Abstract
Schisandra chinensis Bail. polysaccharides from China (CSP) and Russia (RSP) were separated by DEAE-52 cellulose column chromatography. The content of neutral polysaccharide was determined by the phenol concentrated sulfuric acid method, the content of acid polysaccharide by the hydroxybiphenyl method, and the monosaccharide composition and molecular weight arrangement of CSP and RSP by 1-phenyl-3-methyl-5-pyrazolone (PMP) pre-column derivatization HPLC. The effects of CSP and RSP on the exercise endurance of mice were compared by the forelimb grip strength test, rota-rod test and weight-bearing swimming. The results showed that one neutral polysaccharide and three acidic polysaccharides could be eluted from a DEAE-52 cellulose column from CSP and RSP, respectively. The content of acidic and neutral polysaccharides in RSP was higher than that in CSP, and the anti-fatigue effect of RSP was more significant than that of CSP.
Keywords
Introduction
Schisandra chinensis, a Magnoliaceae plant, is widely cultivated in China, Japan, Korea and Russia, and has been used in China for more than 2000 years as a famous traditional medicine. 1 The main functions of S. chinensis include liver protection, sedation and hypnosis, anti-fatigue, anti-oxidation and regulation of immune function, and the active compounds are mainly lignans, polysaccharides and volatile oil. 2 Studies have shown that the main effective anti-fatigue component of S. chinensis is its polysaccharides.3,4
The chemical composition of S. chinensis grown in different geographical environments is significantly different.5,6 S. chinensis is mainly distributed in Jilin, Heilongjiang and Liaoning, especially in mountainous areas in the east of Xiaoxing'an Mountains in China, 7 and in the Russian Far East, Khabarovsky region and Kuril Islands. 8 However, there is no research on the comparison of the polysaccharide composition and its anti-fatigue effect between Chinese and Russian S. chinensis polysaccharides (CSP and RSP, respectively).
In recent years, transplanting wild strains and centralized cultivation have become an effective means to optimize plant resources. Our previous research results showed that S. chinensis planted in Ji'an Schisandra chinensis Planting Base of Jilin Province was better than that of other strains in China. 9 In order to optimize further and develop S. chinensis varieties, the Chinese preferred S. chinensis varieties were compared with the preferred varieties provided by the All-Russian Research Institute of Medicinal and Aromatic Plants, in which S. chinensis polysaccharides were extracted with water, precipitated with ethanol, and fractionated on a DEAE-52 column. The contents of neutral and acidic polysaccharides were compared, the monosaccharide components and their molecular weight arrangement were analyzed by 1-phenyl-3-methyl-5-pyrazolone (PMP) pre-column derivatization HPLC, and the anti-fatigue effect of CSP and RSP were compared.
Results and Discussion
Comparison of Neutral and Acidic Polysaccharides
In this study, an optimized DEAE-52 cellulose column was used to separate polysaccharides from Schisandra chinensis. 1 The results showed that both CSP and RSP could be separated into four polar fractions, and according to the elution order, they were one neutral polysaccharide and three acidic polysaccharide components, respectively. The content of neutral polysaccharide was 48.5% and that of acidic polysaccharide 21.5% in RSP, and the content of neutral polysaccharide 46.6% and acidic polysaccharide 18.6% in CSP. The results of the linear gradient elution test of CSP and RSP are shown in Figure 1.

Elution results of RSP (A) and CSP (B).
Combined with the calculated results of the polysaccharide extraction rate, the total polysaccharide extraction rate, and the neutral polysaccharide and acid polysaccharide contents in Russian S. chinensis were higher than those in Chinese S. chinensis. It has been considered that polysaccharide, a kind of general term composed of polyhydroxyaldehyde or polyhydroxyketone and its derivatives and polymers, is one of the main pharmacodynamic substance bases in traditional Chinese medicine, and its content is closely related to its efficacy.10,11 So, the results of polysaccharide content might indicate that the activity of RSP should be superior to that of CSP, but these results needed to be verified by subsequent anti-fatigue animal experiment.
Comparison of Monosaccharide Composition
Under the selected chromatographic conditions, nine monosaccharide standards {mannose (Man), glucuronic acid (GlcA), rhamnose (Rha), galacturonic acid (GalA), glucose (Glc), galactose (Gal), xylose (Xyl), arabinose (Ara) and fructose (Fuc)} could be separated. The molar percentages of the monosaccharide composition of CSP and RSP are shown in Table 1. The results showed that the monosaccharide molar ratio of Man, GlcA, Rha, GalA, Glc, Gal, Xyl, Ara and Fuc in CSP was about 1.7 : 0.1 : 0.5 : 4.4 : 1.0 : 1.3 : 0.2 : 1.5 : 0.1, and that in RSP was about 1.5 : 0.1 : 0.5 : 3.0 : 1.7 : 1.7 : 0.4 : 1.9 : 0.1.
Comparison of Monosaccharide Composition of CSP and RSP.

As shown in Figure 2 and Table 2, the molecular weight of CSP was mainly concentrated in three fragments, with relative molecular weights of 106, 3.7 and 2.4 kDa, and the molecular weight of RSP was also mainly concentrated in three fragments, with relative molecular weights of 41, 3.9 and 2.4 kDa.

HPLC chromatograms of molecular weight of CSP and RSP.
Molecular Weight Arrangement of CSP and RSP.

The monosaccharide composition is the most basic and important link in the structure analysis of polysaccharides, and due to the complexity of the structure, analysis of the monosaccharide composition of polysaccharides generally needs three steps: degradation of polysaccharides, structural modification of the monosaccharides, and their separation and detection. 12 In this study, S. chinensis polysaccharides were enriched by an optimized chemical technology, and the monosaccharides were obtained by acid hydrolysis, and, after derivatization, the monosaccharide composition of the polysaccharides was determined by HPLC-UV. Nine monosaccharide components were well separated; the contents of Man, GalA, Rha and Fuc in Chinese S. chinensis were higher than those in the Russian, while the contents of Glca, Glc, Gal, Xyl and Ara in Chinese S. chinensis were lower than those in the Russian. Since the bioactivity of polysaccharides is closely related to their molecular weight arrangement and composition, these have been included in the quality standards of polysaccharides in the Pharmacopoeia of the People's Republic of China (2015). 13 The results of this study may help to further clarify the pharmacodynamic material basis of S. chinensis polysaccharide.
Comparison of the Anti-Fatigue Effect Between CSP and RSP
The first and last body weights of mice were recorded, the weight growth rate calculated, and the weight change observed. As shown in Table 3, there was no significant difference in the body weight, weight growth rate, and weight change between the CON group and CSP and RSP groups.
Changes in the Body Weight of Mice (Mean ± SD, n = 15).

As shown in Figure 3, compared with the control (CON) group, the forelimb grip strength and swimming time of mice in the CSP-M, CSP-H, RSP-M and RSP-H groups were significantly increased (P < 0.01), and compared with the CON group, the rod rotation time of mice in both the CSP-H and RSP-H groups was significantly increased (P < 0.01), while that in the RSP-L and RSP-M groups was also significantly increased (P < 0.05); compared with those in the CSP-H group, the grip strength and rod rotation time of mice in RSP-H were significantly increased (P < 0.01), in which the fatigue was mainly manifested as the decreased maximum power output of the energy system and decreased muscle strength during the exercise. Polysaccharides and their complexes are not only used as energy resources and building materials in organisms, but also exist in all cell membrane structures and participate in the activity of various cells. 14 Comparison of the results of forelimb grip strength, weight-bearing swimming and the rota-rod test may objectively reflect the differences in the endurance of mice among the experimental groups. 15 The results of the three tests showed that the anti-fatigue effect of Russian S. chinensis was better than that of the Chinese, which may verify the conclusion based on the polysaccharide composition detection, as described above.

Comparison of forelimb grip strength (A), swimming time (B) and rod rotation time (C) of mice (mean ± SD, n = 15). Note: Compared with the CON group, * P < 0.05, ** P < 0.01; Compared with the same dose CSP group, # P < 0.05, ## P < 0.01.
Effects of RSP and CSP on Histomorphology
As shown in Figure 4, the gastrocnemius muscle cells of the normal mice were arranged in order, the cell membrane was clear, the blood vessels were complete, and the nuclei were evenly distributed. In the CON group, after the fatigue endurance exercise, the muscle cells were deformed and broken due to severe pulling, with a disordered arrangement, an unclear cell membrane and an obvious vascular rupture. The distribution of nuclei was disordered, with an aggregation, rupture or disappearance in many locations.

Effects of RSP and CSP on the histomorphology by HE staining (400 × ). Note: The red arrow indicates the nucleus of the gastrocnemius muscle, and the blue arrow points to the abnormal state of gastrocnemius muscle cells.
Compared with the CON group, the deformation and rupture of muscle cells were significantly reduced, and the cell membrane was clear, but there were still cell arrangement disorder, nuclear aggregation rupture, and vascular rupture in the RSP-L and CSP-L groups; in the RSP-M, RSP-H, CSP-M and CSP-H groups, the arrangement of muscle cells was more regular, the cell membrane was clear, the nuclei were evenly distributed under the cell membrane, and the distribution of blood vessels was more normal; in the RSP-H group, the muscle cell membrane was smoother, the texture was neat and clear, and the arrangement of muscle cells was closer.
Analysis of Relevant Biochemical Indicators
During continuous and intense exercise, the body needs to start anaerobic respiration to consume proteins to provide more energy for the body, and the content of urea nitrogen (BUN) after exercise may reflect the degree of protein decomposition into amino acids to provide energy, 16 while lactate dehydrogenase (LDH) is directly related to the production of lactic acid during glycolysis in anaerobic respiration. 17 Strenuous exercise can accelerate the production of free radicals to induce oxidative stress injury. As an important peroxidase widely existing in the body, glutathione peroxidase (GSH-Px) can catalyze the transformation of GSH into oxidized glutathione to reduce toxic peroxides into non-toxic hydroxyl compounds, and total antioxidant capacity (T-AOC) can show the body's antioxidant capacity.18,19
As shown in Figure 5, compared with those in the CON group, BUN contents in the serum of mice were significantly decreased in the CSP-H, CSP-M and RSP-H groups (P < 0.05, P < 0.01). LDH activities in the serum of mice were significantly decreased (P < 0.01), while GSH-Px activities and T-AOC were significantly increased (P < 0.01) in the CSP and RSP groups, of which the increase in the activity of GSH-Px in RSP-M was the most significant. In addition, the effects on the other indicators were most significant in the CSP-H and RSP-H groups, showing a dose-dependent manner; the decrease in the content of BUN in RSP-H was more significant than that in the CSP-H group (P < 0.05), and the increase in the activity of GSH-Px in the CSP-H group was more significant (P < 0.01).
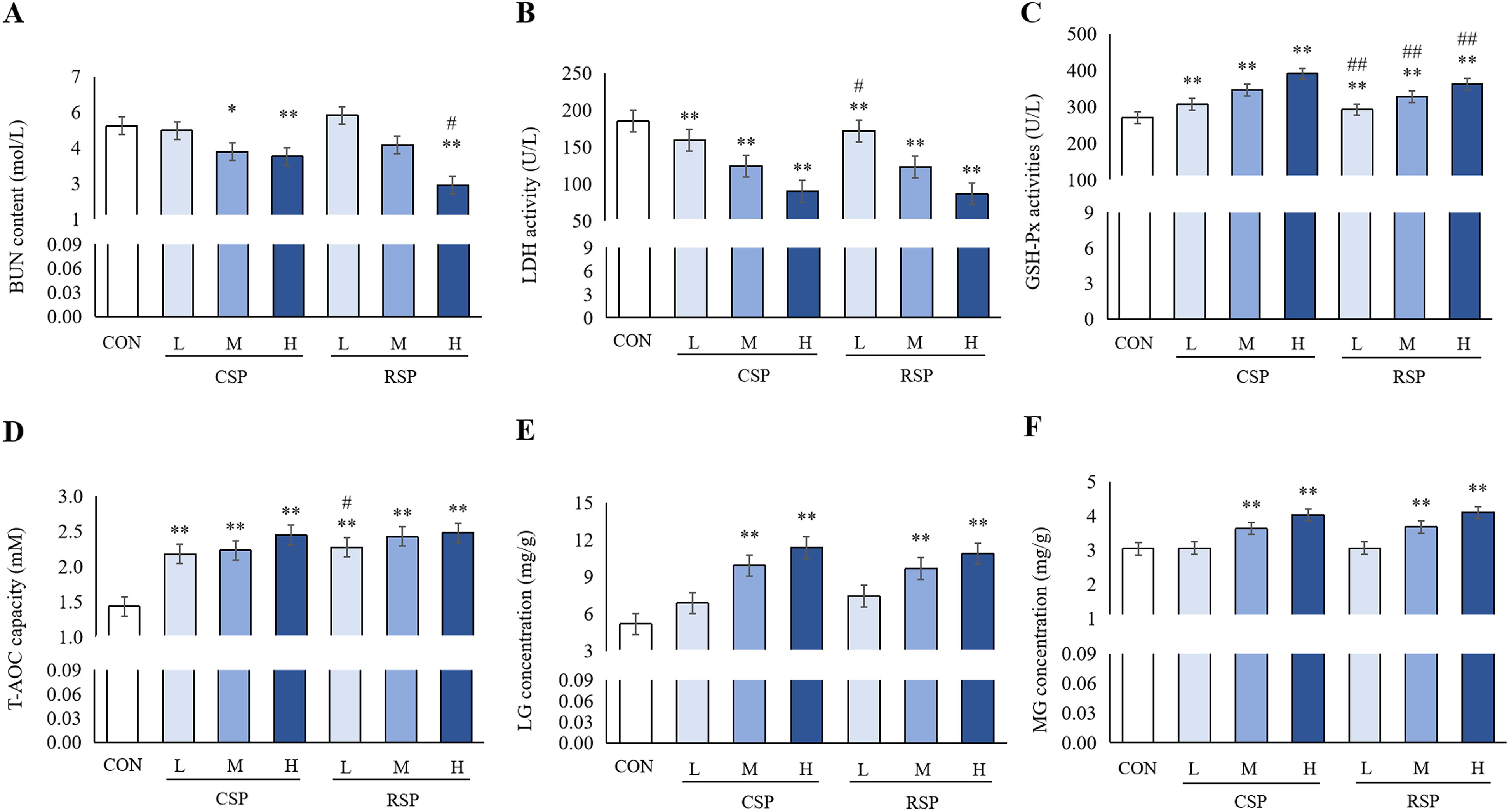
Comparison results of the serum BUN content (A), LDH (B) and GSH-Px activities (C), T-AOC (D), liver glycogen (E) and muscle glycogen (F) of mice (mean ± SD, n = 15). Note: Compared with CON group, * P < 0.05, ** P < 0.01; Compared with the same dose CSP group, # P < 0.05, ## P < 0.01.
Liver glycogen and muscle glycogen are the main substrates for maintaining glycolysis, oxidative phosphorylation and physiological blood glucose levels during exercise. After a period of exercise, the glycogen in the body will decompose into lactic acid to release energy for the movement of muscles. When the glycogen content in the body is not enough to provide energy for body activities, the body will become fatigued. 20
Compared with those in the CON group, the liver glycogen and muscle glycogen of mice were significantly increased in the CSP-H, CSP-M, RSP-H and RSP-M groups (P < 0.01), with the increase being most significant in the CSP-H and RSP-treated groups, showing a dose-dependent manner. There was no significant difference in the glycogen content between the CON group and the RSP-L and CSP-L groups, and there was no significant difference between the RSP and CSP groups at the same doses, suggesting that both CSP and RSP could increase the glycogen reserve in the liver and muscle of mice, and there was no significant difference in the glycogen reserve between them.
Both CSP and RSP could significantly inhibit the increase of BUN content caused by the metabolic transformation of amino acids into urea in the blood to reduce the content of BUN in the serum of fatigued mice after endurance exercise, and reduce the generation of glycolysis products of carbohydrates in anaerobic conditions during strenuous exercise, so as to lower the activity of LDH. At the same time, both CSP and RSP could significantly increase the reserve of liver and muscle glycogen, and reduce their consumption. Both CSP and RSP could also enhance the T-AOC and GSH-Px activity to improve the body's antioxidant level, and RSP could more significantly delay the fatigue and improve the endurance level than CSP, to enhance the anti-fatigue ability in mice.
Conclusion
In this study, differences in the content of Schisandra chinensis acidic and neutral polysaccharides, monosaccharide composition, and molecular weight arrangement of polysaccharides were compared between CSP and RSP. Under the experimental conditions, a DEAE-52 cellulose column could effectively separate the polysaccharides into four uniform components, and, moreover, the PMP pre-column derivatization HPLC method showed good resolution for the determination of the monosaccharide composition of the S. chinensis polysaccharides. The HPLC procedure can be used as a reliable analytical method for the analysis of the monosaccharide composition of S. chinensis polysaccharides. The results indicate that the content of acidic and neutral polysaccharides in S. chinensis polysaccharide from Russia were higher than those from China. The results of HPLC analysis suggest that CSP and RSP both are composed of nine monosaccharides (Man, GlcA, Rha, GalA, Glc, Gal, Xyl, Ara and Fuc), but the molar ratio of each monosaccharide is different, and the molecular weight arrangement of the two polysaccharides was also different.
The anti-fatigue effects of CSP and RSP were compared by the weight-bearing swimming test, rota-rod test, forelimb grip strength test, gastrocnemius histomorphology observation, and serum, liver and muscle biochemical indicators. The results indicate that CSP and RSP could significantly delay fatigue, improve the endurance level to enhance the exercise ability of mice, and at the same doses, the effect of RSP was more significant than that of CSP. It is revealed in this study that there are differences in the composition of polysaccharides between CSP and RSP varieties, which may provide support in basic data for the optimization and development of S. chinensis varieties. In addition to the polysaccharides, S. chinensis contains many other active components such as lignans and volatile oil. We will continue to conduct more in-depth research in order to comprehensively analyze and compare the differences between Chinese and Russian S. chinensis.
Experimental
Instruments
LC-20AT SPD-20A HPLC system (Shimadzu, Japan); Spectrophotometer (Beijing Purkinje Instruments Co., Ltd, Beijing, China); YLS-13A rat and mouse grip-strength meter (Chengdu Taimeng Software Co., Ltd, Chengdu, China); YLS-4C mouse fatigue rotating rod instrument (Shanghai Ruanlong Technology Development Co., Ltd, Shanghai, China); Infinite M200 microplate reader (TECAN, Switzerland).
Materials and Reagents
Schisandra chinensis (Jilin Ji'an Schisandra chinensis Planting Base of Jilin Province, October 2019, Jian, China); S. chinensis (All-Russian Research Institute of Medicinal and Aromatic Plants, October 2019, Russia); DEAE-52 cellulose (Beijing Ruida Henghui Co., Ltd, Beijing, China); chromatographic grade methanol and acetonitrile (Tianjin Yongda Chemical Reagent Co., Ltd, Tianjin, China); anhydrous ethanol (Tianjin Zhiyuan Chemical Reagent Co., Ltd, Tianjin, China); m-hydroxybiphenyl (Shanghai Yansheng Biochemical Reagent Co., Ltd, Shanghai, China); sodium tetraborate [Maryer (Shanghai) Chemical Technology Co., Ltd, Shanghai, China]; concentrated sulfuric acid (Tianjin Kaixin Chemical Industry Co., Ltd, Tianjin, China); phenol (Tianjin Fuchen Chemical Reagent Co., Ltd, Tianjin, China). The above reagents were all analytically pure. LDH assay kit (No. A020-2), T-AOC assay kit (No. A015-2-1), GSH-Px assay kit (No. A005-1) and BUN assay kit (No. C03-2-1) (Nanjing Jiancheng Bioengineering Institute, Nanjing, China); glycogen content test kit (No. 20210426) (Division of Biochemical Kits, Beijing Solarbo Technology Co., Ltd, Beijing, China); Man, GlcA, Rha, GalA, Glc, Gal, Xyl, Ara and Fuc (Sichuan Weikeqi Biological Technology Co., Ltd, Chengdu, China), with a purity more than 98%.
Experimental Animals
Male ICR mice (18-22 g, Chang Chun Yisi Experimental Animal Technology Co., Ltd, Changchun, China) were acclimatized to the laboratory environment for one week before experiments, during which the mice were raised under standard laboratory conditions for one week, with free access to water and food. All the experiments were approved by Beihua University Laboratory Animal Ethics Committee (No. 20191015).
Extraction and Purification of Polysaccharides
Schisandra chinensis was crushed into powder, which was passed through a 40-mesh sieve and then dried to a constant weight in an oven at 50 °C. The dried powders were extracted with a 10 times volume of distilled water, and reflux-extracted for 4 h. The extract was concentrated to 1/5 of its original volume by rotary evaporation and then the concentrated extract was mixed with 4 times its volume of 95% ethanol; 12 h later, the sediment was taken and lyophilized, namely the crude polysaccharide sample of Schisandra chinensis. One g of the crude polysaccharides was dissolved in 100 mL distilled water, then 4% acicular activated carbon granules were added for decoloration. The activated carbon was removed after the solution had been heated on a water bath at 60 °C for 1 h. The solution was added to 20 mL of chloroform-n-butanol (5:1) for extraction and deproteinization, which was repeated 3 times. The organic layer was discarded and the water layer was collected to obtain the S. chinensis polysaccharide by ethanol precipitation. The extraction rates of RSP and CSP obtained by this experimental method were 9.93% and 8.28%, respectively.
Preparation and Separation on DEAE-52 Cellulose Chromatographic Column
Dry DEAE-52 cellulose powders were soaked in a 5 times volume of water overnight, then the solution was filtered, and the upper water was discarded to remove the impurities. The residue was dissolved and soaked in 0.5 mol/L NaOH solution for 2 h, during which the solution was continuously stirred, and the residue was washed with water until the pH test paper showed that it was neutral and then put in a separating funnel for draining. Then, the residue was dissolved in 0.5 mol/L HCl solution for 2 h, during which the solution was continuously stirred, then washed with water until the pH test paper showed that it was neutral, and then put in a separating funnel for draining to obtain the swelling activated DEAE-52 cellulose. After ultrasonic degassing for 30 min, the chromatographic column (1.2 × 65 cm) was loaded, in which the natural settlement reached to a constant height. Seven mg of Schisandra chinensis polysaccharides were dissolved in 1 mL deionized water. The solution was mixed by vortex and then centrifuged at 3000 r/min for 10 min. The precipitate was discarded and the supernatant was loaded into the chromatographic column, and the loading volume was 5% of the column volume. Thirty mL of deionized water and 70 mL of 0.1–0.7 mol NaCl solution were loaded in turn for linear elution, and the flow rate was controlled at 0.5 mL/min in the whole process. Two mL eluent was collected from each tube, in total 50 tubes, and the contents of neutral and acidic polysaccharides in each tube were determined.
Determination of Neutral Polysaccharide by Phenol-Concentrated Sulfuric Acid Method
Five mg of dried glucose standard was diluted with distilled water to a constant volume of 50 mL to prepare a 0.1 mg/mL glucose standard solution, which was diluted with distilled water to produce 0.1, 0.05, 0.04, 0.03, 0.02 and 0.01 mg/mL solutions. From each of the glucose solutions of different concentrations, 0.5 mL was taken and added to 1.0 mL of 5% phenol solution newly prepared, and mixed evenly. The glucose-phenol solution was heated in a water bath at 60 °C for 5 min, then 2.5 mL concentrated sulfuric acid was added, mixed well, and placed in an ice water bath to cool for 20 min. The ultraviolet spectrophotometer was set to zero with distilled water, and the absorbance at 490 nm was measured. A standard curve was drawn with absorbance (A) as abscissa and glucose concentration (mg/mL) as ordinate, and the calculation was performed according to the following equation: Y = 0.117x - 0.018 (R2 = 0.994).
Determination of Acidic Polysaccharide by m-Hydroxybiphenyl Method
Twenty-five mg of dried galacturonic acid was dissolved in water and diluted to a constant volume of 250 mL to prepare a 0.1 mg/mL galacturonic acid standard solution; from this, solutions of 0.1, 0.08, 0.06, 0.05, 0.025 and 0.01 mg/mL were prepared. The galacturonic acid solutions (0.5 mL each) were added to 3 mL of sodium tetraborate-sulfuric acid solution in an ice water bath, mixed by vortex, heated in a boiling water bath for 5 min, cooled, and added to 20 μL of a 3 mg/mL m-hydroxybiphenyl solution. Scanning was performed in the range of 200 to 800 nm, in which the ultraviolet spectrophotometer was set to zero with distilled water, and 523 nm was taken as the maximum absorption wavelength. The absorbance was measured at 523 nm, and the standard curve was drawn with absorbance (A) as abscissa and galacturonic acid concentration (mg/mL) as ordinate. The calculation equation was Y = 0.093x - 0.013 (R2 = 0.993).
Pre-Column Derivatization of PMP
Two mg of the sample prepared in the Section “Extraction and purification of polysaccharides” was dissolved in 1 mL of a methanol solution containing 1 mol hydrochloric acid, the tube was sealed with N2, and the sample hydrolyzed in a 80 °C constant temperature metal bath for 16 h. After drying with an air pump, 1 mL of 2 mol trifluoroacetic acid was added to the sample solution, which was hydrolyzed at 120 °C for 1 h and then dried using an air pump.
The dried sample was dissolved in 0.5 mL of 0.3 mol NaOH solution and 0.5 mL of PMP-methanol reagent, and 0.2 mL of the sample solution were placed in a water bath at 70 °C for 30 min. Then, 0.1 mL of 0.3 mol hydrochloric acid solution was added to the sample solution, mixed well, and 0.7 mL trichloromethane was added to it for extraction, in which the lower layer of trichloromethane was sucked out and discarded, and the water layer was reserved and extracted twice. Finally, the extract was passed through a 0.22 μm filter membrane, and the filtrate used as the test sample for the HPLC analysis of the monosaccharide components of S. chinensis polysaccharides.
Determination of Monosaccharide Composition
Column: COSMOSIL 5C18-PAQ (250 mm × 4.6 mm, 5 μm); mobile phase A: 0.1 mol/L phosphate buffer (pH = 7.0), mobile phase B: acetonitrile/water (81/19, v/v); column temperature: 35 °C; flow rate: 1.0 mL/min; detection wavelength: 245 nm; sample size: 20 μL.
Ten mg of each of Man, GlcA, Rha, GalA, Glc, Gal, Xyl, Ara, and Fuc were accurately weighed and dissolved in 1 mL anhydrous pyridine, then added to 10 mg hydroxylamine hydrochloride, and mixed well. The solutions were heated in a water bath at 90 °C for 30 min, then cooled down to room temperature. One mL anhydrous acetic anhydride was added for acetylation at 90 °C for 30 min, and cooled down in the same way. Finally, the solutions were passed through a 0.22 µm filter membrane, and the filtrates were kept for use.
Determination of Molecular Weight
The molecular weights of the polysaccharides were determined by high-performance size-exclusion chromatography (HPSEC). The sample was dissolved in distilled water, applied to a SHIMADZU LC-20AT HPLC system equipped with a TSK-GEL G3000 PWXL column, eluted with 0.2 mol/L NaCl and detected by a SPD-20A refractive index detector. Dextran standards with different molecular weights (T-2000, T-70, T-40, T-20, and T-10) were used to calibrate the column and establish a standard curve. Molecular standard curve: Y = −0.222x + 1.4051.
Animal Grouping and Administration
Mice were randomly divided into 7 groups, including control group (CON), high-dose Chinese S. chinensis polysaccharide group (CSP-H), medium-dose Chinese S. chinensis polysaccharide group (CSP-M), low-dose Chinese S. chinensis polysaccharide group (CSP-L), high-dose Russian S. chinensis polysaccharide group (RSP-H), medium-dose Russian S. chinensis polysaccharide group (RSP-M) and low-dose Russian S. chinensis polysaccharide group (RSP-L), with 15 mice in each group. Mice in CSP-L, CSP-M, CSP-H, RSP-L, RSP-M and RSP-H were intragastrically given 25, 50 and 100 mg/(kg·d) CSP and RSP, respectively, and those in the CON group were given an equal volume of distilled water in the same way. According to the weight of the mice, the intragastric volume was 0.1 mL/10 g, once a day for 15 days. The weight of mice in each group was measured before administration and 15 days after administration, and the average weight was used for comparison of the difference in the body weight of mice.
Anti-Fatigue Test
A forelimb grip strength tester was used to test the fatigue endurance of mice 30 minutes after the last administration. Each mouse was tested three times, and the average value of the three-time grip strength was recorded.
Three days before the last administration, the mice were put into a rota-rod tester, in which the rotating speed was 30 r/min. The first two days were for adaptation training. After the last administration, the mice completed the forelimb grip strength test and then took a rest for 60 min, and the time of mice falling from the rota-rod was recorded.
After the last administration, the mice were rested for 60 min after completing the rota- rod test, then the tail of each mouse was loaded with lead to 5% of the weight of the mouse, which was then placed in the swimming pool. The time from the mouse entering the water to the time when the head of the mouse could not come out of the water within 8 sec was recorded.
Detection of Biochemical Indicators in the serum and Gastrocnemius Muscle Tissue of Mice
After swimming for 30 min, the mice were anesthetized by intraperitoneal injection of 0.3% pentobarbital sodium (0.5 mg/10 g), and the eyeball blood, liver and gastrocnemius muscle tissues were taken immediately. The blood was centrifuged at 4000 r/min for 10 min, and the serum taken for detecting blood urea nitrogen (BUN), lactate dehydrogenase (LDH) activity, total antioxidant capacity (T-AOC), and glutathione peroxidase (GSH-Px) activity, according to the instructions of the kits. Gastrocnemius muscle tissues from three mice in each group were randomly selected and soaked in 10% formalin for the preparation of pathological sections. HE staining was used for the observation of the morphological changes in the muscle tissue of mice under a microscope (another three mice without any treatment were taken as normal control). The liver and the rest of the gastrocnemius muscle were cut up and homogenized with 4 °C normal saline. The homogenates were centrifuged at 3000 r/min for 10 min, and the supernatants taken and used for the determination of the content of liver glycogen and muscle glycogen. The process of the animal experiment is shown in Figure 6.

Grouping and administration of rats and experimental processes.
Statistical Methods
All values are expressed as mean ± SD. The number of samples in each group is expressed as 'n'. SPSS software (Windows version 19.0) was used for statistical analysis. One way ANOVA was used for the comparison between groups. It was considered that P < 0.05 or P < 0.01 meant a statistically significant difference.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
All animal experiments were approved by the Ethical Committee of Beihua University. All procedures were conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals (China).
Footnotes
Author Contributions
Chen Jianguang, Andrey Tsitsilin and Sun Jinghui contributed to the conception and design of the study; Li Han and Chen Cong carried out the HPLC analysis; Feng Yanbo and Li Han were involved in the determination of SCP; Lin Hao, Chen Cong and Wang Chunmei participated in the animal experiments and data collection; Li He and Wang Chunmei performed the statistical analysis; Li Han, Chen Jianguang and Sun Jinghui wrote the manuscript. All authors read and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
This work was supported by the Natural Science Foundation of Jilin Province (No. 20200404022YY), Jilin Science and Technology Innovation Development Plan (No. 20190601177).
Trial Registration
Not applicable, because this article does not contain any clinical trials.
