Abstract
The polymethoxyflavonoids nobiletin and tangeretin are isolated from peels of shekwasha (
Shekwasha (
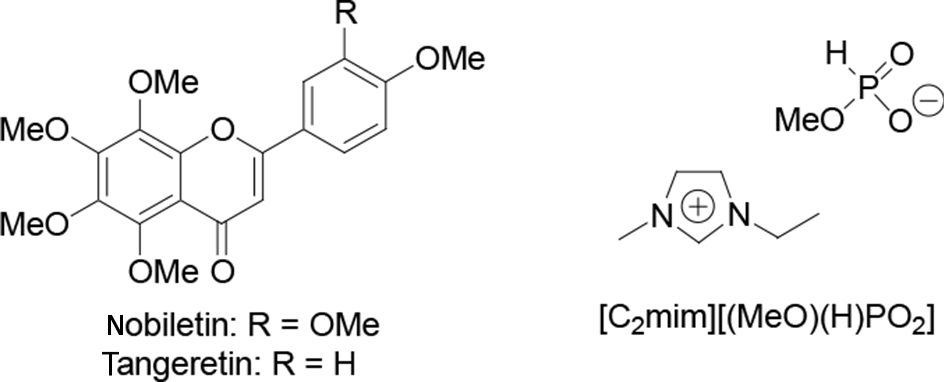
Structures of nobiletin, tangeretin, and [C2mim][(MeO)(H)PO2].
Ionic liquids (ILs) are salts composed of an anion and a cation, which have melting points of less than 100°C. 10 They offer advantages such as low volatility, low flammability, high thermostability, high viscosity, and recyclability. Additionally, some ILs such as 1-ethyl-3-methylimidazolium methylphosphonate ([C2mim][(MeO)(H)PO2], Figure 1) can dissolve cellulose under mild conditions. 11 A plant cell is surrounded and protected by a cell wall, which consists mainly of cellulose. Therefore, the use of an IL as an extraction solvent was expected to result in efficient extraction of natural products from citrus peels. The IL [C2mim][(MeO)(H)PO2] is particularly useful because it dissolves cellulose at room temperature, 12 but is not soluble in ethyl acetate (EtOAc), which could be used to extract or back-extract organic compounds.
A previous report indicated that 1-butyl-3-methylimidazolium chloride ([C4mim]Cl) was an efficient extraction solvent for shikimic acid and bilobalide from
First, the extraction of nobiletin and tangeretin from shekwasha peels was conducted using ethanol (EtOH), acetone, and EtOAc as control experiments (Figure 2). Crushed shekwasha peels and an organic solvent were stirred at 25°C for 75 minutes and then centrifuged for 10 minutes to separate the shekwasha peel residue. After evaporation, the extract was dissolved in methanol (MeOH). Next, extraction using the cellulose-dissolving IL [C2mim][(MeO)(H)PO2] was conducted as follows (Figure 2). The crushed shekwasha peels and [C2mim][(MeO)(H)PO2], [C2mim][(MeO)(H)PO2]/EtOH (1:1 w/w), [C2mim][(MeO)(H)PO2]/acetone (1:1 w/w), and [C2mim][(MeO)(H)PO2]/EtOAc (1:1 w/w) were stirred at 25°C for 60 minutes. After addition of H2O and EtOAc, the extraction solutions were stirred at 25°C for 5 minutes and centrifuged for 1 minute to separate the supernatant. Nobiletin and tangeretin are insoluble in H2O, but soluble in [C2mim][(MeO)(H)PO2] and EtOAc. Thus, addition of H2O decreases the solubility of nobiletin and tangeretin in the [C2mim][(MeO)(H)PO2] layer and increases their solubility in the EtOAc layer. This EtOAc layer containing nobiletin and tangeretin was collected by decantation and, after evaporation, the extract obtained was dissolved in MeOH.
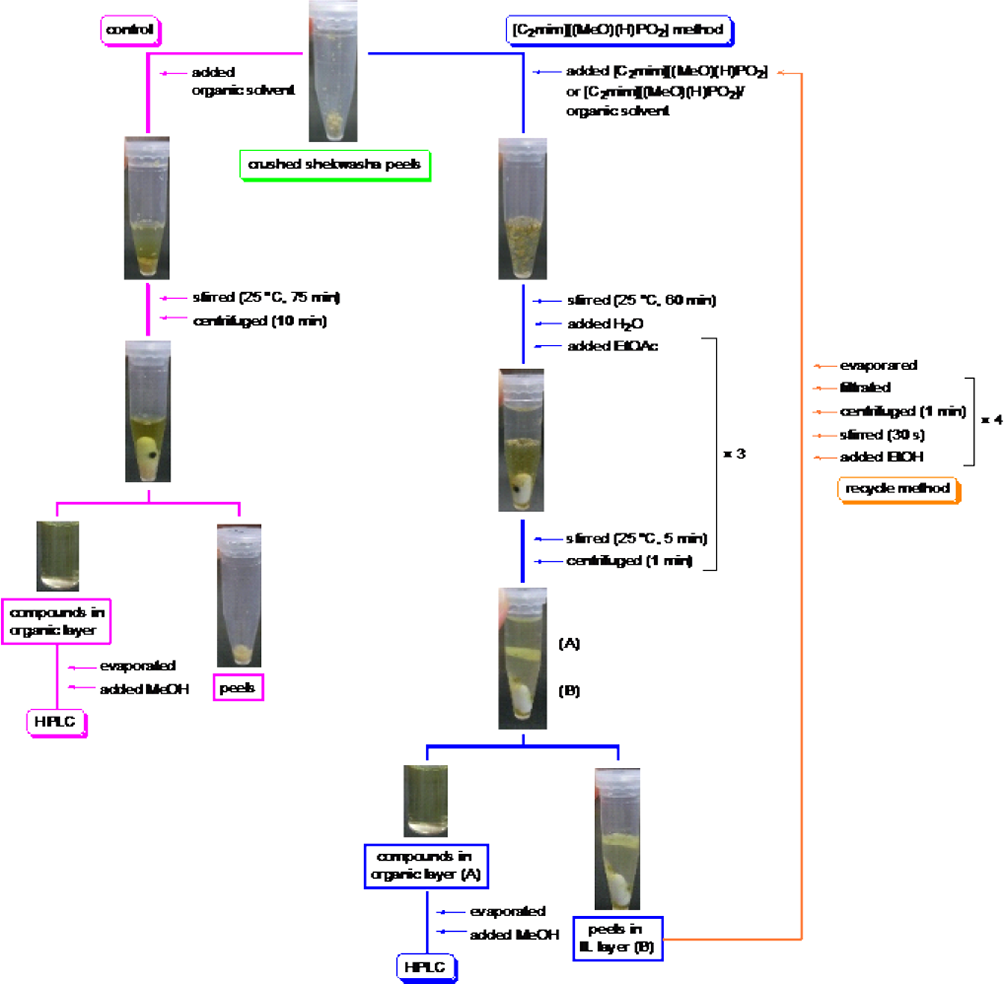
Protocols for extraction of nobiletin and tangeretin from shekwasha peels using an organic solvent or an ionic liquid (IL) and for recycling the IL.
Quantitative analysis of the obtained extracts was performed using reversed-phase high-performance liquid chromatography (RP-HPLC). The quantity of nobiletin and tangeretin in individual extracts was calculated using a calibration curve. Extraction yields were calculated as [amount of natural products]/[original quantity of natural materials] (Figure 3).

Extraction yield of nobiletin and tangeretin from shekwasha peels using organic solvents and ionic liquid.
The extraction yields of nobiletin and tangeretin using [C2mim][(MeO)(H)PO2] and organic solvents were similar when [C2mim][(MeO)(H)PO2]/organic solvents (1:1 w/w) was used as the extraction solvents. These extraction yields were better than those obtained using an organic solvent only, with the greatest yields resulting from the use of [C2mim][(MeO)(H)PO2]/acetone (1:1 w/w) (nobiletin: 2.3%; tangeretin: 0.29%). These yields were 1.5 and 1.4 times higher, respectively, than those using acetone (nobiletin: 1.5%, tangeretin: 0.21%). These results suggest that the proposed method is an effective alternative for the extraction of nobiletin and tangeretin from shekwasha peels.
The solubility of the flavonoids in the organic solvents was similar, as indicated by the similarity of the extraction yields. Extraction using [C2mim][(MeO)(H)PO2] did not improve the yield, probably because of the high viscosity of the IL, which reduced the stirring efficiency. Therefore, a mixture of organic solvents and IL was used, which improved the extraction yield by decreasing the viscosity. The greatest yield was obtained when using [C2mim][(MeO)(H)PO2]/acetone (1:1 w/w). The effect of extraction time was also examined at 30, 90, and 120 minutes. The results showed that an extraction time of 60 minutes was optimal. The identity of nobiletin and tangeretin was confirmed by liquid chromatography-mass spectrometry (LC-MS) (data not shown).
The reusability of [C2mim][(MeO)(H)PO2] was also confirmed (Figures 2 and 4). First, the [C2mim][(MeO)(H)PO2] layer, which included the shekwasha peels and EtOH, was stirred for 30 seconds to allow the EtOH to dissolve [C2mim][(MeO)(H)PO2] and reduce the viscosity. The tube was centrifuged for 1 minute to separate the shekwasha peel residue, followed by filtration of the supernatant to remove the shekwasha peel residue. This operation was repeated 4 times. The filtrate obtained was evaporated and then reused in subsequent extractions after mixing with an equivalent volume of acetone. The [C2mim][(MeO)(H)PO2] could be reused at least 9 times without any effect on extraction yield (Figure 4).

Extraction yield of nobiletin and tangeretin using recycled [C2mim][MeO(H)PO2].
Similarly, the extraction of nobiletin and tangeretin from ponkan peels was also conducted under the conditions optimized for shekwasha peels. First, the extraction of nobiletin and tangeretin from ponkan peels was carried out using EtOH, acetone, and EtOAc as control experiments (Figure 5). The crushed ponkan peels and an organic solvent were stirred at 25°C for 60 minutes. The resulting solution was stirred at 25°C for 15 minutes, followed by centrifugation for 10 minutes to separate the ponkan peel residue. The obtained extract was dissolved in MeOH after evaporation. Next, extraction using IL [C2mim][(MeO)(H)PO2] was conducted (Figure 5). The crushed ponkan peels and [C2mim][(MeO)(H)PO2], [C2mim][(MeO)(H)PO2]/EtOH (1:1 w/w), [C2mim][(MeO)(H)PO2]/acetone (1:1 w/w), and [C2mim][(MeO)(H)PO2]/EtOAc (1:1 w/w) were stirred at 25°C for 60 minutes. The resulting solutions were stirred with H2O and EtOAc at 25°C for 5 minutes and centrifuged for 1 minute to separate the supernatant. Finally, the EtOAc layer containing nobiletin and tangeretin was collected by decantation. The extract obtained was dissolved in MeOH after evaporation. Quantitative analysis of the extracts was performed using RP-HPLC (Figure 6).
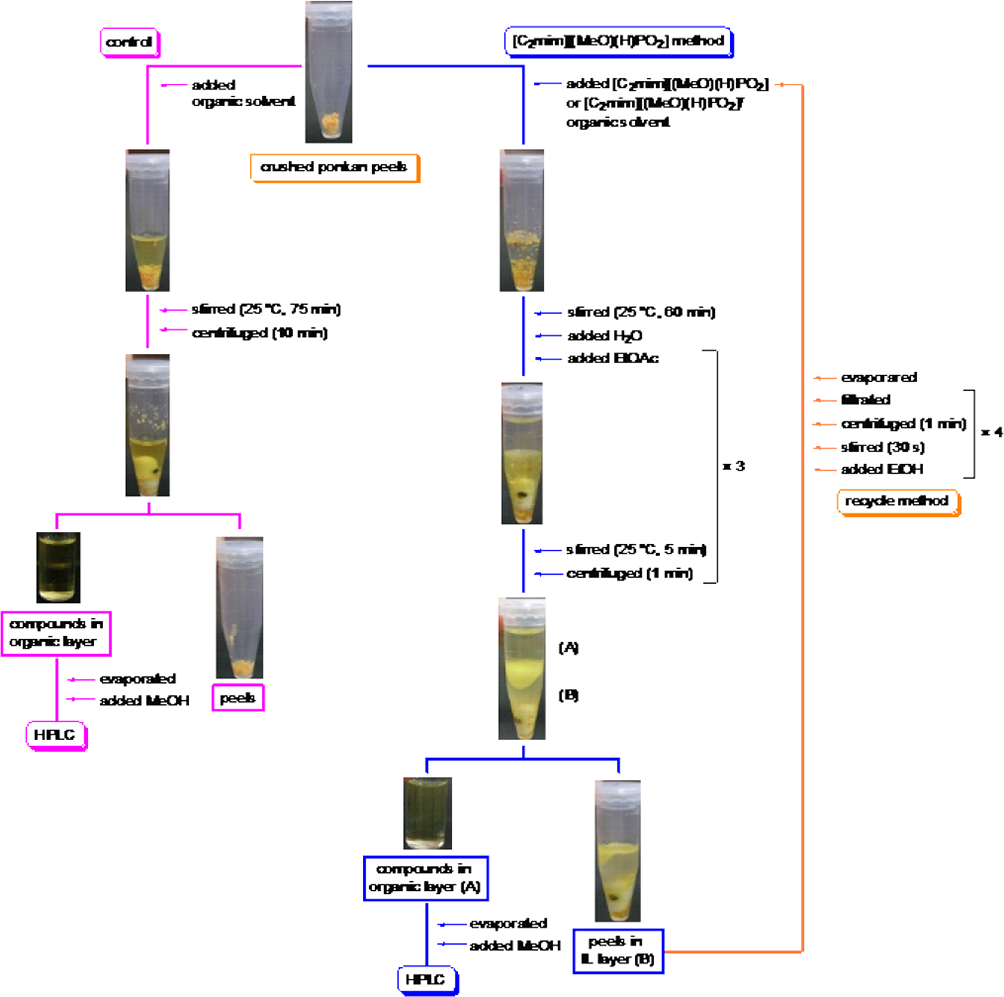
Protocols for extraction of nobiletin and tangeretin from ponkan peels using either an organic solvent or an ionic liquid (IL) and for recycling the IL.
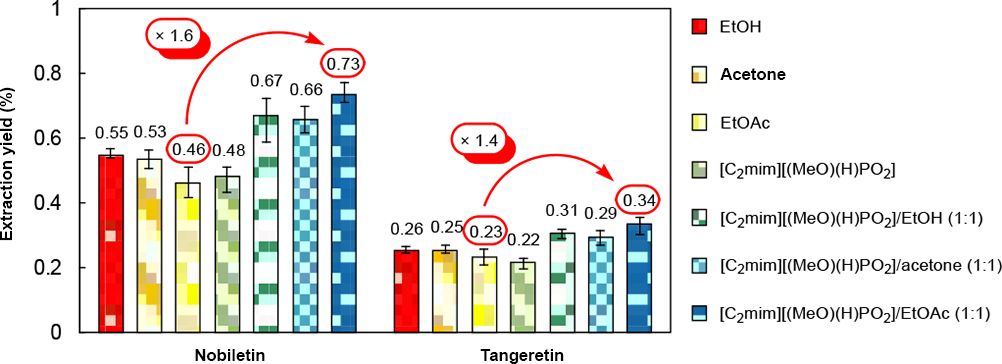
Extraction yield of nobiletin and tangeretin from ponkan peels using organic solvents and an IL.
The extraction yield of nobiletin and tangeretin from ponkan peels using EtOAc (0.46% and 0.23%, respectively) was the lowest among the control experiments. However, the extraction yield using [C2mim][(MeO)(H)PO2]/EtOAc (1:1 w/w) was the highest (0.73% and 0.34%, respectively), 1.6 and 1.4 times greater than that using EtOAc. These results indicated that [C2mim][(MeO)(H)PO2]/EtOAc (1:1 w/w) was the best combination for the extraction of nobiletin and tangeretin from ponkan peels. While EtOAc gave the worst extraction yield, use of [C2mim][(MeO)(H)PO2]/EtOAc (1:1 w/w) afforded the best yield. The poor performance of EtOAc alone was probably due to its inability to extract flavonoids from inside the cell wall. However, EtOAc does not dissolve [C2mim][(MeO)(H)PO2], which is able to maintain cellulose solubility and thus improve the extraction yield. The identity of nobiletin and tangeretin was confirmed by LC-MS (data not shown). Reusability of [C2mim][(MeO)(H)PO2] after use for extraction was confirmed (Figure 5). As previously, [C2mim][(MeO)(H)PO2] could also be reused at least 9 times without reduced extraction yields.
These results demonstrated that the best solvents for extraction of nobiletin and tangeretin from shekwasha and ponkan peels were [C2mim][(MeO)(H)PO2]/acetone (1:1 w/w) and [C2mim][(MeO)(H)PO2]/EtOAc (1:1 w/w), respectively. Differences in the optimized conditions for extraction from peels of shekwasha and ponkan were likely due to differences in the components, component ratios, and pH of individual peels.
In summary, an efficient method for the extraction of nobiletin and tangeretin from shekwasha and ponkan peels was developed using the cellulose-dissolving [C2mim][(MeO)(H)PO2] and centrifugation. The use of [C2mim][(MeO)(H)PO2]/organic solvent (1:1 w/w) allowed more efficient extraction of nobiletin and tangeretin than extraction using organic solvent alone. In addition, the [C2mim][(MeO)(H)PO2] could be reused at least 9 times for extraction without loss of efficiency. This method is expected to be applicable to the extraction and isolation of other natural products.
Experimental
General Methods
All reagents were obtained from commercial suppliers and used without further purification unless otherwise stated. [C2mim][(MeO)(H)PO2], HPLC-grade MeOH, and HPLC-grade distilled H2O were purchased from Kanto Chemicals (Tokyo, Japan), and shekwasha and ponkan from Ryukyu Front (Okinawa, Japan) and Shikisai-En (Wakayama, Japan), respectively. The HPLC analyses were performed using a JASCO instrument equipped with a multiwavelength detector (MD-2010), semi-micro HPLC pump (PU-2085), autosampler (AS-2057), column thermostat (CO-2060), and a Shimadzu instrument equipped with a communications bus module (CBM-20A), UV/Vis detector (SPD-20A), degassing unit (DGU-20A), autosampler (SIL-20AC), liquid chromatograph (LC-20AD), and column oven (CTO-20AC). The LC-MS were recorded on a Shimadzu LCMS-8030 instrument.
Extraction of Nobiletin and Tangeretin From Shekwasha Peels
Control experiments were conducted using organic solvents to extract nobiletin and tangeretin from shekwasha peels. The shekwasha peels were crushed using a mixer. The crushed peels (0.025 g) and an organic solvent (EtOH, acetone, or EtOAc) (422 µL) were added to an Eppendorf tube, which was stirred at 25°C for 60 minutes. The resulting solution was stirred at 25°C for 15 minutes, followed by centrifugation for 10 minutes to remove the shekwasha peel residue. This operation was repeated 3 times. The obtained supernatant was analyzed by RP-HPLC.
Extraction using [C2mim][(MeO)(H)PO2)] was conducted as follows: Crushed shekwasha peels (0.025 g) were added to the extraction solvents (422 µL), either 1:1 (w/w) such as [C2mim][(MeO)(H)PO2)], [C2mim][(MeO)(H)PO2)]/EtOH (1:1 w/w), [C2mim][(MeO)(H)PO2)]/acetone (1:1 w/w), or [C2mim][(MeO)(H)PO2)]/EtOAc (1:1 w/w), and the solution stirred at 25 °C for 60 minutes. The solution was next stirred with H2O (211 µL) and EtOAc (633 µL) at 25°C for 5 minutes, followed by centrifugation for 1 minute to separate the supernatant. This operation was repeated 3 times. The resulting supernatant was analyzed by RP-HPLC.
HPLC Analysis of Nobiletin and Tangeretin From Shekwasha Peels
Quantitative analyses of the extracts obtained were performed using an RP-HPLC system [column: YMC-Pack ODS-AM (150 × 4.6 mm); mobile phase: HPLC-grade distilled H2O/MeOH (gradient), flow rate: 1.0 mL/min; detection: 330 nm; injection amount: 10 µL; temperature: 30°C]. The quantities of nobiletin and tangeretin from individual extraction experiments were calculated using a calibration curve. The equations used to determine nobiletin and tangeretin amounts are as follows, where
nobiletin:

tangeretin:

Extraction yields were calculated as [quantity of nobiletin or tangeretin] per [original quantity of material]. The retention times of nobiletin and tangeretin were 23.0 and 25.3 minutes, respectively.
Extraction of Nobiletin and Tangeretin From Ponkan Peels
Ponkan peels were crushed using a mixer. The crushed peels (0.025 g) and an organic solvent (EtOH, acetone, or EtOAc) (422 µL) were then added to an Eppendorf tube, which was stirred at 25°C for 60 minutes. The resulting solution was stirred at 25°C for 15 minutes, followed by centrifugation for 10 minutes to remove the ponkan peel residue. This operation was repeated 3 times. The obtained supernatant was analyzed by RP-HPLC.
Extraction using [C2mim][(MeO)(H)PO2)] was conducted as follows: Crushed ponkan peels (0.025 g) were added to the extraction solvents (422 µL), either [C2mim][(MeO)(H)PO2)] or [C2mim][(MeO)(H)PO2)]/organic solvents (EtOH, acetone, EtOAc) (1:1 w/w), and the solution stirred at 25°C for 60 minutes. The solution was next stirred with H2O (211 µL) and EtOAc (633 µL) at 25°C for 5 minutes, followed by centrifugation for 1 minute to separate the supernatant. This operation was repeated 3 times. The resulting supernatant was analyzed by RP-HPLC.
HPLC Analysis of Nobiletin and Tangeretin From Ponkan Peels
Quantitative analyses of the extracts obtained were performed using an RP-HPLC system [column: Symmetry C-18 (250 × 4.6 mm); mobile phase: HPLC-grade distilled H2O/MeOH (gradient), flow rate: 1.0 mL/min; detection: 330 nm; injection amount: 10 µL; temperature: 30°C]. The quantities of nobiletin and tangeretin from individual extraction experiments were calculated using a calibration curve. The equations used to determine nobiletin and tangeretin amounts are as follows, where
nobiletin:

tangeretin:
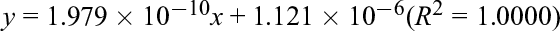
Extraction yields were calculated as [quantity of nobiletin or tangeretin] per [original quantity of materials]. The retention times of nobiletin and tangeretin were 22.4 and 24.4 minutes, respectively.
Recycling of [C2mim][(MeO)(H)PO2)]
The [C2mim][(MeO)(H)PO2] layer, including shekwasha or ponkan peels, was added to EtOH (500 µL), which was centrifuged for 1 minute to allow precipitation after stirring for 30 seconds. The mixture was then filtered. These operations were repeated 4 times. The filtrate was recycled as mixture solvent.
Footnotes
Acknowledgments
We thank Prof M. Yoshizawa-Fujita (Sophia University) for discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Sophia University Special Grant for Academic Research.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
