Abstract
Chrysoeriol is a 3′-O-methoxy flavone, chemically a derivative of luteolin, which is commonly found across the plant kingdom. Chrysoeriol is of great scientific interest because of its promising anti-inflammatory, anti-cancer, antioxidative, anti-lipase, anti-xanthin oxidase, and antimicrobial activities against multidrug-resistant (MDR) bacterial pathogens; however, its effects on melanogenesis have not yet been elucidated. Here, we report a novel effect of chrysoeriol on melanogenesis in B16F10 cells. Chrysoeriol treatment significantly increased the expression of the melanogenic enzymes tyrosinase (TRY), tyrosinase-related protein-1 (TRP-1), and TRP-2 and upregulated the expression of microphthalmia-associated transcription factor (MITF) in a concentration-dependent manner. Furthermore, chrysoeriol suppressed the phosphorylation of extracellular signal-regulated kinase (ERK) and protein kinase B (AKT) in a concentration-dependent manner. In addition, chrysoeriol treatment increased the phosphorylation of p38 mitogen-activated protein kinase (MAPK), glycogen synthase kinase (GSK)-3β, β-catenin, and protein kinase A (PKA) and decreased the production of β-catenin, which is involved in the transcriptional activation of MITF in melanogenesis. Finally, the structure–activity relationship (SAR) of chrysoeriol and its derivatives, including luteolin and apigenin, with regard to their melanin inhibitory activity was also investigated; we identified the significance of the 4′-OH group and C-3′ methoxylation in melanogenesis. Together, these findings indicate that chrysoeriol promotes melanogenesis in B16F10 cells by upregulating the expression of melanogenic enzymes through the MAPK, phosphatidylinositol 3-kinase (PI3K)/AKT, PKA, and Wnt/β-catenin signaling pathways; thus, chrysoeriol may be used as a cosmetic ingredient to promote melanogenesis or as a therapeutic agent against hypopigmentation disorders.
Introduction
Vitiligo is a relatively common acquired hypopigmentation disorder wherein epidermal melanocytes are selectively lost owing to an autoimmune mechanism. Vitiligo is characterized by the absence of pigmentation on the skin and the presence of white spots on different parts of the body. Although the prevalence of vitiligo is reported to vary by country and race, it is reported to occur in 0.1% of the global population.1,2 Vitiligo causes significant emotional and psychological distress and thus reduces the quality of life. Most people with vitiligo develop this disorder between the ages of 10 and 30 years. However, the pathophysiology of this disease is not well understood. 3 Several causes for vitiligo, including genetic, biochemical, immunological, and environmental factors, are being elucidated, and current treatments for vitiligo include the use of excimer lasers, vitamin D analogues, or steroid therapy, which often have unwanted side effects. 3 Thus, the discovery of melanogenesis-stimulating natural compounds is required.
Melanin plays a key role in protecting the skin from ultra violet (UV)-induced damage and oxidative stress caused by various pollutants, but abnormal production and accumulation of melanin can lead to skin cancer and/or skin diseases. Melanin is synthesized through a process called melanogenesis in epidermal melanocytes and is scattered in the basal layer of the epidermis. Melanin is stored within melanocytes in subcellular lysosomal-like organelles called melanosomes, which are then transferred to the surrounding keratinocytes.4,5
In mammals such as humans, melanin is present in two basic forms: brown to black eumelanin and yellow to red peomelanin. The synthesis of these melanin forms is directly controlled by three enzymes: tyrosinase (TYR), tyrosinase-related protein-1 (TRP-1), and TRP-2. 6 In the first two stages of melanogenesis, TRY (considered to be the rate-limiting enzyme of melanogenesis) catalyzes the hydroxylation of tyrosine into L-3,4-dihydroxyphenylalanine (L-DOPA) and of L-DOPA into L-dopaquinone. In contrast, L-dopaquinone can be converted to cysteinyl-DOPA and oxidized further to produce pheomelanin. However, L-dopaquinone can also be converted to L-dopachrome, and TRP-2 can isomerize dopachrome to 5,6-dihydroxyindole-2-carboxylic acid (DHICA); TRP-1 then oxidizes DHICA to indole-5,6-quinone-2-carboxylic acid, which is eventually converted to eumelanin.6–9 Thus, modulating the activity of enzyme cascades consisting of several key enzymes, such as TRY, TRP-1, and TRP-2, is considered an important strategy in the development of skin pigmentation treatments.10,11
Microphthalmia-associated transcription factor (MITF) is involved in the survival, proliferation, and differentiation of melanocytes and is an important transcriptional regulator of TRY, TRP-1, and TRP-2.12,13 The key players in UV-mediated MITF regulation are propiomelanocortin (POMC), α-melanocyte-stimulating hormone (α-MSH), stem cell factor (SCF), and endothelin-1 (ET-1), all of which induce melanogenesis through four major signaling pathways: the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) signaling pathway, and (4) Wnt/β-catenin signaling pathway. 14 To date, many plant extract-derived melanin inducers have been discovered, such as Polygonum multiflorum extract, Gynostemma pentaphyllum saponins, and Argania spinosa fruit shell extract.3,7,15 Several small molecules derived from coumarins and flavonoids have also been reported to promote melanin production. 16 Both of the above types of melanin inducers promote melanogenesis through the modulation of the mitogen-activated protein kinase (MAPK), protein kinase A (PKA), PI3K/AKT, and Wnt/β-catenin signaling cascades.
Chrysoeriol (5,7-dihydroxy-2-[4-hydroxy-3-methoxyphenyl]chromen-4-one) is a 3′-O-methoxy flavone, a derivative of luteolin, which belongs to the flavone group of compounds, and is abundantly present in Flos Lonicerae (Lonicera japonica flowers), Tanacetum vulgare, Artemisia arborescens, Salix matsudana, Aspalathus linearis, and Coronopus didymus.17,18 Furthermore, chrysoeriol has numerous pharmacological properties, such as anti-inflammatory, anti-cancer, and antioxidation activities as well as effects of smooth muscle relaxation, obesity reduction, xanthin oxidase inhibition, immune system regulation, and efflux system inhibition in multidrug-resistant (MDR) bacterial pathogens.18–21 However, there are no reports regarding the effects of chrysoeriol on melanogenesis and the underlying mechanism of action. Therefore, this study aimed to analyze the stimulating effect of chrysoeriol on melanogenic activity in B16F10 murine melanoma cells. Furthermore, this study investigated MITF transcription regulators and kinase regulators of phosphorylation in the MAPK, PKA, PI3K/AKT, and Wnt/β-catenin signaling pathways to evaluate potential mechanisms of action.
Results
Chrysoeriol Stimulates Melanin Production in B16F10 Cells Without Cytotoxic Effects
To determine safe concentrations of chrysoeriol, its cytotoxicity was evaluated in B16F10 melanoma cells using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Cells were treated with chrysoeriol at concentrations of 1 to 16 μM. As shown in Figure 1a, no apparent cytotoxicity was observed in B16F10 cells after treatment with chrysoeriol at concentrations of 1, 2, 4, and 8 μM. At the highest concentration of 16 μM, cell viability was still above 87%. Therefore, we conclude that chrysoeriol is not toxic to B16F10 cells at concentrations below 8 μM. Subsequent experiments were conducted with chrysoeriol concentrations of 1, 2, 4, and 8 μM, which also did not demonstrate cytotoxicity.

Effects of chrysoeriol on the viability, melanin content, and tyrosinase (TRY) activity in B16F10 melanoma cells. Cells were treated with chrysoeriol at the indicated concentration for 72 h. Cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay and is expressed as a percentage relative to the untreated cells. Melanin content and TRY activity are expressed as percentages relative to untreated cells. α-MSH was used as the positive control. Data are presented as mean ± SD of at least three independent experiments (n = 3). * indicates P < .05 and **P < .01 versus control.

Effects of chrysoeriol on tyrosinase (TRY), TRP-1, and TRP-2 expression in B16F10 cells. Cells were treated with chrysoeriol at the indicated concentration for 72 h. Western blotting was performed to verify protein levels. TRY, TRP-1, and TRP-2 levels were quantified by Image J. β-actin was used as the loading control.
Next, we examined the effect of chrysoeriol on melanin synthesis by measuring the melanin content of cells after incubation with chrysoeriol at different concentrations (1, 2, 4, and 8 μM) for 72 h. Alpha-melanocyte stimulating hormone (α-MSH), a major factor that stimulates melanin synthesis in the skin, was used as a positive control. As shown in Figure 1b, melanin content in the positive control group increased by approximately 230% compared to that in the untreated control group; compared to the untreated control group, the chrysoeriol treatment groups (1, 2, 4, and 8 μM) showed an increase in melanin content of 12%, 18%, 137%, and 204%, respectively. These results demonstrate that melanin levels were significantly increased by chrysoeriol in a concentration-dependent manner, indicating that chrysoeriol stimulates melanogenesis in B16F10 cells with no cytotoxicity.
Chrysoeriol Increased Intracellular Tyrosinase Activity in B16F10 Cells
TRY activity was first analyzed to understand the possible underlying mechanisms associated with the chrysoeriol-induced stimulation of melanin synthesis in B16F10 cells. TRY is a rate-limiting enzyme involved in the oxidation of tyrosine and L-DOPA in the initial stage of melanin synthesis. After the incubation of B16F10 cells with chrysoeriol at different concentrations (1, 2, 4, and 8 μM) for 72 h, intracellular TRY activity was measured. As shown in Figure 2c, chrysoeriol significantly inhibited intracellular TRY activity in a concentration-dependent manner. Compared to that in the untreated control group, the TRY activity of the positive control group increased by 160%, whereas that of the chrysoeriol-treated groups (1, 2, 4, and 8 μM) increased by 4%, 120%, 161%, and 219%, respectively. In particular, on treatment with 8 μM chrysoeriol, TRY activity was approximately 59% higher than that in the positive control group.
Chrysoeriol Stimulates the Expression of Melanogenic Enzymes in B16F10 Cells
As chrysoeriol treatment increased TRY activity, the protein expression level of TRY was evaluated in B16F10 cells by western blotting. At 72 h after treatment with chrysoeriol, TRY expression was significantly increased in a concentration-dependent manner. TRP-1 and TRP-2, along with TRY, regulate melanin synthesis; therefore, we also examined the expression of TRP-1 and TRP-2 by western blotting. As shown in Figure 2, chrysoeriol treatment led to an increase in the expression of both proteins. In particular, on treatment with 8 μM chrysoeriol, the protein expression of TRY and TRP-2 was increased by approximately 75% and 50%, respectively, compared to that in the untreated control. In the case of TRP-1, the increase in expression on treatment with 8 μM chrysoeriol was nearly four times that in the untreated control group. Taken together, these results indicate that chrysoeriol stimulates melanogenesis by positively regulating the expression of melanogenic proteins.

Effects of chrysoeriol on MITF expression in B16F10 cells. Cells were treated with chrysoeriol at the indicated concentration for 48 h. Western blotting was performed to verify protein levels. MITF expression was quantified by Image J. β-actin was used as a loading control.
MITF is a master regulator of melanogenesis and can upregulate melanogenic genes (TRY, TRP-1, and TRP-2) by binding to the M-box motif in their promoter regions. 22 Thus, we further investigated the protein expression of MITF after chrysoeriol treatment. As shown in Figure 3, the MITF protein expression levels in B16F10 cells were noticeably induced after chrysoeriol treatment in a concentration-dependent manner. In particular, on treatment with 8 μM chrysoeriol, the MITF expression level was approximately 70% higher than that in the untreated control group. These results suggest that MITF is involved in chrysoeriol-induced activation of melanogenesis.
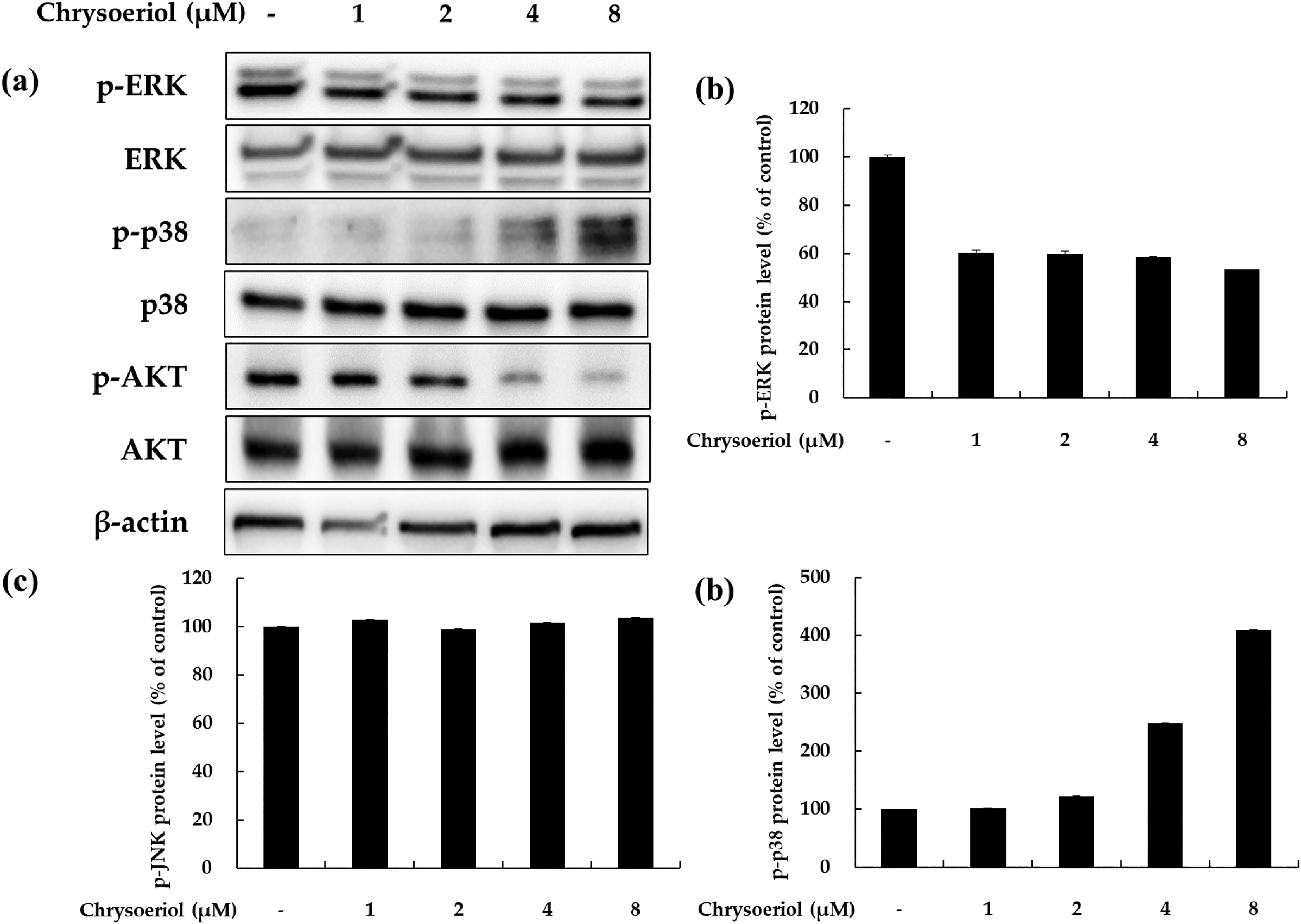
Effects of chrysoeriol on p-ERK, p-p38, and p-AKT expression in B16F10 cells. Cells were treated with chrysoeriol at the indicated concentration for 3 to 4 h. Western blotting was performed to verify protein levels. p-ERK, p-p38, p-AKT, ERK, p38, and AKT levels were quantified by Image J. β-actin was used as a loading control.
Chrysoeriol Inhibits the ERK and PI3K/AKT Signaling Pathways in B16F10 Cells
Previous reports have demonstrated that the extracellular signal-regulated kinase (ERK) and PI3K/AKT signaling pathways negatively regulate melanogenesis in melanocytes and melanoma cells. 23 Therefore, we investigated whether chrysoeriol influences such signaling pathways in B16F10 cells. Western blotting was performed to confirm the effect of chrysoeriol on ERK and AKT phosphorylation. As shown in Figure 4, chrysoeriol treatment tended to decrease ERK and AKT phosphorylation. In particular, treatment with 8 μM chrysoeriol reduced phosphorylation of ERK and AKT by 21% and 70%, respectively, compared to the untreated control group. In addition, we confirmed that phosphorylation of p38 MAPK increased in a concentration-dependent manner following chrysoeriol treatment. These results suggest that chrysoeriol can induce the MAPK and PI3K/AKT signaling pathways in B16F10 cells, which may be responsible for the upregulation of melanogenesis.

Effects of chrysoeriol on p-PKA expression in B16F10 cells. Cells were treated with chrysoeriol at the indicated concentration for 24 h. Western blotting was performed to verify protein levels. p-PKA, PKA, and β-actin levels were quantified by Image J. β-actin was used as the loading control.
Chrysoeriol Stimulates the PKA Signaling Pathways in B16F10 Cells
The cyclic adenosine monophosphate (cAMP)-PKA pathway is also known to be the primary signaling pathway that stimulates melanogenesis. The induction of cAMP production induces the activation of PKA, which phosphorylates the cAMP response element-binding protein (CREB) and CREB-binding protein and consequently upregulates the expression of key melanogenic genes, including MITF, TRY, TRP1, and TRP 2. 24 Therefore, we investigated whether chrysoeriol influences the cAMP-PKA signaling pathway in B16F10 cells. Western blotting was performed to confirm the effect of chrysoeriol on PKA phosphorylation. As shown in Figure 5, on treatment with all concentrations of chrysoeriol, p-PKA expression was increased by more than 30% compared to that in the untreated control group; in particular, p-PKA expression increased by 44% on treatment with 8 μM chrysoeriol compared to that in the untreated control group. These results suggest that chrysoeriol induces melanogenesis through the cAMP/PKA signaling pathway.
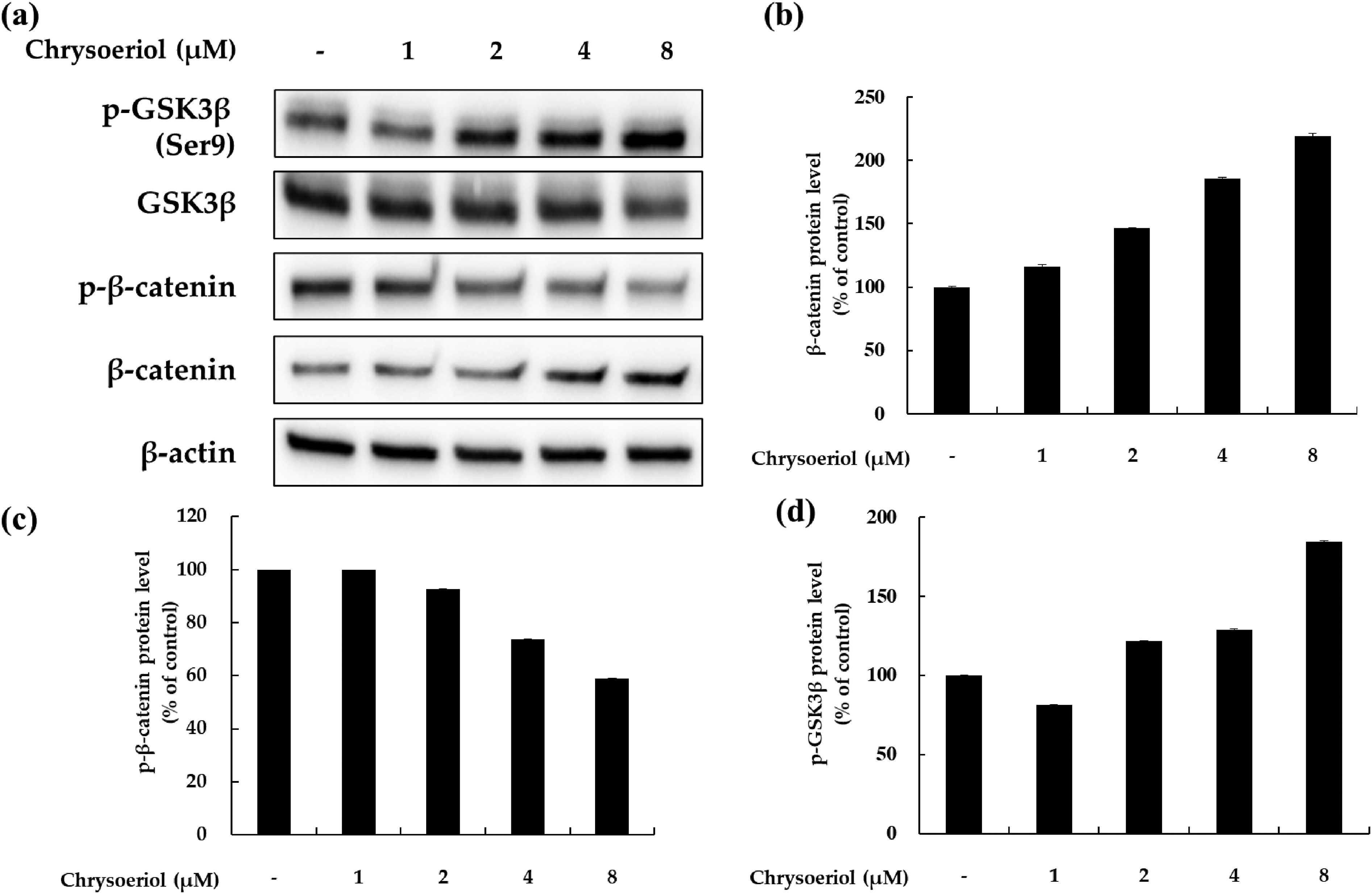
Effects of chrysoeriol on p-GSK-3β, p-β-catenin, and β-catenin expression in B16F10 cells. Cells were treated with chrysoeriol at the indicated concentration for 24 h. Western blotting was performed to verify protein levels. p-GSK-3β, p-β-catenin, β-catenin, and β-actin levels were quantified by Image J. β-actin was used as the loading control.
Effects of Chrysoeriol on the Wnt Signaling Pathway
Previous reports have demonstrated that the Wnt protein binds to its receptor, which phosphorylates and inactivates glycogen synthase kinase 3β (GSK3β), an enzyme that phosphorylates β-catenin that is then destroyed by the proteasome. 25 Through this process, β-catenin accumulates in the cytoplasm and can be transferred to the nucleus; the increased level of β-catenin in the nucleus enhances the expression of MITF to increase melanin production and melanoma cell proliferation. Therefore, we examined whether chrysoeriol influences the modulation of GSK3β/β-catenin expression. As shown in Figure 6, GSK3β and β-catenin levels were increased by chrysoeriol treatment in a concentration-dependent manner. In contrast, the phosphorylation of β-catenin was decreased by chrysoeriol treatment. These data indicate that chrysoeriol increases pigmentation by inducing MITF via the GSK3β/β-catenin signaling pathway.

Effects of chrysoeriol, luteolin, and apigenin on melanin production in B16F10 cells. Cells were treated with chrysoeriol, luteolin, and apigenin at the indicated concentration for 72 h. Melanin content is expressed as a percentage relative to untreated cells. α-MSH-treated cells were used as the positive control. Data are presented as mean ± SD of at least three independent experiments (n = 3). *P < .05 and **P < .01 versus non treated control.
Structure–Activity Relationship of Chrysoeriol and Related Flavonoids
Chrysoeriol (45,7-trihydroxy-3-methoxyflavone) is a flavone; chemically, it is the 3′-methoxy derivative of luteolin, a tetrahydroxyflavone in which the four hydroxy groups are located at positions 3′, 4, 5, and 7. In contrast, apigenin has a 4′,5,7-trihydroxyflavone structure in the absence of a hydroxy group at the C-3′ position of luteolin. The structure–melanogenesis activity relationship of the three flavonoids can be established from the results of melanin production on treatment with 8 µM flavonoid, as shown in Figure 7. Luteolin and apigenin did not show cytotoxicity at the highest concentration of chrysoeriol (8 μM). Chrysoeriol, which has a hydroxyl group at the C-4′ position and a methoxy group at the C-3′ position of the B-ring, increased melanin production in a concentration-dependent manner, similar to the results in the previous experiment. Interestingly, apigenin, which also has a hydroxyl group at the C-4′ position, showed a melanogenesis effect at a concentration of 8 µM, similar to that of chrysoeriol; however, luteolin, which has hydroxyls group at the C-3′ and C-4′ positions, did not show a melanin-increasing effect even at a concentration of 8 µM. The structure–activity relationship (SAR) analysis suggests that a good melanogenic effect requires hydroxyl group substitution at C-4′ and methoxyl group substitution at C-3′ in the B ring.

Chemical structure of chrysoeriol (a) and its related flavonoids, luteolin (b) and apigenin (c).
Discussion
Chrysoeriol has a structure similar to luteolin and apigenin, as indicated by its various synonyms: 4′,5,7-trihydroxy-3′-methoxyflavone, luteolin 3′-methyl ether, 3-O-methylluteolin, and 3-methoxyapigenin (Figure 8). In this study, we investigated the effect of chrysoeriol on melanin biosynthesis and the corresponding molecular mechanisms to understand the relevant signal transduction pathways in B16F10 mouse melanoma cells. Chrysoeriol was not cytotoxic up to a concentration of 8 μM and significantly increased the melanin content and intracellular TRY activity in a concentration-dependent manner in B16F10 cells. Therefore, we hypothesized that the regulation of the melanogenic signaling pathway may be responsible for the stimulation of melanogenesis by chrysoeriol. 25 Thus, the expression of melanogenesis-related proteins was determined by western blot analysis of B16F10 cell lysates obtained after treatment with different doses of chrysoeriol (1-8 μM) for 72 h. In mammals, melanogenesis is controlled by several enzymes, including TRY, TRP-1, and TRP-2. MITF is a major transcriptional regulator of TRY, TRP-1, and TRP-2 and is the most important regulator of melanogenesis in melanocytes.4,5,25 The results presented in Figure 2 demonstrate that chrysoeriol increased the protein expression of TRY, TRP-1, TRP-2, and MITF. MAPKs, including p38 MAPK, extracellular signal-regulated kinase (ERK), and c-Jun N-terminal kinase (JNK), have been reported to be involved in the regulation of MITF expression.4, 25 In addition, previous studies have suggested that melanin synthesis is regulated by a number of signaling pathways, especially those associated with ERK, p38, and PI3K/AKT. Recent studies have shown that some natural compounds upregulate melanogenesis in B16F10 cells through P-ERK downregulation and p38 upregulation. Thus, modulation of the MAPK signaling pathway is a potential means to control melanogenesis. The results shown in Figure 2 indicate that chrysoeriol can activate melanogenesis through the MAPK (ERK and p38) signaling pathways. Additionally, to assess whether chrysoeriol affects the MAPK signaling pathway, we assessed the phosphorylation status of AKT by western blot analysis. As expected, chrysoeriol inhibited AKT phosphorylation (Figure 3d). These results conclusively demonstrate that chrysoeriol downregulates melanogenesis through p-AKT modulation. In previous studies, β-catenin was shown to directly interact with MITF to activate MITF-specific target genes. Both MITF and β-catenin are mediators of Wnt signaling during melanocyte differentiation. Inhibition of GSK3-mediated β-catenin phosphorylation has been reported to be a major event in the Wnt-β-catenin signaling pathway.15,25 Therefore, we used western blotting to confirm the involvement of the GSK3β/β-catenin signaling pathway in the stimulation of melanogenesis by chrysoeriol. As shown in Figure 6, the level of phospho-GSK3β was also upregulated in response to chrysoeriol, leading to inactivation of GSK3β and resulting in increased β-catenin expression and decreased phosphorylation. Melanogenic factors are expressed in response to external stressors and are controlled through mutual regulation by various signaling molecules. It has been reported that the cAMP signaling pathway modulates melanogenesis.6,15 In particular, the cAMP-PKA-CREB pathway activates the expression of melanogenesis-related genes, such as those encoding MITF, TRY, TRP-1, and TRP-2. In this study, chrysoeriol significantly increased PKA activity. These data suggest that the melanogenic effects of chrysoeriol are mediated through the cAMP-PKA-CREB signaling pathway. However, it is necessary to further verify the effect of chrysoeriol on the cAMP/PKA signaling pathway by identifying the levels of cAMP production or CREB phosphorylation.
The biological activity of flavonoids is related to their molecular structure and depends on both the number and location of functional groups such as hydroxyl groups in molecules and the possibility of interacting with the biological membrane, the first natural barrier to the inside of the cell by these components. In nature, flavonoids often occur in plants in the form of glycosylation or methylation. The reason is that this structure is more stable, bioavailability, and bioactive. 26 Numerous scientific studies have shown that methylated flavonoids exhibit much better biological properties than unmethylated forms.26,27 Therefore, the SAR of chrysoeriol and its derivatives including luteolin and apigenin to their melanin stimulating activity were also investigated additionally. It was established that the B-ring of flavone with a hydroxyl group substitution at C-4 led to the most efficient melanin stimulation, and a hydroxyl group substitution at C-3 markedly decreased the melanin-stimulating activity in a reversible manner. Also, comparing the SAR of chrysoeriol and luteolin, it was confirmed that the activation of melanogenesis was restored by replacing the hydroxy group at the C-3 position with a methoxy group.
Taken together, these data demonstrate that chrysoeriol induces melanogenesis by inducing the expression of MITF, a critical protein involved in the expression of other melanogenic proteins, including TRY and TRP. In addition, the chrysoeriol-induced expression of MITF is dependent on the activation of the ERK, AKT, PKA, and Wnt/β-catenin signaling pathways. These results suggest that chrysoeriol is a candidate for use in the treatment of hypopigmentary skin diseases such as vitiligo.
Materials and Methods
Chemicals and Reagents
The chrysoeriol used in this study was purchased from Extrasynthese (Genay CEDEX, France). Dulbecco's modified Eagle's medium (DMEM), penicillin/streptomycin, trypsin-ethylenediaminetetraacetic acid, and bicinchoninic acid assay (BCA) protein kit were purchased from Thermo Fisher Scientific (Waltham, MA, USA). Fetal bovine serum (FBS) was purchased from Merck Millipore (Burlington, MA, USA). α-MSH, protease inhibitor cocktail, sodium hydroxide (NaOH), L-DOPA, sodium phosphate monobasic, and sodium phosphate dibasic were purchased from Sigma-Aldrich (St. Louis, MO, USA). TRY TRP-1, TRP-2, MITF, and β-actin primary antibodies were purchased from Santa Cruz Biotechnology (Dallas, TX, USA). Protease/phosphatase inhibitor cocktail and p-ERK, p-AKT, p-GSK-3β, p-β-catenin, p-PKA, ERK, AKT, GSK-3β, β-catenin, PKA primary antibodies, anti-mouse secondary antibodies, and anti-rabbit secondary antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA). 2 × Laemmli sample buffer and Tween 20 were purchased from Bio-Rad (Hercules, CA, USA). Skim milk was purchased from BD Difco (Sparks, MD, USA). Bovine serum albumin (BSA) was purchased from Bovostar (Bovogen). MTT, dimethyl sulfoxide (DMSO), sodium dodecyl sulfate (SDS), tris-buffered saline (TBS), and enhanced chemiluminescence (ECL) kits were purchased from Biosesang (Seongnam, Gyeonggi-do, Korea).
Cell Culture
B16F10 murine melanoma cells were obtained from ATCC (Manassas, VA, USA). Cells were cultured in DMEM containing 10% (v/v) FBS and 1% (v/v) penicillin-streptomycin in a humidified incubator at 37°C and 5% CO2. The cells were passaged every 3 days.
Measurement of Cell Viability
The MTT assay was performed to determine the effect of chrysoeriol on cell viability. Cells were seeded into a 24-well plate at 8 × 103 cells/well for 24 h, treated with 1, 2, 4, 8, or 16 μM chrysoeriol and α-MSH (100 nM), and incubated for 72 h. The supernatant was removed, and MTT reagent (0.2 mg/mL in DMEM) was added for 4 h. After the reaction time, all MTT reagents and DMSO were removed. Absorbance was measured at 560 nm using a microplate reader (Tecan).
Measurement of Melanin
The release of melanin from the culture was determined as previously described with modifications. 16 B16F10 cells were seeded into a 6-well plate at 5 × 104 cells/well for 24 h, treated with 1, 2, 4, or 8 μM chrysoeriol and α-MSH(100 nM), and incubated for 72 h. Then, the supernatant was removed, and the cells were washed once with 1 × PBS. Radioimmunoprecipitation assay (RIPA) buffer (200 μL) was added, followed by lysis at 4°C for 20 min. The cells were added to a 1.5 mL e-tube with a cell scraper and centrifuged at 15 000 rpm for 20 min at −8°C. The pellets were then obtained. The pellet was dissolved in 200 μL of 1 N NaOH with 10% DMSO at 80°C for 10 min. The absorbance was measured at 405 nm using a microplate reader (Tecan). Protein concentration was quantified using a BCA protein assay kit.
Intracellular Tyrosinase Assay
An intracellular TRY activity assay was performed to determine TRY activity in B16F10 cells. B16F10 cells were seeded into a 6-well plate at a density of 5 × 104 cells/well for 24 h. The cells were treated with the samples at different concentrations and incubated for 72 h. After washing with 1 × PBS, RIPA buffer containing 1% protease inhibitor cocktail was added. Cells were lysed at 4°C for 20 min. The supernatant was separated by centrifugation under the same conditions used for the melanin content assay. Protein concentration was quantified using a BCA protein assay kit. Then, 20 μL of protein (20 μg/mL) and 80 μL of L-DOPA (2 mg/mL) were added to a 96-well plate and incubated at 37°C for 2 h. Absorbance was measured at 490 nm using a spectrophotometric microplate reader (Tecan).
Western Blot Analyses
B16F10 cells were seeded into a 6-well plate at 5 × 104 cells/well for 24 h and treated with 1, 2, 4, or 8 μM chrysoeriol and α-MSH(100 nM). The samples were then treated for 4, 24, 48, and 72 h. After washing with cold 1 × PBS, RIPA buffer with 1% protease inhibitor cocktail or 1% protease/phosphatase inhibitor cocktail and lysed at 4°C for 20 min. After collecting cells in a 1.5 mL e-tube, centrifugation was performed at 15 000 rpm for 30 min at −8°C, and the supernatant was obtained. Protein concentration was quantified using the BCA protein assay kit. The protein was quantified at 20 μg/mL, and the loading sample was heated at 100°C for 5 min with the sample buffer prepared; the protein was quantified in a 1:1 ratio. The loading sample was stored at −20°C before loading. Protein samples were separated by SDS-polyacrylamide gel by size. The isolated proteins were transferred from a gel to a polyvinylidene fluoride (PVDF) membrane. The membrane was washed with TBS containing 1% Tween 20 (1 × TBS-T), blocked with 5% skim milk (in 1 × TBS-T) or 5% BSA (in 1 × TBS-T) for 1 h, and cleaned six times with 1 × TBS-T for 10 min. Antibodies were diluted with 1% skim milk (in 1 × TBS-T, Santa Cruz) or 1% BSA (in 1 × TBS-T, Cell Signaling) at a 1:1000 ratio. The membrane was incubated with the primary antibody for more than 12 h at 4°C. The secondary antibody diluted at 1:1000% to 1% BSA (in 1 × TBS-T) was then incubated at room temperature for more than 2 h and washed with 1 × TBS-T for 10 min for six times. Finally, the ECL solution was attached to the secondary antibody, and proteins were detected using Chemidoc (WL, Vilber Lourmat, France).
Statistical Analysis
All results are presented as the mean ± SD of three replicates. Statistical significance was expressed as a P value using Student's t-test. *P < .05, **P < .01
Footnotes
Acknowledgments
The authors would like to thank all the students in our research group for their helpful cooperation and discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the Ministry of Trade, Industry and Energy (MOTIE), Korea Institute for Advancement of Technology (KIAT) through the Industry Innovation Foundation Project (P0018139).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
