Abstract
Tangeretin (Tan) possesses great anti-oxidation and anti-inflammation bioactivities; however, it is accompanied by poor water solubility, which leads to inefficient cellular internalization. To address this issue, a reactive oxygen species (ROS)-triggered poly (thioether-polyesters) micelle (PDHP, PEG-DTT) was designed and prepared via self-assembly, which consisted of poly (thioether-polyesters) as the hydrophilic shell, and the drug Tan as the hydrophobic inner core. The micelles (Tan@ PDHP), with a 63.15% loading efficiency of Tan, showed negligible cytotoxicity, high stability in phosphate-buffered saline buffer (pH = 7.4), and continuous release of Tan with the stimulation of H2O2. In addition, this Tan loading micelle was more efficient in responding to the formation of ROS in the lipopolysaccharide-stimulated RAW264.7 cells compared to that of the free Tan. In short, the strategy of encapsulating the low solubility Tan in ROS-triggered poly (thioether-polyesters) micelles provides an effective assay of enhancing Tan's antioxidative activity.
Introduction
Tangeretin (4′, 5, 6, 7, 8-pentamethoxyflavone, Tan) is a natural poly-methoxylated flavonoid extracted from citrus peel with more than one mechanism of antioxidant and anti-inflammation activity.1‐4 Its calculated absolute oral bioavailability is only 27.11% due to its poor aqueous solubility.5,6 As we know, poor solubility of a drug is often unfavorable in long-term administration because of its unrealistic administration for clinical practice.
7
The good news is that drug delivery systems (DDSs) can improve the solubility and absorption of the drugs and target more drugs to the tissue compared to drug solutions.
8
Nano polymeric micelles have been widely applied in the field of DDSs. They provide improved water solubility, stability, and bioavailability of the encapsulated compounds.9‐12 For example, methoxyl poly(ethyleneglycol)-
In tumor or inflamed tissues, reactive oxygen species (ROS) levels are usually higher than those of normal tissues (1.0 × 10−4 mol/L at the tumor site vs 2.0 × 10−8 mol/L at the normal site).20,21 Therefore, ROS is regarded as a meaningful trigger signal for the responsive DDSs. Till now, numerous ROS-triggered chemical polymers have been reported.22‐24 Among them, thioether bond containing polymers seem to be the simplest. Under the oxidation of ROS, the hydrophobic thioether will be transformed into hydrophilic sulfoxide or sulfone.25,26 The assembly structure of the delivery polymers will be broken with the transformation of hydrophobic/hydrophilic thioether bonds, and the encapsulated drugs will be eventually released. For example, the thioether groups of polyethylene glycol (PEG)-based polymers are oxidized to hydrophilic sulfoxide and sulfone. 27 This transformational property encouraged us to design thioether-based ROS-responsive Tan delivery polymers. Combined with the good biocompatibility of PEG, the PEG-based thioether delivery polymers may possess great potential for ROS-targeted DDSs.
In this study, we prepared PEG-based polymeric micelles (PDHP, PEG-DTT) via a Michael addition reaction. Tan encapsulated micelles (Tan@ PDHP) were fabricated by the self-assembly and dialysis assay. The properties of water solubility, chemical stability, and Tan release rate were investigated under simulated
Materials and Methods
Materials
Tan (CAS#: 481-53-8) was isolated from aged tangerine peels using a published method, 28 with a minimum of 98% purity. Pentaerythritol triacrylate (PETA, #3524-68-3), hexamethylene diacrylate (HDDA, #28393-01-3), methoxy polyethylene glycol mercapto (PEG-SH, MW = 2 kDa, #JKA13015), LPS (#L2880), and Cell Counting Kit-8 (#96992) were purchased from Sigma-Aldrich, and DL-dithiothreitol (DTT, #3483-12-3) from Aladdin. RAW264.7 cells were obtained from the China Center for Type Culture Collection. Fetal bovine serum (#A3161001C), Gibco Dulbecco's Modified Eagle Medium (DMEM) high glucose medium (#11965092), trypsin (#12605010), and 100 × concentrated penicillin-streptomycin stock mixture (#10378016) were purchased from Gibco. The primary antibodies of phospho-PI3 Kinase (#17366), PI3 Kinase (#34050), Akt (#4691), phospho-Akt (#4060), COX-2 (#12282), iNOS (#13120), and β-Actin (#4970) were purchased from Cell Signaling Technology, RIPA lysis (#sc-24948A) from Santa Cruz, DNase I (#2270A) and Advantage RT-for-PCR kit (#639505) from Takara, and SYBR Green Master Mix (Cat#: KM4101) from Kapa Biosystems.
Determination of the Loading Content of Tan in Micelles
High-performance liquid chromatography (Agilent 1290 HPLC) was used to analyze the loading content (LC) of Tan in the prepared micelles. A Thermo C18 HPLC analytical column (4.6 mm × 25 cm, 5 μm particle size) was used with a column temperature of 30 °C and an injection volume of 20 μL. The mobile phase (acetonitrile:10 mM phosphate buffer = 90:10, v/v) was run at a flow rate of 1 mL/min. A linear equation was plotted with Tan as a standard compound, taking the concentration (C) as the
Chemical Synthesis and Characteristics
PDHP monomers were synthesized following a previous similar report. 13 Briefly, PETA (110 mg, 0.3 mmol) and HDDA (46 mg, 0.2 mmol) were dissolved in 10 mL dimethyl sulfoxide. Subsequently, 10 μL triethylamine (0.1 mmol) was added and then stirred with N2 bubbling for 15 min. Then, DTT (30 mg, 0.2 mmol) was added to the mixture quickly and the solution reacted in a 60 °C sealed oil bath for 72 h. Then, 73 mg PEG-SH was added for another 48 h reaction. Afterwards, the solution was dialyzed (MWCO 3500, Spectrum) against ultrapure water for 48 h. The gray micelle PDHP were obtained with the freeze-dried assay. 29 and confirmed by nuclear magnetic resonance (NMR) spectroscopy (Bruker, Germany 500 MHz NSL2300).
Preparation of Tan-Loaded Micelles
Tan-loaded Tan@ PDHP micelles were prepared by emulsion-solvent evaporation. 30 Briefly, gray PDHP monomer powder (20 mg) and Tan (15 mg) were dissolved in 10 mL of chloroform: methanol (1:1, v/v) mixture (pH 7.2-7.4), and the mixture was homogenized in succession via an ultrasonic probe (Scientz-IID, Ningbo Scientz) at 120 W (4 s on and 4 s off) under ice-water bath conditions. After 10 min sonication, free Tan was removed by gel filtration with a MicroSpin G-25 column (GE Healthcare Life Sciences). The emulsion was stirred at 30 °C for 6 h in order to volatilize the organic solvent and then centrifuged at 4000 rpm for 10 min to obtain the final Tan@ PDHP micelles, according to the dehydration-rehydration method. 31 The amount of micelle-encapsulated Tan was quantified by HPLC analysis. Finally, Tan@ PDHP micelles were fabricated successfully after low-temperature freeze-drying. Particle sizes and Zeta potential of the micelles were analyzed by a ZS90 laser particle size meter and Zeta potentiometer Nano ZS. Then, morphological characteristics of all micelles were examined after negative staining with 0.5% w/v uranyl acetate solution and imaged by FEI Tecnai G2 F20 S-TWIN TEM (FEI Co.).
Chemical Characterization of PDHP
The basic characteristics of PDHP monomers were recorded on a BRUKER AVANCE III 500 M NMR spectrometer (Bruker Corporation), and analyzed by MestReNova software (Bruker Corporation), gel permeation chromatography (GPC) mass spectrometry (1515/2414 GPC, Waters) and Fourier transform infrared (FT-IR) spectroscopy (ThermoiS50-Nicolet, Thermo Fisher). All spectra were obtained in absorbance mode with 32 scans at an infrared spectral resolution of 4 cm−1 from 4000 cm−1 to 400 cm−1, respectively. The images of Tan@ PDHP were observed by a TEM (JEM 2100F, Jeol, Japan).
Ex Vivo Tan Release

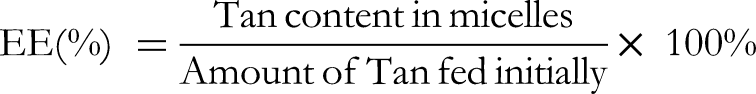
In Vitro Experimental Grouping and Treatment
RAW264.7 cells were cultured in DMEM high glucose medium with the addition of 10% fetal bovine serum and a mass ratio of 1‰ tetrahydrofuran at 37 °C in 5% CO2, and saturated humidity. Under these conditions, 5 cell treatment groups were set up as follows: control group (CON), LPS group (LPS, 500 ng/mL LPS), LPS + Tan group (Tan, 500 ng/mL LPS + 12 µg/mL Tan), LPS + PDHP micelles group (PDHP, 500 ng/mL LPS + 28.85 µg/mL PDHP micelles), and LPS + Tan@ PDHP micelles group (Tan@ PDHP, 500 ng/mL LPS + 40.85 µg/mL Tan@ PDHP micelles).
Detection of Cell Viability
RAW264.7 cells were inoculated into a 96-well plate at 1 × 104 cells per well and incubated with the addition of 0 to 50 µmol/mL Tan@ PDHP micelles for 24 h. Then, the cells were incubated continuously at 37 °C for 2 h after 10 μL of cell counting kit-8 solution had been added. The absorbance values (A450) were measured by an MK3-Microplate Reader (LabSystems).
Reverse Transcription-Polymerase Chain Reaction
RAW264.7 cells were inoculated into a 6-well plate at 5 × 105 cells per well. After culturing according to the experimental grouping for 24 h, the cells were collected, and the total RNA was extracted. DNase I without RNase was used to eliminate the remained genomic DNA, and then the mRNA was reversely transcribed into cDNA. The transcription levels of tumor necrosis factor (TNF)-α, interleukin (IL)-6, and inducible nitric oxide synthase (iNOS) were analyzed with the SYBR Green Master Mix (Cat#: KM4101, Kapa Biosystems) under 39 cycles of amplification (95 °C for 5 s, 56 °C for 10 s, and 72 °C for 25 s) in a Bio-Rad Real-Time PCR System (Bio-Rad). The relative expression levels were calculated via the 2−ΔΔCt assay. The primer sequence is shown in Table 1.
Sequences of Primers Used in This Study.

Detection of Intracellular ROS
Cells in the logarithmic growth phase were washed twice with Dulbecco's PBS (NaCl 8 g/L, KCl 0.2 g/L, Na2HPO4 1.15 g/L, KH2PO4 0.2 g/L, CaCl2 0.1 g, MgCl2 6H2O 0.1 g/L, pH 7.4), then 100 μL/well working solution of 1000 × diluted 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA, S0033S, Beyotime) was added, and then incubated continuously for 20 min. After that, the fluorescence signal was imaged using a BH2 optical microscope (Olympus), and quantified by Image J (NIH).
Protein Collection and Western Blotting Assay
After culture for 24 h, the total protein of each group was extracted, respectively, by RIPA lysis, and then denatured after quantitative analysis. After separation by sodium dodecyl sulfate-polyacrylamide gel electrophoresis, the proteins were transferred onto polyvinylidene difluoride membranes. Combination of the associated primary or secondary antibodies was carried out by standard laboratory procedures. The luminous reaction was performed with enhanced chemiluminescence Western blotting substrate. Both image collection and the quantitative gray value of the target bands were carried out by the imaging system of BIO-RAD ChemiDoc XRS.
Statistical Analysis
Data were analyzed using 1-way analysis of variance and Dunnett's test. SPSS Statistics 17.0 (SPSS Inc.) was applied for statistical analysis, and the graphs were plotted with Excel 2007. All results are presented as mean ± SEM. A
Results
Chemical Characteristics of the Micelles
PDHP micelles were synthesized via a 2-step Michael addition reaction of coupling with PETA, HDDA, DTT, and PEG-SH (Figure 1A). Characterization by 1H NMR, FT-IR, GPC, and TEM clearly revealed the successful synthesis of PDHP (Figure 2A to C, and Table 2).

Synthesis and characterization of micelles of PDHP and Tan@ PDHP. Synthesis route (A) and 1H NMR spectrum (B), and FT-IR spectrum (C) characterization of PDHP and Tan@ PDHP.

Characterizations of micelles. Representative TEM image with micelles of PDHP (A), Tan@ PDHP (B), and (C) the size distribution of micelles. * The size distribution was determined by measuring the hydrodynamic diameter using DLS in PBS (pH 7.4) at 25 °C (n = 3). Scale bar stands for 300 nm.
Size Distribution and Other Relative Data of Micelles.

Abbreviations: PDI, polydispersity index; CMC, critical micelle concentration.
Tan loading efficiency of the PDHP micelles was calculated by the Tan standard curve formulation, in which S = 35.38298C-9.04361 (
According to the results of the dynamic light scattering, we deduced that the PEG-SH layer was exposed on the surface of Tan@ PDHP micelles. 33 The diameter of the PDHP particles was about 205 nm (Figure 2C), which showed a near agreement with the TEM results (Figure 2B).
1H NMR Characterization
1H NMR spectroscopy was used to determine the structure of PDHP polymeric micelles. As shown in Figure 1B, the signals between
IR Characterization
According to the Michael's self-assembly addition reaction, a weak peak of sulfhydryl-SH of the DTT molecules occurred between
Physical Characterization of Micelles
As shown in Figure 2, PDHP and Tan@ PDHP micelles all had ellipse morphologies with an average diameter of 150 ± 11.0 and 196 ± 14.0 nm, respectively, which showed a near agreement with the TEM results. We deduced that the PEG-SH layer was exposed on the surface of Tan@ PDHP micelles. 33 The polydispersity index of PDHP was 2.27 (Mw/Mn = 190087/83738), which implied that the micelles were relative uniform in mass distribution without the residues of the impurity.
The Zeta potentials of PDHP and Tan@ PDHP were + 25.7 ± 1.0 and + 18.4 ± 1.9 mV, respectively, which revealed that the surface of the micelles is charged with the complexes of cationic molecules, and the positively charged micelles were stable and possessed the characteristic of a long cycle time
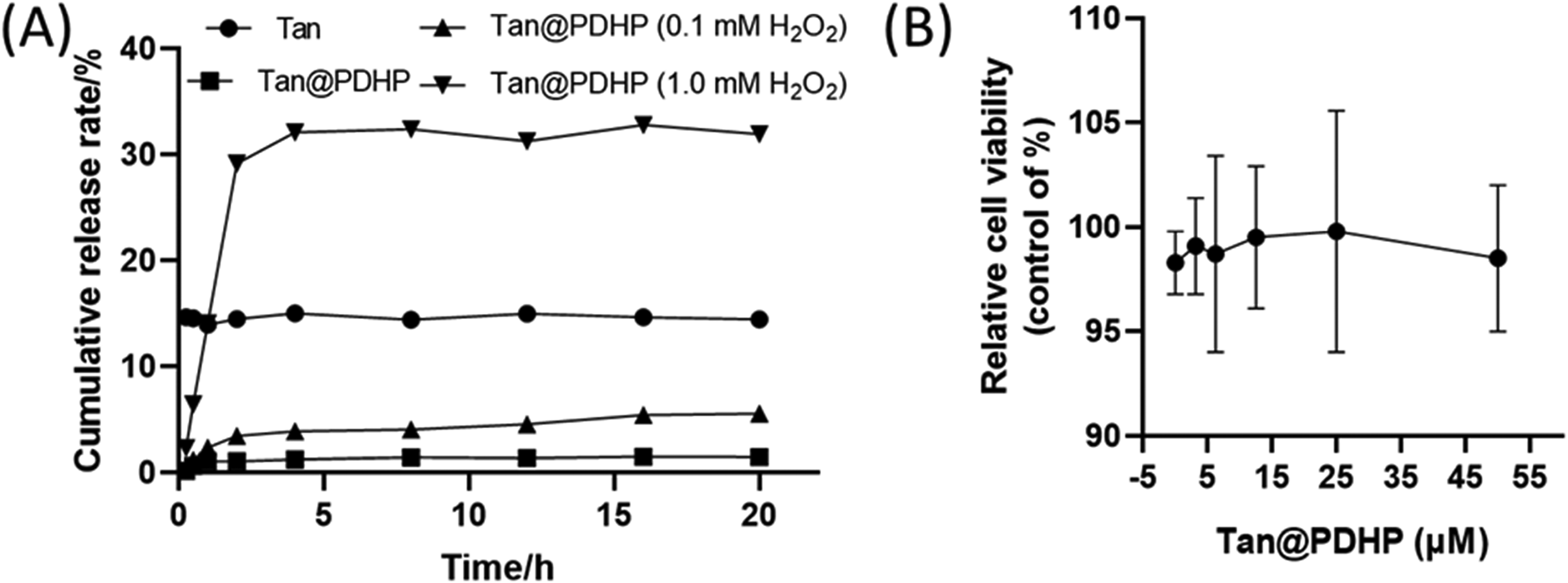
Ex Vivo Release of Tan
The results shown in Figure 3A showed that the cumulative release rate of Tan was 1.44%, 5.55%, and 31.93%, respectively, of the Tan@ PDHP micelles after treatment for 20 h with 0, 0.1, and 1.0 mmol/L H2O2, respectively. These results indicated that the solubility of Tan was poor (200 μg/mL × 15% = 30 μg/mL). Tan@ PDHP micelles remained stable under nonoxidative stress conditions, but Tan was released rapidly from the Tan@ PDHP micelles under the stimulation of 1.0 mmol/L H2O2. The highest cumulative release rate was only 32.80%, which was consistent with the maximum solubility of Tan (200 μg/mL × 0.328 [release rate] × 0.46 [entrapment efficiency] = 30.33 μg/mL). These results indicated that Tan@ PDHP micelles possess the characteristics of releasing the encapsulated Tan under the stimulation of oxidative stress.
Tan Inhibited LPS-Induced Inflammation of RAW264.7 Macrophages
After treatment with 0 to 50 µmol/mL of Tan@ PDHP micelles for 24 h, the survival of the RAW246.7 cells showed no obvious difference (
Tan@ PDHP Micelles Inhibited the Production of Inflammatory Cytokines.

Different letters (a-c) above the columns represent significant differences of the same indexes for different groups when
Under the stimulation of LPS, a large amount of endogenous ROS was produced in RAW264.7 cells, which mediated the phagocytic function of macrophages and the transmission of inflammatory signals (Figure 4A and B). Western blot detection results (Figure 4C-G) showed that the relative level of iNOS was 1.00 (CON), 2.77 (LPS), 1.17 (Tan@ PDHP), 2.17 (PDHP), and 1.75 (Tan), respectively. The relative level of COX-2 was 1.00 (CON), 3.67 (LPS), 1.07 (Tan@ PDHP), 1.97 (PDHP), and 1.56 (Tan), respectively. At the same time, the administration of Tan@ PDHP micelles also down-regulated the ratio of p-PI3K/PI3K (0.89 of Tan@ PDHP micelles vs 2.89 of LPS) and p-Akt/Akt (2.94 of Tan@ PDHP micelles vs 6.08 of LPS). These results showed that Tan effectively suppressed the expression of the LPS-stimulated PI3K-Akt pathway.

Effect of Tan@ PDHP micelles intervention on the formation of ROS in LPS-stimulated RAW264.7 cells. (A), (B) Detection of ROS in different treatment groups and (B) quantified by image J. (C) The result of Western blot and (D-G) the relative protein levels quantified via Image J and normalized to β-actin. (n = 3, mean ± SEM. Differences are shown as **
Discussion
Tan is a highly hydrophobic natural product with a poor water solubility and low oral bioavailability. After oral administration of 50 mg/kg BW Tan, the concentration of Tan in the rats’ plasma was only 0.49 μg/mL. 36 Accordingly, the shortage of its low bioavailability limited Tan's incorporating into foods. Therefore, it is necessary to improve the utilization efficiency of this compound. Hu et al. 6 found that after encapsulated with the emulsion-based delivery systems, the bioaccessibility of Tan raised from around 36% to 90%. Zhan et al. 37 also obtained similar results. Therefore, embedding will improve the utilization rate of Tan. As a DDSs, nano-micelles prevented the encapsulated drugs from metabolic degradation and provided a controlled release. It has aroused emerging attention. 38 In the present study, electrostatic repulsion was observed between the side chain hydrophobic group and the polymer chain of PDHP micelles, which leads to the formation of a spacious hydrophobic core in the micelles. 32 Moreover, the Tan loading capacity of the micellar cavity was largely increased with the hydrogen-bonding interactions between the oxyether (-C-O-) and the ester bonds (-C(O)-O-) of PDHP micelles and the methoxy groups of Tan. Besides, electrode-deficient diene structures of the micelles possess an enormous number of positive charges, which contributed greatly to its binding with the cytoplasmic membrane through electrostatic interaction. 39 Our previous studies 4 revealed that Tan inhibited the formation of ROS and downregulated the degree of inflammation. Meanwhile, it was confirmed that Tan down-regulated the expression of COX-2 and alleviated the generation of ROS.36,37 Therefore, this study examined the inhibitive activity of Tan@ PDHP micelles on the formation of LPS-induced ROS. The results revealed that high-level endogenous ROS formed in RAW264.7 cells with the stimulation of LPS, mediated the phagocytic function of macrophages and the transmission of inflammatory signals. 40 The Western bolt results of iNOS and COX-2 revealed that the ROS level was significantly inhibited with the administration of Tan or Tan@ PDHP micelles. Furthermore, Tan@ PDHP micelles exhibited stronger ROS inhibitory activity than that of the unencapsulated Tan, which means the transformation of the oxidative thioether groups to the sulphoxide and sulphone ones 41 under the high ROS environment. Accordingly, ROS-triggered disintegration of the Tan@ PDHP micelles occurred with the oxidation of the thioether ester. Therefore, Tan was targeted-released intracellular under the function of LPS-stimulated oxidative stress. Conversely, the high content of intracellular Tan significantly depressed the level of intracellular ROS. Due to the high ROS concentration of the inflammatory, cancer tissues and so forth, we believe that the prepared Tan@ PDHP micelles will be rapidly internalized in these tissues and the utilization efficiency of the encapsulated Tan is higher than the free one.
In summary, a ROS-responsive prodrug micelle, consisting of poly (thioether-polyesters) segment and a hydrophobic compound Tan, was prepared via a 2-step Michael addition reaction. The prodrug micelles exhibited excellent drug release efficiency under the stimulation of 1.0 mmoL/L H2O2. In addition, cytotoxicity assays demonstrated good biocompatibility of the prepared Tan@ PDHP. Both Tan@ PDHP and Tan administration significantly down-regulated LPS-stimulated PI3K/Akt signaling pathway. Thus, prodrug micelles are expected to be applied to overcome the shortcoming of the poor solubility for free Tan. In short, this micellar encapsulation strategy offers some useful pointers on the efficient utilization of low water-soluble compounds.
Footnotes
Acknowledgments
This study was supported by the key scientific research projects of Natural Science Foundation of Hunan Province (No. 2021JJ31154) and the National Nonprofit Institute Research Grant of CAFINT, China (No. CAFYBB2018GA001).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
