Abstract
Spirulina are multicellular and filamentous cyanobacteria that have achieved considerable popularity in the health sector, food industry, and aquaculture. In the present study, we aimed to evaluate the antifatigue effects of Spirulina-derived peptides on Institute for Cancer Research mice and explore the association between antifatigue activity and fat metabolism involving the adenosine monophosphate-activated protein kinase (AMPK) signaling pathway. We extracted the peptides from Spirulina by enzymatic hydrolysis and ultrafiltration. The mice were orally administered with Spirulina peptides (0.125, 0.5, and 2 mg/g bw/day) daily for 4 weeks. We found that Spirulina peptides, especially the high-dose group, significantly prolonged the swimming time by 126.1%, increased the activities of antioxidant enzymes, and decreased the content of malondialdehyde by 60.2% compared with the glutathione (GSH) group. The levels of some indicators of exercise fatigue, including lactic dehydrogenase, blood lactic acid, and creatine phosphokinase, were reduced. In the high-dose group, these indicators were reduced by 40.7%, 22.3%, and 11.3% compared with the GSH group. Spirulina peptides did not excessively consume blood sugar or glycogen in the liver and muscle to produce energy. However, the triglyceride level was reduced, and the level of free fatty acids was increased. Besides, the proteins in the AMPK signaling pathway were activated. Taken together, these findings indicated that Spirulina peptides could effectively alleviate physical fatigue by reducing the production of lactic acid and improving antioxidant capacity. Spirulina peptides also helped increase the energy resources by activating the AMPK signaling pathway to utilize fat metabolism.
Fatigue is best defined as difficulty in either initiating or sustaining voluntary activities and can be classified into mental and physical fatigue. 1 Fatigue marks not only a temporary decline in work efficiency but also a precursor to certain diseases, 2 such as cancer, multiple sclerosis, depression, and Parkinson’s disease. 3,4 Although fatigue has become an increasingly common health issue due to the rapid development of modern society, 5 drugs or therapies for its effective treatment remain largely unsatisfied. 6
It is well known that production of free radicals, depletion of energy, and accumulation of detrimental metabolites all lead to fatigue, and oxidative stress plays a significant role in the pathophysiology of fatigue caused by exercise. 7 Some studies have indicated that adequate superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and catalase (CAT) activities play an essential role in preventing and reducing oxidative damage induced by fatigue. 8 However, these antioxidant defense mechanisms will become weaker during chronic fatigue or under other disease conditions. 9 Therefore, improvement in these defense mechanisms is of great significance to help relieve fatigue.
In recent years, bioactive peptides have been shown to ameliorate fatigue by providing energy, enhancing antioxidant capacity, and scavenging metabolic waste and excess free radicals. 10 For instance, a peptide termed SH200, isolated from seahorse extracts, can improve the exercise performance of mice, enhance the mitochondrial function, and protect the muscle fibers. 11 Wheat peptide supplementation can effectively improve the exercise capacity of one-time exhaustive exercise and maintain long-term emotional stability, 12 and the mechanism underlying the antifatigue properties of peptide extracted from sheep placenta is related to the improvement of antioxidant activities, glycogen reserves, and the ultrastructures in gastrocnemius muscle cells. 13
Spirulina species are edible and multicellular blue-green cyanobacteria, 14 which contains 60%-70% of proteins 15 . Most studies on Spirulina-derived peptides focus on antihypertensive activity 16 and antitumor, antiaging, antimicrobial, and antiproliferation properties. 14,16 -19 For example, Sun et al have isolated an antibacterial peptide SP-1 (the amino acid sequence is KLVDASHRLATGDVAVRA) from Spirulina platensis, which can inhibit Escherichia coli and Staphylococcus aureus. 15 Lu et al have purified an angiotensin I-converting enzyme inhibitory peptide Ile-Gln-Pro from S. platensis. 16 An antitumor polypeptide Y2 from trypsin digest of S. platensis proteins has a maximum inhibition of 97% on MCF-7 and HepG2 cells. 20 Therefore, we conducted an in-depth study on whether Spirulina peptide had antifatigue activity.
In the present study, Institute for Cancer Research (ICR) mice were treated with Spirulina peptides for 28 days and were then evaluated for weight-loaded forced swimming time, energy consumption, protective effects, metabolite accumulation, and energy acquisition mechanisms based on the adenosine monophosphate-activated protein kinase (AMPK) signaling pathway. We first found that Spirulina peptides could activate the fat metabolism pathway through the AMPK signaling pathway to relieve fatigue, instead of consuming either blood sugar or glycogen in liver and muscle excessively.
Materials and Methods
Materials
Fresh S. platensis BR01 was provided by Chenghai Baoer Corporation, Yunnan. The suitable temperature for S. platensis is within 25-27 °C, and the optimum pH 8.5-10.5. Spirulina platensis BR01 was harvested when the number of trichome rings reached 6-8, and the OD value 0.8 or more.
Protamex was purchased from Beijing Novozymes Company; alkaline proteinase from Beijing Solarbio Company; glutathione (GSH) from China Sinopharm Chemical Reagent Co., Ltd.; kits of blood glucose (GLU), malondialdehyde (MDA), SOD, GSH-Px, and CAT from Nanjing Jiancheng Biological Engineering Research Institute, and ELISA kits of liver glycogen (GLY), muscle glycogen (MG), hexokinase (HK), free fatty acid (FFA), adipose triglyceride lipase (ATGL), and carnitine palmitoyltransferase 1 (CPT1) from Shanghai Kexing. Anti-β-actin was purchased from America Santa Cruz Biotechnology, anti-p-AMPK, anti-p-acetyl CoA carboxylase (ACC), anti-Forkhead box O1 (FoxO1), and anti-Sirtuin1 (SIRT1) from America Cell Signaling Technology; horseradish peroxidase (HRP)-conjugated secondary antibody from America Thermo Fisher Scientific, and the 5000 and 2000 Mw ultrafiltration membranes from Germany Sartorius Group; the model of the ultrafiltration membranes was VF20H9.
Preparation of Spirulina Peptides
Fresh Spirulina was dispersed into distilled water. Solutions were stirred and ultrasonicated at 500 W for 5 minutes, followed by centrifugation. The supernatant was collected and freeze-dried to obtain the protein powder. According to the previously described methods, 16,21,22 Spirulina protein was successively hydrolyzed by protamex and alkaline proteinase. Briefly, the solution containing 3% protamex and protein at 1:30 (m/v) was reacted at pH 7 °C and 45 °C for 6 hours. After heat inactivation, 6% alkaline proteinase was added to the solution at a ratio of 1:30 (m/v), followed by reaction at pH 9 °C and 45 °C for 6 hours. The supernatant of the enzymatic hydrolysate was obtained by centrifugation and then successively ultrafiltered through membranes with molecular cut-offs of 5000 and 2000 Mw to obtain the peptides with molecular weight less than 2000 Mw. Finally, the peptides were collected and freeze-dried. The sequences of the peptides were analyzed by online nanospray LC1000 system (Thermo Fisher Scientific, MA, USA), and identified by PEAKS Studio X+ (Bioinformatics Solutions Inc. Waterloo, Canada) referring to PEAK DATABASE method. 23 The 10 peptides with the highest percentage in the database searches were listed in Supplemental Table S1.
Animal-Experimental Design
ICR mice (male, aged 6-8 weeks, weighing between 25 and 30 g, SCXK 2018-0004) were purchased from Shanghai Jiesijie Experimental Animal Co., Ltd. (SCXK 2018-0004, Shanghai, China), and housed in the Animal Center of Ningbo University Medicine College (Ningbo, China). Briefly, 5 mice of the same sex were housed in a cage, maintained under a 12-hour light/12-hour dark cycle (08:00 AM lights on), and given free access to food and water. Housing and experimental environments were temperature and humidity controlled (21 ± 2 °C and approximately 60%, respectively). All experimental procedures were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals, and animal-related experiments were approved by the Ethical Committee of Animal Use and Protection of Ningbo University Health Science Center.
After an adaptation period of 1 week, ICR mice (SCXK 2018-0004) were randomly divided into 5 groups (n = 16) as follows. Mice in the negative control group were fed with 10 mL/kg of distilled water by oral gavage. In the positive control group, mice were administered with 0.5 mg/g of GSH by oral gavage. Spirulina peptides were given to the mice at doses of 0.125, 0.5, and 2 mg/g by oral gavage, which was referred to as the animal-experimental design of Guo et al. 5 All solutions were given once a day. Mice in all groups were weighed once per week for 4 weeks.
Swimming Exhaustion Experiment
After the last oral gavage, 8 mice from each group were taken out for the swimming exhaustion experiment. Briefly, mice were individually placed in a swimming pool (30 cm in depth and 25 cm in diameter) filled with fresh water at 30 ± 2 °C. A lead block (7% of body weight) was loaded on the tail root of the mice. The water was constantly agitated to ensure the continuous exercise of the mice. The swimming time to exhaustion was used as the index of forced swimming capacity. When the mice failed to float to the surface of the water for 10 seconds, they were assessed to be exhausted, and the time was recorded. The mice were rescued from the pool, dried with paper towels, and placed back into their cages.
Determination of Physiological and Biochemical Indices
After the last oral gavage, the mice were allowed to rest for 30 minutes. Then they were placed in a swimming pool (30 cm in depth and 25 cm in diameter) filled with fresh water at 30 ± 2 °C for 30 minutes. The water was constantly agitated to ensure the continuous exercise of the mice. The GLU level was examined by an Accu-Chek Active blood glucose meter and paper (Roche, Mannheim, Germany). Blood samples were respectively collected with anticoagulant by removing the eyeball, and livers and skeletal muscles were collected via biopsy. Whole blood was centrifuged at 3000 × g for 10 minutes and then stored at −80 °C. Contents of GLU, MDA, SOD, GSH-Px, and CAT were determined according to the manufacturer’s instructions. GLY, MG, HK, FFA, ATGL, and CPT1 were detected using ELISA kits. Except for GLY and MG, serum was used for the detection of the other indices. TG, creatine phosphokinase (CK), lactate dehydrogenase (LDH), blood lactic acid (BLA), and blood urea nitrogen (BUN) were evaluated using the biochemistry analyzer ADVIA 2400 (Siemens, Berlin, Germany).
Western Blotting Analysis
The liver tissue was lysed in lysis buffer. Subsequently, equal amounts of extracted proteins (100 µg) were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then electrotransferred onto a polyvinylidene fluoride membrane. The membrane was blocked with blocking solution (5% skim milk) at room temperature for 1 hour and then incubated with the following specific primary antibodies under the same conditions: anti-β-actin (1:8000), anti-p-AMPK (1:1000), anti-p-ACC (1:1000), anti-FoxO1 (1:1000), and anti-SIRT1 (1:1000). Blots were washed with Tris-buffered saline containing 0.05% Tween-20 and then incubated with HRP-conjugated secondary antibody at room temperature for 40 minutes. Immunoreactive bands were visualized by Western Bright ECL (Advansta, California, America), followed by densitometry analysis.
Statistical Analysis
All data are expressed as means ± SD. Differences between groups were determined by analysis of variance and Tukey’s test. A P value less than 0.05 was considered as statistically significant.
Results
Effect of Spirulina peptides on body weight of mice
There was no significant difference in the body weight of mice in the treatment groups compared with the negative and positive control groups during the whole period of the experiment (P > 0.05, Figure 1). Our data demonstrated that Spirulina peptides had no significant effect on body weight.

Effect of Spirulina peptides on body weight of mice. Positive refers to mice that were treated with glutathione at 0.5 mg/g. Low, medium, and high refer to mice that were treated with Spirulina peptides at 0.125, 0.5, and 2 mg/g for 28 days, respectively.
Effects of Spirulina Peptides on Weight-Loaded Forced Swimming Endurance Time
The swimming time to exhaustion of the Spirulina peptide treatment groups was significantly longer compared with the negative and positive control groups (P < 0.05, Figure 2). Compared with the negative control group, the swimming time in the low-dose group was increased by 177.7% and by 264.9% in the medium-dose and high-dose groups. The improvement of exercise endurance was the most powerful representation of the antifatigue effect.
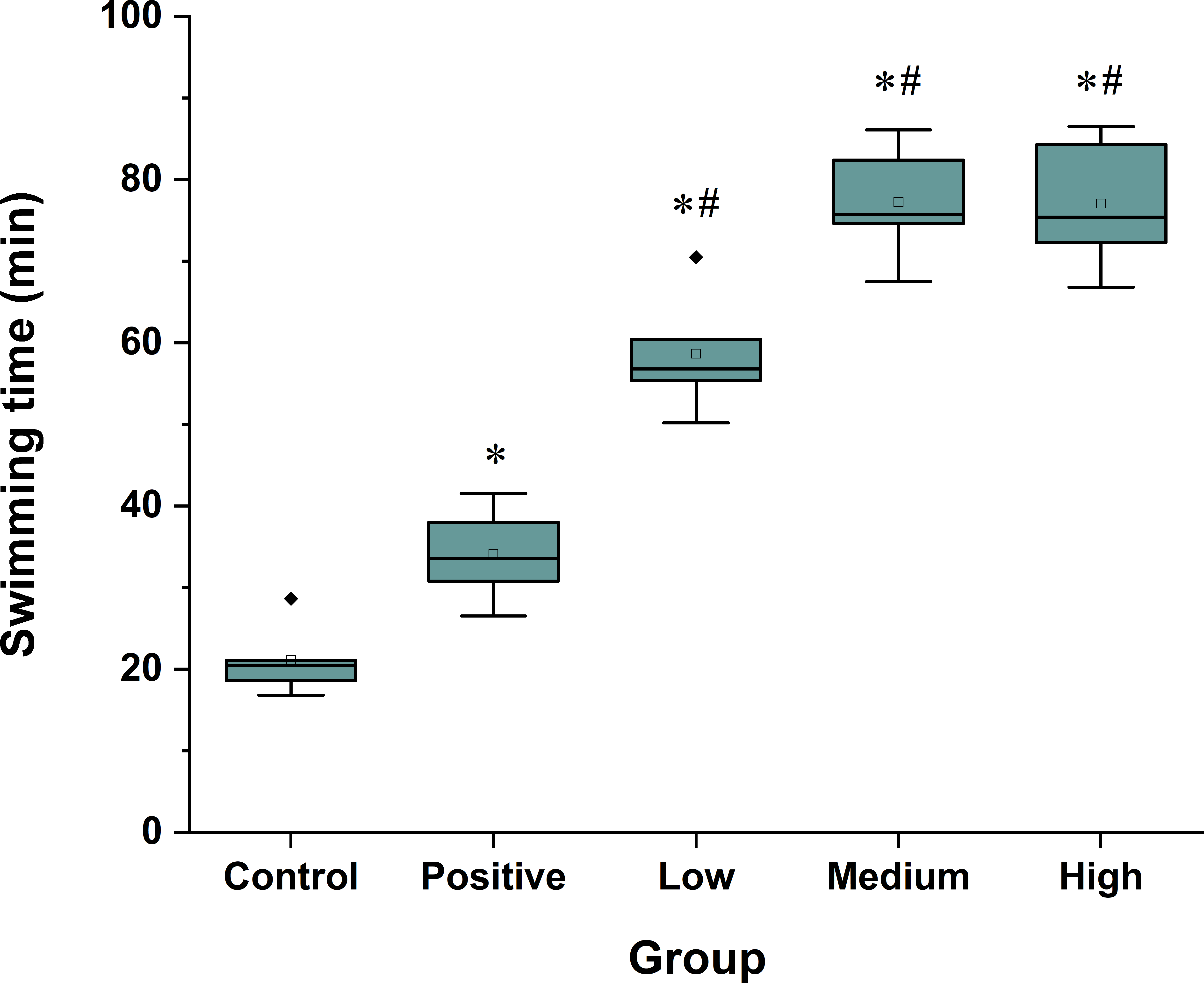
Effects of Spirulina peptides on weight-loaded forced swimming endurance time in mice. All the values are presented as mean ± SD (n = 8); *P < 0.05 versus the negative control group and #P < 0.05 versus the positive control group. Positive, 0.5 mg/g GSH group; low, 0.125 mg/g Spirulina peptide group; medium, 0.5 mg/g Spirulina peptide group; and high, 2 mg/g Spirulina peptide group. GSH, glutathione.
In Vivo Antioxidant Activity of Spirulina Peptides
GSH-Px, SOD, and CAT are 3 important antioxidant enzymes. Figure 3(A) shows that the SOD activity in serum of the 3 spirulina peptide treatment groups was significantly increased by 26.3%, 26.5%, and 26.8% compared with the negative control group (P < 0.05), respectively. Moreover, it was a little higher in the positive control group compared with the peptide treatment groups. The GSH-Px content in mouse serum was significantly increased, and its level in the middle-dose and high-dose groups was increased by around 50% compared with the negative control group (Figure 3(B), P < 0.05) and by 38.5% and 38.2% compared with the positive control group, respectively. However, there were no significant changes in the CAT activity among the Spirulina peptide treatment groups.

In vivo antioxidant activity of Spirulina peptides in serum. (A) SOD; (B) GSH-Px; (C) MDA; (D) CAT. All the values are presented as mean ± SD (n = 8), *P < 0.05 versus the negative control group and #P < 0.05 versus the positive control group. Positive, 0.5 mg/g GSH group; low, 0.125 mg/g Spirulina peptide group; medium, 0.5 mg/g Spirulina peptide group; and high, 2 mg/g Spirulina peptide group. CAT, catalase; GSH-Px; glutathione peroxidase; MDA, malondialdehyde; SOD, superoxide dismutase,
The content of serum MDA can also illustrate the speed and degree of fatigue development. Its level in the medium-dose and high-dose groups was significantly decreased by 47.2% and 65.6% compared with the negative control group, respectively, and by 38.9% and 60.2% compared with the positive control group, respectively (Figure 3(C), P < 0.05).
Protective Effects of Spirulina Peptides
Figure 4(A) shows that the LDH activity in the serum of the 3 peptide treatment groups was significantly decreased compared with the negative control group (P < 0.05). For example, the LDH activity in the high-dose group was reduced by 53.5%. However, only the high-dose group showed a significant decrease of 40.7% compared with the positive control group (P < 0.05). Figure 4(B) indicates that the CK activity in mouse serum in the positive control and Spirulina peptide treatment groups was decreased (P < 0.05), and its activity in the high-dose group was decreased by 37.2% compared with the negative control group.

The protective effects of Spirulina peptides. (A) LDH; (B) CK; (C) BLA. All the values are presented as mean ± SD (n = 8), * P < 0.05 versus the negative control group and # P < 0.05 versus the positive control group. Positive, 0.5 mg/g GSH group; low, 0.125 mg/g Spirulina peptide group; medium, 0.5 mg/g Spirulina peptide group; and high, 2 mg/g Spirulina peptide group. BLA, blood lactic acid; CK, creatine phosphokinase; GSH, glutathione; LDH, lactate dehydrogenase.
The accumulation of BLA is closely associated with fatigue. Figure 4(C) shows that the BLA levels of the positive control group were significantly decreased by 27.7% compared with the negative control group (P < 0.05), and its level in the Spirulina peptide treatment groups was decreased by 38.2%, 45.6%, and 43.8% of the negative control group, respectively (P < 0.05). These results indicated that both GSH and Spirulina peptides reduced the accumulation of BLA, and the latter was more effective.
Effects of Spirulina Peptides on Energy Metabolism
The GLU level in the negative control group was lower compared with the normal range (3.7-6.9 mmol/L) 5 and was lower compared with the other groups (P < 0.05, Figure 5(A)). The GLU levels of the peptide treatment groups were all within the normal range but were higher in the Spirulina peptide treatment groups compared with the positive control group (P < 0.05).
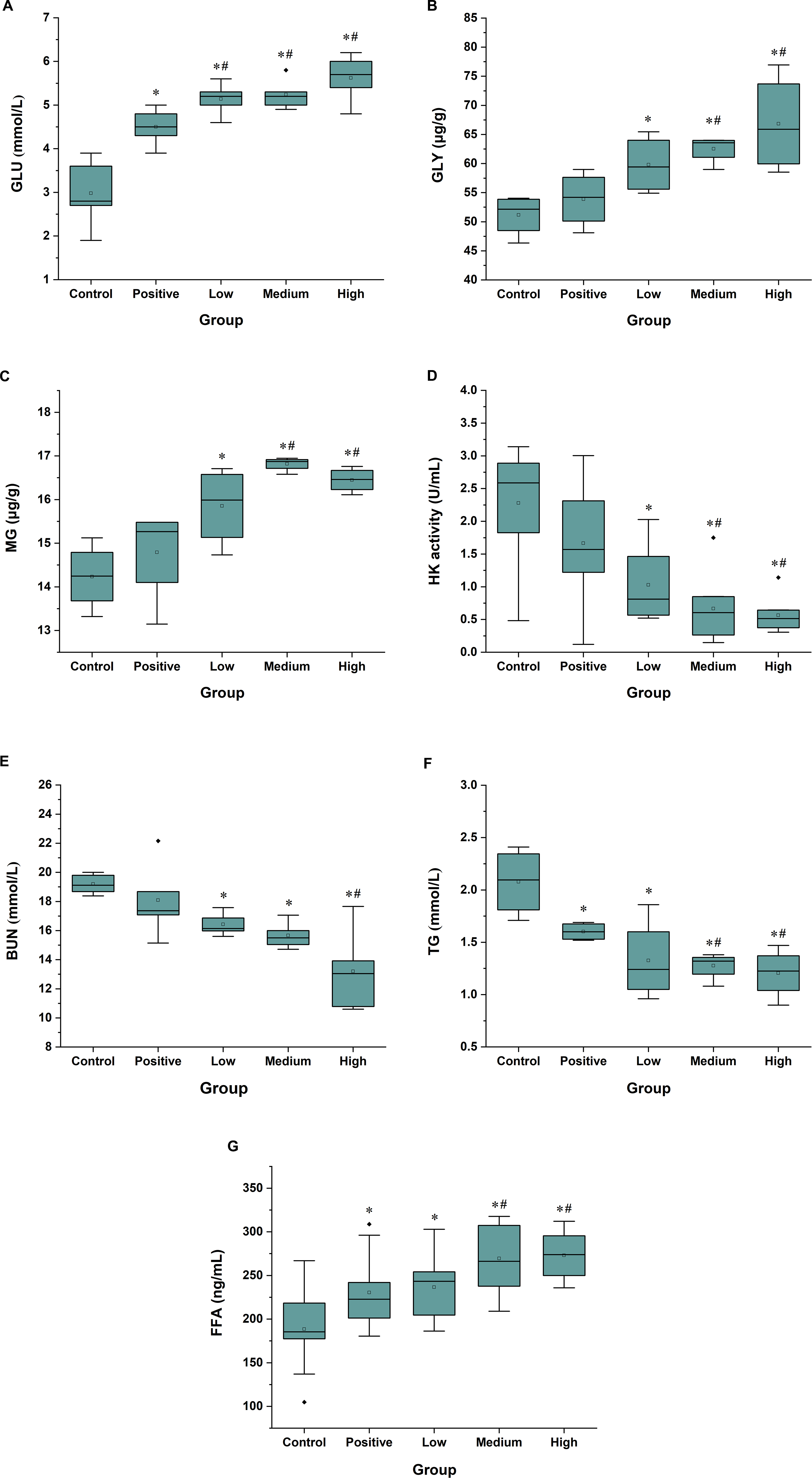
Effects of Spirulina peptides on energy consumption. (A) GLU; (B) GLY; (C) MG; (D) HK; (E) BUN; (F) TG; (G) FFA. All the values are presented as mean ± SD (n = 8), *P < 0.05 versus the negative control group and #P < 0.05 versus the positive control group. Positive, 0.5 mg/g GSH group; low, 0.125 mg/g Spirulina peptide group; medium, 0.5 mg/g Spirulina peptide group; and high, 2 mg/g Spirulina peptide group. BUN, blood urea nitrogen; GLU, glucose; GLY, glycogen; GSH, glutathione; FFA, free fatty acid; HK, hexokinase; MG, muscle glycogen; TG, trigtyceride.
The contents of MG and GLY in the Spirulina peptide treatment groups were significantly higher compared with the negative control group. For example, the value of the high-dose group was 30.6% higher compared with the negative control group after forced swimming (P < 0.05). Moreover, the values of the medium-dose and high-dose groups were increased by 16.1% and 24% compared with the positive control group, respectively (Figure 5(B) and (C)). However, the HK activity was decreased in the Spirulina peptide treatment groups compared with the negative control group (P < 0.05), and its value for the middle-dose and high-dose groups was decreased by 70.7% and 75.2% compared with the negative control group, respectively. The HK activity of the middle-dose and high-dose groups was also different from that of the positive control group (P < 0.05, Figure 5(D)).
The content of BUN can reflect the level of blood urea. Figure 5(E) shows that the BUN level of the Spirulina peptide treatment groups was reduced by 14.4%, 18.4%, and 31.2% compared with the negative control group (P < 0.05), respectively, clearly indicating that the peptides weakened the increased BUN level induced by catabolism of proteins or amino acids.
In the mammalian organism, most energy is stored in adipose tissue in the form of TG. Once this is hydrolyzed to FFA, FFA enters the common FFA pool. If FFA moves from the adipocytes into the blood, it can be used as an energy source in the tissues. 24 Figure 5(F) and (G) reveal a significant decrease of TG and an increase of FFA in the treatment groups (P < 0.05), and such changes of the medium-dose and high-dose peptide groups were even more pronounced compared with the positive control group (P < 0.05). For example, in the high-dose group, the TG level was reduced by 42%, while the content of FFA was increased by 39.9% compared with the negative control group (P < 0.05).
Spirulina Peptides Activate AMPK Signaling
In the present study, we showed that the TG level was decreased in the peptide treatment groups, and the FFA level increased, while no reduction of GLU and MG was found. Therefore, we speculated that the energy required for exercise might come from the oxidation metabolism of fatty acids. It is known that ATGL initiates the hydrolysis of TG and produces diacylglycerol and fatty acids. AMPK is an upstream regulator of ATGL. 25 Figure 6(B) shows that the phosphorylation levels of AMPK and ACC were significantly increased in livers of mice in the positive control and Spirulina peptide treatment groups (P < 0.05). We next determined whether peptides induced CPT1 expression. Figure 6(C) indicates that 3 doses of Spirulina peptides increased the CPT1 level in serum by 15.5%, 24.7%, and 35.3% compared with the negative control group, respectively, and such a tendency was consistent with p-AMPK and p-ACC (P < 0.05). This finding proved that GSH and Spirulina peptides might activate the AMPK/ACC signaling pathway.

Spirulina peptides activate AMPK signaling. (A) The expressions of phosphorylated AMPK, phosphorylated ACC, SIRT1, and FoxO1 proteins. Western blotting analysis of p-ACC, p-AMPK, SIRT1, and FoxO1 in liver protein (B). (C) Serum CPT1 and (D) ATGL. All the values are presented as mean ± SD (n = 8), *P < 0.05 versus the negative control group and #P < 0.05 versus the positive control group. Positive, 0.5 mg/g GSH group; low, 0.125 mg/g Spirulina peptide group; medium, 0.5 mg/g Spirulina peptide group; and high, 2 mg/g Spirulina peptide group. ACC, acetyl CoA carboxylase; AMPK, adenosine monophosphate-activated protein kinase; ATGL, adipose triglyceride lipase; CPT1, carnitine palmitoyltransferase 1; FoxO1, forkhead box O1; SIRT1, sirtuin1.
FoxO1 is one of the well-known direct targets of SIRT1, which is activated by deacetylation. 26 The levels of SIRT1 and FoxO1 were increased in livers of mice in the positive control and Spirulina peptide treatment groups (P < 0.05, Figure 6(B)). The peptide-induced increase of FoxO1 was almost directly proportional to the increase of SIRT1 expression in livers. The content of serum ATGL of the positive control and Spirulina peptide treatment groups was significantly higher compared with the negative control group. For example, it was increased by 34.6% in the high-dose group compared with the negative control group (P < 0.05). All of the above-mentioned expressions, except for p-ACC, in the medium-dose and high-dose groups were significantly higher compared with the positive control group.
Discussion
Spirulina has been studied by several authors due to its various biological activities, including angiotensin-converting enzyme inhibition, antitumor, antiaging, antimicrobial, and antiproliferation properties. 14 -20 However, no studies have reported the antifatigue activities of Spirulina peptides. The weight-loaded forced swimming test is a valid experimental protocol widely used to assess the degree of exercise of endurance and fatigue in laboratory animals. 27 In the present experiment, we proved that Spirulina peptide treatment significantly prolonged the time to exhaustion of mice, showing that the peptides possessed an antifatigue effect, and 2 mg/g was the optimal dose. The level of BLA is one of the significant indicators of exercise fatigue. During intensive exercise, muscle produces massive BLA through anaerobic glycolysis. 28 LDH catalyzes the formation of lactic acid from pyruvate. The accumulation of lactic acid results in a decrease in pH in muscle tissue, which can induce fatigue and hamper exercise performance. 29 In this study, Spirulina peptide treatment significantly decreased the LDH activity and BLA levels. In addition, plasma CPT1 level can also serve as a key index for evaluating the cell damage and subsequent extent of muscle disruption induced by intense exercise. 30 In the present study, the levels of CPT1 in the Spirulina peptide treatment groups were also significantly decreased, suggesting that Spirulina peptides could mitigate the cell damage caused by fatigue, thereby deferring the occurrence of fatigue.
Unaccustomed and/or exhaustive exercise can generate excessive reactive oxygen species, causing impaired muscle contractility and oxidative stress-related tissue damage. 31 Adequate SOD and GSH-Px activities, as well as low levels of MDA, can alleviate oxidative stress and ameliorate exercise-induced fatigue. 32 In this study, peptide treatment significantly increased the SOD activity, elevated the GSH level, and decreased the MDA content, strongly indicating that the peptide treatment was able to protect the cells from lipid oxidation.
Muscle movement causes rapid ATP consumption, and energy deficiency is a significant factor for physical fatigue. 33 GLU is expeditiously available and can be directly oxidized to supply ATP. 34 Furthermore, glycogen primarily exists in liver and muscle cells, which are the main storage sites of carbohydrate energy for ATP generation. 35 However, consumption of too much glycogen indicates fatigue. Here, the levels of GLU and glycogen storage in the liver and skeletal muscles in the Spirulina peptide treatment groups were higher compared with the negative control group, indicating that the fatigue level was decreased.
In order to meet the energy requirement, proteins and amino acids will become alternative sources for catabolic metabolism when the body fails to derive energy from carbohydrates. 35 However, the BUN levels were reduced after peptide treatment, indicating that neither proteins nor amino acids were effectively used to relieve the energy deficiency. Moreover, increased BUN levels also reflect the jeopardy of the contractive strength of muscle and typical indexes related to fatigue. In the present study, we showed that peptides inhibited the accumulation of BUN, and it remained unclear what provided the energy needed for the exercise.
AMPK is a metabolic energy sensor that plays a key role in regulating energy homeostasis. 36 Activation of the AMPK signaling pathway can regulate lipid metabolism in liver cells, and AMPK phosphorylation stimulates ACC phosphorylation, 37 leading to increased β-oxidation by promoting the CPT1 activity. 38 AMPK activation can inhibit FoxO1 phosphorylation from promoting ATGL expression for lipolysis. 39 ATGL is a TG hydrolase, which initiates hydrolysis of TG and produces fatty acids. 40 When TG is hydrolyzed to FFA, more FFA may undergo beta-oxidation for ATP production. 41 In the present study, we observed the reduction of TG and elevation of FFA after Spirulina peptide treatment. Phosphorylation of AMPK and ACC and contents of CPT1, SIRT1, FoxO1, and ATGL were all increased. Taken together, the Spirulina peptides activated the AMPK signaling pathway to induce the hydrolysis of TG into FFA, which entered into the mitochondria to generate more ATP with the help of the AMPK signaling pathway to delay exercise-induced fatigue (Scheme 1).

The proposed model of AMPK signaling pathway upon reduction of triglyceride and elevation of free fatty acid.
Conclusions
Our present experimental results showed that Spirulina peptides possess an antifatigue activity in mice to prolong the weight-loaded forced swimming time. Moreover, Spirulina peptides could relieve fatigue by reducing the production of lactic acid, leading to improved antioxidant capacity. However, Spirulina peptides did not excessively consume blood sugar or glycogen in the liver and muscle to produce energy. Instead, the energy was generated by activating the fat metabolism pathway through the AMPK signaling pathway. In this study, we found that Spirulina peptides with a molecular weight below 2000 had antifatigue activity. However, the specific peptide of Spirulina with antifatigue activity requires further investigation.
Supplemental Material
Supplementary Material 1. - Supplemental material for In Vivo Antifatigue Activity of Spirulina Peptides Achieved by Their Antioxidant Activity and by Acting on Fat Metabolism Pathway in Mice
Supplemental material, Supplementary Material 1., for In Vivo Antifatigue Activity of Spirulina Peptides Achieved by Their Antioxidant Activity and by Acting on Fat Metabolism Pathway in Mice by Yuhao Chen, Feng Wang, Jiawei Zhou, Tingting Niu, Rongrong Xuan, Haimin Chen and Wei Wu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by National Key R&D Program of China (2018YFD0901103); NSFC (31872540), Natural Science Foundation of Zhejiang (LY18C190004, LY19H030001, LQ20D060001), China Agriculture Research System (CARS-50), Ningbo Programs for Science and Technology Development (2017C110026), Medical Science and Technology of Zhejiang Province (2018KY726, 2018KY710), K.C. Wong Magna Fund in Ningbo University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
