Abstract
Purification of the petroleum ether and ethyl acetate fractions from Ludwigia hyssopifolia yielded 9 natural products, ethyl gallate (
Ludwigia hyssopifolia is widely distributed in Guangxi Zhuang Autonomous Region, China. It has the effects of relieving fever, swelling, and pain, and is used to treat diseases such as dysentery, infectious hepatitis, and edema.
1
Pharmacological studies have shown that the plant exhibits a variety of biological activities including anticancer, antibacterial, antidiarrheal, and anti-ulcer activities.
2
-4
Phytochemical studies have shown that L. hyssopifolia contains piperine,
3
palmitic acid, isovanillin, β-sitosterol, stigmasterol-3-O-β-d-glucopyranoside, gallic acid, ethyl gallate, oleanolic acid, 2,4,6-trihydroxybenzoic acid, ursolic acid,
5
kaempferol, ginsenoside Rb1,
6
6β,24-hydroxy tormnetic acid, xanthyletin, (+)trans-decursidinol, β-sitosterol-β-d-glucopyranoside, 6β,23-hydroxy tormentic acid, 23-hydroxy tormentic acid, and 6β,23-hydroxy tormentic acid.
7
In the present study, 7 known phenols and 2 known macrolides were isolated from L. hyssopifolia. On the basis of spectroscopic data (Figure S1-S25), they were identified as ethyl gallate (
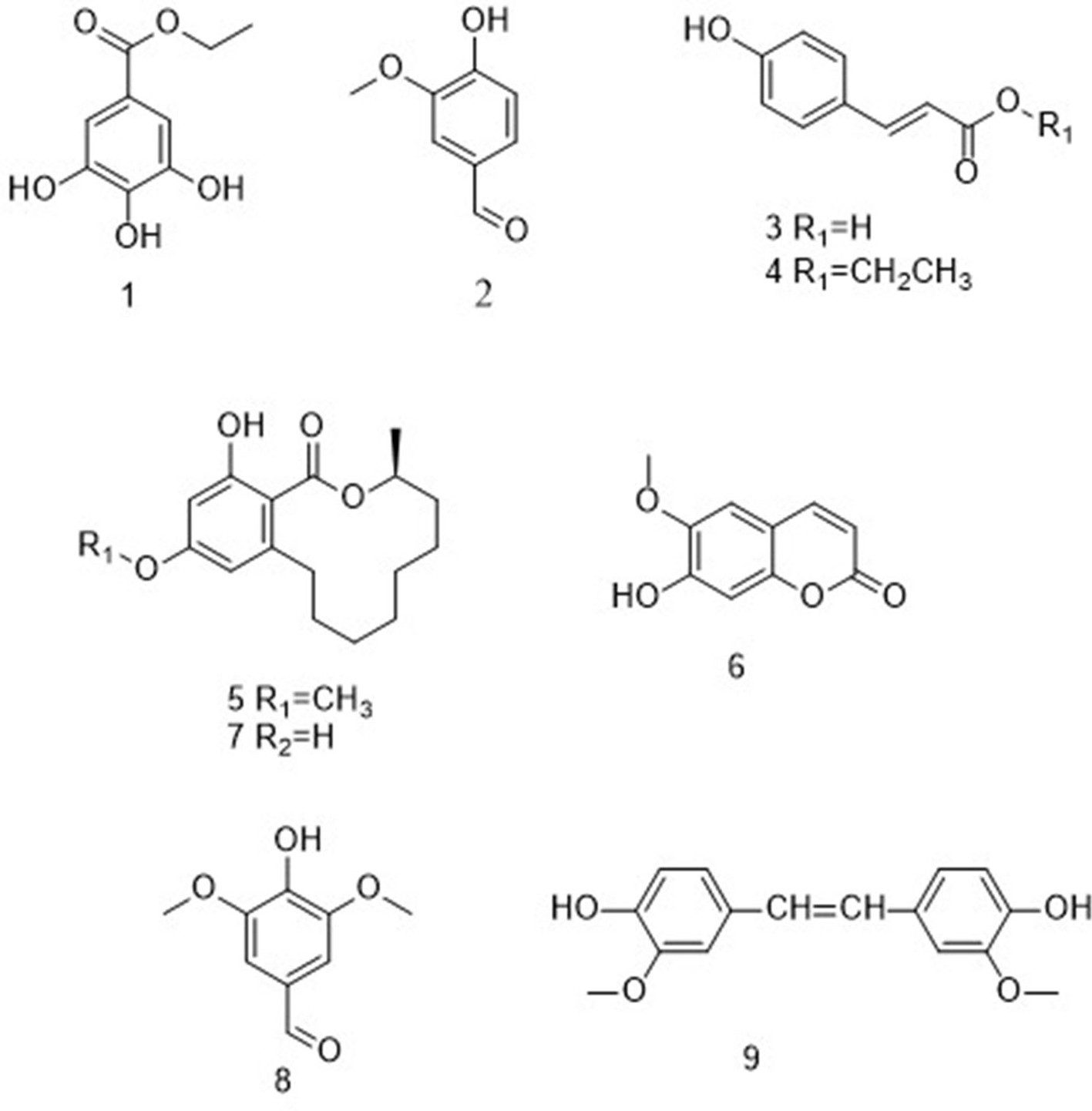
Structures of compounds 1 to 9.
Cytotoxic Activity of Compounds 1 to

Compounds
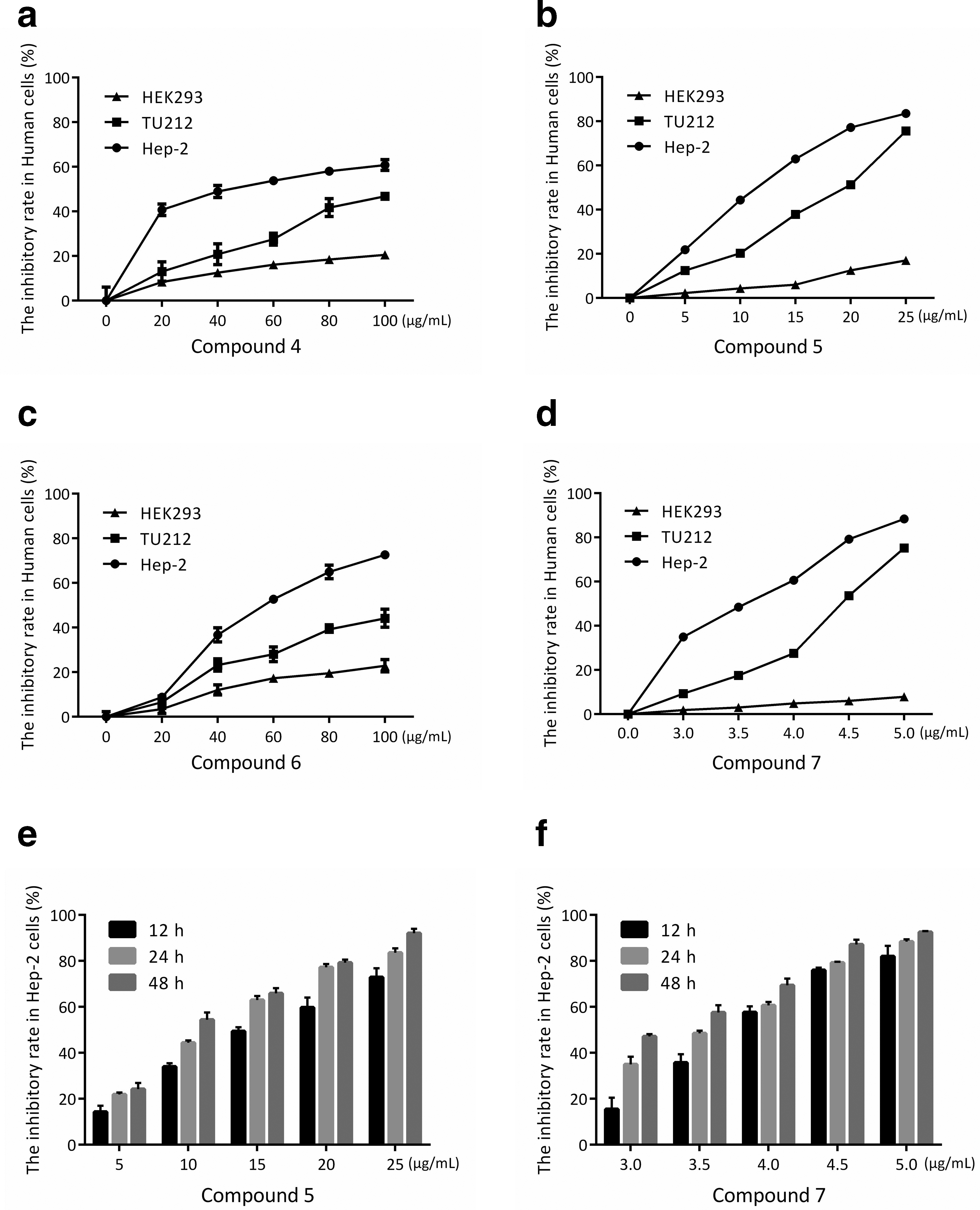
Compounds 4-7 inhibited human laryngeal cancer cell proliferation. (a-d) Hep-2, TU212, and HEK293 cells were treated with indicated concentrations of compounds 4-7 for 24 h and determined by MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. (e, f) Hep-2 cell line was treated with indicated concentrations of compounds 5 and 7 for 12, 24, and 48 h and determined by MTT assay.
Cytotoxic Activity of Compounds 5 and 7 Against Hep-2 (IC50, μg/mL; mean ± SD; N = 3).

The morphological changes of Hep-2 cell line treated with different concentrations of compounds

Observation of morphological changes in Hep-2 cell line. (a, c) The morphology of the cells treated with compounds 5 and 7 was photographed under an inverted-phase contrast microscope. (b, d) The morphology of the cells treated with compounds 5 and 7 was photographed under a fluorescence microscope. The cell nuclei were stained for Hoechst 33258 and the apoptotic bodies were indicated by yellow arrows.
In order to examine whether compounds

Compound 5 induced apoptosis of Hep-2 cell line. (a) Hep-2 cells were treated with compound 5 for 24 h, then stained with Annexin V/propidium iodide staining and evaluated by flow cytometry. (b) Hep-2 cells were treated with compound 5 for 24 h, then stained with Annexin V/propidium iodide staining and observed by fluorescence microscope. Annexin V-fluorescein isothiocyanate fluorescence was green and propidium iodide fluorescence was red.

Compound 7 induced apoptosis of Hep-2 cell line. (a) Hep-2 cells were treated with compound 7 for 24 h, then stained with Annexin V/propidium iodide staining and evaluated by flow cytometry. (b) Hep-2 cells were treated with compound 7 for 24 h, then stained with Annexin V/propidium iodide staining and observed by fluorescence microscope. Annexin V-fluorescein isothiocyanate fluorescence was green and propidium iodide fluorescence was red.
According to the anticancer mechanisms, regulating apoptotic proteins is an attractive therapeutic strategy for laryngeal cancer.
18
Therefore, we investigated the effects of compounds
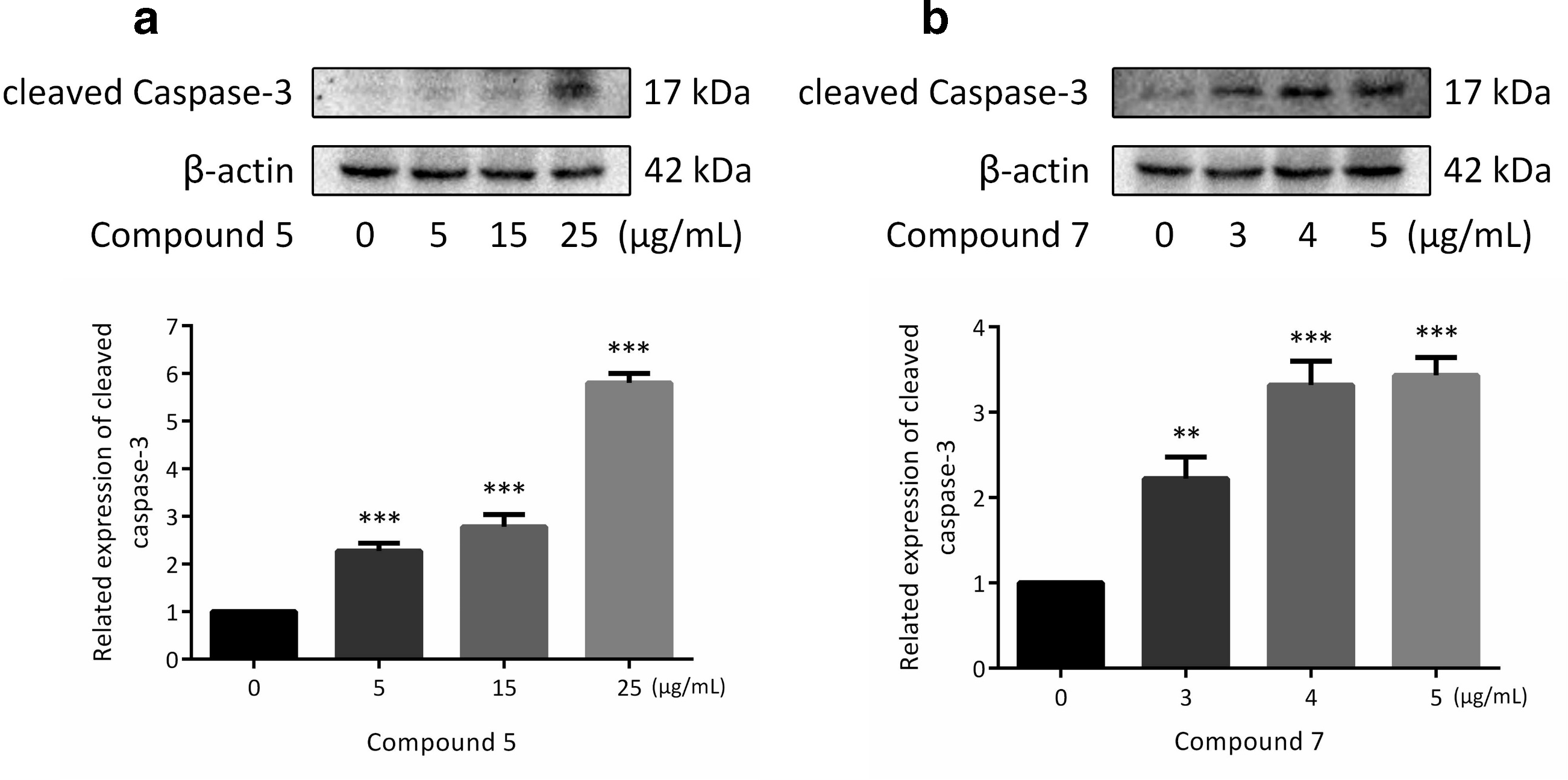
Effects of compounds 5 and 7 on the apoptosis regulatory protein of Hep-2 cell line. (a, b) The related expression of cleaved caspase-3 was detected in Hep-2 cells treated with compounds 5 and 7 by western blotting. β-actin was used as a control (*P < 0.05, **P < 0.01, or ***P < 0.001).
In conclusion, 7 known phenols and 2 known macrolides were isolated from L. hyssopifolia. On the basis of the spectroscopic data, they were identified as ethyl gallate (
Experimental
General Experimental Procedures
Semipreparative High Performance Liquid Chromatography (HPLC) was carried out on a Waters 2535 HPLC connected with a 2998 Photodiode Array Detector, a 2707 Autosampler (Waters) and a Waters Sunfire C18 column (5 µm, 10 × 150 mm) (Waters, Ireland). Optical rotations were measured on a Perkin-Elmer polarimeter 341. Infrared Spectrophotometry (IR) spectra were determined on a Nicolet Magna FT-IR 750 spectrometer (ν max in cm−1). The NMR spectra were recorded on the Bruker DRX-600 NMR spectrometer for 1D and 2D NMR. Direct injection ESIMS and HP High Performance Liquid Chromatography-Photo-Diode Array-Electrospray Ionization Mass Spectrometry (HP LC PDA-ESIMS) analyses were recorded on a Waters ACQUITY SQD MS system (Waters, United States) connected to a Waters 1525 HPLC with a 2998 Photodiode Array Detector (Waters, United States). Commercial silica gel (Qing Dao Hai Yang Chemical Group Co., 200-300 and 300-400 meshes) was used for column chromatography. Precoated silica gel plates (Yan Tai Zi Fu Chemical Group Co., GF-254) were used for analytical Thin Layer Chromatography (TLC).
Plant Material
The whole plants of L. hyssopifolia were collected from Luchuan County, Yulin, Guangxi Zhuang Autonomous Region, China, in August 2016. The plant was identified as L. hyssopifolia (G. Don) Exell by Professor Songji Wei of Guangxi University of Chinese Medicine, China. A voucher specimen (No. SC0864) has been deposited in the Herbarium of South-Central University for Nationalities, Wuhan, China.
Extraction and Isolation
Dried whole plants of L. hyssopifolia (16.5 kg) were ground and then extracted sequentially by maceration with 80% EtOH 3 times (16 L each, 4 days) at room temperature. The solvent was evaporated under reduced pressure to obtain a crude extract (1403.4 g). The extract was suspended in warm water and then partitioned successively with petroleum ether, ethyl acetate, and n-butyl alcohol to afford PE fraction (120.4 g), EtOAc fraction (474.6 g) and n-BuOH fraction (251.9 g), respectively. The PE fraction (85.0 g) was subjected to D101 macroporous resin column chromatography (2500 g, Sinopharm Chemical Reagent Co., Ltd., Shanghai, China) eluted with 10%, 30%, 50%, 70%, 80% and 95% aq. ethanol in a gradient manner to give 8 fractions (A1-A8). A2 (720 mg) was purified on a Sephadex LH-20 column eluting with MeOH (containing 0.1% formic acid) to afford 5 fractions (A2.1-A2.5). A2.3 (252 mg) was further purified by semipreparative HPLC (CNCH3-H2O, 15%:85% → 70%:30%, 20 minutes, containing 0.1% formic acid in both phases) to afford compounds
Cell Culture
Human laryngeal cancer cell lines Hep-2 and TU212 and human embryonic kidney cell line HEK293 were obtained from American Type Culture Collection (ATCC). Hep-2, TU212, and HEK293 cells were grown in Dulbecco's Modified Eagle Medium (DMEM) medium (Sigma-Aldrich, St. Louis, MO, United States), supplemented with 10% fetal bovine serum, 1% penicillin and streptomycin (Hyclone, Logan, UT, United States). Cells were cultured in an incubator containing a humidified atmosphere with 5% CO2 at 37°C.
MTT Assay
The MTT method was used to test the cytotoxicity of compounds
Morphological Changes of Apoptosis
The cells were seeded in 6-well plates and incubated for 24 hours to confluence, then exposed to different concentrations of compound
Annexin V-FITC/PI Double Staining
The cells were seeded in 6-well plates and incubated for 24 hours to confluence, then exposed to different concentrations of compound
Western Blotting
The cells were seeded in 10 cm cell culture dishes and incubated for 24 hours to confluence, then exposed to different concentrations of compound
Supplemental Material
Supplementary data - Supplemental material for Cytotoxic Compounds From Ludwigia hyssopifolia
Supplemental material, Supplementary data, for Cytotoxic Compounds From Ludwigia hyssopifolia by Jinyan Zhang, Chang Liu, Jianhua Wei, Bing Li, Xin Zhan, Yunqing Li, Ji Hao, Rumei Lu and Xinzhou Yang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was financially supported by the grants of Innovation Project of Graduate Education of Guangxi University of Chinese Medicine(No. YCSZ20190014), Guangxi First-class discipline (No. 2019XK114), the Applied Basic Research Programs of Science and Technology Commission Foundation of Wuhan (No. 2017060201010217), and National Natural Science Foundation of China grants (81774000).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
