Abstract
Phytochemical study of the roots of Erythrina senegalensis led to the isolation of a new α-sophoradiol glycoside, erythrinoside (
Introduction
The Fabaceae is a large family comprising over 16 000 species, which are grouped into about 480 genera. Erythrina senegalensis DC, a member of this family, is a tree (6 to 7 meters high) 1 found in Cameroon among the savannah woods and shrubs of the Sudano-Guinean woodland of the Adamawa Region and is equally spread in many other tropical and subtropical zones.1,2 In African countries, E. senegalensis is a medicinal plant used as a general tonic and as remedy for several ailments such as cough, pneumonia, bronchial infection, malaria, snake bites, stomach ache, wound healing, normal fever and yellow fever, toothache, gastrointestinal disorders, jaundice, nose bleed, female infertility, gonorrhea and other venereal diseases, abdominal pain, and microbial and parasitic infections.2,3-6 Previous scientific studies have shown biological activities possessed by this plant such as antimicrobial, antidiabetic, antioxidant, analgesic, anti-inflammatory, antiviral, antimalarial and antitumor.2,7-10 The reported constituents of E. senegalensis are mainly phenolic compounds and prenylated flavonoids such as erythrinasinate, octacosyl (E)-fenilate, auriculatin, 2,3-dihydroauriculation, lonchocarpol A, scandenone, erysenegalensein D-M, 4′,5,7-trihydroxy-6,8-diprenylisoflavone, 6,8-diprenylgenistein, alpumisoflavone,2,3,11,12, and alkaloids such as erysocline, erysopine, erysotrine, erythratidine, 11-hydroxyerysodine, 11-hydroxyerysovine, 11-oxyerysodine and glucoerysodine.2,13,14
There are many documented medicinal benefits of Erythrina senegalensis, and, thereby, it is necessary to investigate scientifically the chemical compounds contained in this plant, as well as their biological activities. In this study, the roots of Erythrina senegalensis were investigated for their chemical composition, and the extracts, together with the isolated compounds, were evaluated for their antidiabetic potential through α-amylase and α-glucosidase inhibitory activities. To the best of our knowledge, the chemical compounds from the roots of E. senegalensis have not been the subject of any phytochemical and pharmacological studies.
Results and Discussion
Column chromatographic separation on silica gel of the ethyl acetate extract (25 g) of the roots of E. senegalensis led to the isolation of a new α-sophoradiol glycoside, named erythrinoside (
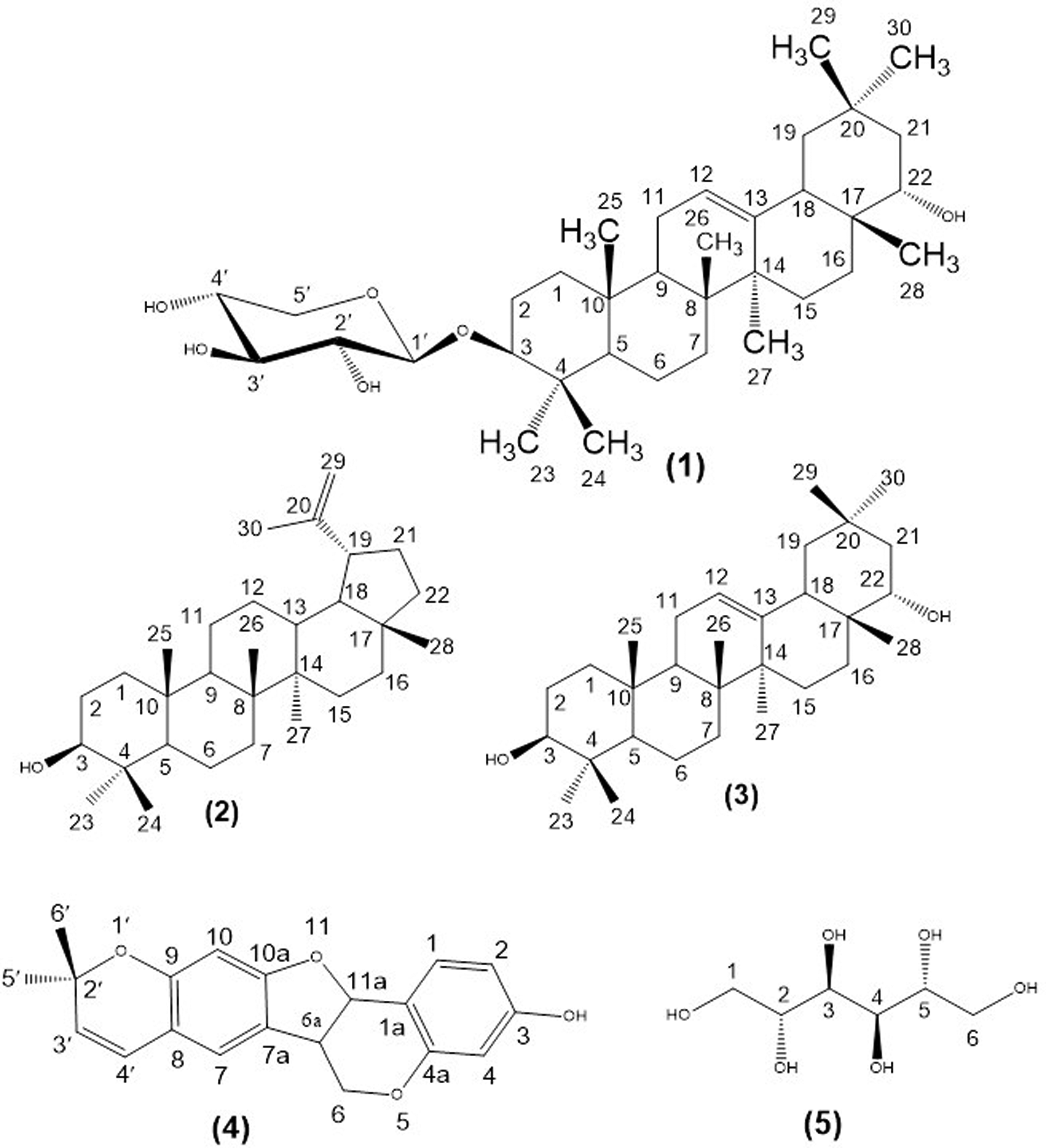
Structures of compounds (
Compound
The known compounds have been previously described in many other plants, lupeol (
The ethyl acetate and methanol extracts and the isolated compounds from Erythrina senegalensis were evaluated for their antidiabetic capacity by measuring their α-amylase and α-glucosidase inhibition (Table 1); the results are reported as inhibition percentages and IC50 values of α-amylase and α-glucosidase inhibition. The methanol extract showed higher inhibition (46.3%) of α-amylase at the dose of 400 mg/mL than the ethyl acetate extract (39.9%) at the same dose. Both extracts showed moderate activity on the inhibition of α-amylase. Likewise, among the tested compounds, erythrinoside (
α-Amylase and α-Glucosidase Inhibition by Extracts and Tested Compounds.

Material and Methods
General Experimental Procedure
Column chromatography (CC) was performed on silica gel 60 (70-230 mesh, Merck), and thin layer chromatography (TLC) on silica gel pre-coated plates F-254 Merck (20 × 20 cm). Compounds were visualized under UV light (254 and 365 nm), then sprayed with dilute sulfuric acid, and heated. The 1H and 13C NMR data were recorded on Bruker Avance AV-500 and 600 spectrometers, with trimethylsilane (TMS) as standard. Chemical shifts are given in ppm (δ) and coupling constants (J) in Hz. HR-TOF-MS LD+ spectra were registered on a QTOF Spectrometer (Bruker, Germany).
Plant Material
The roots wood of Erythrina senegalensis was collected in Ngaoundere in the Adamawa Region during July 2020. The plant was identified at the National Herbarium of Cameroon (NHC) with the voucher number: N° 50119 NHC.
Extraction and Isolation Procedure
The roots of Erythrina senegalensis were collected, dried at room temperature then ground into powder. Two Kg of the plant material was successively extracted by maceration at room temperature with 10 L of ethyl acetate and 8 L of methanol. For each solvent, extraction was made three times at the rate of one extraction every 72 h before moving on to the next solvent. The resulting solutions were evaporated using a rotary evaporator to obtain 30 g of ethyl acetate crude extract and 33 g of methanol crude extract. Twenty-five g of each extract was separated through column chromatography on silica gel using a gradient system of n-hexane/ethyl acetate (0 → 100%) and ethyl acetate/methanol (0 → 100%). From the ethyl acetate extract, 352 fractions were obtained and grouped into sixteen sub-fractions (A-P), according to their TLC profile. Sub-fractions B, C, D and G crystallized and were filtered and washed to obtain compounds
General NMR Data of Compounds 1 to 5
Erythrinoside (1)<PE: Please Check Heading Level.>
Amorphous beige solid; HR-ESI-MS( + ) m/z = 597.4129 [M + Na]+ (Calcd. for C35H58O6 + Na, 597.4131); 1H NMR (CD3OD, 600 MHz) δH ppm : 5.26 (H-12, t, J = 7.6 Hz), 4.20 (H-1′, d, J = 7.6 Hz), 3.49 (H-4′, m), 3.42 (H-3, dd, J = 8.0, 3.1 Hz), 3.32 (H-3′, m), 3.18 (H-22, dd, J = 13.6, 4.2 Hz), 3.17 (H-2′, m), 3.14 (H-5′a, m), 3.85 (H-5′b, m), 2.11 (H-9, m), 2.03 (H-18, m), 1.90 (H-16a, m), 1.93 (H-16b, m), 1.80 (H-11a, m), 1.03 (H-11b, m), 1.78 (H-15a, m), 1.04 (H-15b, m), 1,76 (H-19, m), 1.63 (Ha-21, m), 1.57 (Hb-21, m), 1,58 (H-6a, m), 1,45 (H-6b, m), 1,53 (H-1a, m), 1,36 (H-1b, m), 1.52 (H-2a, m), 1.36 (H-2b, m), 1,39 (H-7, m), 1.15 (H-27, s), 1.05 (H-23, s), 1.02 (H-26, s), 1.00 (H-25, s), 1.00 (H-28, s),0.93 (H-29, s), 0.90 (H-30, s), 0.81 (H-24, s), 0.77 (H-5, m).
13C NMR (CD3OD, 150 MHz) δC ppm: 36.4 (C-1), 26.5 (C-2), 81.9 (C-3), 38.4 (C-4), 55.3 (C-5), 18.1 (C-6), 32.7 (C-7), 39.4 (C-8), 44.8 (C-9), 36.6 (C-10), 23.2 (C-11), 122.4 (C-12), 143.8 (C-13), 41.8 (C-14), 25.4 (C-15), 27.3 (C-16), 25.4 (C-17), 45.4 (C-18), 46.1 (C-19), 29.8 (C-20), 38.6 (C-21), 78.2 (C-22), 27.5 (C-23), 14.7 (C-24), 14.9 (C-25), 16.2 (C-26), 24.2 (C-27), 27.3 (C-28), 31.2 (C-29), 19.7 (C-30), 101.3 (C-1′), 73.5 (C-2′), 76.5 (C-3′), 69.9 (C-4′), 65.3 (C-5′).
Lupeol (2)
White solid; ESI-MS (+) m/z = 449.9 [M + Na]+ for C30H50O; 1H NMR (CDCl3, 500 MHz) δH ppm: 4.55 (H-29, d, J = 1,6 Hz), 3.21 (H-3, m), 2.45 (H-19, m), 1.93 (H-21a, m), 1.32 (H-21b, m), 1.67 (H-30, s), 1.66 (H-13, m), 1,52 (H-6a, m), 1.63 (H-1a, m), 0.92 (H-1b, m), 1.51 (2H, m, H-2a and 2b), 1.38 (H-6b, m), 1,37 (2H, m, H-7a and 7b),1.47 (Ha-21, m), 1.35 (H-16a, m), 1,45 (H-16b, m), 1.25 (H-9, m), 1.37 (H-22a, m), 1,18 (H-22b, m), 1.03 (H-26, s) 0,95 (H-27, s), 0.90 (H-23, s), 0.85 (H-25, s), 0.79 (H-28, s); 0.77 (H-24, s). 13C NMR (CDCl3, 125 MHz) δC ppm: 38.7 (C-1), 27.5 (C-2), 78.9 (C-3), 38.8 (C-4), 55.2 (C-5), 18.4 (C-6), 34.3 (C-7), 41.0 (C-8), 50.5 (C-9), 37.2 (C-10), 20.9 (C-11), 25.3 (C-12), 38.1 (C-13), 42.8 (C-14), 27.4 (C-15), 35.3 (C-16), 42.4 (C-17), 48.4 (C-18), 47.9 (C-19), 151.1 (C-20), 29.6 (C-21), 39.9 (C-22), 27.9 (C-23), 15.7 (C-24), 16.2 (C-25), 15.9 (C-26), 14.7 (C-27), 17.8 (C-28), 109.5 (C-29), 19.4 (C-30).
α-Sophoradiol (3)
White powder; ESI-MS ( + ) m/z = 465.8 [M + Na]+ for C30H50O2; 1H NMR (CD3OD, 500 MHz) δH ppm: 5.27 (H-12, t, J = 7.5 Hz), 3.41 (H-3, dd, J = 8.0, 3.1 Hz), 3.17 (H-22, dd, J = 13.6, 4.2 Hz), 1.55 (H-9, m), 1.02 (H-16a, m), 0.99 (H-16b, m), 1.91 (2H, m, H-11a and 11b), 1.78 (H-15a, m), 1.04 (H-15b, m), 1,76 (2H, m, H-19a and 19b), 1.47 (Ha-21, m), 1.35 (Hb-21, m), 1,58 (H-6a, m), 1,45 (H-6b, m), 1,66 (H-1a, m), 1,00 (H-1b, m), 1.64 (H-2a, m), 1.57 (H-2b, m), 1,41 (2H, m, H-7a and 7b), 2.03 (H-18, m), 1.14 (H-27, s), 1.05 (H-23, s), 1.02 (H-26, s), 0.80 (H-25, s), 1.31 (H-28, s), 0.92 (H-29, s), 0.85 (H-30, s), 0.99 (H-24, s), 0.75 (H-5, m). 13C NMR (CD3OD, 150 MHz) δC ppm: 38.4 (C-1), 26.5 (C-2), 75.5 (C-3), 38.6 (C-4), 55.3 (C-5), 18.1 (C-6), 32.8 (C-7), 39.4 (C-8), 48.2 (C-9), 36.6 (C-10), 23.1 (C-11), 122.3 (C-12), 143.8 (C-13), 41.9 (C-14), 25.5 (C-15), 27.3 (C-16), 25.4 (C-17), 45.3 (C-18), 46.1 (C-19), 29.9 (C-20), 40.8 (C-21), 78.2 (C-22), 27.6 (C-23), 14.7 (C-24), 14.9 (C-25), 16.2 (C-26), 24.0 (C-27), 28.4 (C-28), 31.1 (C-29), 19.0 (C-30).
Isoneorautenol (4)
Gray powder; ESI-MS (+) m/z = 345.5 [M + Na]+ for C20H18O4; 1H NMR (CD3OD, 500 MHz) δH ppm: 7.29 (H-1, d, J = 8.4 Hz), 6.92 (H-7, s), 6.51 (H-2, dd, J = 8.4, 2.4 Hz), 6.32 (H-4, d, J = 2.4 Hz), 6.30 (H-4′, d, J = 9.8 Hz), 5.51 (H-3′, d, J = 9.8 Hz), 5.43 (H-11a, d, J = 6.8 Hz), 4.22 (H-6eq, dd, J = 10.7, 4.8 Hz), 3.53 (H-6ax, m), 3.43 (H-6a, m), 1.39 (H-5′, s), 1.37 (H-6′, s). 13C NMR (CD3OD, 125 MHz) δC ppm: 111.3 (C-1a), 131.7 (C-1), 109.3 (C-2), 158.7 (C-3), 102.7 (C-4), 156.6 (C-4a), 66.1 (C-6), 39.4 (C-6a), 119.5 (C-7a), 121.9 (C-7), 114.8 (C-8), 154.2 (C-9), 98.3 (C-10), 160.1 (C-10a), 78.6 (C-11a), 76.0 (C-2′), 127.0 (C-3′), 121.9 (C-4′), 26.8 (C-5′), 26.7 (C-6′).
D-Mannitol (5)
White solid; ESI-MS (+) m/z = 205.06 [M + Na]+ for C6H14O6; 1H NMR (DMSO-d6, 600 MHz) δH ppm: 4.50 (2H, d, J = 5.8 Hz, HO-2 and 5), 4.40 (2H, t, J = 6.0 Hz, HO-1 and 6), 4.25 (2H, d, J = 7,3 Hz, HO-3 and 4), 3.60 (2H, m, H-1a and 6a), 3,55 (2H, t, J = 7,1 Hz, H-3 and 4), 3.45 (2H, m, H-2 and 5), 3,40 (2H, m, H-1b and 6b). 13C NMR (DMSO-d6, 150 MHz) δC ppm: 64.3 (C-1 and 6), 71.7 (C-2 and 5), 70.0 (C-3 and 4).
In Vitro α-Amylase Inhibition Assay
The α-amylase inhibition assay was performed using the 3,5-dinitrosalicylic acid (DNSA) method.
21
Extracts and compounds (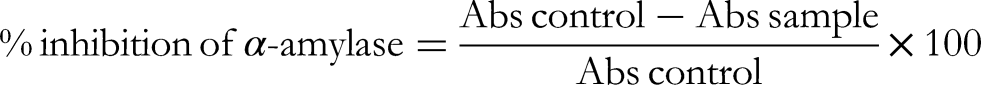
In Vitro α -Glucosidase Inhibition Assay
β-Glucosidase inhibitory activity was determined as described elsewhere.
22
Briefly, mixtures of 20 μL sodium phosphate buffer (pH 5.0), 20 μL p-nitrophenyl-β-D-glucopyranoside (Sigma Chemical Co., 1 mg/mL) and 10 μL of the sample at different concentrations (dissolved in DMSO) were incubated in a 96-well plate at 37 °C for 10 min, followed by the addition of 10 μL β-glucosidase solution from almonds (Sigma Chemical Co., 5 mg/mL) to each well, and incubation at 37 °C for 30 min. The reaction was terminated by adding 140 μL of sodium carbonate buffer, pH = 10. Absorbance was determined at 410 nm using a microplate reader (iTecan Microplate). To the control and blank were added 10 μL DMSO instead of the sample solution. The system without β -glucosidase was used as blank, and acarbose was used as positive control. The β-glucosidase inhibitory activity was expressed as the percentage of inhibition and calculated by the following equation:
% inhibition of
Conclusion
Investigation of the chemical constituents from the roots of Erythrina senegalensis resulted in the isolation and characterization of five compounds, including one new oleanane-type triterpenoid glycoside (erythrinoside) and four known compounds. The antidiabetic potential of the extracts and the isolated compounds were evaluated by measuring the inhibition of α-amylase. Compounds
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211044564 - Supplemental material for An α-Sophoradiol Glycoside from the Root Wood of Erythrina senegalensis DC. (Fabaceae) with α-Amylase and α-Glucosidase Inhibitory Potential
Supplemental material, sj-docx-1-npx-10.1177_1934578X211044564 for An α-Sophoradiol Glycoside from the Root Wood of Erythrina senegalensis DC. (Fabaceae) with α-Amylase and α-Glucosidase Inhibitory Potential by Cyrille Tchuente Djoko, Isaac Silvère Gade, Alex De Theodore Atchade, Alfred Ngenge Tamfu, Rodica Mihaela Dinica, Eliezer Sangu, Madeleine Tchoffo Djankou, Celine Henoumont, Sophie Laurent and Emmanuel Talla in Natural Product Communications
Footnotes
Acknowledgments
The authors are grateful to the Laboratory of NMR and Molecular Imaging of the University of Mons for conducting NMR analysis of the isolated compounds. We are also grateful to the Department of Chemistry, Physics and Environment of ‘Dunarea de Jos’ University, Galati for the material support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
