Abstract
Introduction
The skeletal system is a dynamically balanced system that plays a crucial role in providing support and protection for muscles and other organs, facilitating body movement, and ensuring blood production. 1 Additionally, the aging of the skeleton is closely associated with age. Skeletal disorders such as osteoarthritis, osteoporosis, and bone tumors have emerged as a significant global public health concern. Primary osteoporosis and secondary osteoporosis are both categorized as metabolic bone diseases, impacting prognosis and increasing patient mortality. 2 Osteoporosis is typically characterized by decreased bone mass, strength, and structural deterioration of bone tissue leading to increased fragility and fracture susceptibility. Its pathogenesis involves an imbalance between osteoblast-mediated bone formation and resorption, resulting in activity limitations and a severe reduction in the quality of life, imposing a significant healthcare burden on the individual and society.3–5 Excessive osteoclast formation disrupts the balance of bone turnover, resulting in pathological bone loss associated with a range of diseases. 6
Most bones are composed of dense cortical shells and internal reticular trabecular bone networks. Estrogen signaling plays a crucial role in the development and maintenance of healthy bones, with bones exhibiting differential sensitivity to both internal and external cues such as hormonal signals. Therefore, the enhancement of osteoblast differentiation and matrix mineralization can be achieved through modulation of estrogen levels.7,8 Estrogens, such as estradiol, estriol, and estrone, are crucial hormones that directly or indirectly regulate bone and skeletal muscle metabolism and function through the estrogen receptor. They also play a significant role in maintaining skeletal and articular cartilage homeostasis. 9 Osteoporosis is a common condition in older individuals, with numerous studies demonstrating that metabolic disorders, increased osteoclast activation, and decreased bone mineral density in menopausal women due to insufficient estrogen secretion and premature ovarian failure further contribute to the development of osteoporosis and sarcopenia.10,11 It has been noted that around 5000 wrist fractures and 3500 vertebral fractures occur each year in Portuguese women in their 50 s, resulting in substantial healthcare expenses. 12
In this study, we modified the structure of TSG to obtain a new derivative acetylated-TSG (Ac-TSG), and evaluated the effects of the two compounds on osteoblast activity, pharmacokinetics in rats and anti-osteoporosis in ovariectomized rats.
Materials and Methods
Materials
TSG (purity 98%) was purchased from Shanghai yuanye Bio-Technology Company Limited (Shanghai, China). Methanol, acetonitrile, n-butanol, and ethyl acetate were HPLC-grade products purchased from Tianjin Kermel Chemical Reagent Company Limited (Tianjin, China). Mouse embryonic osteoblasts cells MC3T3-E1 were purchased from Shanghai Zhongqiao Xinzhou Biotechnology Co. 1,2-Propanediol, gentiopicroside, carboxymethylcellulose sodium (CMC-Na) were purchased from Shanghai Aladdin Biochemical Technology Company Limited (Shanghai, China). Acetic anhydride and hydrochloric acid were purchased from Fuchen (Tianjin) Chemical Reagent Company Limited (Tianjin, China). Pyridine and petroleum ether were purchased from Tianjin Tianli Chemical Reagents Company Limited (Tianjin, China). The experimental water used throughout was purified water (Merck KGaA, Darmstadt, Germany).
The Synthesis of Ac-TSG and the Effect of TSG and Ac-TSG on the Viability of MC3T3-E1 Cells Were Detected by CCK-8 Method
According to the ratio of 1:4:5, TSG, pyridine, and acetic anhydride were weighed (METTLER TOLEDO, Zurich, Switzerland) and measured in a round-bottomed flask with a rotor in an oil bath at 110 °C for 4 h. Hydrochloric acid, pH 3–4, was prepared with purified water, and added 4 times the reaction solution. After constant stirring, the solution was poured into the liquid separator funnel and extracted with ethyl acetate. The obtained ethyl acetate layer was washed with water and then dried using a rotary evaporator (Ya Rong, Shanghai, China). After purification by recrystallization with petroleum ether, a white solid was obtained, the yield was calculated, and the purity was determined by HPLC. The structure was confirmed by 1H NMR, 13C NMR, and HEMS.
MC3T3-E1 cells were cultured in MEM medium containing 10% fetal bovine serum and 1% double antibody in an incubator containing 5% CO2 at 37 °C. The oxidative stress model of MC3T3-E1 cells was established by H2O2, and the control group was cultured normally without any treatment. TSG and Ac-TSG with concentrations of 1 μM, 3 μM were prepared. MC3T3-E1 cells were given a series of concentrations of TSG and Ac-TSG. After 24 hours, 48 hours, and 72 hours, the viability of MC3T3-E1 cells was measured by CCK-8.
Expression of CUL2, CDC23, ANAPC10, Smad5, BMP-2, and Runx2 mRNA in MC3T3-E1 Cells
Four concentrations of TSG (1 μM, 3 μM) and Ac-TSG (1 μM, 3 μM) were selected for the experiments. Total RNA was extracted according to the instructions of RNA extraction kit, and the purity of RNA was measured in UV–visible spectrophotometer to calculate the concentration of RNA. The cDNA product was produced by reverse transcription according to the instructions of the reverse transcription kit. The cDNA was diluted in an ice bath and added to the samples according to the instructions. Gene expression was calculated by the relative quantification 2−ΔΔCt method. The primers were synthesized by Shanghai Sangong Bioengineering Co., Ltd with β-actin as the internal reference gene and CUL2, CDC23, ANAPC10, Smad5, BMP-2 and Runx2 as the target genes. Primer sequences are shown in Table S1.
Pharmacokinetics of TSG and AC-TSG in Rats
The animal experiments involved in this study were approved by the Animal Ethics Committee of Shaanxi university of Chinese medicine (Shaanxi, China) (Approval number: Sucmd20200220001 and SUCMDL20220401006). Twenty-four Sprague Dawley (SD) rats were randomly divided into 4 groups (
Twenty microliters of gentiopicroside (101 μg·mL−1) was added to 0.2 mL rat plasma. After vortexing for 30 s, 1 mL ethyl acetate:n-butanol mixture (5:1) was added, then vortexed for 3 minutes and centrifuged at 12 500 rpm for 10 minutes. 900 μL of the supernatant was blown dry with a nitrogen blowing instrument (Tianjin Automatic Scientific Instruments, Tianjin, China) in a 40 -°C fume hood, then 200 μL of methanol was added to dissolve and vortex shake for 3 minutes, and centrifuged at 12 500 rpm for 10 min. 20 μL of supernatant was used for HPLC analysis (Thermo Fisher Scientific, New York, USA). The following parameters were used: column: Inertsil ODS-3 (150 mm × 4.6 mm, 5 µm); mobile phase: acetonitrile:water (20:80); flow rate: 1.0 mL min−1; detection wavelength: 290 nm; injection volume: 20 L; and column temperature: 30 °C. The standard solution and internal standard stock solution were TSG and gentiopicroside methanol solution with a concentration of 0.1 g mL−1, respectively.
The plasma of the blank group was mixed with the standard solution and the internal standard solution, and the final concentrations were 10.99, 2.199, 1.099, 0.4398, 0.2199, and 0.1099 μg mL−1, respectively. The working curve was plotted with the standard solution concentration C (μg·mL−1) as the abscissa and the ratio of TSG (A1) to the internal standard chromatographic peak area (A2) as the ordinate.
The plasma samples with TSG concentrations of 8.796, 3.282, and 0.328 μg mL−1 were prepared according to the standard curve preparation method, and analyzed by HPLC for 5 times in the same day. The peak area was measured and the intra-day relative standard deviation (RSD) of TSG was calculated. The inter-day RSD of TSG was calculated after continuous analysis for 3 days. At the same time, the stability data of three concentration samples were investigated, and the recovery rates of several different concentration samples were calculated.
The Drug and Statistics (DAS) software developed by Anhui Provincial Drug Clinical Evaluation Center was used for automatic calculation, and the C-T data of rats were processed in batches. Then the C-T data of each rat were intelligently processed to fit the compartment model, and then the pharmacokinetic parameters were obtained. The evaluated pharmacokinetic parameters included area under the infinite time curve (AUC0−∞),
Effects of TSG and Ac-TSG on Ca, P, BGP and ALP in Serum of Ovariectomy (OVX) Rats
A total of 84 SPF female SD rats were purchased from Chengdu Dashuo Experimental Center. After one week of ordinary feeding and adapting to the environment, they were randomly divided into Control group, Sham group, OVX group, E2 group, TSG group, Ac-TSG group. The control group did not do any treatment. Osteoporosis model was made according to ovariectomy. 21 The rats were anesthetized and the hair on the back of the rats was removed. An incision of about 1.5 cm was made on the upper middle spine of the back, and the skin and muscle membrane were separated. Then, the peritoneum was cut under the bilateral ribs at a distance of about 1.5 cm from the middle of the back. After exposing the abdominal cavity, the “V” uterus was found. Along the uterus, the white adipose tissue next to the two kidneys was found. A mulberry-like ovary was isolated from it, and the blood vessels were ligated to remove the entire ovary. The tissue was put back into the abdominal cavity of rats, sutured layer by layer, and disinfected with iodophor again.
One week after the operation, the vaginal secretions of female rats were fixed and stained every day. The results were observed under an optical microscope and the images were collected to observe the modeling. At the end of the experiment, serum, liver and kidney, femur and tibia of rats were collected, and the indexes were detected according to the instructions of relevant kits.
The Effects of TSG and Ac-TSG on Osteoclasts and Femoral Tissue in OVX Rats Were Observed by Staining
After the working solution was configured, paraffin sections of femoral tissue were taken after decalcification with EDTA, and after routine dewaxing and hydration, the sections were incubated with pure water at room temperature and then discarded and added with TRAP incubation solution, and then placed in 37 °C for a certain period of baking. After washing, hematoxylin re-staining nuclei for 40 s, 1% hydrochloric acid alcohol disintegration, ammonia return to blue, with neutral gum sealing.
The 10% formalin-fixed femoral samples were removed and treated with EDTA decalcification solution for 15 days. The decalcified samples were subjected to hematoxylin–eosin (HE) staining manipulation, and the results were visualized under a light microscope and images were captured.
The Effect of TSG and Ac-TSG on Bone Tissue of OVX Rats Was Detected by Micro-CT
The right femur specimens of rats in the fixation fluid were taken for detection. The samples were washed with 75% ethanol and scanned in a Micro-CT instrument. The original scan data were reconstructed and selected by three-dimensional reconstruction processing software NRecon and Data Viewer. The same position area in the reconstructed image was quantitatively analyzed by CT Analyzer.
The Effect of TSG and Ac-TSG on the Expression of Osteogenic Factor mRNA in OVX Rats
Tibia were ground and powdered, and the total RNA was extracted according to the instructions of the RNA extraction kit and the concentration of RNA was calculated. Reverse transcription was performed according to the instructions of the Reverse Transcription Kit under the following conditions: 65 °C for 5 min, high-speed centrifugation in an ice bath for 5 s, addition of Reverse Transcription 2 reagent and mixing, 25 °C for 5 min; 42°C for 60 min; and the cDNA product. The cDNA product was diluted in ice bath and added to the samples according to the instructions. The cDNA was diluted in an ice bath and added to the samples according to the instructions. Gene expression was calculated by the relative quantification 2−ΔΔCt method. The primers were designed and synthesized by Shanghai Sangong Bioengineering Co., Ltd with β-actin as the internal reference gene and CUL2, CDC23, BMP-2 and Runx2 as the target genes. Primer sequences are shown in Table S2.
Histological Examination
The liver and kidney tissues of each group were taken for routine paraffin embedding, and the slices were subjected to HE staining operation, and the results were observed under a light microscope and images were captured.
Statistical Analysis
The obtained measurements were expressed as (
Results
The Synthesis of Ac-TSG and the Effect of TSG and Ac-TSG on the Viability of MC3T3-E1 Cells
Synthesis of Ac-TSG
The synthesis of acetyl-TSG according to the method under 2.2 measured 0.060 g of white solid, and normalized purity by HPLC was greater than 98% (Figure S1). HR-ESI-MS m/z: 723.2161 [M + Na]+(calculated value 700.20), combined with the 1H-NMR spectrum (Figure S2), 13C-NMR spectrum (Figure S3 and S4) and 2D NMR (Figure S5-S8), to determine the molecular formula is C34H36O16, and the degree of unsaturation was 17. The results of the identification of the chemical structure are displayed in Table 1 and Figure 1A.

Structural formula of Ac-TSG and the effect of TSG and Ac-TSG on the viability of MC3T3-E1 cells. Compared with control group. #
Chemical Shifts and Attribution of Ac-TSG.

The Effect of TSG and Ac-TSG on the Viability of MC3T3-E1 Cells
An
The Effect of TSG and Ac-TSG on mRNA Expression of MC3T3-E1 Cells
According to the PCR results Figure 2, the relative expression of BMP-2, Smad5, Runx2, CUL2, CDC23, and Anapc10 mRNA in each group was counted using the 2−ΔΔCt method. Compared with the CONTROL group, the relative expression of BMP-2, Smad5, Runx2, CUL2, CDC23, and Anapc10 mRNA in the H2O2 group was significantly down-regulated (
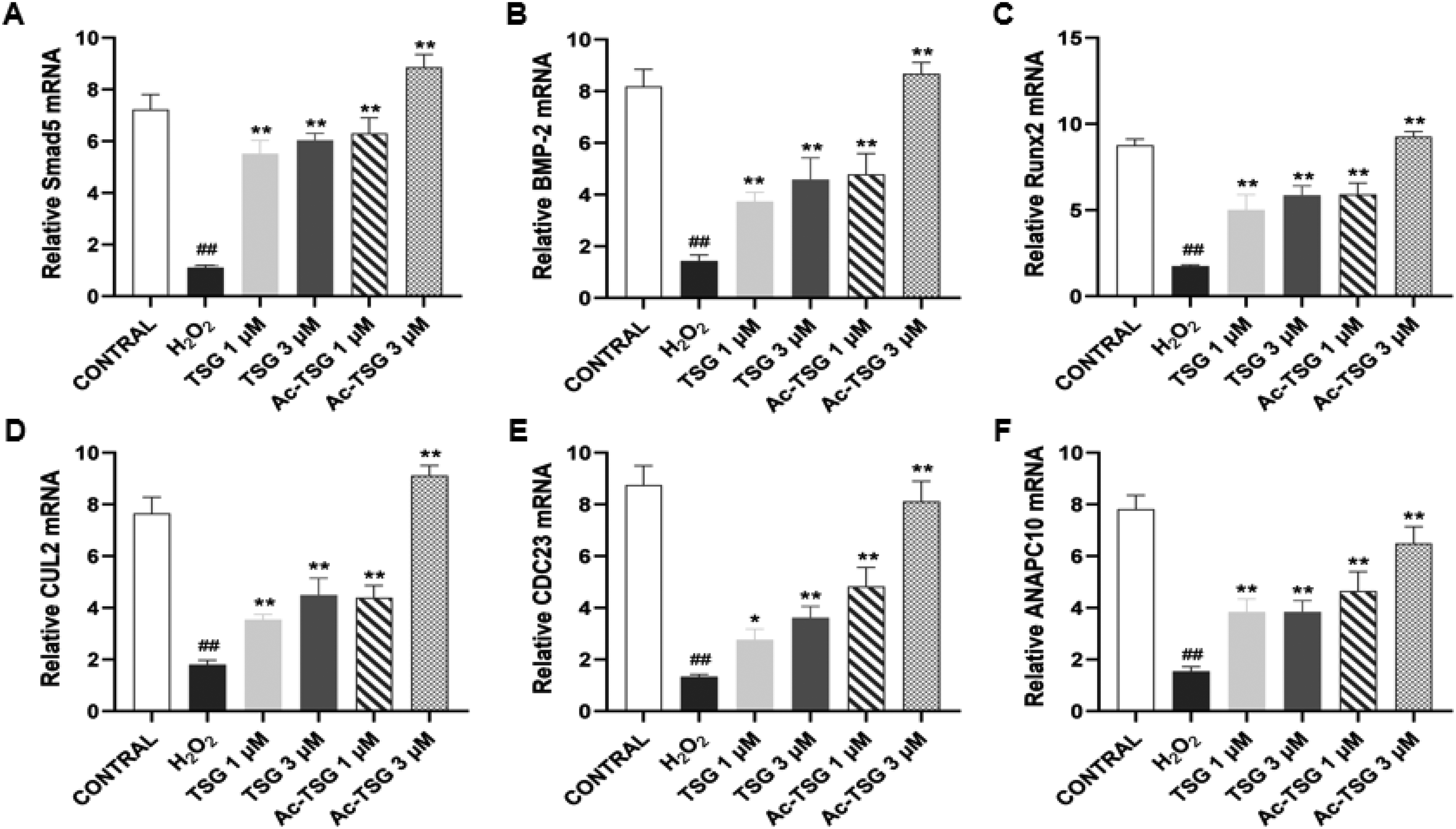
The effect of TSG and Ac-TSG on the expression of osteogenic factor mRNA in MC3T3-E1 cells. Compared with control group. #
Pharmacokinetics of TSG and AC-TSG in Rats
The method was validated according to section “The Effect of TSG and Ac-TSG on Bone Tissue of OVX Rats Was Detected by Micro-CT,” and no detectable interference peak was found in the sample, indicating that endogenous substances in blank plasma did not interfere with the determination of TSG and internal standard (Figure S9). The retention time of TSG was 12.55 min, and the retention time of internal standard was 8.29 min. The method was simple, sensitive and reproducible. It had good linearity in the concentration range of 0.1099–10.99 g mL−1 (Figure S10). The intra-day and inter-day precision RSD was less than 10%, and the recovery rate was 85.91%-88.07%. The UV detection of TSG had strong absorption at 290 nm, which met the basic requirements of pharmacokinetic study (Tables S3-S5).
As shown in Table 2 and Figure 3A, B, the t1/2 was 7.7 times that of TSG, and the AUC0−∞ was 6.9 times that of TSG. Indicating that the action time of Ac-TSG

Pharmacokinetic characteristics of TSG and Ac-TSG in OVX rats after oral administration. (A) C-T curves of instilled TSG in SD rats. (B) C-T curves of instilled Ac-TSG in SD rats. (C) C-T curves of low, medium and high concentrations of Ac-TSG in single instillation in SD rats.
Pharmacokinetic Parameters of Equimolar TSG and Acetyl-TSG in SD Rats by Single Gavage.

The pharmacokinetic parameters of acetyl TSG after single oral administration of 123.1 mmol kg−1, 246.3 mmol kg−1, and 492.6 mmol kg−1 in rats showed that the
Pharmacokinetic Parameters of Low, Medium and High Concentrations of Ac-TSG in SD Rats by Single Gavage.

Effects of TSG and Ac-TSG on Serum Bone Metabolic Markers in OVX Rats
The rats were in good condition and gained significant body weight after ovariectomy (The complete experimental process and rat modeling example are shown in Figure 4A and B). After seven days of modeling, vaginal smears of rats in the OVX model group and the sham-operated group were randomly selected on three consecutive days (The flow chart of
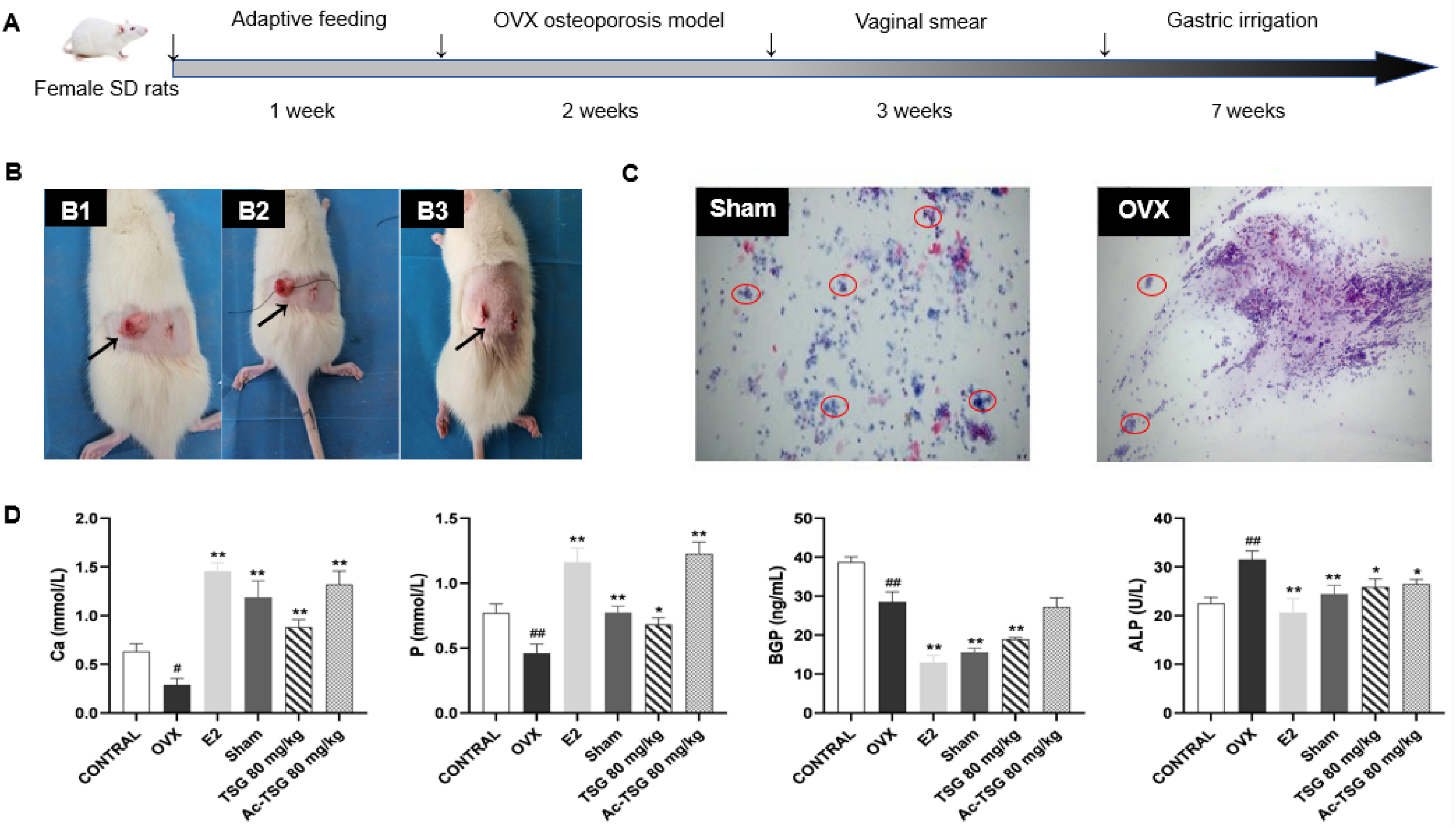
Effects of TSG and Ac-TSG on serum bone metabolic markers in OVX rats compared with CONTROL group. #
As shown in Figure 4D, the serum Ca and P contents of rats in the OVX group were lower than those in the Sham group, but the ALP and BGP contents were higher than those in the Sham group (
The Effects of TSG and Ac-TSG on Osteoclasts and Femoral Tissue in OVX Rats
As shown in Figure 5A of the rat bone histopathological sections, the histopathological sections of the rat femur in the Sham group were structurally intact and did not show trabecular bone loss, but in the OVX group, it was clearly seen that the structure of the bone tissue was disrupted and the number of trabeculae was reduced. However, under the intervention of E2, TSG 80 mg/kg and Ac-TSG 120 mg/kg groups, the structure within the rat bone was moving towards good and intact, and the number of bone trabeculae increased significantly, suggesting that these two drugs have a better anti-osteoporotic effect.

The effects of TSG and Ac-TSG on osteoclasts and femoral tissues in OVX rats and Micro-CT images. (A) Rats femur tissue staining (the arrow refers to the bone trabeculae) scale bars = 50 μm; (B) Rat osteoclast tissue staining (the arrow refers to the osteoclasts) scale bars = 50 μm; (C) Micro-CT three-dimensional reconstruction of bone tissue in OVX rats.
Tartrate-resistant acid phosphatase (TRAP) staining was mainly used to observe the development of osteoclasts. The staining results showed that the number of osteoclasts in the bone tissue of rats in the Sham group was low, and the number of osteoclasts on the bone surface of rats in the OVX group was significantly elevated compared with that in the Sham group. Compared with the OVX group, the number of osteoclasts on the bone surface of rats in the E2 group was significantly decreased, and the number of osteoclasts on the bone surface of rats in the TSG 80 mg/kg and Ac-TSG 120 mg/kg groups was decreased, as shown in Figure 5B.
Micro-CT Images of TSG and AC-TSG in Bone Tissue of OVX Rats
Bone tissues were scanned and imaged and analyzed using micro-CT, and 3D images were processed by software for advanced processing. As shown in Figure 5C, compared with the Sham group, the femur of the OVX group showed a decrease in bone mass, and there was a gap between the bone structures and poor continuity. The microstructure of the femoral bone tissue of the rats after administration was significantly improved, and the bone tissue became dense. Bone mass increased, and continuity was also restored to a certain extent, and the improvement trend of acetylation at the same dose was more obvious.
As shown in Figure S12, compared with the Sham group, the bone volume fraction (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th), trabecular connection density (Conn.D) in the OVX group were significantly decreased (
The Effect of TSG and Ac-TSG on the Expression of Osteogenic Factor mRNA in OVX Rats
RT-PCR results revealed that the expression of CUL2, CDC23, BMP-2, and Runx2 mRNA in the OVX group was significantly lower than that in the Sham group, and the difference was statistically significant (
Safety Evaluation of TSG and AC-TSG on Liver and Kidney Tissues of OVX Rats
The safety of TSG and Ac-TSG was monitored during a gavage period of up to 12 weeks. HE staining of liver and kidney tissues of rats at each dose was performed, and the results, as shown in Figure S14, showed that the structure of rat liver cells was normal, the hepatic cords were arranged neatly and orderly, no steatosis, and no inflammation was found. And kidney tissues were intact, and did not produce an injurious response in liver and kidney tissues of SD rats during the dosing cycle.
Discussions
As the population ages, the number of cases of postmenopausal osteoporosis is steadily rising, making its prognosis and prevention crucial. Postmenopausal osteoporosis is identified as a systemic metabolic bone disorder marked by a reduction in bone mass, destruction of bone tissue microstructure, increased bone fragility, decreased bone strength, and susceptibility to fracture, 22 and its development is a multifactorial, multigenic, multistage biological process. Osteoporosis is the most prevalent chronic bone disease in China and globally, with its extended duration placing a significant strain on the physical and mental well-being of patients, as well as the economy, affecting the prognosis and increasing the mortality rate of patients. 23 Estrogen plays a key role in the prevention and treatment of postmenopausal osteoporosis, inducing osteogenesis through activation of the estrogen receptor signaling pathway and up-regulation of the expression of bone morphogenetic protein (BMP). 24 However, insufficient estrogen secretion in postmenopausal women results in premature ovarian failure and enhanced osteoclast activation, which leads to an imbalance in bone homeostasis, ie, an imbalance in the complex dynamic process of bone resorption and bone formation leading to disorders of bone metabolism and contributing to the onset and progression of osteoporosis.
In rats, postmenopausal osteoporosis is classified as primary osteoporosis and is primarily caused by ovariectomy. Currently, the most frequently used ovariectomy methods in rats include abdominal incision, back incision, and inguinal incision. This study confirmed that the operation time of the back incision method was significantly shortened, the mortality rate was small, the weight loss was stable, the operation was simple and easy, and the risk of wound infection was reduced. Therefore, in this study, a female rat model of ovariectomy was used to study the effects of TSG and Ac-TSG on osteoporosis in rats, and to explore its possible molecular mechanism.
In recent years, it has become increasingly important to scientifically understand how Chinese medicine treats osteoporosis due to its internationalization and modernization. Chinese medicines offer unique advantages in the treatment of osteoporosis due to their lower incidence of side effects, cost-effectiveness, and effectiveness, as well as their multi-component, multi-targeted, and symptomatic modes of action when compared to Western medicines. He Shou Wu has received social attention due to its hepatotoxicity, but the TSG it contains is an effective antioxidant with unique active components and a wide range of pharmacological effects, and it has been mainly used in the clinic in recent years for the treatment of anti-aging.
25
Studies have shown that TSG has good hypolipidemic activity and anti-inflammatory and antioxidant effects26–28 and can play a role in neuroprotection,29–33 cardioprotection, and intestinal homeostasis.34,35 Studies of TSG
The acetyl group, acting as a hydrophobic entity, adds amphiphilic properties to compounds containing multiple hydroxyl groups, thereby broadening the range of natural compounds found in organic organisms and different chemical environments. The diversity of compound structures can be influenced by acetylation, where variations in acetyl groups at different locations and quantities can alter the molecular weight and spatial configuration of compounds, leading to changes in their physical, chemical, and biological properties including water solubility, emulsification, and crystallinity. A study modified tamarind wood glucan by acetylation, which increased thermal stability and solubility.
40
Acetylation of polysaccharides extracted from the seeds of Artemisia annua improved the emulsification properties of the macromolecules, reduced their surface tension to a greater extent, and showed higher stability.
41
Acetylation modification of crude polysaccharides from Cordyceps militaris modified the melting properties of polysaccharides and retained their bioactivity.
42
Meanwhile, acetylation can reduce molecular polarity and significantly improve fat solubility.
43
Research has demonstrated that acetylated Ganoderma lucidum polysaccharides exhibit potent antioxidant abilities in neutralizing DPPH free radicals, as well as boosting macrophage phagocytosis and secretion of tumor necrosis factor-α.
44
Currently, the research on acetylation modification is primarily centered on polysaccharides from plants, animals, and microbes, demonstrating significant potential in delivery systems. After undergoing acetylation modification, the compounds become more stable and can also improve their antioxidant, immune regulation, and anti-tumor capabilities. It offers valuable insights for the development and utilization of natural products, particularly in the realm of food health and natural product development. As a result, this study opted to utilize TSG for the synthesis of Ac-TSG via acetylation modification. On one hand, it enhanced the metabolism rate of TSG in living organisms. Conversely, TSG was acetylated to enhance its stability and improve its suitability for osteoporosis treatment. The experiment was carried out by acetylating TSG and synthesizing acetyl-TSG, and the pharmacokinetic and pharmacodynamic parameters of the two were compared, and the results showed that the prototype drug could be detected in the blood 5 min after intragastric administration of TSG in rats, and the peak concentration can be reached at about 40 min. The half-life is 97.32 ± 13.4 min, and the AUC0−∞ is 59.08 ± 5.57 mg·h·L−1, indicating that TSG is rapidly absorbed in the gastrointestinal tract of rats. After intragastric administration of Ac-TSG, the decomposition product TSG was detected at 15 min, and the peak time was 120 min, which was greater than the peak time of TSG, indicating that Ac-TSG decomposed rapidly but was absorbed slower than TSG. The Cmax of 0.455 ± 0.037 µg mL−1 was lower than that of TSG, indicating that the absorption distribution of acetyl-TSG was smoother than that of TSG. The t1/2 was 750.9 ± 176.5 min, which was 7.7 times that of TSG, and the AUC0−∞ was 410.47 ± 84.5 mg h L−1, which was 6.9 times that of TSG. Indicating that the action time of Ac-TSG
Numerous studies have demonstrated the significant advantages of Chinese herbal medicines and the active ingredients or extracts they contain in the treatment of postmenopausal osteoporosis, including Cistanche, 45 Gleditsiae fructus, 46 Psoralea corylifolia, 47 Salvia miltiorrhiza (Dihydrotanshinone I), 48 Phellodendron amurense, 49 Ligustrum Lucidum alcohol extract, 50 Polygonum sibiricum polysaccharide, 51 Emodin, 52 Resveratrol, 53 and so on. However, no uniform standard has been established for the clinical application of traditional Chinese medicine, so a comprehensive determination of bone mineral density, serum bone metabolism-related biochemical indexes, bone tissue morphology, ie, pathological observation, bone microstructure, bone biomechanics, and other testing indexes is needed in the treatment of postmenopausal osteoporosis.
BMP is derived from the transforming growth factor-β (TGF-β) super-family of multifunctional growth factors that are essential for the developmental processes of the body, including the heart, nerves, and osteogenesis.
54
BMP induces osteoblasts to proliferate and increases the mineralizing activity of osteoclasts, which play a key role in bone homeostasis and repair.
55
The dual effect of BMP on bone resorption and mineralization highlights the BMP signaling important role in bone homeostasis, making it a therapeutic target for diseases such as osteoporosis.
56
BMP has a wide range of roles, among which BMP-2 is one of the most pronounced proteins in osteogenic induction among this growth factors,57,58 which is important in embryonic development as well as skeletal remodeling and homeostasis
Osteoblast differentiation is regulated by the sequential activation of signaling molecules through complex interactions of extracellular signals such as bone morphogenetic proteins (BMPs) and Wnt ligands, and many studies have identified natural and synthetic compounds that have osteogenic activity through the regulation of BMPs/SMADs or Wnt/β-catenin pathways. 66 Numerous studies have shown that BMPs/Smads are important signaling pathways that regulate osteogenic differentiation, BMPs have important roles in promoting osteogenesis and other cellular functions, and activation of BMP-2 promotes the expression of osteoblast marker molecules. 67 Smad1, 5, and 8 are the downstream target genes of BMPs receptors, which are the core of BMPs signaling, and their mechanism of action is due to the ability of BMP-2 ligand to bind to BMPR type II receptor, which in turn phosphorylates BMPR type I receptor, and the activated BMPR type I receptor transmits BMP signals by phosphorylating the BMP-specific R-Smads1, 5, and 8, 68 and these Smads proteins play the activation and repression functions on target genes’ transcripts within the cell. 69 When the Wnt pathway is activated, β-catenin enters the nucleus, binds to transcription factors such as TCF/LEF, and then regulates the expression of downstream genes such as Runx2, which participates in the regulation of osteoblast formation, differentiation and maturation processes. 70 In this experiment, BMP-2, Runx2, CUL2 and CDC23 mRNA expressions were detected by RT-PCR in bone tissues of OVX rats after intervention with TSG and Ac-TSG. The results showed that the expression of BMP-2, Runx2 CUL2 and CDC23 mRNA in the bone tissues of rats in the OVX group was significantly decreased, whereas the gene expression in each group increased after the drug interventions of TSG 80 mg/kg and Ac-TSG 80 mg/kg, among which the up-regulation in the Ac-TSG 80 mg/kg group was the most significantly, indicating that TSG and Ac-TSG have anti-osteoporotic effects, and the anti-osteoporotic effect of Ac-TSG was stronger than that of TSG, and showed concentration dependence within a certain concentration range. This study shows that AC-TSG and TSG have the same pharmacological activity of anti-osteoporosis. Therefore, it is concluded that the mechanism of AC-TSG anti-osteoporosis is mainly related to the regulation of mitochondrial function and reactive oxygen species production involving Nrf2 activation, inhibition of osteoclast differentiation and bone resorption, promotion of osteogenic differentiation, and regulation of osteogenesis and osteoclastogenesis.25,71,72 In addition to BMP-2/Smad5/Runx2 signal transduction, the immune system and chemokine signaling pathways may contribute to the bone protection of AC-TSG and TSG. 73
This study explored the effects of TSG and Ac-TSG on osteoporosis from three perspectives: cellular, pharmacokinetic, and in OVX rats. Overall, we discovered the potential of Ac-TSG for the first time, which was achieved through acetylation modification of TSG, One active ingredient found in the medicinal plant He Shou wu can act as a primary compound for developing anti-osteoporosis treatments, showcasing the plant's value as a potential source for new drugs to combat osteoporosis. Nevertheless, there could be certain constraints to this research. Initially, we were unable to test the isolated compounds for other biological activities, which would have provided a more thorough understanding of their properties. Furthermore, osteoporosis involves numerous mechanisms, and we have only examined the pharmacodynamic aspect for its anti-osteoporotic effects, further studies are needed to fully elucidate the exact molecular mechanisms of the anti-osteoporotic effects exerted by TSG and its derivatives contained in He Shou Wu.
Conclusions
The current study was confirmed through
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241296419 - Supplemental material for The Acetylated 2,3,5,4′-Tetrahydroxystilbene-2-O-β-d -Glucoside Improved Pharmacokinetics and Enhanced Anti-Osteoporosis Effects
Supplemental material, sj-docx-1-npx-10.1177_1934578X241296419 for The Acetylated 2,3,5,4′-Tetrahydroxystilbene-2-O-β-
Footnotes
Acknowledgments
The author sincerely thanks Dr Liangliang Chen for his invaluable guidance and support throughout the experimental process. His insights and expertise have been instrumental in advancing this research. Special thanks are also due to Dr Juanjuan Yang for her assistance with the data and her constructive feedback, which greatly contributed to the quality of this study.
Author Contributions
Conceptualization, P.F. Wei; Methodology, F. Gao; Software, X.Y. Li; Validation, J. Wang; Formal Analysis, W.X. Wei; Investigation, H. Wang; Resources, F. Liu; Data Curation, J.S. Liu; Writing – Original Draft Preparation, J. Wang; Writing – Review & Editing, Y.Q. Yang; Visualization, W. Lan; Supervision, F. Gao; Project Administration, P.F. Wei; Funding Acquisition, P.F. Wei.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethical Committee of Shaanxi University of Chinese Medicine (SUCM), Ethical Certificate No.: Sucmd20200220001 and SUCMDL20220401006
Funding
Shaanxi Province ' Special Support Plan ' regional development talent project, Shaanxi Province Chinese medicine ' double chain integration ' young and middle-aged scientific research innovation team construction project, The national key R & D plan project, (grant number 2022-SLRH-YQ-008, 2017YFC1703901, 2017YFC1703902).
Statement of Animal Rights
Rats were handled according to international regulation for animal use in laboratory. The animal handling procedure was in accordance with international guidelines. 74
Statement of Informed Consent
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
