Abstract
Because the number of people suffering from allergies has significantly increased, improved ways of treating these conditions by medical, pharmaceutical, and dietary means are required. Large numbers of studies on allergy have been conducted, and many anti-allergic compounds have been found. Phenolic compounds from terrestrial plants, including catechins and flavonoids, possess anti-allergic properties. Although polyphenols are present in some brown algae, their anti-allergic activities were not studied in detail before the 1990s. The focus was on the algal polyphenols, collectively called phlorotannins (eg., eckol, 6,6′-bieckol, 8,8′-bieckol, dieckol, and phlorofucofuroeckol-A), and research was conducted to clarify their anti-allergic activities. This review summarizes the anti-allergic effects of phlorotannins isolated from the brown alga, Eisenia nipponica, and related reports by other research groups.
Introduction
The number of allergic patients increases worldwide, particularly in developed countries. Therefore, allergy is a serious issue. Platts-Mills mentioned that the various hypotheses on the increase in allergy include hygiene improvement, indoor lifestyle, dietary habit alterations, and immunological deterioration. 1 According to the report, hay fever increased in the first half of the twentieth century, and asthma, primarily among children, in the second half. 1 He also highlighted the increase in food allergy after the 1990s. 1 In Japan, epidemiological research by the Ministry of Health, Labour and Welfare in 2005 showed that 47.2% of adults suffered from allergic symptoms. 2 Moreover, asthma, atopic dermatitis, and food allergy were found in about 10% of infants from the research in 2005 to 2008. Although there was a difference among regions, 30%–50% of all age groups were estimated to suffer from hay fever. 2 Thus, allergy has been a societal issue. Symptomatic therapies and dietary remedies were investigated in the medical and pharmaceutical fields to approach the allergy issue. Meanwhile, many studies on anti-allergic foods and their active principles were performed arduously in the field of food science. The studies revealed various healthy foods and components for their anti-allergy effects, and the typical anti-allergic and anti-inflammatory compounds were polyphenols.
At the beginning of the 1970s, the anti-inflammatory effect of taxifolin, a quercetin derivative, was reported. 3 In the 1980s, Japanese research groups reported the anti-allergic and anti-inflammatory effects of flavonoids 4 and catechins. 5 After the 1990s, the anti-allergic effects of catechins were vigorously investigated. Epigallocatechin gallate (EGCG) and methylated catechins were the typical active compounds, and details of their activity expression mechanisms were elucidated. 6
Although polyphenols are primarily obtained from terrestrial products, marine brown algae contain polyphenols called phlorotannins. 7 The brown algae Ecklonia cava, E. cava subsp. kurome, E. cava ssp. stolonifera, Eisenia bicyclis, and E. nipponica are edible 8 and grow off the coast of Japan.9,10 The algae were expected to contain many phlorotannins composed of a unique phloroglucinol skeleton, 7 and their local and chemical distribution in brown algae, including Ecklonia cava, E. cava subsp. kurome, and E. bicyclis, were discovered. 11
In 1973, Whitaker released the first report on the anti-inflammatory effect of E. bicyclis extract, rich in phlorotannins. 12 In the 2000s, Shibata et al reported that phlorotannins from E. cava ssp. kurome and E. bicyclis inhibited the activities of inflammation-related enzymes, such as phospholipase A2 (PLA2), 13 cyclooxygenase (COX)-2, 13 lipoxygenase (LOX), 13 and hyaluronidase (HA). 14 After these studies, many reports on the anti-inflammatory and anti-allergic effects of phlorotannins were published.
Although many studies on the anti-allergic effects of terrestrial polyphenols had already been published, studies on the impact of phlorotannins were few at the beginning of the 2000s. This caused us to focus on the activity of phlorotannins and to investigate their active components.
This review discusses the results of our research on the anti-allergic and anti-inflammatory effects of phlorotannins, and related reports by other researchers, such as those by Shibata et al.13,14
The Anti-Allergic Effects of the Brown Alga, Eisenia nipponica, and Related Studies by Other Research Groups
Screening of Seaweeds Possessing an Anti-Allergic Effect
To investigate seaweeds possessing anti-allergic effects, seaweed samples were obtained, from April to May 2001, from the Karasu fishing port in Tsu (latitude, 34.660123; longitude, 136.543755) to Zaga Island (latitude, 34.276119; longitude, 136.804116) and Mugizaki (latitude, 34.260573; longitude, 136.844467) in the Ise-Shima region, located on the coast of Mie Prefecture, Japan. Forty-two samples, identified by Dr M. Maegawa and Dr A. Kurashima, Mie University, were obtained as follows; 21 brown algae, five green algae, 15 red algae, and one seagrass. 15 A method for screening for anti-allergic effects was considered, following sample collection. Because there were many anti-allergic studies using rat basophilic leukemia (RBL)-2H3 cells, 16 they were used as mast cell-like cultured-cells, which possess a histamine release property, a typical-allergic marker. The measurement of histamine content in RBL cells in culture supernatant was performed using high-performance liquid chromatography (HPLC) with fluorescent detection. 17
Eighty percent methanol (80M) extracts from the 41 seaweeds and one seagrass were evaluated for their suppressive effects on histamine release in RBL cells using the procedure. From the results of the experiments, 80M extracts from seven brown algae (Ecklonia cava, Eisenia nipponica, Ishige foliacea, I. okamurai, Sargassum micracanthum, S. ringgoldianum, and S. thunbergia) suppressed histamine release. Moreover, resulting from cell death detection by Trypan blue exclusion, the extracts of E. nipponica and S. thumbergia did not cause RBL cell death. 15 The 80M extract of E. nipponica had a high amount of phlorotannin (89.2%; original data). Therefore, E. nipponica, which is used as a food, 18 was screened as the most suitable research subject. 15 The brown alga contains abundant phlorotannins, 8 which are deemed to be anti-allergic components.
Investigation of Active Compounds
Because the 80M extract of E. nipponica showed an anti-allergic effect, an investigation to identify its active principles began. Nakamura et al reported that the methanol/chloroform (2/1) extract of E. bicyclis was partitioned using distilled water to identify phlorotannins in the methanolic water layer. The methanolic water solution was extracted with diethyl ether to obtain a crude mixture of phlorotannins. 19 Nagayama et al reported using ethyl acetate instead of diethyl ether to obtain a phlorotannin mixture from the methanol/chloroform (2/1) extract of E. cava ssp. kurome. 20 They used normal-phase chromatography on a silica gel column and reverse-phased HPLC on an octadecylsilyl (ODS) column to isolate each phlorotannin.19,20
According to the reports, methanol/chloroform (M/C; M/C = 1/2) extraction was applied instead of 80M extraction of dried E. nipponica powder. The M/C extract was partitioned with water. The aqueous methanol fraction was partitioned with either diethyl ether or ethyl acetate (Figure 1). At a concentration of 1 mg/mL, the two fractions obtained were assessed to determine the presence of suppressive effects on degranulation in RBL cells using the method of degranulation amount measurement 21 attributable to the correlation between the level of degranulation in RBL cells and β-hexosaminidase activity. 22 The diethyl ether fraction (suppression ratio, 95.4%) was more potent than that of the ethyl acetate fraction (suppression ratio, 72.8%). The diethyl ether fraction was fractionated using silica gel chromatography and HPLC with an ODS column. The anti-degranulation test was applied to the obtained fractions, and the active fractions were exposed to nuclear magnetic resonance (NMR) spectral analyses. Each tested fraction was contaminated by a large amount of pigment, which forced the re-evaluation of the strategy to isolate and identify the active components of the alga. The pigments had to be removed during the early stage of purification. Thus, the dried algal powder was defatted and depigmented using n-hexane and ethyl acetate before M/C extraction. According to the above-mentioned procedure, the M/C extract was purified. By optimizing the HPLC conditions, six compounds with strong anti-degranulation activity were isolated. Mass and NMR spectrometric analyses of these compounds clarified their chemical structures; eckol, 6,6′-bieckol, 6,8′-bieckol, 8,8′-bieckol, phlorofucofuroekol (PFF)-A, and PFF-B, namely, phlorotannins. The structures are indicated in Figure 2. 23 Among these phlorotannins, PFF-B was a new entry. 24

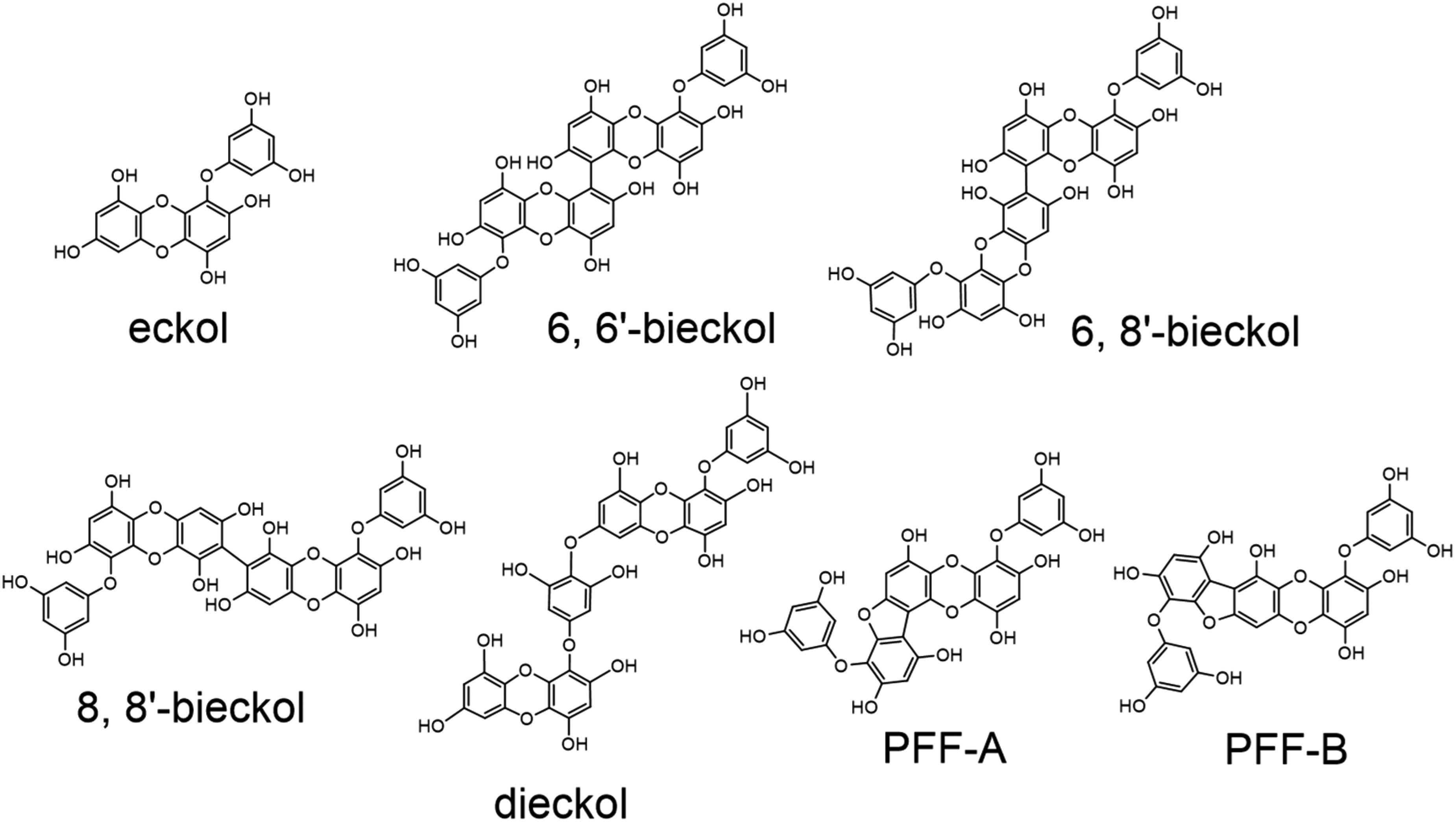
Chemical structures of anti-allergic phlorotannins isolated from E. nipponica. Abbreviation: PFF, phlorofucofuroeckol.
The remaining problem was whether the chloroform fraction of the M/C-water partition (Figure 1) possessed any anti-allergic compounds. A bioactivity test using an allergic inflammatory mouse model showed the anti-inflammatory effect of the fraction. 25 A similar purification procedure for the aqueous methanol fraction was applied to the chloroform fraction. It was fractionated first using a normal-phase silica gel column and then by reverse-phase HPLC. A LOX activity inhibition test used on the purified compound indicated that it possessed anti-allergic effects. Mass and NMR spectral analyses clarified its structure as dieckol, a phlorotannin (Figure 2). 26
The seven phlorotannins were isolated from not only E. nipponica, but also from other Ecklonia and Eisenia species.13,14,19,20,23,26‐33 For example, dieckol was isolated from the ethyl ether and ethyl acetate fractions of E. cava ssp. kurome13,20 and E. bicyclis;13,14,19 the amounts of dieckol in dried E. cava ssp. stolonifera and E. bicyclis were < 2.4% and < 2.2%, respectively.30,33 Therefore, dieckol would be the primary phlorotannin found in Ecklonia and Eisenia species. The seven phlorotannins isolated from the edible Ecklonia and Eisenia species are summarized in Table 1.
The Seven Phlorotannins from Five Edible Brown Algae, Ecklonia sp. and Eisenia sp.

Abbreviation: PFF, phlorofucofuroeckol.
In Vivo Assay Using Allergic Model Mice and in Vitro Mechanism-Related Study
Anti-allergic evaluation of the seven isolated phlorotannins was performed using the Institution of Cancer Research (ICR) strain mice, an allergic inflammation model, 34 to assess the efficacy of in vivo experiments. Arachidonic acid (AA), 12-O-tetradecanoylphorbol 13-acetate (TPA), and oxazolone (OXA) served as the sensitizer for allergic inflammation induction. Acute, acute chronic, and delayed-type (type IV allergic) inflammation were induced by the sensitizaters.35,36 When the seven phlorotannins were percutaneously administered to the ICR mouse‘s ear stimulated by sensitizers, suppressive effects on ear swelling were observed.26,37,38 For oral administration, the phlorotannins also showed suppressive effects26,39 (Table 2).
The Anti-Allergic Effects of the Seven Phlorotannins Isolated from E. nipponica.

Abbreviations: PFF, phlorofucofuroeckol; AA, arachidonic acid; TPA, 12-O-tetradecanoylphorbol-13-acetate; OXA, oxazolone; PLA2, phospholipase A2; SBL, soybean lipoxygenase; COX-2, cyclooxygenase-2; HA, hyaluronidase.
Data were calculated from the results of multiple experiments (in vivo ear swelling test, n = 4; in vitro enzymatic activity and degranulation test, n = 3). Values are presented as means ± standard deviations. These data were converted from published data.26,37‐39,47,48 The dose of phlorotannins in the ear swelling test was 75-nmol/mouse. aInhibition ratio of 30.2% ± 14.7% at 2000-µM concentration; b,cinhibition ratios of 9.8% ± 0.9% and 40.4% ± 3.4% at 500-µM concentration, respectively.
The ear swelling induced by AA and TPA is involved in the AA cascade. 40 Enzymes for chemical mediator synthesis are activated in the cascade, and the related enzymes are primarily PLA2, COX-2, and LOX. 41 Because the ear swelling induced by TPA accompanies rapid COX-2 mRNA expression, 42 TPA action was considered to be gene associated. Alternatively, OXA that induces type IV allergic inflammation causes topical inflammation, such as AA and TPA. 43 OXA also causes immunity imbalance accompanied by excessive pro-inflammatory cytokine production, including interleukin (IL)-6 and tumor necrosis factor (TNF)-α. 44 For ear swelling induced by the sensitizers, chemical mediators released from inflammatory lymphocytes provoke inflammation, which results from the activation of the AA cascade.43,44 The ear swelling is involved with degranulation through inflammatory lymphocytes, 45 and HA activation corresponding to the degranulation reaction 46 should be considered.
Considering the above-mentioned mechanisms in ear swelling induction, further investigations for mechanistic elucidation were discussed based on the suppressive effects of the seven phlorotannins on ear swelling. When the inhibitory effects of the phlorotannins on inflammation-related enzymatic activities (PLA2, COX-2, LOX, and HA) were assessed, the phlorotannins inhibited the enzymatic activities (Table 2).26,39,47,48 These data confirmed the inhibitory effects of phlorotannins isolated from E. cava ssp. kurome and E. bicyclis on enzymatic activities.13,14 In degranulation experiments using RBL cells stimulated by the antigen-antibody reaction, each phlorotannin also exhibited its anti-degranulation activity (Table 2).23,26,39 The phlorotannins also suppressed the chemical mediators [histamine, leukotriene B4 (LTB4), and prostaglandin E2 (PGE2)] which were released.26,39 Moreover, the phlorotannins suppressed COX-2 mRNA expression in RBL cells stimulated by calcium ionophore, A23187.26,39 Additionally, dieckol suppressed IL-6 and TNF-α mRNA expression. 26 Efficacies and suppressive mechanisms of the seven phlorotannins are summarized in Figure 3 and Table 3.

Diagram of the anti-allergic and anti-inflammatory mechanisms of the seven phlorotannins. Abbreviations are indicated in Table 3.
Anti-Allergic and Anti-Inflammatory Mechanisms of the Seven Phlorotannins.

Abbreviations: AA, arachidonic acid; COX, cyclooxygenase; HA, hyaluronidase; LOX, lipoxygenase; IL, interleukin; OXA, oxazolone; PLA2, phospholipase A2; TNF, tumor necrosis factor; PFF, phlorofucofuroeckol; SBL, soybean lipoxygenase; TPA, 12-O-tetradecanoylphorbol 13-acetate.
1We hope that you change a layout of Table 3 likeTables 4 and 5.
2Please change to 14.
3Please change to 13.
Thus, the involvement of the above-mentioned suppressive mechanisms was associated with phlorotannin suppression of ICR mouse ear swelling. Therefore, it was discovered that the seven phlorotannins from E. nipponica can suppress acute inflammation through type I allergic reactions and chronic inflammation by type IV allergy in four allergy types.
Comparison of Efficacy Between the Isolated Phlorotannins and a Synthetic Anti-Allergic Reagent
As mentioned above, the seven phlorotannins are useful for anti-allergic health food components. In comparison with dexamethasone, which is widely used as a synthetic anti-allergic reagent and suppresses mouse ear swelling, 49 the suppressive effects of the phlorotannins were either comparable to and/or weaker than those of the reagent (Tables 2 and 4). Notably, 6,6′-bieckol in percutaneous administration and 6,8′-bieckol in both administrations exhibited noticeable suppression similar to that of dexamethasone. Regarding structure-activity correlations, the number of hydroxy groups in the three bieckols is greater than those in the other four phlorotannins, and the locations of the hydroxy groups vary among the phlorotannins (Figure 2). Since the amount and locations of the hydroxy groups in terrestrial polyphenols, including tea catechins and flavonoids, are critical,50,51 the amount and locations of the hydroxy groups in 6,6′-bieckol and 6,8′-bieckol may explain their higher efficacy. The suppressive or inhibitory effects of the phlorotannins on degranulation in RBL cells and inflammation-related enzymatic activities were also compared to those of synthetic anti-allergic reagents [ie., wortmannin (degranulation suppresser), 52 Me-Indoxam (secretory PLA2 inhibitor), 53 zileuton (5-LOX inhibitor), 54 celecoxib (COX-2 inhibitor), 55 and disodium cromoglycate (HA inhibitor) 56 ]. As indicated in Tables 2 and 4, the inhibitory effects of the phlorotannins on PLA2, LOX, and COX-2 activities were weaker than those of the reagents. The inhibitory effects on HA activity were greater than and/or comparable to those of disodium cromoglycate. Regarding anti-degranulation, the suppressive effects of PFF-A and PFF-B were similar to that of wortmannin (Tables 2 and 4). Generally, the efficacy of these synthetic anti-allergic reagents would be comparable to and/or higher than those of the phlorotannins. Considering the potential of phlorotannins as healthy food ingredients with anti-allergic properties and food safety, a more moderate effect than that seen in the aforementioned anti-allergic agents might be desirable. In parallel, according to previous studies and a report by Shibata et al,13,26,48 the phlorotannins inhibited secretory PLA2, 5-LOX, and COX-2 involved in inflammatory reactions, such as allergy.57‐59 The phlorotannins would specifically suppress allergic inflammation. Therefore, it is proposed that phlorotannins are useful components of healthy foods.
The Efficacy of Typical Synthetic Anti-Allergic Reagents.
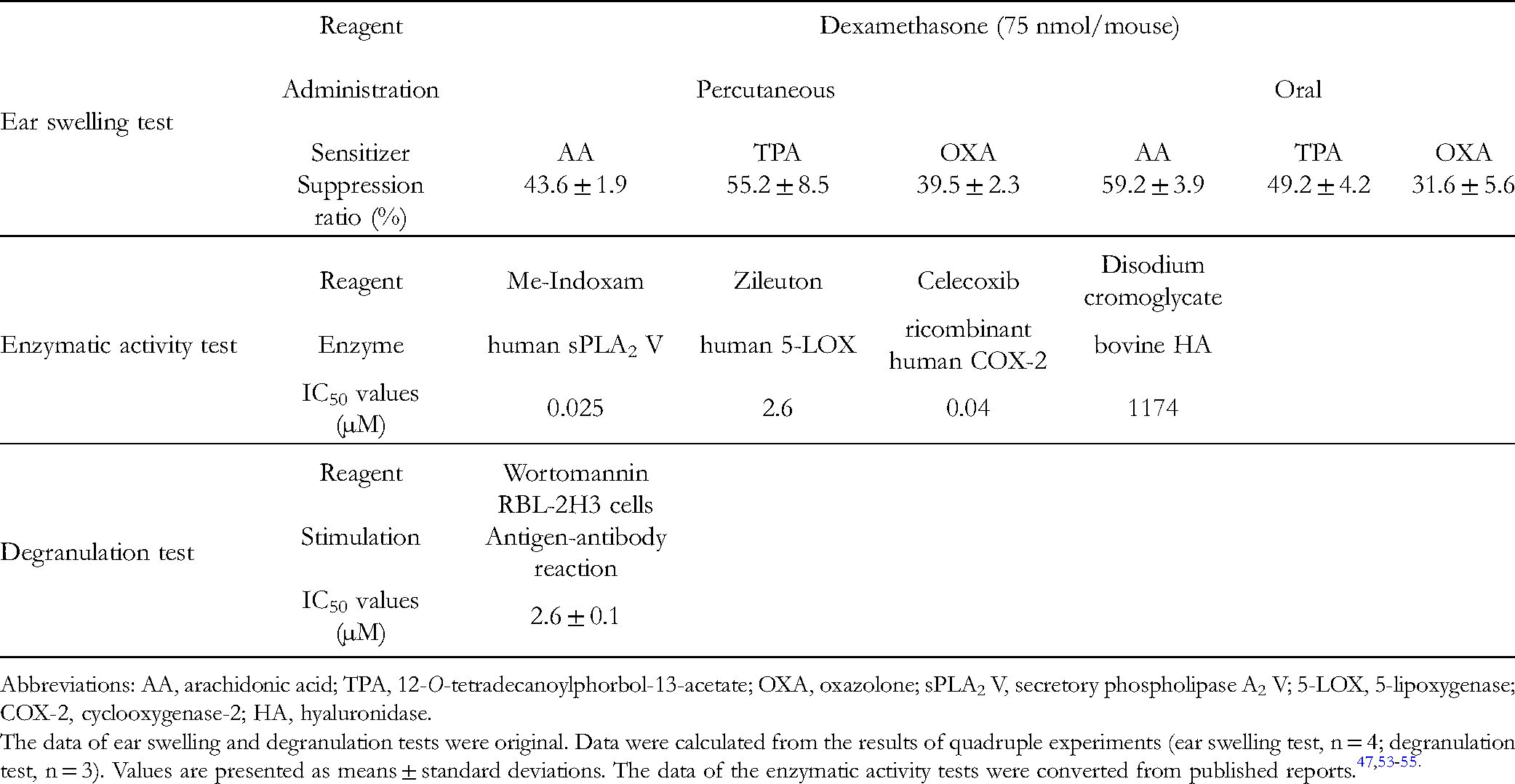
Abbreviations: AA, arachidonic acid; TPA, 12-O-tetradecanoylphorbol-13-acetate; OXA, oxazolone; sPLA2 V, secretory phospholipase A2 V; 5-LOX, 5-lipoxygenase; COX-2, cyclooxygenase-2; HA, hyaluronidase.
The data of ear swelling and degranulation tests were original. Data were calculated from the results of quadruple experiments (ear swelling test, n = 4; degranulation test, n = 3). Values are presented as means ± standard deviations. The data of the enzymatic activity tests were converted from published reports.47,53‐55.
The Anti-Allergic or Anti-Inflammatory Effects of Typical Phlorotannins
Previous studies revealed the anti-allergic mechanisms for suppressing degranulation and the attenuation of the AA cascade. The following account describes the anti-allergic and anti-inflammatory effects of phlorotannins as reported by other research groups. Among phlorotannins isolated from brown algae, eckol, 6,6′-bieckol, 8,8′-bieckol, dieckol, and PFF-A (Figure 2) have been isolated from Ecklonia and Eisenia species,13,14,19,20,23,27‐30 and are the most typical phlorotannins. Other research groups reported their anti-allergic and anti-inflammatory effects with different mechanisms. Therefore, in the following section, the reported effects of typical phlorotannins are discussed.
Anti-Degranulation
The suppressive effects of 6,6′-bieckol, dieckol, and PFF-A on histamine release by RBL cells were observed and associated with binding inhibition between IgE and IgE receptors (FcεRI) expressed on the cell membrane surface.60,61 Moreover, in an experiment using human mast cell lines (KU812 F), PFF-A suppressed degranulation and Ca2+ influx, and the mechanism was the suppression of FcεRI expression on the cell surface 62 (Table 5).
Anti-Allergic and Anti-Inflammatory Mechanisms of Five Typical Phlorotannins.

Abbreviations: Akt, protein kinase B (PKB); AP-1, activator protein-1; CAMs, cell adhesion molecules; COX-2, cyclooxygenase-2; ERK, extracellular signal-regulated kinase; GATA3, GATA binding protein 3; HMGB-1, high mobility group box-1; HO-1, heme oxygenase-1; I-κB, inhibitor of NF-κB; IFN-γ, interferon-γ; IgE, immunoglobulin E; IKK, I-κB kinase; IL, interleukin; iNOS, inducible nitric oxide synthase; JNK, c-Jun N-terminal kinase; MAPK, mitogen-activated protein kinase; MDC/CCL22, macrophage-derived chemokine/C-C motif chemokine ligand 22; NF-κB, nuclear factor-kappa B; NO, nitric oxide; Nrf2, nuclear factor erythroid 2-related factor 2; PCA, passive cutaneous anaphylaxis; PGE2, prostaglandin E2; PI-3K, phosphatidylinositol-3 kinase; ROS, reactive oxygen species; SMAD, contraction of Sma and Mad (Mothers against decapentaplegic); SOD, superoxide dismutase; STAT 1, signal transducer and activator of transcription 1; TARC, thymus and activation-regulated chemokine; T-bet, Th1-specific T box transcription factor; TGF-β1, transforming growth factor-β1; Th17, T helper 17 cell; Treg, regulatory T cell; TNF-α, tumor necrosis factor-α; TSLP, thymic stromal lymphopoietin; PFF, phlorofucofuroeckol.
Attenuation of the MAPK and NF-κB Signaling Pathway
Mitogen-activated protein kinase (MAPK) and nuclear factor-kappa B (NF-κB) signaling play a vital role in inflammatory reactions, such as allergic reactions. The signaling pathway complexly includes the production of chemical mediators, such as nitric oxide (NO) and PGE2; the expression of inflammation-related enzymes, such as COX-2 and inducible nitric oxide synthase (iNOS); and the expression and production of pro-inflammatory cytokines, such as IL-1β, IL-6, and TNF-α. These phenomena are attributed to phosphorylation and activation of the signal transduction factors [extracellular signal-regulated kinase (ERK), I-κB kinase (IKK), c-Jun N-terminal kinase (JNK), and MAPK] and the expression and nuclear translocation of transcription factors [inhibitor of NF-κB (I-κB) and NF-κB]. Thus, the signaling pathway is a well-accounted suppression target.63‐67
The five phlorotannins attenuated the MAPK and NF-κB signaling pathways to suppress chemical mediator production, such as NO, PGE2, and reactive oxygen species (ROS), after the regulation of inflammation-related enzymes (COX-2 and iNOS) and pro-inflammatory cytokines [interferon (IFN)-γ, IL-1β, IL-6, and TNF-α]. The attenuation was attributed to the suppression of activation and nuclear translocation of NF-κB, activation of signal transduction factors [protein kinase B (Akt), activator protein-1 (AP-1), JNK, and p38 MAPK], and phosphorylation of transcription factors (NF-κB and I-κBα) and signal transduction factors [Akt, ERK, IKK-α/β, JNK, p38 MAPK, and phosphatidylinositol-3 kinase (PI-3K)].32,68‐85 Regarding the other suppressive mechanism, 6,6′-bieckol and PFF-A suppressed the signal transduction of the promoter region involved in COX-2, iNOS, IL-6, and TNF-α expression.72,84 Dieckol caused superoxide dismutase (SOD) production and activation of heme oxygenase-1/nuclear factor erythroid 2-related factor 2 (HO-1/Nrf2) signaling because of the above-mentioned suppression of phosphorylation and nuclear translocation, and this regulation mechanism caused anti-inflammation.79,80 Although these reports resulted from in vitro studies, there is a report on in vivo studies. According to Yang et al, dieckol suppressed inflammation in 1,2-dimethylhydrazine (DMH)-induced inflammation model rats attributable to the suppression of COX-2, iNOS, and pro-inflammatory cytokine (IL-1β, IL-6, and TNF-α) expression 82 (Table 5).
Immunomodulation
Commonly, an allergic reaction includes diverse lymphocytes and different immunological factors, including cytokines and chemokines. Regulation of these lymphocytes and immunological factors lead to the alleviation of inflammation and allergic reactions. There are some reports on the immunomodulating effects of eckol, dieckol, and PFF-A. Kim et al discovered that eckol and dieckol exerted anti-inflammatory effects by attenuating inflammatory lymphocyte‘s migration and adhesion by suppressing high mobility group box-1 (HMGB-1) release, barrier disruption caused by HMGB-1, and cell adhesion molecules (CAMs) expression. 86 These effects include suppression of chemokine [macrophage-derived chemokine/C-C motif chemokine ligand 22 (MDC/CCL 22)] production; and attenuation of signal transducer and activator of transcription 1‘s (STAT 1) phosphorylation and nuclear translocation. 87 Passive cutaneous anaphylaxis (PCA) in BALB/c mice was alleviated by eckol and dieckol through either the expression or production suppression of pro-inflammatory cytokines and chemokines, including IL-4, 5, 6, 13, and thymus and activation-regulated chemokine (TARC).88,89 Additionally, there are some reports on dieckol‘s effect. Yang et al reported that its immunomodulation alleviated atopic dermatitis in NC/Nga mice. The immunomodulating mechanism included suppression of IgE, pro-inflammatory cytokine (IL-4, 5, and 33), and chemokine [thymic stromal lymphopoietin (TSLP)] production; suppression of GATA binding protein 3 (GATA3) expression; increase in IFN-γ production; and the induction of Th1-specific T box transcription factor (T-bet) expression. 90 Moreover, dieckol relieved inflammation model rats through decreased serum IL-17A level and improved Th17/Treg cell balance. 91 There is a report by Heo et al on PFF-A. Binding inhibition between transforming growth factor (TGF)-β1 and its receptor by PFF-A caused the degradation of MAPK signaling through the suppression of signal transduction factor [ERK, JNK, and contraction of Sma and Mad (SMAD) 2/3] phosphorylation 92 (Table 5).
Other Phlorotannins
Other research groups have reported on the anti-inflammatory effects of several phlorotannins, except for the above-mentioned typical phlorotannins. Dioxinodehydroeckol, diphlorethohydroxycarmalol, fucofuroeckol-A, and trifuhalol-A attenuated MAPK and NF-κB signaling pathways through the suppression of signal transduction factor‘s [muscle RING-finger protein (MuRF)-1, muscle atrophy F-box (MAFbx)/Atrgoin-1, suppressor of cytokine signaling 1 (SOCS1), and STAT 5] expression; and degradation of NF-κB‘s phosphorylation and nuclear translocation. Resulting from those effects, they suppressed the production of PGE2, IL-1β, IL-6, TNF-α, and monocyte chemoattractant protein-1 (MCP-1) and expression of COX-2 and iNOS.93‐97 Although PFF-B‘s effects were mentioned above, there are reports by other research groups. PFF-B suppressed expression of COX-2 and iNOS and production of NO, IL-1β, and IL-6 and the effect was attributed to the degradation of the NF-κB signaling pathway through the suppression of Akt, ERK, and JNK phosphorylation.32,68,98 Moreover, 2′-phloroeckol suppressed NO production, 32 and fucophlorethol C inhibited LOX activity. 99
Conclusion
Phlorotannins are phenolic compounds abundantly present in brown algae, such as Ecklonia sp. and Eisenia sp., that possess a chemical structure different from that of terrestrial products. Phlorotannins alleviate allergic inflammation, the PCA reaction, atopic dermatitis, and DMH-induced inflammation through AA cascade attenuation, regulation of the MAPK/NF-κB signaling pathway, and immunomodulation. Thus, phlorotannins would decrease acute type I allergic and chronic type IV allergic reactions. The suppressive mechanisms of the five typical phlorotannins are summarized in Figure 4. The above-mentioned mechanisms are indicated as follows: Frame A. anti-degranulation; Frame B. attenuation of AA cascade; Frame C. attenuation of MAPK and NF-κB signaling; Frame D. immunomodulation.

Diagram of the anti-allergic and anti-inflammatory mechanisms of typical phlorotannins. Frame A. anti-degranulation; Frame B. attenuation of AA cascade; Frame C. attenuation of MAPK and NF-κB signaling; Frame D. immunomodulation. Abbreviations such as AA, COX-2, HA, LOX, and PLA2 are indicated in Table 3, and other abbreviations are indicated in Table 5.
Footnotes
Acknowledgments
We thank Dr T. Shibata (Mie University), Guest Editor of Special Collection on Phlorotannins for Natural Product Communications, for the invitation to this article‘s author. Regarding the outcome of this study on the anti-allergic effect of E. nipponica, we thank collaborators from Mie University, Kanehatsu Food Co., Ltd, and National Fisheries University for their technical assistance. The authors would like to thank Enago (www.enago.jp) and MARUZEN-YUSHODO Co., Ltd (![]() ) for the English language review.
) for the English language review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the JSPS KAKENHI (grant number 17K07951, 23880036, 25850099).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
