of the pharmacological properties of T chebula extracts obtained using various types of solvents.32,33 However, the effects of T chebula aqueous extract have been inconclusive in some studies regarding liver cancer, 34 inflammation, 35 and skin lightening. 36 While water extraction may not be as effective as many organic solvents for separating certain compounds or plant materials, it has been demonstrated to be a safer, more environmentally friendly, and cost-effective alternative. 37 More research is needed to assess the effects of the water extract of T chebula fruits. Skin carcinogenesis, which is the abnormal growth of skin cells, is a multistep process that involves DNA damage, oxidative stress, and inflammation. While the potential of T chebula as an antioxidant and anti-inflammatory agent has been reported, 20 its effects on skin carcinogenesis have not been well studied. In this study, we aimed to evaluate the antioxidant and antitumorigenic effects of the aqueous extract of T chebula fruit. To detect the bioactive compounds in the T chebula extract, we used a high-performance liquid chromatography (HPLC) assay which allowed us to identify and quantify the presence of gallic acid and protocatechuic acid in the extract. To assess the antioxidant activity, we measured the ability of the extract to scavenge free radicals using DPPH (2,2-diphenyl-1-picrylhydrazyl) and ferric reducing antioxidant power (FRAP) assays. We also measured the level of ROS in U-937 monocytic cells treated with the T chebula extract using the DCFH-DA assay. The U-937 cell line was chosen as a model because it is derived from human monocytes and has been widely used in studies related to inflammation and cancer. 38 In addition, we evaluated the antitumorigenic activity of the T chebula extract using a DMBA (7,12-dimethylbenz(a)anthracene)-induced mouse model of cancer and analyzed tumor volume, weight, and histopathological changes. Using the 2 models highlighted the potential anticancer and antioxidant properties of T chebula aqueous extract and provided insights into its mechanism of action, which could pave the way for further investigation and potential clinical use.
Material and Methods
Chemical Material
The following were purchased from Sigma Chemical Co (St. Louis, MO, USA): acetonitrile for HPLC (No.34851), 7,12-dimethylbenz[a]anthracene (DMBA; No. D3254), ferric chloride (FeCl3; No. 931527), ferrous sulfate (FeSO4; No. 1270355), hydrochloric acid (HCl; No. H1758), sodium acetate (No. S2889), phorbol-12-myristate-13-acetate (PMA; No. P8139), 2-thiobarbituric acid (TBA; No. T5500), 12-O-tetradecanoylphorbol 13-acetate (TPA), 2,4,6-tripyridyl-striazine (TPTZ), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox®; No. 391913), and sodium carbonate (Na2CO3; No. 223484). Merck KGaA (Darmstadt, Germany) supplied acetone, 2′,7′-dichlorofluorescein diacetate (2,7-DCF-DA; No. 35845), Folin-Ciocalteu reagent (No. 47641), gallic acid (3,4,5-trihydroxybenzoic acid, No. 91215), hydrogen peroxide (No. 386790-M), methyl alcohol (No. 1424109), and protocatechuic acid (3,4-dihydroxybenzoic acid, protocatechuic acid, No. PHL89766). Thermo Fisher Scientific (Waltham, MA, USA) provided fetal bovine serum (No. 16000044), Gibco™ RPMI 1640 Medium (No. 1TFS-1CC-11875093), Hanks’ Balanced Salt Solution (HBSS, No. 1TFS-1CC-14175095), and penicillin-streptomycin (1TFS-1CC-15140122).
Plant Material and Preparation
T chebula plants were grown between May and July and harvested between September and December in the deciduous forests of Soeng Sang District, Nakhon Ratchasima Province, and Pakham District, Buri Ram Province following the WHO Guidelines on Good Agricultural and Collection Practices (GACP) for Medicinal Plants. 39 Identification, authentication, and preparation of the T chebula fruit was performed by Assoc. Prof. Dr Noppamas Soonthornchareonnon of the Faculty of Pharmacy, Mahidol University. A voucher herbarium specimen (number PBM-00485) of the T chebula fruit is kept in the Herbarium of Pharmaceutical Botany, Faculty of Pharmacy, Mahidol University. Approximately 360 kg of T chebula fruits was steamed for 40 min, then sun-dried for 2 weeks. The dried fruits (68 kg) were then boiled in water for an hour. The liquid was filtered to remove any solid residues, and the extracted liquid was mixed with additives as carriers before undergoing the spray drying process. The dried T chebula extract was stored in a sterilized, sealed container and refrigerated at 4 °C to prevent exposure to air and moisture until it was needed for further experiments.
Quality Control of T chebula
The quality of the T chebula sample was determined following Thai Herbal Pharmacopoeia 40 and WHO guidelines on assessing the quality of herbal medicines concerning contaminants and residues. 41 To ensure the quality of the T chebula extract, pharmacognostical, physicochemical, and microbiological investigations were performed. In addition, physical properties were also assessed, including percent weight loss of sample, the total ash and insoluble-acid ash content. A microbiological analysis was conducted to verify the absence of aflatoxins, and to measure concentrations of heavy metals and other chemical substances. Evaluation of active phytoconstituents of T chebula was carried out in compliance with the previously published approaches. 42
Determination of the Bioactive Components of T chebula by HPLC
HPLC was used to analyze the potential bioactive components of the extract of T chebula. The test samples were separated using a Brownlee Spheri-5 RP-18 column, measuring 220 mm × 4.6 mm, with a 5 μm particle size supplied by PerkinElmer Inc. (MA, USA). An isocratic elution was carried out with acetonitrile + 1 mM phosphoric acid (H3PO4) and 1 mM H3PO4 (8:92), at a flow rate of 0.7 mL/min. The absorption wavelength for the T chebula extract was set at 216 nm, with an injection volume of 20 μL. In order to detect pharmacologically active components, peak area and retention time were compared with standard curves of varying concentrations of gallic acid and protocatechuic acid.
Determination of Total Phenolic Content
The total phenolic content of the T chebula extract was determined using the Folin-Ciocalteu approach.43,44 A combination of 10 mg/mL T chebula extract, 10% v/v Folin-Ciocalteu reagent, and 700 mM Na2CO3 solution was incubated for 2 h before being transferred to a 96-well microplate. Absorption was measured at 765 nm with a microplate reader. A standard curve of gallic acid concentrations ranging from 2 to 8 mg/L was constructed to calculate the amount of gallic acid (in g) in 100 g of T chebula extract.
Determination of Antioxidant Activity of T chebula Extract Using DPPH Radical Scavenging Assay
T chebula extract was evaluated for its ability to scavenge free radicals using a 1, 1-diphenyl-2-picrylhydrazyl (DPPH) assay.45,46 The experiment was conducted 3 times, each time with 3 mL of DPPH solution dissolved in methanol mixed with 200 μL of T chebula extract at concentrations ranging from 10-200 μg/mL in 50% methanol. The tubes were incubated at room temperature for 30 min in the dark, after which absorbance at 517 nm was measured using a Synergy HT, Bio-Tek microplate reader (Santa Clara, CA, USA). Gallic acid acted as the positive control, while 50% methanol was used as the vehicle control. The antioxidant activity of T chebula extract against DPPH radicals was calculated as follows:
Determination of Antioxidant Activity of T chebula Extract with FRAP Assay
The reducing ability of T chebula extract and Trolox was evaluated using a ferric ion reduction assay, as described previously. 47 The FRAP reagent was freshly prepared by combining 30 mL of 0.3 M sodium acetate buffer (pH 3.6), 3 mL of 10 mM TPTZ solution in 40 mM hydrochloric solution, and 3 mL of 20 mM ferric chloride hexahydrate (FeCl3.6H2O). Standard solutions of FeSO4.7H2O, ranging in concentration from 0.1 to 25 μmol/10 mL in 50% methanol, were used to determine the standard curve. One hundred µL of either T chebula extract or Trolox standard reference, ranging from a concentration of 2 to 6 mg/10 mL in methanol, was combined with 0.3 M acetate buffer (pH 3.6) and added to the FRAP reaction in a 96-well plate. The mixture was left to sit in the dark at room temperature for 30 min, before being measured for absorbance at 595 nm using a microplate reader. The antioxidant activity of 1 mg of T chebula extract was stated in terms of the equivalent concentration to that of ferric-TPTZ reduction ability (μmole per mg) calculated from the following equation: X = (Y-0.088)/0.1297 (R2 = 0.9986), where × is the equivalent concentration to that of ferric-TPTZ reduction capacity and Y is the absorbance of each sample measured in the FRAP assay (Supplemental Figure S1). Each experiment was replicated 3 times.
Determination of Cellular Antioxidant Activities of T chebula Extract
Cell Culture
Human monocytic U-937 cells (American Type Culture Collection, Manassas, VA, USA), in RPMI 1640 media were supplemented with 10% (v/v) heat-inactivated fetal bovine serum, 100 IU/mL of penicillin G, 100 μg/mL of streptomycin, and 2 mM L-glutamate, in a 5% CO2 environment at 37 °C.
Cellular Antioxidant Activities
The U-937 cells were suspended in an RPMI medium containing 2.5 ng/mL of phorbol-12-myristate-13-acetate (PMA) and incubated at 37 °C for 3 days in order to induce differentiation from human monocytic cells into active macrophage-like cells.
48
The differentiated cells were rinsed twice with Hank's balanced salt solution (HBSS) containing no phenol red. To evaluate intracellular peroxide levels, U-937 cells at a density of 2.5 × 105 cells/mL were exposed to different amounts of T chebula extract in a serum-free RPMI medium for 24 h at 37 °C. Intracellular ROS of U-937 cells treated with T chebula extract were determined by incubating with 10 μM of fluorescent dye carboxy-H2DCFDA in phenol red-free HBSS for 30 min in the dark at 37 °C. After that, the U-937 cells were exposed to hydrogen peroxide (H2O2) for 30 min in order to provoke oxidative stress. The presence of endogenous ROS was determined by analyzing fluorescence value before and after H2O2 induction using fluorescence spectroscopy with 485-nm excitation and 530-nm emission wavelengths (Synergy HT, Bio-Tek, CA, USA). The cells treated with H2O2 were used to measure stress and background fluorescence levels, and untreated cells in phenol red-free HBSS acted as a control. The ability of T chebula extract to inhibit the formation of ROS was determined according to the following equation:
Experimental Animals
Male MLAC: ICR mice (Mus musculus), approximately 6-8 weeks old and weighing between 25 and 40 g, were procured from the National Laboratory Animal Center (NLAC) at Mahidol University (Nakhon Phathom, Thailand). The mice were placed in polypropylene cages filled with sterilized corncob bedding and kept in an environment of 25 ± 1 °C, with a 50-70% humidity level, and a 12/12-h light-dark cycle. Before the experiments, a 7-day acclimatization period was provided to allow the animals to adjust to their new environment. During the acclimatization period, they were fed commercial laboratory rodent diets and had access to pathogen-free water ad libitum. The animals were then deprived of food overnight before being administered drugs, but still had free access to clean water. Sodium pentobarbital was used for euthanasia. 49 The experiment adhered to standard guidelines concerning the ethical use of animals in laboratories50‐53 and was approved by the Animal Ethics Committee of the Faculty of Medicine at Thammasat University (Protocol approval number: 0001/2006).
DMBA/TPA-Induced Skin Tumorigenesis
To evaluate the effectiveness of T. chebula extract against skin carcinogenesis induced by DMBA/TPA, an experiment was conducted using methods described previously. 54 In brief, 3 days before the experiment started, hair was shaved from a 2 × 4 cm area in the middle of the mice's backs. The 30 mice were then randomly divided into 5 groups of 6 mice each, as depicted in Figure 1. There was no repetition within each group. Group 1 was administered 100 μL of acetone twice a week for 20 weeks, as a control group. The other 4 groups (2-5) received a single topical application of 390 nmol of DMBA dissolved in 100 μL of acetone using a micropipette to initiate carcinogenesis. An hour after application of 390 nmol of DMBA, group 2-5 were treated with 4 nmol TPA in 100 μL of acetone using a micropipette. Seven days after DMBA application, the mice in group 3-5 were administered 1, 2, and 4 mg, respectively, of T chebula extract in acetone via micropipette twice a week for 20 weeks. The size of the nodules on the backs of the mice was accurately measured and recorded weekly. Following the completion of the experiment, the animals were put under deep anesthesia and the skin with tumors was removed and fixed in 10% neutral buffered formalin for 48 h. The upper layer of the skin and the tumors were then excised and stained with hematoxylin and eosin for microscopic analysis of their histopathology. The percentage of mice with tumors and the average number of tumors per mouse were used to assess the effects of T chebula extract on DMBA/TPA-induced tumorigenesis. The percentage of tumor-bearing mice was calculated by dividing the number of mice showing a tumor response by the total number of mice in each group and multiplying by 100. The average number of tumors per mouse was calculated by dividing the total number of tumors by the number of mice showing a tumor response. Additionally, the mean volume of the tumor mass was calculated using a formula (mean volume tumor mass = 4/3πr3) that considers the mean radius (r) of each tumor in mm.

Treatment schedule for assessing the effect of T chebula extract on the growth of tumors triggered by DMBA/TPA. Abbreviations: DMBA, 7,12-dimethylbenz[a]anthracene; TPA, 12-O-tetradecanoylphorbol-13-acetate.
Histopathological and Immunohistochemical Examinations
The tissue samples were preserved in 10% neutral-buffered formalin, processed, and embedded in paraffin blocks. A rotary microtome was used to prepare 4 µm thick section of the samples. Hematoxylin and eosin (H&E) stain was applied to the sections which were then viewed under a microscope using a digital camera system.
Statistical Analysis
Data are shown as mean ± standard deviation of mean (SD). IBM SPSS Statistics (version 22.0) was used to compare differences between the normal group, the standard control group, and the groups treated with T chebula through 1- or 2-way analysis of variance (ANOVA). Further comparison was made using the least significant difference (LSD) test. P-values less than .05 were considered statistically significant.
Results
Quality of T chebula Extract
The aqueous extract yield from T chebula fruits was 9.62% (w/w) of the dried fruit powder. The total polyphenol content was equivalent to 26.75 ± 1.02 g gallic acid per 100 g of water extract. Gallic acid standards were used to create a calibration curve. The calibration curve (y = 0.0327x + 0.0225) had a coefficient of determination (R2) of 0.9997, indicating high linearity within the studied concentration range. A physicochemical analysis of the extract was performed to evaluate its properties including total ash (6.37 ± 0.8%), acid insoluble ash (2.61 ± 0.12%), and loss on drying (6.76 ± 1.1%). The levels of tannins, gallic acid, total carbohydrates, and uronic acid were 26.75 ± 5.2%, 4.37 ± 0.81%, 19.63 ± 2.1%, and 5.39 ± 0.9%, respectively. No contamination with aflatoxin, heavy metals, or microbes eg, Enterobacteriaceae (Escherichia coli), Salmonella sp., Clostridium sp., and Staphylococcus aureus, was found (Supplemental Table S1).
HPLC Chemical Composition of T chebula Extract
High-performance liquid chromatography (HPLC) was used to examine the water extract of T chebula for the presence of gallic acid and protocatechuic acid. Figure 2A shows the appearance of the dried fruit of T chebula. The HPLC profile (Figure 2D) revealed the presence of gallic acid and protocatechuic acid, with retention times of 6.189 and 12.173 min, respectively, which correspond with the retention times of the standard compounds (Figure 2C). The amount of gallic acid was 2.43% w/w according to the calibration curve. These results indicate that gallic acid and protocatechuic acid are the major compounds in T chebula extract and likely contribute to its activity (Figure 2B). The calibration curve is shown in Supplemental Figure S2 and Table S2.

(A) External appearance of dried T chebula fruit. (B) Chemical structure of gallic acid and protocatechuic acid. (C) high-performance liquid chromatography (HPLC) chromatograms of gallic acid and protocatechuic acid. (D) HPLC chromatogram of T chebula extract.
Antioxidant Activities of T chebula Extract in DPPH Assay
The IC50 value of the T chebula extract in the DPPH assay was 109.0 ± 14.5 μg/mL, whereas the standard compound (gallic acid) had an IC50 of 8.5 ± 0.7 μg/mL, indicating that the standard (gallic acid) was significantly more potent in scavenging DPPH radical compared to the T chebula extract. The relative potency of T chebula extract compared to the standard was calculated to be 0.079 ± 0.010.
Antioxidant Activities of T chebula Extract in FRAP Assay
The antioxidant capability of T chebula extract was assessed using a FRAP assay which measures its capacity to reduce ferric ions to ferrous sulfate. The result for the T chebula extract was 2.39 ± 0.17 μmole of FeSO4/mg, approximately 29% of the activity of the Trolox standard (8.35 ± 0.16 μmole of FeSO4/mg). The relative antioxidant activity of T chebula extract was 0.29 ± 0.02 when compared to the Trolox standard.
Intracellular Antioxidant Activities of T chebula Extract on U-937 Cells
The carboxy-H2DCFDA assay was used to measure the influence of T chebula extract on H2O2-induced ROS production in human monocytic U-937 cells. T chebula extract was effective in suppressing the generation of ROS in the cells. The IC50 value of the extract was 0.53 ± 0.23 μg/mL, while the IC50 value of the standard gallic acid was 4.28 ± 1.32 μg/mL. This suggests that T chebula extract was more effective at reducing the production of ROS than the standard gallic acid.
Antitumor Activity on DMBA/TPA-Induced Tumorigenesis in Mice

The suppression of DMBA/TPA-induced mouse skin tumorigenesis by T chebula extract. (A) Total tumors per mouse. (B) Percentage of mice showing tumors (tumor-bearing mice). (C) Total tumor volume (mm3). (D) Tumor size increase (percent). Data, presented as mean ± SD, analyzed using 1-way ANOVA followed by LSD post-hoc test, demonstrate that the extract treatments had a statistically significant effect compared to the DMBA/TPA group (p < .05; n = 8). Abbreviations: DMBA, 7,12-dimethylbenz[a]anthracene; TPA, 12-O-tetradecanoylphorbol-13-acetate; LSD, least significant difference; ANOVA, analysis of variance.
Examination of tissue samples from the control group showed the skin to be normal with clear boundaries between the outermost layer, the epidermis, the dermis, and underlying subcutaneous tissue (Figure 4A). After twenty weeks of the study, the skin of the mice in the DMBA/TPA group had formed nodules which were identified as squamous cell carcinoma based on their histological characteristics (Figure 4B). The use of DMBA/TPA on the mouse skin caused a thickening of the keratinocytes and the over-production of epidermal cells, also known as hyperkeratosis. In the dermis layer, the DMBA/TPA-treated mice had abnormal squamous keratin cells, suggesting the presence of invasive or malignant carcinoma. Specifically, histopathological analysis showed that the skin thickness in the T chebula group decreased by 35% at week 20 compared to the DMBA-induced group without treatment. This suggests that T chebula extract has notable inhibitory effect on epidermal growth and hyperkeratosis induced by DMBA/TPA (Figure 4C). Together, these results suggest that T chebula extract can successfully inhibit skin tumorigenesis caused by DMBA/TPA in mice.

The effect of T chebula extract (4 mg) on DMBA/TPA-induced epidermal hyperproliferation as seen with H&E staining. Histopathology of the control group's normal skin architecture visible at 40 × magnification (scale bar = 100 μm) (A), DMBA/TPA-induced tumorigenesis in a mouse (B), and T chebula extract treated mouse (4 mg). Abbreviations: DMBA, 7,12-dimethylbenz[a]anthracene; TPA, 12-O-tetradecanoylphorbol-13-acetate; H&E, hematoxylin and eosin.
Discussion
Oxidative stress, caused by the excessive production of ROS, plays a crucial role in various pathophysiological conditions, including cardiovascular diseases, diabetes, and cancers.55‐57 An imbalance of ROS can trigger mutations and damage proteins and nucleic acids, resulting in abnormal DNA linkage and aberrant gene expression.58‐60 In order to maintain redox homeostasis, antioxidants act as scavengers of ROS and free radicals. 61 There is ongoing research on the relationship between antioxidants and tumor growth, 62 with some studies suggesting that antioxidants can prevent cancer by neutralizing harmful free radicals in the body. 63 However, other studies have shown that high levels of antioxidants may actually promote cancer growth. 64 This study determined the antioxidant capacity of T chebula water extract through DPPH and FRAP assays and evaluated its antitumor effect in a mouse model induced with DMBA/TPA.
The antioxidant capabilities of the T chebula extract were analyzed using DPPH assay. 65 The water extract was able to effectively scavenge DPPH radicals in a concentration-dependent manner (IC50 109 ± 14.5 μg/mL). Previous studies also found that the ethanolic extract of the fruit of T chebula that was cultivated in India had strong DPPH scavenging activity, with an IC50 value of 42.14 μg/mL. 66 In addition, Birur Eshwarappa et al (2015) showed that leaf gall extracts of T chebula exhibited DPPH scavenging activity with IC50 values of 274 ± 2, 201 ± 3, 143 ± 6, and 96 ± 2 μg/mL when extracted with light petroleum, chloroform, aqueous, and ethanol solvents, respectively. 67 Chen et al (2011) reported that triethylchebulate, an aglycone isolated from T chebula, had a strong free-radical scavenger effect with an IC50 value of 2.4 × 10−5 M 68 , while Lee et al (2005) reported that the aqueous extract of the fruit of T chebula scavenged radical DPPH with an EC50 of 127.1 ± 1.8 μg (dry matter)/mL. 69 Suksaeree et al 12 investigated the synergistic antioxidant activity of T chebula, T arjuna, T bellirica, and Phyllanthus emblica using response surface analysis. T chebula had the weakest antioxidant activity among the 4 species studied, with an IC50 value of 14.50 μg/mL. In contrast, the current study observed a different IC50 value for T chebula extract. The difference in IC50 values between Suksaeree et al and this study could be due to several factors. One possible factor is the different extraction methods to prepare the plant samples. The extraction method used can affect the amount and composition of the bioactive compounds extracted, which could, in turn, affect the antioxidant activity. Additionally, the prevalence of the plant samples used in this study and in previous studies was different. Even within the same species, variations in geographical location, growing conditions, and harvest time can affect the composition of bioactive compounds and antioxidant activity. Furthermore, differences in experimental conditions such as temperature, pH, and duration of reaction can also influence the IC50 values obtained. Therefore, it is important to consider these factors when comparing the results of different studies. In this study, we performed an accurate and simple FRAP assay to confirm the antioxidant activity of T chebula extract. The FRAP value of T chebula extract was less than that of Trolox, a result similar to previous findings that the conventional reflux extraction of T chebula fruit definitely has lower ferric-reducing antioxidant power than ascorbic acid. 70
In order to ascertain the inhibitory effect of T chebula water extract on intracellular ROS, we conducted an assay using the fluorescent dye 2, 7-DCF-DA. 71 Our study found that T chebula extract suppressed ROS production in H2O2-induced human monocytic U-937 cells. This is consistent with previous findings that T chebula extract, a source of polyphenols, tannins, and triterpenes, showed a marked decrease in intracellular ROS production in H2O2-induced human leukemia monocytic THP-1 cells 72 and in H2O2-induced rat pheochromocytoma PC12 cells. 73 It is noteworthy that the antioxidant effects of T chebula extract are mediated by its polyphenol compounds and flavonoids, 32 such as 2,4-chebulyl- β-D-glucopyranose, 1,6-di-O-galloyl-β-D-glucose, 74 quercetin, 75 gallic acid, 17 ellagic acid, 18 chebulinic acid, casuarinin, and chelanin. 19 One potential limitation of this intracellular ROS assay in this study is that the experiments were conducted in vitro using cell lines. Although these assays provide valuable information about the potential biological activity of T chebula extract, they do not necessarily translate to the same effects in vivo in human subjects. Therefore, further research, eg, animal and/or clinical studies, is needed to confirm the potential health benefits of T chebula extract in humans.
The effect of T chebula water extract on suppressing carcinogenesis was investigated using a mouse model with DMBA/TPA-induced skin tumorigenesis. Our study found that the water extract significantly reduced tumor incidence, volume, and number per mouse. Similarly, a previous study showed that T chebula extract blocked nickel chloride-induced tumorigenesis in male Wistar rats. 76 In another study, T chebula aqueous extract was evaluated as a chemopreventive treatment for hepatocellular carcinoma in rats and was found to significantly decrease lipid peroxidation and pathophysiological marker enzyme levels, as well as to increase enzymatic antioxidant status. 34 Furthermore, phenolic-, triterpenoid-, and glucoside-compounds isolated from methanol extract of the gall of T chebula had shown inhibitory effects against melanogenesis, Epstein-Barr virus activation, TPA-induced inflammation, and cancer cell cytotoxicity. 33 The water extract may be more suitable as well as being safer for use on the skin than extracts obtained using organic solvents. In terms of safety, our previous study on the acute and chronic toxicities of the water extract derived from the dried fruits of T chebula indicated that oral administration of the same water extract to both male and female rats had no acute or chronic toxic effects. 77 The current study observed inhibitory effects on skin tumor promotion in an in vivo 2-stage mouse-skin carcinogenesis experiment. Additionally, histopathological analysis showed that the T chebula extract was effective in reducing the excessive thickness of the epidermis and the number of abnormal cells in the dermis layer which are indicative of the presence of malignant carcinoma. Therefore, we propose that the antitumorigenesis effects of T chebula extract are likely due to its high antioxidant activity. Moreover, it is very useful and effective for skin care: a standardized T chebula fruit extract has previously been reported to be efficacious in the treatment of inflammation, 66 photodamage, skin ageing, and wound healing, 78 which may also contribute to its antitumorigenesis effects.
Our HPLC study indicated that gallic acid and protocatechuic acid are the 2 major active components of T chebula extract, both of which are known to possess antioxidant and anti-inflammatory properties. 66 It is well known that gallic acid (3,4,5-trihydroxybenzoic acid), which contains 3 adjacent hydroxyl groups, has several pharmacological effects, including antioxidant, antitumorigenic, and cytotoxic properties, which have been demonstrated in both laboratory studies and in living organisms. 79 Additionally, it has been shown that derivatives of gallic acid such as ethyl gallate, 2,3,4-trihydroxybenzoic acid, and ellagic acid are able to induce apoptosis in KATO III cells from human stomach cancer and COLO 205 cells from colon adenocarcinoma. 80 Protocatechuic acid (3,4-dihydroxy benzoic acid), a phenolic compound present in T chebula extract, has antioxidant and chemopreventive properties which directly block the binding sites between carcinogens and DNA molecules. 81 Therefore, in addition to its triterpenoid compounds, T chebula extract also potentially has high antioxidant and antitumorigenic effects due to its active compounds, including tannins and related phenolic compounds.
Conclusions
The current study suggests that T chebula water extract effectively suppresses intracellular ROS and scavenges antioxidants as measured by DPPH and FRAP assays. Moreover, 4 mg of T chebula extract can significantly decrease skin thickness as well as the incidence, size, and number of tumors in mice with DMBA/TPA-induced skin tumorigenesis. This study provides insights into the antioxidative activities and antitumorigenesis effects of T chebula water extract. Further clinical study is needed to establish the level and efficacy of these activities in humans.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231176925 - Supplemental material for Antioxidant and Antitumorigenic Activities of the Standardized Water Extract From Fruit of Terminalia chebula Retz. var. chebula
Supplemental material, sj-docx-1-npx-10.1177_1934578X231176925 for Antioxidant and Antitumorigenic Activities of the Standardized Water Extract From Fruit of Terminalia chebula Retz. var. chebula by Mingkwan Na Takuathung, Ariyaphong Wongnoppavich, Kanjana Jaijoy, Noppamas Soonthornchareonnon and Seewaboon Sireeratawong in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank the Faculty of Medicine, Chiang Mai University, and the National Research Council of Thailand for their financial support. We would also like to express our sincere gratitude to Dr Lamar Robert for his assistance in proofreading the manuscript.
Author Contributions
Conceptualization was done by M.N., A.W., K.J., N.S., and S.S.; methodology was designed by K.J. and S.S.; software was handled by M.N; validation was carried out by M.N., A.W., and S.S.; formal analysis was performed M.N; investigation was carried out by M.N and S.S; re-sources were handled by S.S.; data curation was done by M.N., A.W., K.J., N.S., and S.S.; writing—original draft preparation was done by M.N; writing—review and editing was done by M.N., A.W., K.J., N.S., and S.S.; visualization was done by M.N; supervision was done by S.S.; project administration was handled by S.S.; funding acquisition was obtained by S.S. All authors have read and agreed to the published version of the manuscript.
Data Availability
The data that support the findings of this study are available from the corresponding author, [SS], upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by Faculty of Medicine, Chiang Mai University, and by the National Research Council of Thailand. The funding sources had no role in the design of the study including the collection, analysis, and interpretation of the data; the preparation, review, and approval of the manuscript; or the decision to submit this manuscript for publication.
Ethical Approval
This study was approved by the Animal Ethics Committee of the Faculty of Medicine, Thammasat University (Protocol approval number: 0001/2006).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the International and Thai National Guidelines for Ethical Conduct in the Care and Use of Animals of Thammasat University, Thailand and were approved by the Animal Ethics Committee of the Faculty of Medicine, Thammasat University, Bangkok, Thailand.
Informed Consent
There were no human subjects in this study and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.