Abstract
Background of the study
This study established the antidiabetic efficacy of Alternantera brasiliana in vitro and in vivo, identified the most active fraction and profiled the chemical constituents of the most active fraction.
Methodology
The leaf of A. brasiliana was air-dried, pulverized and extracted with methanol and evaluated for its α-amylase activity at different concentrations using in vitro method. The in vivo antidiabetic activity of the extract and glibenclamide was evaluated in glucose loaded and streptozotocin-induced diabetic rats using various doses, while the most active partitioned fraction was identified using glucose loaded method followed by identification of its chemical constituents through GC-MS analysis.
Results
The extract elicited good α-amylase inhibitory potential with IC50 = 0.45 mg/mL. Also, the 50 mg/kg dose demonstrated the highest percentage blood glucose level reduction and significantly (P > 0.05) comparable to glibenclamide (5 mg/kg) from 0.5 to 4 h. The streptozotocin-induced antidiabetic assay showed that the 50 mg/kg demonstrated better activity than glibenclamide (5 mg/kg) and was significantly comparable on days 10 and 14. The aqueous methanol fraction was the most active fraction with 53% blood glucose level reduction at 4 h. The secondary metabolite profiling identified 30 chemical compounds whereby 1H-Indole-2-carboxylic acid, 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-, isopropyl ester, Benzo[h]quinoline, 2,4-dimethyl-, thymol and 13,5-trioxane as the major constituents.
Conclusion
The antidiabetic activity demonstrated by the A. brasiliana extract established its antidiabetic efficacy
Keywords
Introduction
Diabetes mellitus is a multifarious disorder of carbohydrate, fat and protein, which contributes tremendously to the decrease in life expectancy of the world population.1,2 The disease characterized by consistently high blood glucose levels has been projected to be the major cause of death in different regions of Africa.2,3 A typical diabetic condition is caused by insufficient insulin release, insensitivity of the body cells to insulin or failure in pancreatic beta-cell effectiveness. 4 Globally, a surge in diabetes prevalence was recorded with 151 million people diabetic in 2000 to 537 million in 2021 and about 784 million people have been predicted to become diabetic by 2045, with more prevalence in Africa, South America and the Asia continent.5,6 Type 2 and type 1 diabetic patients contributes more than 90-95 and 5%-10% of the total diabetes mellitus cases reported worldwide. The immune cell-mediated infiltration of the pancreatic beta cells causes its destruction thereby causing to Type 1 diabetes, while type 2 diabetes occurs in older populations with sedentary lifestyles, genetic mutation, family history, obesity, overweight, damage to pancreas and insulin resistance.5,7-9
The severe complications of diabetes like heart attack, stroke, blindness and death have resulted in the development of several conventional medications which has proven effective over the years.10-12 However, the usage of these drugs is presented with issues of affordability for low to middle-income countries inhabitants, serious adverse effects and patients’ compliance to the therapeutic routines.13,14 Considering the increasing need to achieve effective glycaemic control to prevent complications, efforts are consistently made to identify new therapeutic arsenals and targets. Medicinal plants are recognized as effective sources of drugs and also act as therapeutic agents against various life-threatening ailments due to the abundance of pharmacologically active secondary metabolites.15,16
Ethnomedicinally, plants belonging to the Alternantera genus and Amaranthaceae family are known to elicit excellent antidiabetic efficacy.15,16 Alternantera brasiliana (Amaranthaceae), also known as Brazil joy weed, is a medicinal plant native to sub-tropic and tropical regions of Australia, South America and Africa, including Nigeria.17-19 The plant is used traditionally in the treatment of pain, wound, tumor, cough, night blindness, hazy vision, dysentery, post-natal complication, diarrhea and the management of diabetes mellitus (personal communication) and treatment.13,19 Pharmacologically, the lymphocyte has proliferative, analgesic, wound healing, antibacterial, anti-inflammatory, antioxidant and antiviral activities.18-27 Despite the rich pharmacological studies performed on various extractives of A. brasiliana, its antidiabetic ethnomedicinal claim is yet to be investigated. Therefore, this study investigates the antidiabetic effect of the extract and fractions of A. brasiliana and added to the knowledge of its chemistry through identification of the chemical constituents of its active fractions.
Methodology
Rotary evaporator (RE301/601/801 model, Yamato Scientific America, Inc., U.S.A), chiller (Churchill, Instrument Co. Ltd, U.K), vacuum pump (MB 338618 model, Edwards High Vacuum Int., England), oven (Hearson & Co. Ltd, London), Mettler electronic weighing balance (AB 54 model, Mettler Toledo, U.S.A), ultraviolet (UV) lamp (254 and 366 nm) (Grant Instrument, U.K), Oral cannula, ACCU-CHEK Glucometer (model GB 11558973, Roche, Germany) with ACCU-CHECK test strips (Roche, Germany), UV spectrophotometer, Dutrao (Model SM 600, Shang Yhai Yong Chuang Medical Instrument Co. Ltd) spetrophotometric microplate reader, sodium citrate, citric acid, streptozotocin, acarbose, Glibenclamide® (Sigma-Aldrich Co. LLC, U.S.A), and sodium hydroxide.
Plant Collection
The leaves of A. brasiliana were collected on May 15, 2024 by Peter T. Ayoade on Obafemi Awolowo University campus, Ile-Ife, Nigeria. The plant material was identified and authenticated by Mr Ademoriyo and a voucher specimen (IFE 18300) was deposited at the Department of Botany Herbarium, Obafemi Awolowo University.
Extraction and Partitioning
The leaves of the plant were air-dried, pulverized and 100 g of the powdered material was extracted with methanol for 48 h. The methanol solution was filtered and concentrated using rotavapour at 40 oC to afford an extract (15 g). The methanol extract was resuspended in 100 mL of methanol/water (1:1/ v:v) and partitioned into n-hexane (4 × 100 mL), dichloromethane (3 × 100 mL), ethyl acetate (5 × 100 mL) to afford their corresponding fractions namely n-hexane (4.2 g), dichloromethane (3.3 g), ethyl acetate (2.7 g) and aqueous methanol (3.8 g).
Alpha Amylase Activity
The assay was evaluated using modified procedure of McCue et al
28
A volume of 100 µL of A. brasiliana extract or acarbose (positive control) and 100 µL of 0.02 M phosphate buffer (pH 6.9 with 0.006 M sodium chloride) containing α-amylase from Aspergillus oryzae (0.5 mg/mL) were added to each tube and incubated at 25 °C for 10 min. After pre-incubation, 100 µL of 1% starch solution in 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M sodium chloride) was added to each tube. The reaction was stopped with 200 µL of dinitrosalicylic acid colour reagent (1.0 g of 3,5-dinitrosalicylic acid, 20 mil of 2 M NaOH and 30 g of sodium potassium tartarate in 100 ml distilled water). The test tubes were incubated in a boiling water bath for 5 min, then cooled to room temperature. The reaction mixture was then diluted by adding 1.5 mL distilled water, and absorbance was measured at 540 nm using a microplate reader (SpectraMax, USA) by adding 200 µL in 96-well plates. The α-amylase inhibitory activity was expressed as % inhibition and also the concentrations of extract/Acarbose resulting in 50% inhibition of enzyme activity (IC50) were determined.
28
Antidiabetic Activity
Health male and female Wistar albino rats aged 8-10 weeks and weighing between 120-150 g were bred under standard condition (relative humidity 56%, temperature 27 ± 3 oC), standard feed and water at the animal house of the Department of Pharmacology, Obafemi Awolowo University, Ile-Ife. The animal experimental protocol was approved by the Health Research and Ethics Committee of the Institute of Public Health, Obafemi Awolowo University, Ile-Ife, Nigeria. All procedures adhered to the guidelines outlined in the NIH Guide for the care and use of laboratory animals (NIH Publication, No. 83-123, revised 1985). Additionally, the antidiabetic study was approved by the board of Post graduate committee, Obafemi Awolowo University with registration number: SCP13/14/H/0598. The rats were acclimatized for a minimum of two weeks before the commencement of experiments.
Acute Toxicity
Rats weighing between 120 and 150 g were fasted for 24 h before receiving oral doses of A. brasiliana methanol extract (ABE) (10-5000 mg/kg). The experiment was conducted in two phases. Nine (9) rats total were used in Phase 1 of the trial. The rats were split into three groups of three rats each, and A was given to them at doses of 10, 100, and 1000 mg/kg. The animals were then monitored for toxicity and/or mortality for a full day in each group. Eight (8) rats were used in the phase 2 test, which was based on the findings of the phase 1 test. The rats were split into four groups of two each. The extract was administered at doses of 1000, 1600, 2900, and 5000 mg/kg to each group, respectively. For a whole day, the animals were monitored for toxicity and/or mortality. The geometric mean of the doses that produced 100% lethality and no lethality at all was utilized to compute the LD50. 29
Glucose Lowering Effect of Extract and Fractions of A. brasiliana
Prior to the glucose lowering experiment, normoglycaemic rats were fasted for 18 h. The fasted rats were orally induced with glucose solution (10 g/kg) and their blood glucose level was checked after 30 min to ascertain their hyperglycemic state. Rats that were considered hypergycaemic ≥7 mmol/L were divided into five groups of five rats and administered with extract (50, 100, 200 and 400 mg/kg), fractions (50 mg/kg), glibenclamide (5 mg/kg) (positive control) and 1% tween 80 (negative control). The blood glucose level of the grouped rats were checked by taking blood from the tip of the tail and dropped on the glucometer strip after 0.5, 1 h, 2 h and 4 h of administration.3,30
Effect of Methanol Extract of A. brasiliana on Streptozotocin-Induced Diabetic Rats
Normoglycaemic rats used for the streptozotocin-induced diabetic study were fasted overnight before the diabetes induction. The fasted rats were intraperitonially induced with streptozotocin prepared in 0.1 M sodium citrate buffer (pH = 4.5) solution. The hyperglycemic condition of the rats was confirmed after 72 h. Rats with fasting blood glucose level of 14 mmol/L or more were considered diabetic, selected and divided into three groups of 12 rats each. They were administered with extract (50 mg/kg), glibenclamide (5 mg/kg) and 1% tween 80 in normal saline (negative control) for 15 days. The fasting blood glucose level of the treated and non-treated was checked on day 1, 4, 7, 10 and 14, respectively.3,31,32
Statistical Analysis
The data obtained from the study were analysed with GraphPad Prism 5.01 software package. They were expressed as the mean ± SEM for the number (N) of animals in the group. Analysis of variance (ANOVA) was first used followed by Bonferroni t-test to determine the source of significant differences for all determinations and P < 0.05 were considered significant.
GC-MS Profiling
The aqueous methanol fraction of A.brasiliana underwent GC-MS analysis using a Varian 3800/4000 gas chromatograph mass spectrometer featuring an Agilent fuzed silica capillary CP-Sil 5 CB column (30 m × 0.25 mm i.d.). This system was connected to a Varian 4000 mass spectrometer operating in the Electron Impact (EI) mode (70 eV; m/z 30-1000 amu; source temperature 230 °C and a quadruple temperature 150 °C). The initial temperature was maintained at 150 °C, reaching a maximum of 300 °C. A 1 μL sample was injected with split mode (10:1). Nitrogen gas served as the carrier gas at a flow rate of 0.8 mL/min, and the total run time was set at 40 min.
For identification of phytochemical components, the National Institute Standard and Technology MS library (NIST-MS library 2009) were utilized, comparing the obtained spectrum through GC–MS to identify compounds present in the samples. Retention indices (RI) were determined in relation to a homologous series of n-alkanes (C6–C28) on the CP-Sil 5 CB column under the same chromatographic conditions. The relative concentrations of components were determined through peak area normalization, and no response factors were calculated.
All samples and their replicates were injected as a single batch in a random order to distinguish technical variations from biological ones. Moreover, the pooled samples prepared were employed as quality controls (QCs) and injected at regular intervals during the analytical run. This approach facilitated the generation of a dataset for assessing repeatability.
Results
α-Amylase Inhibitory Activity of A. brasiliana Methanol Extract
Figure 1 showed the α-amylase inhibitory activity of the methanol extract of A. brasiliana and acarbose (standard drug) as tested at varying concentrations. A. brasiliana extract demonstrated the highest percentage inhibition of the enzyme at 1.0 mg/ml with 75.29% and IC50 value of 0.45 mg/mL, while the highest percentage inhibition of acarbose was obtained at 79.31% and IC50 value of 0.17 mg/mL.

Alpha-amylase inhibitory activity of A. brasiliana methanol extract.
Acute Toxicity
The acute toxicity study showed no mortality up to 5000 mg/kg during observation, indicating that the median lethal dose for the extract was greater than 5000 mg/kg. Also, the rats showed no behavioural or physical abnormalities throughout the period of observation.
Glucose Lowering Activity of the Extract of A. brasiliana
Table 1 shows the results of the glucose lowering effect of the methanol extract of A. brasiliana on glucose induced rats. The results obtained for the negative control group showed consistent increase in the blood glucose level up to the fourth hour. Treatments with 50 mg/kg of the extract gave 40.67% after 4 h of administration, followed by the 200 mg/kg which gave 38.50%, 100 mg/kg at 28.70% and 400 mg/kg at 25.96%, respectively. Also, the activity of the extract at all tested doses showed no significant difference (P > 0.05) with the glibenclamide (5 mg/kg) after 4 h, while only 50 mg/kg gave time dependent blood glucose lowering activity similar to the positive control.
Effect of the Methanol Extract of A. brasiliana on Glucose Loaded Rats.

Data show the mean ± SEM blood glucose levels at the different time interval expressed as percentages of levels at 0 h (To), n = 5. Values in parentheses represent the percentage reductions in blood glucose levels relative to negative control for each time point. Values with different superscripts (a and b) within columns are significantly different (P < 0.05, one way analysis of variance followed by Bonferroni t test). ABE: Extract of A. brasiliana, GLI: Glibenclamide (positive control).
Antidabetic Effect of A. brasiliana Methanol Extract in Streptozotocin-Induced Diabetic Rats
The blood glucose levels of treated and untreated diabetic rats were measured on days 1, 4, 7, 10 and 14 are presented in Table 2. The diabetic rats treated with 50 mg/kg of A. brasiliana methanol extract gave blood glucose levels that were not significantly different (P > 0.05) from those in the positive control group (diabetic treated with glibenclamide) on days 10 and 14. Also, the results showed that the extract demonstrated time dependent activity with 81% blood glucose level reduction on day 14.
Effect of A. brasiliana Extract on Streptozotocin-Induced Diabetic Rats.
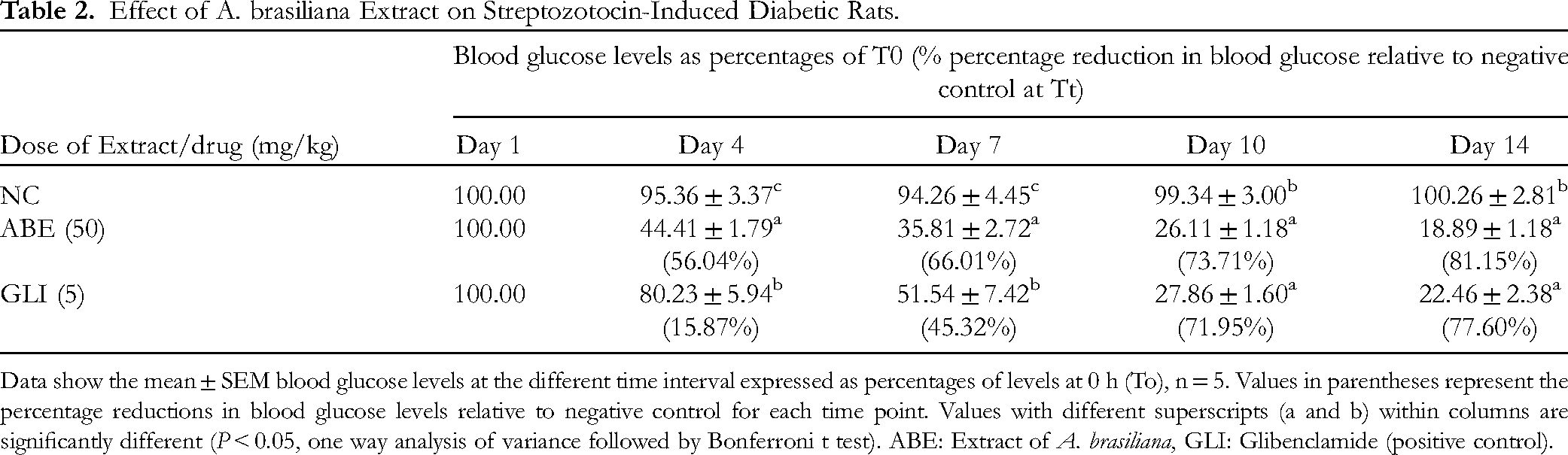
Data show the mean ± SEM blood glucose levels at the different time interval expressed as percentages of levels at 0 h (To), n = 5. Values in parentheses represent the percentage reductions in blood glucose levels relative to negative control for each time point. Values with different superscripts (a and b) within columns are significantly different (P < 0.05, one way analysis of variance followed by Bonferroni t test). ABE: Extract of A. brasiliana, GLI: Glibenclamide (positive control).
Effect of Partitioned Fractions of A. brasiliana Methanolic Extract on Glucose Loaded Rats
The results obtained for the effect of the partitioned fractions on glucose loaded rats are presented in Table 3. The 50 mg/kg dose was selected for the study since it was the most active dose of the extract. In this study, the aqueous methanol fraction gave 53.50% blood glucose reduction after 4 h of administration followed by the n-hexane fraction at 49.64%, dichloromethane fraction at 47.52% and the ethyl acetate fraction at 42.03%. Also, all the fractions assayed showed no significant difference when compared with glibenclamide at 4 h, while only ethyl acetate fraction gave time dependent blood glucose reduction up to the fourth hour.
Effect of the Partitioned Fractions of A. brasiliana Methanol Extract on Glucose Loaded Rats.

Data show the mean ± SEM blood glucose levels at the different time interval expressed as percentages of levels at 0 h (To), n = 6. Values in parentheses represent the percentage reductions in blood glucose levels relative to negative control for each time point. Values with different superscripts (a and b) within columns are significantly different (P < 0.05, one way analysis of variance followed by Bonferroni t test). NC: Glucose in 1% of tween 80 normal saline (negative control), HABE: n-Hexane fraction, DABE: Dichloromethane fraction, EABE: Ethyl acetate fraction, AABE: Aqueous fraction, GLI: Glibenclamide (5 mg/kg).
Secondary Metabolite Profiling of Aqueous Methanol Fraction of A. brasiliana
A total of 30 phytochemicals were identified from the aqueous methanol fraction of A. brasiliana when the peaks were matched with the library. The phytochemicals together with their retention time (RT), molecular weight, molecular formula and peak area (%) are presented in Table 4. 1H-Indole-2-carboxylic acid, 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-, isopropyl ester, Benzo[h]quinoline, 2,4-dimethyl-, Thymol and 13,5-trioxane were identified as secondary metabolites with the highest concentration in the aqueous methanol fraction.
Secondary Metabolites Identified from the Aqueous Methanol Fraction of A.brasiliana.

Discussion
Diabetes mellitus has been identified as one of the diseases attracting considerable attention due to the deadly complications and uncomfortability that arises from its emergence in diabetic subjects.33,34 In many African countries, including Nigeria, both type 1 and type 2 diabetes has become a major health burden that contributes to the decline in life expectancy of the entire population and increasing the chances of the development of various cardiovascular diseases.35,36 Though some of the existing synthetic hypoglycaemic drugs have made some tremendous successes in blood glucose regulation, their side effects and unaffordability is a major source of concern to people living in middle and low-income countries.14,36 Therefore, extensive contemporary research is needed to identify new therapeutic agents that can effectively manage the disease with lesser side effects.
Nigeria is among the African countries richly blessed with beautiful flora and plants with therapeutic attributes. 36 In this study, we sought to evaluate the antidiabetic activity of A. brasiliana leaf extract in vitro through α-amylase inhibitory assay before proceeding to the in vivo oral glucose tolerate test and streptozotocin-induced diabetes models. The α-amylase inhibitory activity is among the major approaches to evaluate the postprandial hyperglycemia lowering efficacy of plant extracts, fractions, isolated and synthetic compounds. Also, α-amylase inhibitor helps to manage type 2 diabetes and reduce blood glucose level of prediabetic patients.37,38 The methanol extract of A brasiliana demonstrated increasing percentage inhibition as the tested concentration increased and also gave a higher IC50 value of 0.44 mg/mL compared to acarbose (0.17 mg/mL). The results obtained is in line with the study performed by Poovitha and Parani 39 and Ayele et al, 40 where Momordica charantia, Phlomis bruguieri, Stachys byzantine and Scutellaria tournefortii methanol extract gave comparable percentage inhibition and IC50 values compared to the positive control.
The in vitro results obtained for the methanol leaf extract of A. brasiliana at varying concentrations of 62.5, 125, 250, 500 and 1000 mg/mL indicated that the plant may be a good candidate for diabetes therapy. Hence, further studies were performed to establish its efficacy in vivo using type 2 diabetes models (glucose lowering and streptozotocin-induced methods). Glucose lowering assay is an established method used in extrapolating the effect of extracts, fractions and phytochemicals as therapeutic agents against type-2 diabetes.3,30 The glucose lowering effect of the extract on glucose induced normoglycaemic rats showed that A. brasiliana methanol extract exhibit good antidiabetic activity with the highest activity obtained at the lowest tested dose of 50 mg/kg. The results further showed that the activity was non-dose dependent and the extract possessed good extrapancreatic and insulinotropic mechanism of action. Similar reports on Otostegia integrifolia leaf, Rubus erlangeri leaf and Uvaria afzelii root showed that extracts at different doses elicits good glucose lowering activity with similar mechanisms of action.30,41,42 However, A. brasiliana at a lower dose of 50 mg/kg showed more promising antidiabetic activity since it exhibited both insulin stimulating and extrapancreatic mechanisms, moreso, extracts with good activity at lower dose tends to be more recommendable than those at higher doses.
Streptozotocin is a known reagent that causes permanent induction of diabetes mellitus in experimental animals by destroying their pancreatic beta cells and drastically reducing insulin production, which in turn results in disruption of carbohydrate, protein and fat metabolism thereby increasing blood glucose level. 43 In this study, the 50 mg/kg dosage of the extract demonstrated good antidiabetic activity and the results obtained were statistically comparable with the positive control. The results also indicated that A. brasiliana extract at 50 mg/kg exhibited both extrapancreatic and insulin stimulating mechanism of actions. Similar results were reported for the antidiabetic effect of Thymus. schimperi leaves and Parquetina nigrescens fruit.43,44 In their reports, Ayoola et al 44 and Melesie et al 45 reported comparable antidiabetic activity with glibenclamide up to day 14 at 200 and 500 mg/kg, respectively. A. brasiliana leaf extract gave better antidiabetic activity at lower dose of 50 mg/kg and could be more preferred than some other herbal remedies.
In an effort to investigate the influence of solvent partitioning and identify the most active fraction of A. brasiliana extract on glucose loaded rats, the extract was partitioned into n-hexane, dichloromethane, ethyl acetate and aqueous methanol fractions and assayed for their glucose lowering efficacy. In this study, the solvent partitioning of the extract improved the activity appreciably with the aqueous methanol fraction eliciting the highest percentage blood glucose reduction followed by n-hexane, dichloromethane and ethyl acetate fractions. These results further showed that the polar and non-polar chemical constituents of the extract are responsible for the activity obtained with the extract. Various studies has shown that thymol, 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-, isopropyl ester, benzo[h]quinoline, 2,4-dimethyl-, thymol and 13,5-trioxane identified from the aqueous fraction to exhibit pharmacological properties like antidiabetic, anticancer, antioxidant, anti-inflammatory and anticonvulsant properties.43-48 Hence, the phytochemical among others may be among the active component of the most active fraction.
Limitation of the Study
The study evaluated the antidiabetic activity of the methanol extract, partitioned fractions of A. brasiliana and identified the chemical constituents of its active fraction. A bioassay guided isolation of antidiabetic compounds from the active fractions could be carried out. Also, in silico, in vitro and in vivo antidiabetic studies of the chemical compounds could be performed to establish their activity and mechanisms of action.
Conclusion
This study used alpha amylase, glucose lowering and streptozotocin-induced methods to study the effect of A. brasiliana as a therapeutic agent in type 1 and 2 diabetes subjects. The extract demonstrated good alpha amylase inhibitory activity in vitro and considerably high antihyperglycaemic activity at the lowest tested dose of 50 mg/kg which was comparable with the reference drug (glibenclamide). Also, the good activities exhibited by A. brasiliana extract in the in vitro alpha amylase and in vivo antihyperglycaemic studies were replicated in the streptozotocin-induced diabetic rats. The study showed that A. brasiliana may be a good therapeutic candidate against type 1 and 2 diabetes. The aqueous methanol fraction gave the highest antihyperglycaemia activity, indicating that the antidiabetic constituents reside largely in the polar fraction. Extensive isolation, characterization and structure elucidation of chemical compounds as well as their in vitro and in vivo antidiabetic studies is recommended.
Footnotes
Acknowledgment
The authors extend their appreciation to the Researchers Supporting Project number (RSPD2024R620), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Postgraduate Ethical committee of Obafemi Awolowo University, Ile-Ife, Nigeria, with registration number: SCP13/14/H/0598.
Funding
This work was supported by the Researchers Supporting Project number (RSPD2024R620), King Saud University, Riyadh, Saudi Arabia.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Right
All procedures in this study were conducted in accordance with the Animal Care approved protocols of the Postgraduate Ethical committee of Obafemi Awolowo University, Ile-Ife, Nigeria.

