Abstract
The Chinese herbal medicine Rosa roxburghii Tratt (RRT) is widely used in the treatment of malignant tumors, including gastric cancer (GC), but its pharmacological mechanism remains unclear. The purpose of this research was to identify the mechanisms of RRT on treating GC by using network pharmacology and molecular docking, combined with the analysis of differential expressed genes in GEO gene chips and TCGA database. We first defined the effective components of RRT and their potential targets for the treatment of GC, and identified core targets according to the topology analysis by constructing a protein-protein interaction network. Furthermore, molecular docking was used to verify the docking between the core active ingredients and the key targets. The results showed that the effect of RRT may be closely associated with multiple signal pathways, including pathways in cancer, phosphatidylinositol 3-kinase-AKT serine/threonine kinase (PI3K-Akt), tumor necrosis factor (TNF), hypoxia-inducible factor 1 (HIF-1), and mitogen-activated protein kinase (MAPK). It is suggested that RRT may play an effect by regulating hypoxia, improving the tumor microenvironment, inhibiting inflammatory reactions and promoting apoptosis. The mechanism of RRT in the treatment of GC is revealed here for the first time based on network pharmacology analysis, which may provide a new direction for further exploration of the mechanisms of RRT in the treatment of GC and a new perspective for research on anti-tumor drugs.
Background
Gastric cancer (GC) is the one of the most common malignant tumors, with nearly half of new cases occurring in eastern Asia 1 . Due to the insidious onset and early misdiagnosis, most GC patients miss the optimal operating time. The median overall survival (MOS) of patients with middle and advanced GC after receiving combined chemotherapy is only 7.6 to 11.6 months 2 . In recent years, although targeted precision medicine, immunotherapy and other therapeutic methods have improved the efficacy of GC, there has been no significant breakthrough in clinical endpoint data 3 , and chemotherapy is still the main treatment method 4 . However, chemotherapy often produces adverse effects such as nausea, fatigue, panocytopenia, and severe gastrointestinal toxicities 5 .
The application of traditional Chinese medicine (TCM) has a long history in cancer treatment 6 . With the increasing scientific evidence in biology, chemistry, medical research and clinical trials, it has been gradually recognized globally as a complementary and alternative therapy 7 . With the accumulation of tumor multidisciplinary comprehensive treatment experience and a large number of clinical cases, TCM therapy has been proved to be able, not only to alleviate fatigue, pain, vomiting, diarrhea, and leukopenia caused by surgery and chemotherapy, but also can help to shrink the tumor volume, improve symptoms, immunity and quality of life 8 .
Rosa roxburghii Tratt (RRT), a traditional food and medicinal plant in southwest China, belongs to the family Rosaceae. Phytochemical studies have indicated that RRT is rich in flavonoids, triterpenes, and organic acids, which are widely used in the treatment of diabetes, malignant tumors, abdominal distension, hyperlipidemia, hypertension and vitamin C deficiency 9 . Some included RRT prescriptions, such as Kang Ai Fu Zheng pills, Jin Ci Shen Jiu Zheng He Ji and Fu Fang Ci Li He Ji, have been commonly used in the treatment of leukopenia, thrombocytopenia, dysphagia, epigastric distension and other symptoms caused by tumor radiotherapy and chemotherapy. Extensive studies have pointed out that RRT has apparent therapeutic effects on malignant tumors, especially GC 10 . However, the molecular mechanism of RRT on treating GC remains unclear.
It is difficult to analyze the complicated mechanisms of TCM only by traditional methods because of the synergistic effect of its multiple ingredients, and multiple targets 11 . The application of network pharmacology combined with high throughput data from public databases such as Gene Expression Omnibus database (GEO) and The Cancer Genome Atlas (TCGA) to mine the mechanism of TCM has aroused extensive attention in recent years. In this study, we used network pharmacology and molecular docking techniques combined with GEO gene chips and TCGA database to identify the molecular mechanism of RRT in the treatment of GC. The workflow is shown in Figure 1.

Network pharmacology workflow to identify the mechanisms of RRT on treating GC.
Material and Methods
Collection of RRT Active Components
According to the published literature on the components of RRT, and combined with traditional Chinese medicine and chemical component database (http://www.chemcpd.csdb.cn/cmpref/main/tcm_introduce.asp), the chemical ingredients of RRT were collected, and the structures were derived from the PubChem database 12 (https://pubchem.ncbi.nlm.nih.gov/). Based on the Swiss ADME database 13 (http://www.swissadme.ch/), the physicochemical properties, pharmacokinetics and drug similarity of the collected components were calculated to screen out the active components. Oral Bioavailability (OB) was indicated by the level of gastrointestinal absorption (GA). Five drug similarity indexes with different rules (Lipinski, Ghose, Veber, Egan, Muegge) were used to screen the drug likeness (DL). Therefore, according to the filtering method reported in the literature 14 and the pharmacokinetic parameters in the Swiss ADME database, GA, OB and Lipinski pharmacokinetic principle were selected as the screening indexes of active ingredients in this study.
Mining Potential Targets of RRT
In order to obtain the potential targets of RRT active components, we searched Swiss Target Prediction 15 (http://www.swisstargetprediction.ch/) and the Traditional Chinese Medicine System Pharmacology database (TCMSP, https://tcmspw.com/tcmsp.php) 16
Identification of Differentially Expressed Genes of the TCGA Database
Genome wide transcriptome profiles of 344 GC tissues and 30 adjacent non-tumor tissues were downloaded from the TCGA database 17 (https://portal.gdc.cancer.gov/) by using the following keywords: “stomach”, “adenomas and adenocarcinomas”, “transcriptome profiling” and “RNA-Seq”. Since the data were obtained from the TCGA database, our research did not require further ethical approval. Collection and processing procedures conformed with the TCGA data access and protection of human subject policies (http://cancergenome.nih.gov/publications/publications guidelines). Genes with an adjusted P < .01 and │log2Fold Change≥3│were identified as the significant differentially expressed genes (DEGS). All the data were normalized and processed by R software.
Collection of GC Targets
The GC targets were collected in the database, including the OMIM database 18 (https://omim.org/), TTD database 19 (http://db.idrblab.net/ttd/), DrugBank database 20 (https://drugbank.com/), GeneCards 21 (https://www.genecards.org/), and GEO database (https://www.ncbi.nlm.nih.gov/geo/). Microarray data in stomach between the GC group and the normal group were acquired from the GEO database (https://www.ncbi.nlm.nih.gov/geo/), series: GSE29272, GSE54129. The LIMMA package of R software (4.0.2) was used to perform the analysis of multiple chips and correct data batches, and identified the DEGS between the GC and normal tissues. The DEGS’ cutoff value was set as│log2Fold Change>1│and FDR<0.05.
PPI Interaction Network Analysis
The intersection of RRT targets and GC targets was the targets of RRT on treating GC. These targets were introduced into the STRING database; the research species was limited to “Homo sapiens”, the lowest interaction score was set to medium confidence (0.4), and the rest was set to the default value. Cytoscape3.7.2 (https://www.cytoscape.org/) was used for visual analysis. The CytoNCA plugin performed topology analysis based on “Closeness (CC)”, “Betweenness (BC)”, “Local Average Connectivity (LAC)” and “Degree”. These four parameters represented the topological importance and can reflect the influence of the corresponding nodes in the whole network.
“Herbs-Components-Targets” Network Analysis
The “Herbs-Components-Targets” network of RRT was constructed using Cytoscape 3.7.2 according to the PPI interaction.
GO and KEGG Enrichment Analysis
After obtaining the therapeutic targets, the online tool of Annotation, Visualization and Integrated Discovery 22 (DAVID, https://david.ncifcrf.gov/) was used for GO and KEGG enrichment analysis. GO mainly analyzes the molecular function, cellular composition and biological process of the targets, whereas KEGG analyzes the potential biological pathways and functions.
Molecular Docking
The crystal structures of proteins and the core active components were downloaded from the RCSB protein (http://www1.rcsb.org/) and PubChem databases, respectively. PyMOL software was used to remove water, add hydrogen, remove ligands, and optimize amino acids. Finally, we used the AutoDock Vina software to calculate the minimum binding energy of the active components and candidate proteins, and visualized and analyzed the results of molecular docking. The location and size of the grid box for protein were required to contain amino acid domains related to bind with the reference compound, which was enclosed with a grid point number in X × Y × Z direction and a grid spacing of 0.375 Å in the box. Particularly, the grid box parameter for AKT1 comprised 20.918 × 2.534 × 16.36 points; −58.241 × 10.637 × −12.139 points for TP53.
Results
Active Ingredients and Target Predictions of RRT
Based on our review of the published literature and screening the database of Chinese medicine and chemical composition, we obtained 196 components of RRT (Table S1). A total of 18 active components were screened following the criteria (Table S2). Although myricetin and β-sitosterol do not meet the screening criteria, the two compounds have been reported to have therapeutic effects on GC, so they were identified as active ingredients of RRT. The TCMSP database and Swiss Target Prediction database were used to search RRT targets. A total of 307 targets were confirmed after integrating the targets of 20 compounds and removing duplicates (Table 1). The “Herbs-Components-Targets” network of RRT was constructed and visualized by Cytoscape 3.7.2 (Figure 2), including 328 nodes and 598 edges.

Herb-Components-Targets (H-C-T) network. Triangle nodes represent RRT, rhombus nodes represent core active ingredients, and hexagon nodes represent targets.
Basic Information on Active Components of RRT.

Quercetin has the most potential therapeutic targets, with a total of 186, followed by kaempferol (64) and ellagic acid (61). The components with multiple potential targets may be the key active components of RRT in the treatment of GC. All the active components in the network acted on 5 or more targets, and about 50% of the targets were regulated by two or more active components in RRT, indicating that the active components of RRT act in the whole network system, and different targets can be regulated by one or more active components, which vividly reflects the integrity, complexity and multiple targets of RRT against GC.
Targets Collection and Analysis of GC
In order to fully mine genes related to GC, we collated and analyzed data from multiple databases, combined with GEO gene chips and the TCGA database. Joint analysis of two GEO gene chips (GSE29272, GSE54129) have identified 554 DEGS related to GC. We downloaded 374 DNA expression datasets from the TCGA database and obtained DEGs by calculating the different gene expression between 30 normal tissues and 344 GC tissues. Using | log 2-fold change | ≥ 3 and P < .01 as screening cutoff conditions, 1436 DEGs were screened, containing 930 upregulated genes and 506 downregulated genes; a volcano diagram is shown in Figure 3. In addition, we synthesized the OMIM database, TTD database, DrugBank, GeneCards targets and, combined with the differentially co-expressed genes between GEO and TCGA to eliminate duplicates, 940 GC targets were identified.
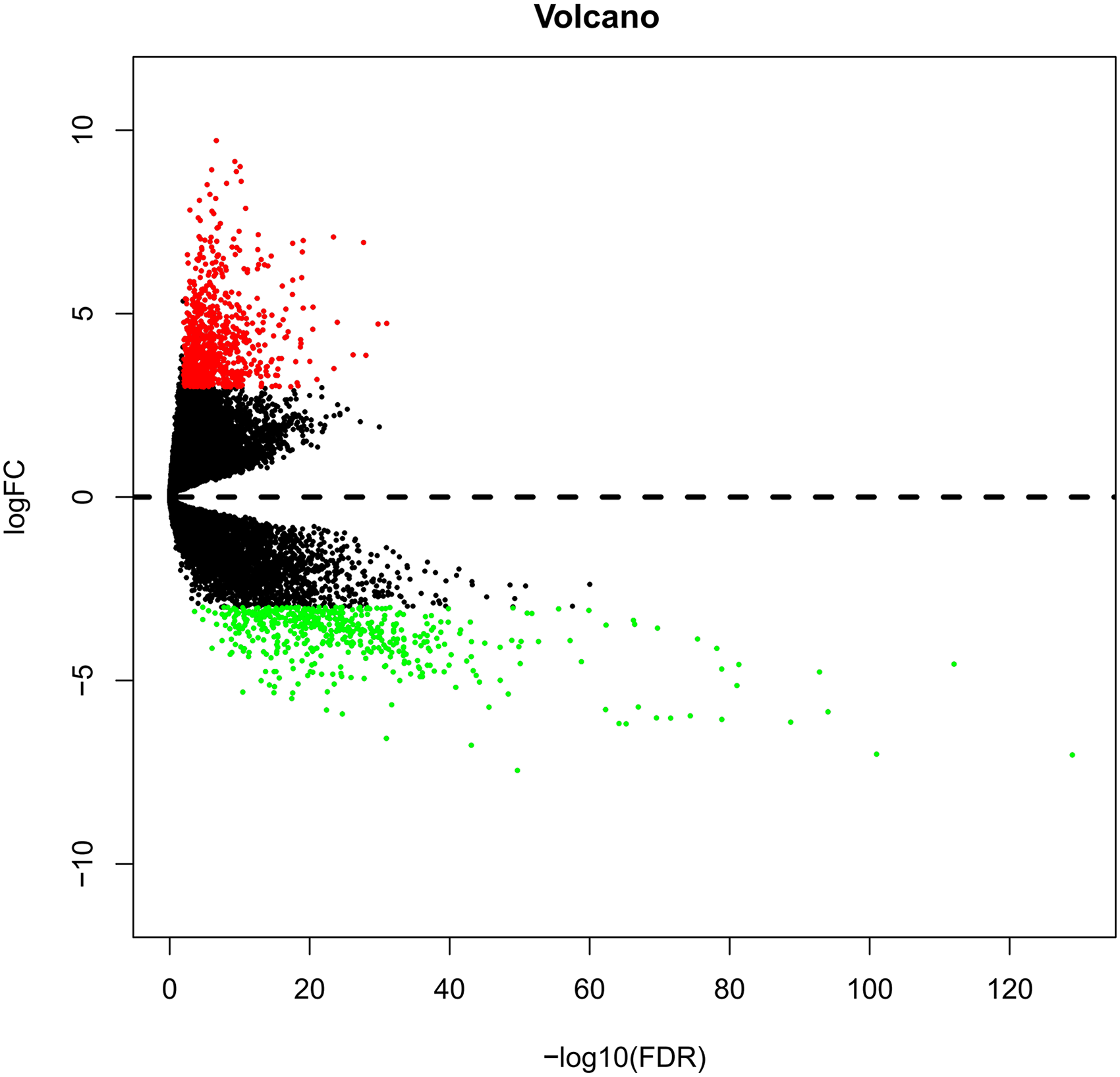
Volcano diagram of differentially expressed genes in GC. Red dots represent upregulated DEGs, and green dots represent downregulated DEGs.
PPI Interaction Network Analysis
The 93 intersection targets were introduced into STRING for PPI analysis, and the results were imported into Cytoscape 3.7.2 for visualization and network topology analysis. There were 93 nodes and 1793 edges in the network. The value of BC, CC, LAC and degree were used for screening the core targets of RRT against GC (Figure 4). Core targets were filtered using NetworkAnalyzer (Table 2). These 21 core targets may play a vital role in the efficacy of RRT against GC.
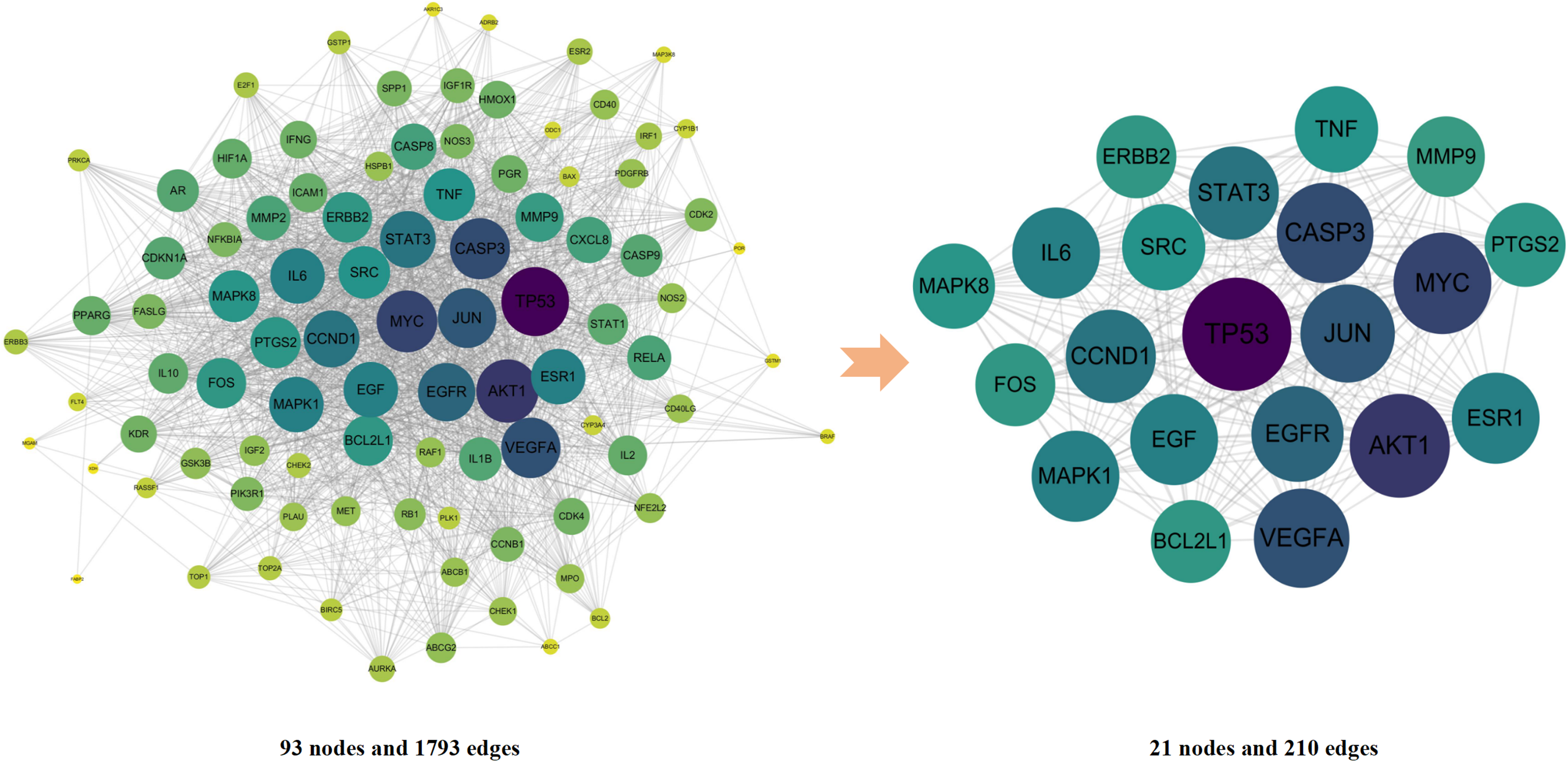
The process of topological screening for the PPI network. The PPI network diagram of 21 core targets was obtained by screening 93 targets through Degree, BC, CC, LAC. The larger the circle and the darker the color, the more important are the nodes in the network.
Information on 21 Core Targets.

Analysis of the Expression of Therapeutic Targets in GC Tissues and Normal Tissues
The expression of 93 therapeutic targets in GC tissues and normal tissues were contrastively analyzed (P < .05) (Figure 5), and it was found that 2 targets were up-regulated genes (CXCL8, SPP1) and 2 were down-regulated genes (MGAM, CYP3A4). CXCL8 and SPP1 have become potential biomarkers and therapeutic targets for GC 20 , and CYP3A4 may be a biomarker for predicting poor prognosis of GC 23 , and its abnormal expression is involved in the occurrence and development of GC 24 . This also fully demonstrated the multiple target characteristics of RRT against GC.
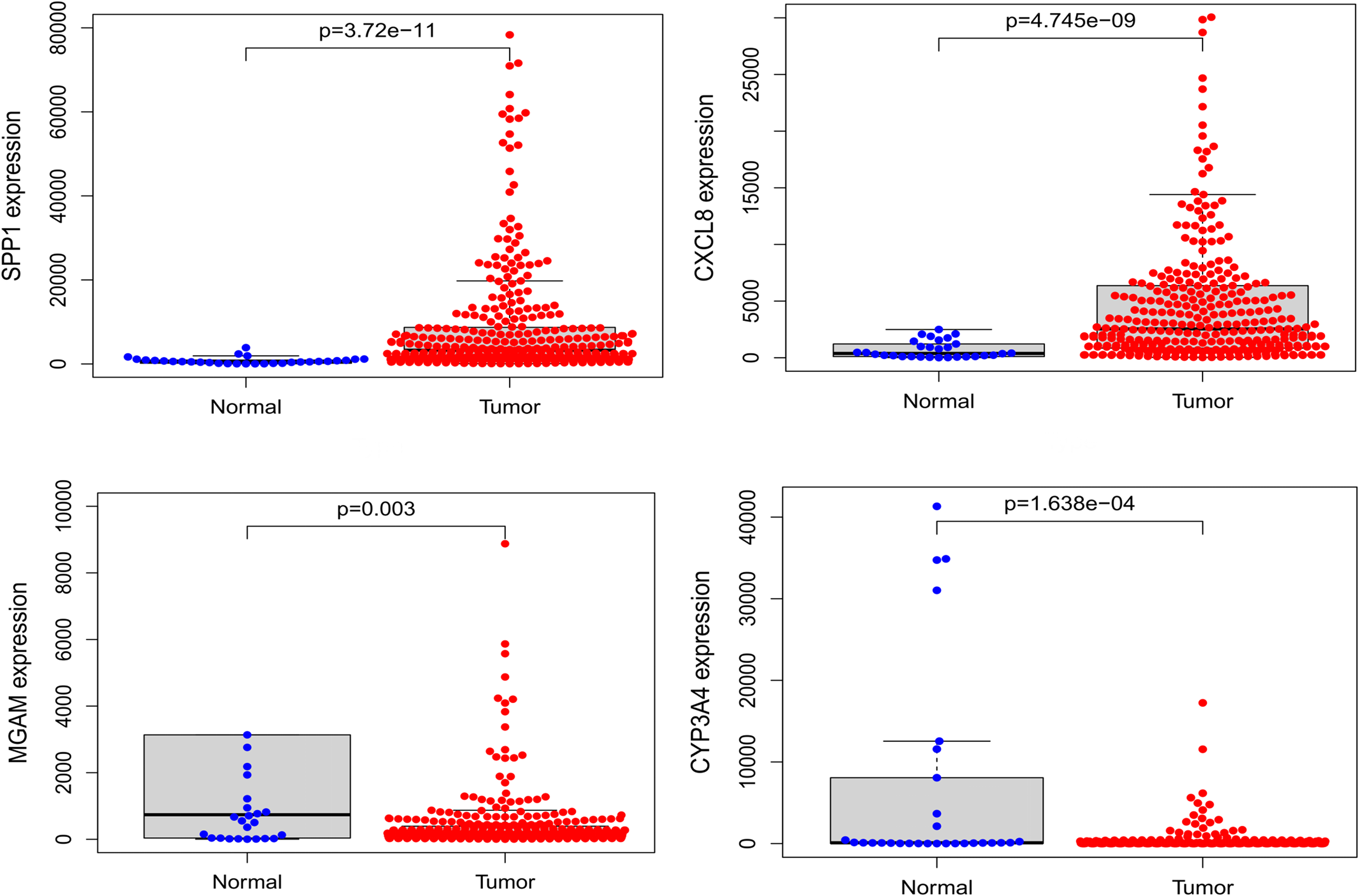
The expression of 4 therapeutic targets (SPP1, CXCL8, MGAM, CYP3A4) in GC tissues and normal tissues. (blue dot: normal tissues, red dot: GC tissues).
GO and KEGG Analysis
In terms of biological process, the 93 targets were mainly relevant to the regulation of cell response, apoptosis and protein phosphorylation, such as the negative and positive regulation of the apoptotic process (GO:0043066,GO:004306), cellular response to hypoxia (GO:0071465), cellular response to DNA damage stimulus (GO:0006974), cellular response to lipopolysaccharide (GO:0071222), positive regulation of protein phosphorylation (GO:0001934), protein phosphorylation (GO:0006468), and peptidyl-serine phosphorylation (GO:0018105). The molecular function mainly involves protein kinase activity, cytokine activity, nuclear transcription, enzyme binding, such as GO:0019899, protein kinase activity (GO:0004672), transmembrane receptor protein tyrosine kinase activity (GO:0004714), transcriptional activator activity, RNA polymerase II core promoter proximal region sequence specific binding (GO:0001077), transcription factor binding (GO:0008134), and tumor necrosis factor receptor binding (GO:0005164). Currently, RRT has also been proved to have anti-oxidant damage activity 25 ; it can inhibit tumor cell proliferation 26 , and promote the activity of pepsin and inhibit gastric acid secretion 27 . These results were consistent with the conclusions of GO enrichment analysis.
In addition, we defined the main GC pathways by KEGG enrichment analysis, and screened out the top 25 pathways relevant to GC and drew a lollipop diagram and a bubble diagram, as shown in Figure 6. These pathways remarkably enriched by the potential targets of RRT were strongly associated with cell proliferation, cell cycle, apoptosis, and angiogenesis, most of which play a key role in the development and progression of cancers, such as pathways in cancer (hsa05200), HIF-1 pathway (hsa04066), PI3K-AKT pathway (hsa04151), TNF pathway (hsa04668), vascular endothelial growth factor (VEGF) pathway (hsa04370), MAPK pathway (hsa04010), cell cycle (hsa04110), apoptosis (hsa04210), MicroRNAs in cancer (hsa05206), and forkhead box O (FoxO) pathway (hsa04068). We thereby speculated that the mechanisms of RRT for treating GC maybe mainly attributed to its coordinate regulation on pathways relevant to cancer.

Lollipop diagrams of the GO enrichment analysis. Bubble diagrams of the KEGG pathway analysis.
Components-Targets Docking Analysis
The three active components of RRT, namely quercetin, kaempferol, and ellagic acid, were selected for molecular docking with cell tumor antigen p53 (TP53) (PDBID: 3Q05) 28 , and AKT serine/threonine kinase 1 (AKT1) (PDBID: 4EKL)29–31. We analyzed these proteins because they were high degree nodes with many functional connections in the network, indicating that they play an important role in the GC treatment. The docking parameters and minimum binding energy are shown in Table 3, and the molecular docking mode is shown in Figure 7. The minimum binding energies between the active core components and target proteins are all less than −6 kcal·mol-1, indicating that the receptor proteins have good binding activity to the active components. Molecular docking analysis successfully showed docking between the three active ingredients of RRT and the binding pocket of the two tested target proteins. Quercetin was predicted to dock into the binding pocket of AKT1 via hydrogen bond interactions with Ala230 and Glu234, and hydrophobic interactions with Met227, Glu228, Met281, Thr211, Thr291, Glu278, Gly159, Lys158, Gly157, Val164, Leu156, Ala177 and Phe438. Similarly, quercetin binds to TP53 by forming hydrophobic interactions with Pro219, Pro151, Thr150, Pro152, Pro153 and Gly154, and hydrogen bond interactions with Arg202, Tyr220 and Thr155. Kaempferol also was predicted to dock into the binding pocket of AKT1 via hydrogen bond interactions with Gly162 and Lys179, and hydrophobic interactions with Phe161, Lys163, Leu181, Asp292, Gly294, Leu295 and His194. In the same way, hydrophobic interactions with Pro219, Thr150, Pro153, and Gly154, and hydrogen bond interactions with Tyr220, Arg202, Pro151, and Thr155 were predicted between kaempferol and TP53. Lastly, ellagic acid binds to AKT1 by forming hydrophobic interactions with Ala177, Val164, Leu156, Gly157, Phe438, Met281, Thr291, Thr211, Met227 and Tyr229, and hydrogen bond interactions with Glu228. An additional hydrophobic interaction with Tyr233, Pro142, Ser116, Lys120, and Cys124, and hydrogen bond interactions with Thr123 and Thr140 were predicted between ellagic acid and TP53. In conclusion, these results provide further evidence that these two proteins act as potential targets of the RRT active ingredients in GC.
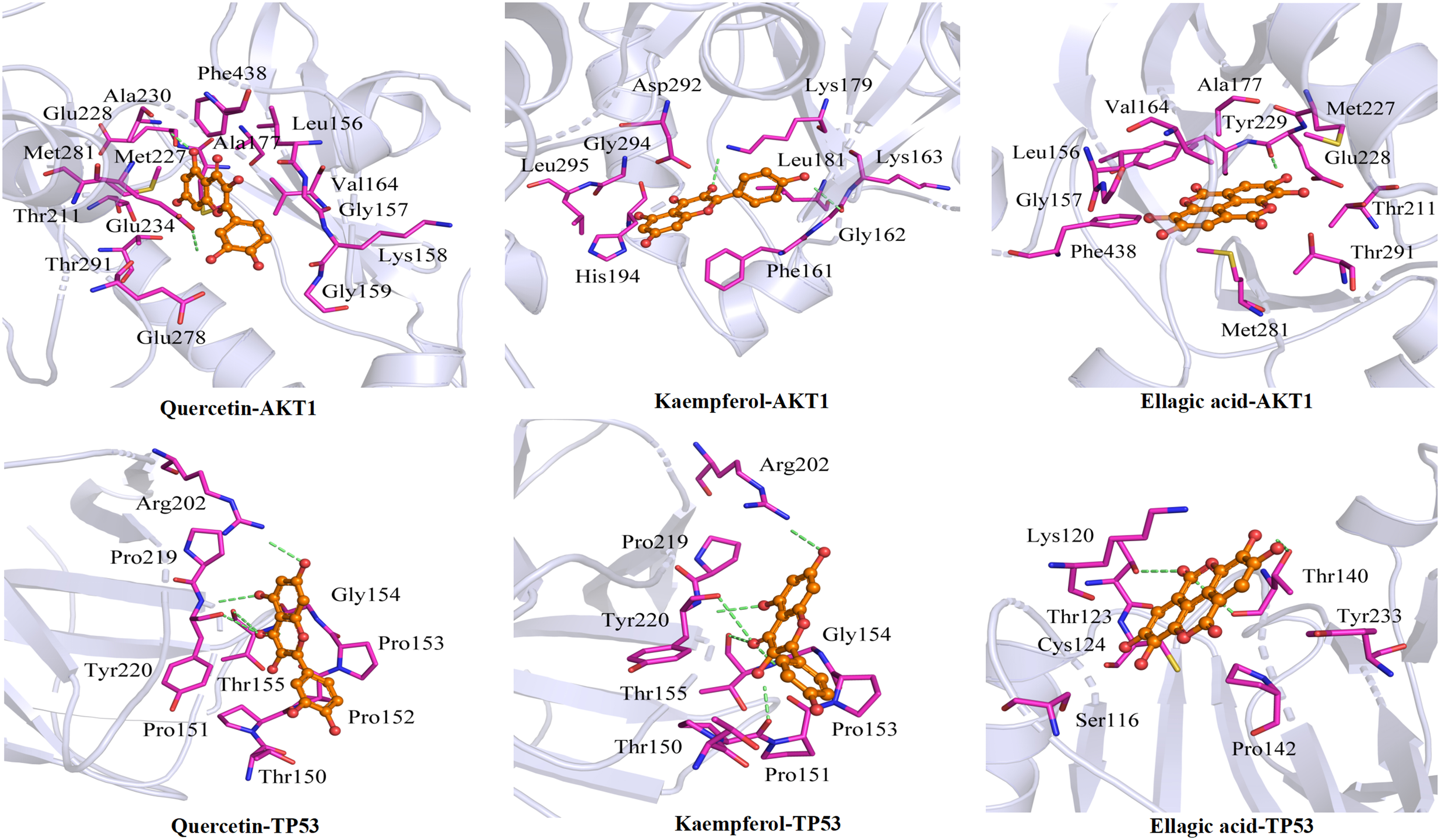
Molecular models of RRT core active components (quercetin, kaempferol, ellagic acid) binding to the target proteins AKT1, TP53.
Docking Parameters and Minimum Binding Energy Between Active Core Components and Target Proteins (Kcal·mol−1).

Discussion
RRT, a precious plant found in Guizhou Plateau in southwest China, has high nutrition and medicinal values. As recorded in Compendium of Materia Medica, “the flowers, fruits, leaves and roots can be used as medicine to relieve tiredness, eliminate stagnation, invigorate the stomach, digest food, nourish, relieve diarrhea, relieve cough and treat bronchitis”. Modern pharmacological studies have demonstrated that RRT has many properties, such as anti-aging, anti-oxidation, regulating immunity, anti-atherosclerosis, anticancer, and detoxification 32 . RRT is one of the main drugs of a variety of evidence-based, clinically efficacious preparations, such as Kang Ai Fu Zheng pills, Jin Ci Shen Jiu Zheng He Ji, and Fu Fang Ci Li He Ji, which are often used to treat malignant tumors. It has been pointed out that RRT juice has a good inhibitory effect on the synthesis of endogenous N-nitrosoproline in the population with a high incidence of GC 33 . RRT has been found to increase gastric mucosal blood flow and superoxide dismutase (SOD) activity, reduce pepsin activity, and the ulcer index, and has an obvious curative effect on gastric ulcer 27 . RRT extracts have been proved to have inhibitory effects on the growth of gastric cancer cells SGC-7901 and MNK-45 in vitro 34 . It has been revealed that RRT mixture can make stable lesions in 78% of cancer patients, and improves the condition of a quarter of the patients with pathological changes 35 . It has been demonstrated that Jin Ci Shen Jiu Zheng He Ji can significantly improve the quality of a patient's life, reduce the adverse effects of chemotherapy, and has been claimed to be a better anticancer TCM because it has a good taste, and easily accepted by patients 36 . Kang Ai Fu Zheng pills have been found to have an attenuating effect on malignant tumor patients during radiotherapy and chemotherapy, and a good alleviating effect on digestive tract and systemic toxic reactions such as anorexia, nausea, vomiting, and fatigue 37 . In addition, a meta- analysis of the literature has been conducted and it was found that Jin Ci Shen Jiu Zheng He Ji was effective, safe and less toxic in the treatment of cancer 38 . A lot of clinical studies have verified that prescriptions containing RRT are effective for GC. However, the molecular mechanism of RRT on treating GC remained unclear. Therefore, it was important to investigate the mechanism of RRT and to use the pharmacology of the TCM network for predictive analysis.
In this study, we analyzed the following active ingredients of RRT: quercetin, palmitic acid, kaempferide, protocatechuic acid, 4-hydroxycinnamic acid, gallic acid, syringic acid, 4-hydroxybenzoic acid, caffeic acid, α-linolenic acid, euscaphic acid, tormentic acid, roxburic acid, ellagic acid, kaempferol, l-glutamic acid, lauric acid, myricetin, β-sitosterol, and arjunolic acid. These components are flavonoids, triterpenes, sterols, polyphenols, fatty acids and amino acids, which are consistent with the effective components of RRT reported in the literature 39 . Modern pharmacological studies have found that quercetin has anti-inflammation, anti-oxidation, anti-virus and anti-tumor effects. Quercetin can induce apoptosis of colon cancer cells through the Akt pathway 11 . It also has anti-angiogenesis activity and can inhibit VEGF secretion by leukemic HL-60 cells 40 . It can exert its anti-tumor effect by regulating the expression of tumor suppressor genes, blocking the cell cycle, inducing apoptosis, and interfering with cell signal transduction 41 . Kaempferol has anti-oxidant, anti-inflammation and anti-tumor effects. It can significantly increase the expression level of p53 protein in gastric cancer cells. It can effectively promote the occurrence of apoptosis of gastric cancer cells, so as to inhibit the malignant proliferation of cancer cells 42 . Ellagic acid suppresses acidity enhanced migration and invasion of gastric cancer cells by inhibiting the expression of multiple factors (COX1, COX2, snail, twist1, and c-myc) 43 . Myricetin has been shown to inhibit the proliferation of GC HGC-27 and SGC7901 cells in vitro and in vivo, and induce apoptosis and cell cycle block in these cells via RSK2 and Mad1 44 . β-Sitosterol has been found to induce apoptosis and inhibit the proliferation of gastric cancer cells 45 .
Through PPI analysis of targets, it was found that TP53, AKT1, Myc proto-oncogene protein (Myc), caspase-3 (CASP3) and vascular endothelial growth factor A (VEGFA) might be the key targets of RRT in GC treatment. TP53, as a transactivator involved in cell cycle regulation, negatively regulates cell division by controlling a set of genes required for this process. It plays an important role in cell cycle arrest, senescence, apoptosis, differentiation and metabolism. TP53 gene changes were found in more than 50% of tumors 46 . AKT1 is an important downstream target of the PI3K-AKT pathway and belongs to the serine / threonine kinase family. These proteins are involved in the cellular signal transduction of downstream targets activated by growth factors and cytokines 47 . AKT1 can promote cell survival by inhibiting apoptosis. Abnormal activation of AKT1 exists in a variety of cancers, which can promote the metabolism, growth, proliferation, survival and angiogenesis of cancer cells 11 . Myc is a nucleoprotein protooncogene, encoding a transcription factor that promotes cell proliferation after binding to nuclear DNA. C-Myc is an important member of this family and most GC patients have c-Myc overexpression, and its abnormal activation can promote the development of GC 48 . It has been proved that quercetin and kaempferol can reduce the level of c-Myc protein and promote the apoptosis of many kinds of tumor cells 49 . In addition, quercetin has been reported to interact with the c-myc gene promoter region to form a stable structure, and may be a potential anti-tumor drug candidate 50 . CASP3 is a cysteine protease located downstream in the cascade of apoptosis. Activated CASP3 performs the process of cell apoptosis through lysis, destruction of cell structure and DNA fragmentation 51 . It has been found that quercetin and kaempferol can activate CASP3 protein through a variety of mechanisms, and then induce apoptosis of gastric cancer cells 52 . VEGFA can induce endothelial cell proliferation, promote cell migration, inhibit apoptosis and induce permeabilization of blood vessels. The expression of VEGFA in tubular glands was increased in gastric cancer tissues 53 .
The KEGG pathway enrichment results showed that RRT could play a therapeutic role by regulating multiple signaling pathways, mainly in cancer, apoptosis, hypoxia, metabolism and inflammation, including the PI3K-Akt, HIF-1, TNF, and MAPK pathways. The predicted mechanisms for RRT against GC are shown in Figure 8. The PI3K-Akt pathway was considered to be one of the most important pathways of RRT against GC. It is a carcinogenic signaling pathway, as well as one of the important pathways regulating cell proliferation, differentiation and apoptosis. Its dysregulation is involved in the development of various cancers. PI3 K exists widely in a variety of tissues and cells, which can activate cytokines, promote cell proliferation and differentiation, and inhibit cell apoptosis. As an important target molecule downstream of PI3 K in the pathway, the activation of Akt is closely related to the occurrence and development of tumors. Quercetin, the main active component of RRT, inhibits the proliferation of gastric cancer cells through apoptosis. In vivo experiments showed that quercetin induced lazy apoptosis of mitochondria by inhibiting the PI3 K/Akt pathway 54 .

Predicted mechanisms for RRT against GC.
An hypoxia microenvironment can lead to tumor proliferation, angiogenesis, metastasis and drug resistance. HIF-1α is a hypoxia inducible factor, which upregulates the expression of MMP-2 and MMP-9 and promotes tumor angiogenesis. At the same time, the abnormal expression of HIF-1α regulates the transformation of immune related cells to an immunosuppressive phenotype and promotes tumor immune escape 55 . There were 19 targets of RRT acting on the HIF-1 pathway (P < .05), suggesting that RRT may play a role in immune regulation by regulating the immune cell phenotype through this pathway. There is a large number of inflammatory mediators in the inflammatory microenvironment, which can cause cell gene mutation and promote tumor development and metastasis. The TNF pathway is related to cell proliferation, inflammation and immune response, and after activation, NF-κB and MAPK pathways can be further activated to promote the release of inflammatory cytokines such as TNF and IL-6, which in turn can promote the activation of inflammatory pathways, forming a vicious cycle 56 . In this study, 17 and 20 targets of RRT were enriched in the TNF and MAPK pathways, respectively (P < . 05), suggesting that RRT may regulate the TNF and MAPK pathway through related enrichment targets to inhibit the release of inflammatory factors, thus playing an anti-tumor effect.
Conclusion
In this present study, we adopted a network pharmacology approach to predict, clarify, and identify the potential mechanisms of RRT on treating GC by integrating targets prediction, network construction, and molecular docking analysis, and combining with the analysis of differential expressed genes in GEO gene chips and the TCGA database. We defined the effective components of RRT and their potential targets for the treatment of GC for the first time. In conclusion, the present research provides a scientific method to reveal that the potential pharmacological mechanisms of RRT on GC might be strongly associated with its collaborative modulation on inhibiting inflammation, cell proliferation and angiogenesis, improving the tumor microenvironment, and promoting apoptosis. This study provides a new direction for further exploration of the mechanism of RRT in the treatment of GC and a new perspective for the research of anti-tumor drugs. Furthermore, this study should be conducive to give clues to understand and assess the synergy of TCM in treating complex diseases and for accelerating the application of network pharmacology in identifying the potential mechanisms of anticancer TCMs.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the scientific research project of major agricultural industry of Guizhou province (KY[2019]009); the social development project of Guizhou Department of Science and Technology ([No.2020]4Y214); and the college students innovation and entrepreneurship training program in Guizhou Province (S202010672041).
