Abstract
Bupleurum chinense DC, a traditional medicinal plant in China that has many pharmacological effects, contains polysaccharide as one of its active components. In this study, we isolated and structurally characterized the polysaccharide from B. chinense. Water-soluble polysaccharides (termed WBCP) were extracted from the plant and fractionated by anion-exchange and size exclusion chromatographies. From this procedure, we obtained a homogeneous acidic polysaccharide (WBCP-A2) and determined its monosaccharide composition. Analysis by FT infrared and 13C NMR spectroscopies, along with enzymatic hydrolysis, indicated that WBCP-A2 is a pectic polysaccharide, composed of rhamnogalacturonan I, rhamnogalacturonan II, highly methyl-esterified homogalacturonan (HG), and either non- or low methyl-esterified HG domains. These different fractions may be covalently linked through HG segments to form the complex pectin molecules.
Bupleurum chinense DC (family Apiaceae) is a well-known traditional Chinese medicine that has been used in China for more than a thousand years. 1 Polysaccharides are one of the bioactive constituents of this plant. Several studies have reported on the preparation, structural analysis, and biological activity of polysaccharides derived from B. chinense. A neutral, water-soluble polysaccharide, BCPS-1, was shown to have a significant antioxidant effect. This polysaccharide has a backbone of 1,5-linked Ara, 1,4-linked Gal, and 1,3-linked Gal residues with occasional branches at O-6 of 1,4-linked Glc and terminal Gal residues. 2 Water-soluble polysaccharides from B. chinense (BCPs) contain Gal, Ara, Glc, Rha, and Man residues in the ratio of 13.43:11.57:4.02:1.02:1.00 with 48.5% uronic acid. BCPs significantly attenuate lung injury by improving lung morphology and reducing complement deposition, 3 and exert modulatory effects on macrophage function. 4 Another water-soluble polysaccharide from B. chinense (WBCP), with 21.4% uronate, shows promising hepatoprotective effects. 5 Although any of these polysaccharides from B. chinense possess various biological activities, their detailed structures, especially in terms of their acidic polysaccharides, remain unclear. Therefore, in this study, we extracted, purified, and structurally characterized a water-soluble, acidic pectic polysaccharide from B. chinense.
The water-soluble polysaccharide from B. chinense (termed WBCP) was extracted with hot water and precipitated with ethanol; the yield was 6.0% (w/w), similar to the 6.5% yield reported by Sun et al. 2 Other water-soluble polysaccharides from B. chinense have been reported with yields of 0.93% 3 and 11.5%. 5 Differences in these yields might be due to the demography of B. chinense and the use of different extraction methods. WBCP contains 75.5% total carbohydrate, 20.2% uronic acid, 1.4% protein, and 12.0% ash, and is composed of Glc (36.8%), GalA (25.7%), Ara (15.8%), Gal (11.1%), Rha (4.5%), Man (4.3%), and GlcA (1.8%) residues (Table 1).
Yield and Monosaccharide Composition of Polysaccharides From Bupleurum chinense.

aYield in relation to weight of Bupleurum chinense.
bYield in relation to WBCP.
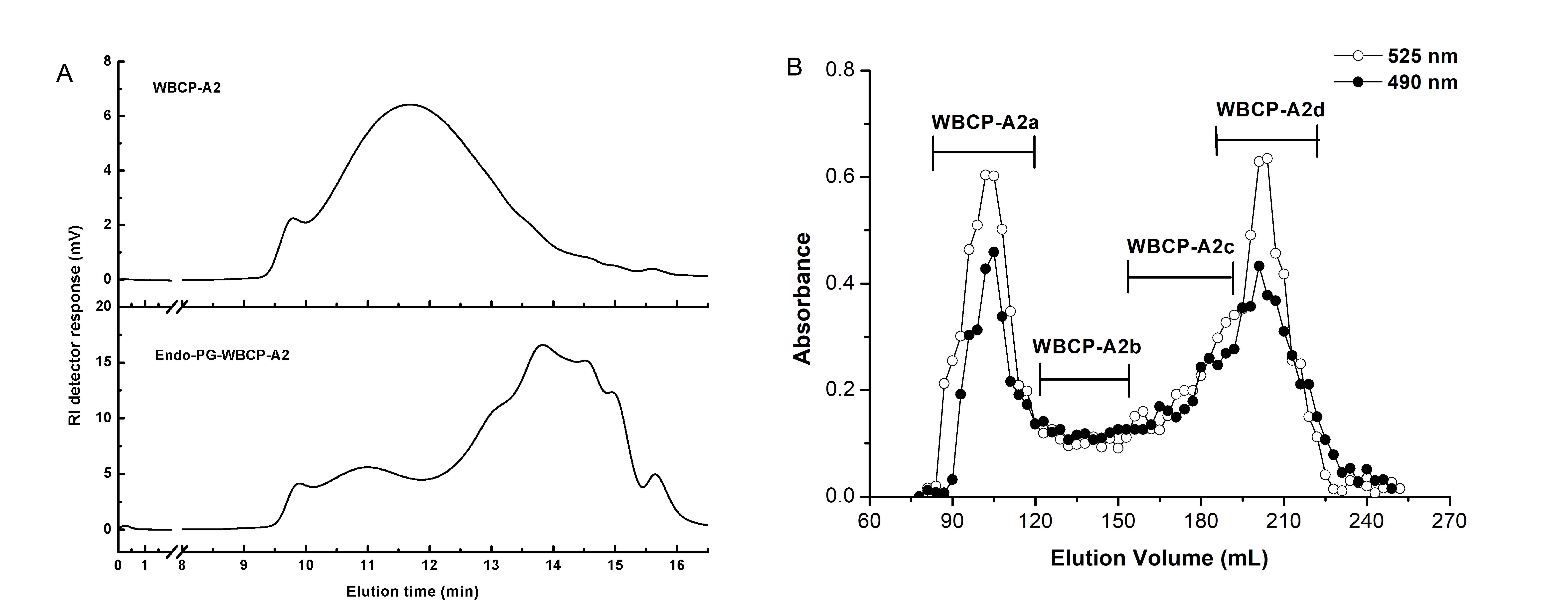
WBCP was initially separated by Diethylaminoethyl-cellulose (DEAE-cellulose) column into neutral (WBCP-N) and acidic (WBCP-A) polysaccharide fractions, with yields of 60.2% and 13.8%, respectively. WBCP-N is mainly composed of Glc (56.6%), Gal (14.4%), and Ara (18.5%) residues, with small amounts of Rha (5.4%) and Man (4.2%) residues (Table 1). The I2-KI assay was positive, indicating that the polysaccharide contains starch and some arabinogalactans. In this study, we did not further investigate WBCP-N, but rather focused our attention on WBCP-A. WBCP-A is composed of GalA (52.0%), Gal (11.3%), Ara (22.7%), Rha (7.4%), and GlcA (1.5%) residues (Table 1), and has a homogeneous charge distribution on a DEAE-cellulose column (Figure 1(a)). WBCP-A was further purified by size-exclusion chromatography, and one major fraction (WBCP-A2) was obtained (Figure 1(b)). WBCP-A2 (Mw 29.7 kDa) is composed of GalA (56.7%), Gal (9.8%), Ara (20.0%), Rha (7.0%), and GlcA (1.4%) residues (Table 1), reflecting a characteristic monosaccharide composition of pectic polysaccharides.

(a) Elution profile of WBCP-A on DEAE-cellulose column. (b) Elution profile of WBCP-A on Sepharose CL-6B column.
The FT-IR spectrum of WBCP-A2 is shown in Figure 2. The strong band around 3424.77 cm−1 was attributed to O-H stretching due to intermolecular and intramolecular hydrogen bonds. The absorption at 2935.00 cm−1 was attributed to C-H stretching. The bands at 1742.36 and 1610.35 cm−1 were from methyl-esterified and free carboxyl groups, respectively, suggesting that GalA residues in WBCP-A2 are methyl-esterified. The bands in the region from 1200 to 1000 cm−1 were assigned to the absorption of skeletal C-O and C-C vibrations of glycosidic bonds and the pyranoid ring. The weak bands at 900.05 and 825.62 cm−1 indicate the presence of β-linked and α-linked sugar residues. 6

FT-IR spectrum of WBCP-A2.
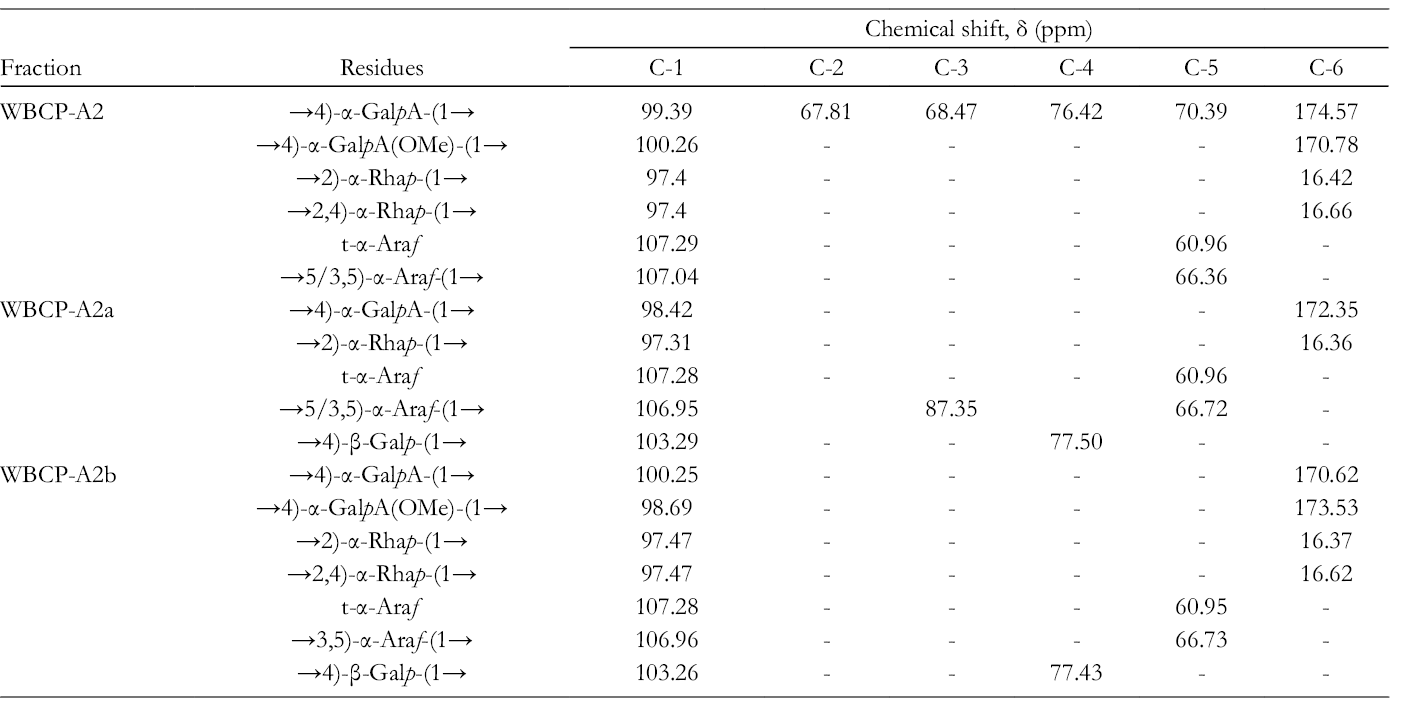
In the 13C NMR anomeric spectral region (Figure 3), signals at 100.26 and 99.39 ppm were assigned to C-1 of esterified and nonesterified α-1,4-GalpA, and signals at 170.78 and 174.57 ppm to C-6 of esterified and nonesterified α-1,4-GalpA. 7 The intense signals at 52.74 and 20.07 ppm were attributed to methyl and acetyl groups, respectively. 8 Signals at 67.81, 68.47, 76.42, and 70.39 ppm were assigned to C-2, C-3, C-4, and C-5 of α-1,4-GalpA, respectively (Table 2). 7 These results indicate that WBCP-A2 is composed of highly methyl-esterified homogalacturonan (HG)-type pectin. 8 The signals at 16.42 and 16.66 ppm were assigned to C-6 of α-1,2-Rhap and α-1,2,4-Rhap, with an anomeric signal at 97.40 ppm. The anomeric signals at 107.29 and 107.04 ppm were assigned to terminal- and α-1,5/1,3,5-linked Araf residues, respectively. The resonance at 60.96 ppm was from C-5 of terminal-Araf, and that at 66.36 ppm was from α-1,5/1,3,5-Araf (Table 2). 9 Therefore, rhamnogalacturonan I (RG-I)-type pectin that is composed of α-1,2-Rhap and α-1,4-GalpA disaccharide repeating units 10 is also present in this fraction, with branching of arabinan side chains at O-4 of α-1,2-Rhap. A purified acid polysaccharide from B. chinense (BC-PS1) also contains terminal and 1,5-linked Araf and 1,4-linked Galp (GalpA) residues. 11

13C NMR spectrum of WBCP-A2.
13C NMR Spectral Assignments of WBCP-A2 and its Degraded Products.
To analyze further the structure of WBCP-A2, we hydrolyzed the polysaccharide using endo-polygalacturonanase, which can degrade unesterified α-
(a) The molecular weight distribution of WBCP-A2 and its enzymatic hydrolysates. (b) The elution profile of enzymatic hydrolysates of WBCP-A2 on Sephadex G-75 column.
Yield, Mw, and Monosaccharide Composition of Subfractions From WBCP-A2.
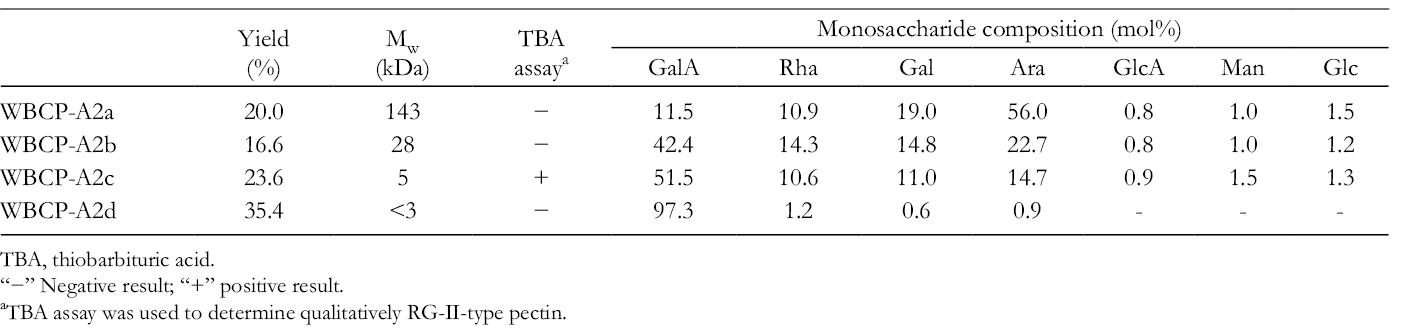
TBA, thiobarbituric acid.
“−” Negative result; “+” positive result.
aTBA assay was used to determine qualitatively RG-II-type pectin.
WBCP-A2a contains Ara (56.0%), Gal (19.0%), GalA (11.5%), and Rha (9.9%) residues as the major sugars. The ratio of Rha/GalA was almost 1.0 (actually 0.95), suggesting that it is a highly branched RG-I-type pectin. 13 WBCP-A2b also contained GalA (42.4%), Rha (14.3%), Gal (14.8%), and Ara (22.8%) residues as the major sugars, but the ratio of Rha/GalA was only 0.34, suggesting that it may contain both HG and RG-I structures. WBCP-A2c showed a positive result in the thiobarbituric acid (TBA) assay, indicating that it is an rhamnogalacturonan II (RG-II)-type pectin that is complex, yet structurally conserved. 14 The monosaccharide composition of WBCP-A2c (GalA [51.5%], Rha [10.6%], Gal [11.0%], and Ara [14.7%]) and its molecular weight of 5 kDa are consistent with a RG-II type pectin. WBCP-A2d (molecular weight <3 kDa) is primarily composed of GalA (97.3%), suggesting that it results from degradation of non- or low-methyl-esterified HG.
The structures of WBCP-A2a and WBCP-A2b were further analyzed by 13C NMR spectroscopy (Figure 5). Due to low content, resonances from GalA and Rha residues in WBCP-A2a were relatively weak (Figure 5(a)). Signals at 98.42 and 172.35 ppm were assigned to C-1 and C-6 of α-1,4-GalpA, and signals at 97.31 and 16.36 ppm to C-1 and C-6 of α-1,2/1,2,4-Rhap, respectively (Table 2), which indicated that WBCP-A2a was an RG-I. 9 The intense signals at 107.28 and 106.95 ppm were attributed to terminal- and α-1,5/1,3,5-linked Araf residues, 60.96 and 66.72 ppm were from C-5 of terminal-Araf and α-1,5/1,3,5-Araf, respectively, and 83.75 ppm was assigned to C-3 of α-1,3,5-Araf. Signals at 103.29 and 77.50 ppm were attributed to β-1,4-Galp (Table 2). 15 Therefore, these results indicate that in WBCP-A2a, α-1,5/1,3,5-arabinan and β-1,4-galactan side chains are linked to the RG-I backbone. In the spectrum of WBCP-A2b (Figure 5(b)), signals at 100.25 and 170.62 ppm were assigned to C-1 and C-6 of esterified α-1,4-GalpA, whereas signals at 98.69 and 173.53 ppm were assigned to C-1 and C-6 of nonesterified α-1,4-GalpA. 7 The intense signal at 52.72 ppm and the weak signal at 20.08 ppm were attributed to methyl and acetyl groups, respectively (Table 2). 8 These results suggest that WBCP-A2b is composed of highly methyl-esterified HG, consistent with it not being hydrolyzed by endo-polygalacturonanase. The signals at 16.37 and 16.62 ppm were assigned to α-1,2-Rhap and α-1,2,4-Rhap, with an anomeric signal at 97.47 ppm. The anomeric signals at 107.28 and 106.96 ppm were attributed to terminal- and α-1,5/1,3,5-linked Araf residues, respectively, signals at 60.95 and 66.73 ppm were from C-5 of terminal-Araf and α-1,5/1,3,5-Araf, respectively, and signals at 103.26 and 77.43 ppm to β-1,4-Galp (Table 2). 15 These data support the idea that branched RG-I is also present in WBCP-A2b and might be linked to the highly methyl-esterified HG region.
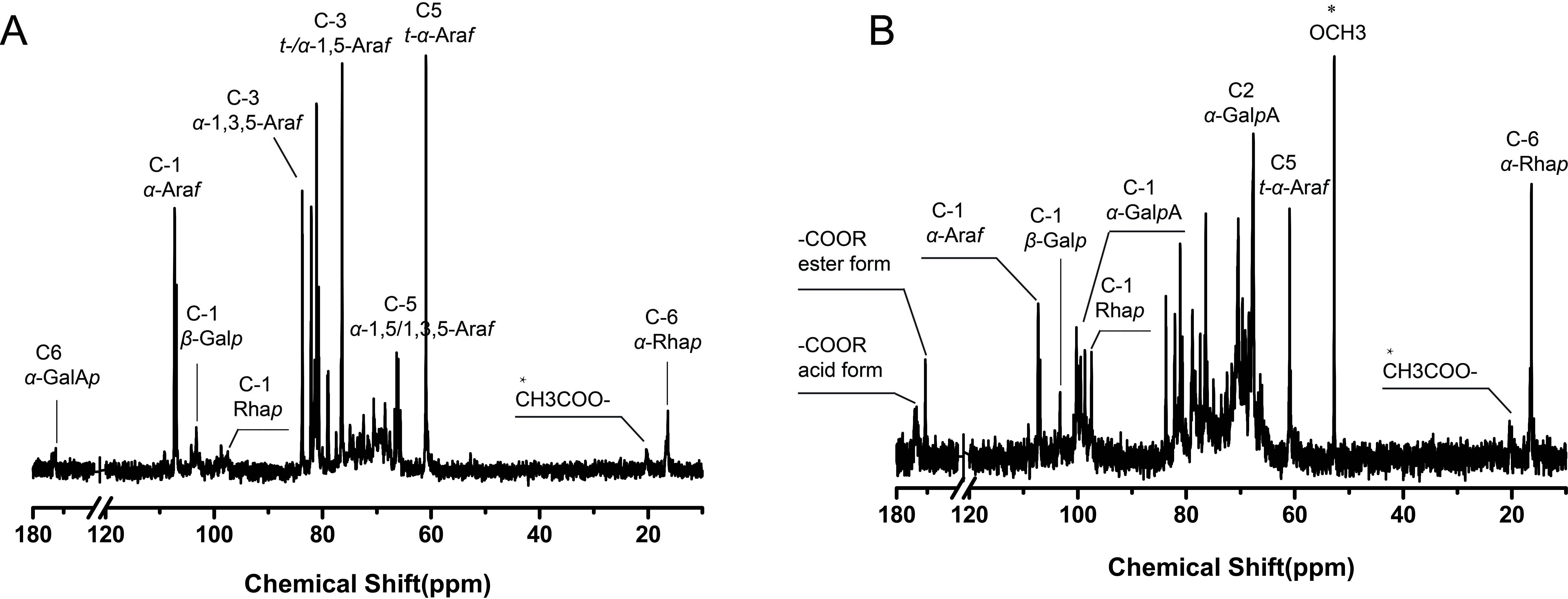
13C NMR spectrum of (a) WBCP-A2a and (b) WBCP-A2b.
According to this analysis, WBCP-A2 is composed of RG-I, RG-II, highly methyl-esterified HG, as well as either non- or low-methyl-esterified HG regions. As different pectic regions can be released following endo-polygalacturonanase hydrolysis, these structural regions might be covalently linked through HG regions to form the complex pectin polysaccharide. Similar results have been found in pectins from citrus peel 16 and elderflower 17 upon endo-polygalacturonanase hydrolysis. Pectin plays an important role in the formation of higher plant cell walls that provide strength and support to a plant. Homogalacturonan, RG-I, and RG-II are structurally diverse polysaccharides that contribute to primary wall function with regard to cell strength, cell adhesion, stomatal function, and the defense response. Therefore, pectin domains isolated from B. chinense should have an important role in the cell wall of this medical plant.
In summary, we extracted water-soluble polysaccharides from B. chinense and purified a homogeneous acidic polysaccharide. Results from monosaccharide compositional analysis, enzymatic hydrolysis, FT-IR, and 13C NMR allowed us to conclude that this acidic polysaccharide is a pectin that contains RG-I and RG-II domains, along with high methyl-esterified HG and non- or low methyl-esterified HG regions. These different pectic structures might be covalently linked by HG to form the complex pectin molecules. Our results have provided crucial structural information of polysaccharides from B. chinense.
Experimental
General
Total carbohydrate content was determined by using the phenol-sulfuric acid method, 18 with glucose (Glc) as the standard. Uronic acid content was determined by the m-hydroxyl diphenyl method, 19 with galacturonic acid (GalA) as the standard. Rhamnogalacturonan II was qualitatively determined by the modified TBA assay. 20 Monosaccharide composition was analyzed by 1-phenyl-3-methyl-5-pyrazolone (PMP) precolumn derivatization and HPLC detection. 21 Weight average molecular weights (Mw) were determined by high-performance size exclusion chromatography on a TSK-gel G-3000 PWXL column (7.8 × 300 mm, TOSOH, Tokyo, Japan) precalibrated by using standard dextrans and coupled to a Shimadzu HPLC system (Tokyo, Japan). 21
Materials and Chemicals
Roots of B. chinense were purchased from a local medicinal herb market. DEAE-cellulose was provided by Shanghai Chemical Reagent Research Institute (Shanghai, China). Sepharose CL-6B and Sephadex G-75 were purchased from GE healthcare (Pittsburgh, United States), and endo-polygalacturonase M2 (E.C.3.2.1.15) from Megazyme (Bray, Ireland). All chemicals and reagents were produced in China and were of analytical grade.
Preparation of Polysaccharides From Bupleurum chinense
One kilogram of dried B. chinense roots were extracted with 15 L hot water at 100°C for 3 hours. After filtration, the solid material was extracted twice again under the same conditions. The extracts were combined, concentrated, and centrifuged at 4500 rpm for 15 minutes. The supernatant was precipitated overnight with a final ethanol concentration of 60%. The polysaccharide was collected by centrifugation and dried by 95% ethanol and absolute ethanol. Water-soluble polysaccharide from B. chinense, termed WBCP (56 g), was obtained.
WBCP was dissolved in distilled water (30 mg/mL) and loaded onto a DEAE-cellulose column (Cl−, 8.0 × 20 cm) pre-equilibrated with distilled water. The column was first eluted with distilled water to give the neutral polysaccharide fraction (WBCP-N), and then with 0.5 M NaCl to give the acidic polysaccharide fraction (WBCP-A). After desalting by dialysis (MWCO 3500 Da) and lyophilized, WBCP-A was further purified on a Sepharose CL-6B column (3.0 × 90 cm). This was eluted with 0.15 M NaCl at 0.5 mL/min. Fractions (10 mL/tube) were collected and assayed for total sugar and uronic acid content. The appropriate fractions were combined, desalted by dialysis (MWCO 3500 Da), and freeze-dried. Two subfractions, WBCP-A1 and WBCP-A2, were obtained.
Enzymatic Hydrolysis of WBCP-A2
WBCP-A2 (50 mg/mL) was dissolved in 50 mM sodium acetate buffer (pH 4.5), and incubated with endo-polygalacturonase (10 U per mL sample) at 40°C for 12 hours. This procedure was repeated once. After inactivation of the enzyme at 100°C for 10 minutes, the hydrolysate was centrifuged at 4500 rpm for 20 minutes. The supernatant was applied to a Sephadex G-75 column (2.6 × 100 cm) and eluted with 0.15 M NaCl at a flow rate of 0.4 mL/min. Four subfractions (WBCP-A2a, WBCP-A2b, WBCP-A2c, and WBCP-A2d) were obtained.
FT-IR Spectroscopy
Polysaccharides were ground with KBr powder and pressed into a 1 mm pellet for FT-IR measurements. FT-IR spectra (4000-400 cm−1) were obtained using a Spectrum Two FT-IR spectrometer (Perkin Elmer, United States). 22
NMR Spectroscopy
13C NMR spectra were acquired at 20°C on a Bruker Avance 600 MHz NMR spectrometer (Germany). The sample (20 mg) was dissolved in D2O (0.5 mL). Data were analyzed using standard Bruker software. 22
Footnotes
Acknowledgment
The authors would like to thank Prof. Kevin H. Mayo from the Department of Biochemistry, Molecular Biology and Biophysics, University of Minnesota for careful reading and editing of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for financial support from the National Science & Technology Major Project “Key New Drug Creation and Manufacturing Program,” China (2019ZX09735001), National Natural Science Foundation of China (Grant nos. 31770852 and 81703461), and the Scientific and Technologic Foundation of Jilin Province (20180101004JC).
