Abstract
Knowledge of the bioactivity of Alphonsea tonkinensis A.DC is limited. We have investigated the in vitro acetylcholinesterase inhibitory and antioxidant activities of extracts and pure compounds isolated from stems and leaves of this species collected from Dakrong district, Quang Tri Province, Vietnam. Extracts and isolated compounds were obtained by using an in-house extraction and chromatographic technique. The in vitro acetylcholinesterase inhibitory and antioxidant activities were evaluated using an Ellman test and 2,2-diphenyl-1-picryl-hydrazyl test, respectively. The total MeOH and CH2Cl2 extracts, the MeOH portion of the CH2Cl2 extract, pseudocolumbamine, and pseudopalmatine showed potential inhibitory activity against acetylcholinesterase with IC50 values of 22.7, 32.9, 14.6, 18.9, and 8.6 μM, respectively. The aqueous phase (pH 9), MeOH portion of the CH2Cl2 extract, and N-trans-feruloyltyramin exhibited significant antioxidant activities with IC50 values of 24.5, 72.1, and 61.2 µM, respectively. This is the first study showing such bioactivities of various extracts obtained from A. tonkinensis.
Alzheimer’s disease (AD), a progressive neurodegenerative disorder and the most common cause of dementia, is reaching epidemic proportions worldwide. Dementia is an organic disorder that is indicated by a gradual loss of memory, language, and intellectual abilities affecting daily life. 1 The decline appears to be due to the damage of nerve cells in the brain associated with cognitive function. To go along with the increase in the number of elderly people, AD is assumed to rise markedly in the coming decades. 1 Noticeable, AD is characterized by a reduction of acetylcholine (ACh) levels in the brain of AD patients. ACh is a neurotransmitter that plays a pivotal role in processing memory and cognitive skills, and acetylcholinesterase (AChE) is an enzyme catalyzing rapid ACh hydrolysis in numerous cholinergic pathways. 2 The decline of the cholinergic system resulting in cognitive impairment is one of the most important aspects of neurodegeneration in AD. 3 Accordingly, the inactivation of AChE by inhibitors leads to ACh accumulation in the brain, thus enhancing cholinergic functions in AD patients. 4 There are many therapies including AChE inhibition, anti-amyloid aggregation, modification of monoamine levels and antioxidant activities that have been used for AD symptoms. Of these, one of the most promising approaches is to employ AChE inhibitors. 5 Naturally occurring compounds from plants have been discovered to be potential sources of useful AChE inhibitors. 6
Oxidative stress is caused by reactive oxygen species exceeding the antioxidant capacity of a cell. It has been reported that oxidative stress is an early event in AD. 7 The brain consumes a higher level of oxygen than other organs due to its high ATP demand, and consequently, the brain produces a relatively high amount of reactive oxygen species (ROS). This leads to neurons being exposed to a high-ROS environment in the human brain. Recent studies have indicated that oxidative stress and damage may play a pathologically important role in AD due to the activation of cell signaling pathways contributing to lesion formation. 7 Today, natural antioxidants attract particular interest and attention in preventive medicine. These compounds possess the ability to scavenge free radicals and ROS to inhibit cellular damage. 8
The genus Alphonsea, family Annonaceae, comprises about 30 species, which are distributed in northeastern India, southern China, and Southeast Asia. Biological properties reported for this genus include antioxidant, anti-inflammatory, cytotoxic, anti-fungal, and antitrypanosomal activities, and alkaloids, sesquiterpenes, monoterpenes, steroids, and lignans have been recorded. However, only a few phytochemical and bioactivity studies of Alphonsea species have been reported, which may be due to their narrow distribution.9,10
The aim of this study was to investigate and gain an understanding of the acetylcholinesterase inhibitory and antioxidant activities of extracts and pure compounds of Alphonsea tonkinensis A. DC.
Materials and Methods
General Experimental Procedures
Nuclear magnetic resonance (NMR) spectra were obtained with a Bruker Avance 500 NMR spectrometer (1H NMR at 500 MHz, 13C NMR at 125 MHz) (Bruker BioSpin Corporation). Tetramethylsilane (TMS) was used as an internal reference. The column chromatography studies were conducted with YMC RP-18 from Fuji Silysia Chemical Ltd, silica gel (Kanto Chemical Co., Inc.), and Sephadex LH-20 (Dowex 50WX2-100, Sigma-Aldrich, St. Louis). Precoated silica gel 60F254 and RP-18 F254 plates (0.25 or 0.50 mm thickness, Merck) were exploited to conduct analytical thin-layer chromatography (TLC). An Agilent 1260 Infinity II (Agilent) preparative high-performance liquid chromatography (HPLC) system was used, equipped with a DAD detector and a Zorbax SB–C18 column (5 µm particle size, 9.4 × 250 mm).
Plant Material Collection and Identification
A. tonkinensis stems and leaves (8.0 kg dried samples) were collected from Dakrong district (geographical coordinates: 16°28′45.2″N; 107°00′49.9″E), Quang Tri Province, Vietnam, and identified by Dr Chinh Tien Vu, Viet Nam National Museum of Nature, VAST, Viet Nam. A voucher specimen under the code AT-01 was deposited in the Faculty of Pharmacy, Hue University of Medicine and Pharmacy, Hue University, Vietnam.
Preparation of Crude Extracts and Isolated Compounds
The total MeOH extract (0.79 kg) of the dried powdered stems and leaves of A. tonkinensis was suspended in HCl solution (pH 2.0), and extracted with ethyl acetate to afford the ethyl acetate-soluble portion (EA extract, 455.70 g). The aqueous phase was adjusted to pH 9 (aqueous phase pH 9) using saturated ammonia solution and extracted with CH2Cl2 to obtain 19.26 g of CH2Cl2 extract and an alkaline solution layer (aqueous phase). The CH2Cl2 extract was then partitioned into n-hexane (0.14 g), ethyl acetate (EC extract, 1.34 g), and MeOH portions (14.59 g), as shown in Figure 1.

Schematic diagram of the extraction and isolation of compounds from A. tonkinensis.
Chromatographic purification of the EC extract, the MeOH portion, and EA extracts resulted in the isolation of liriodenine (
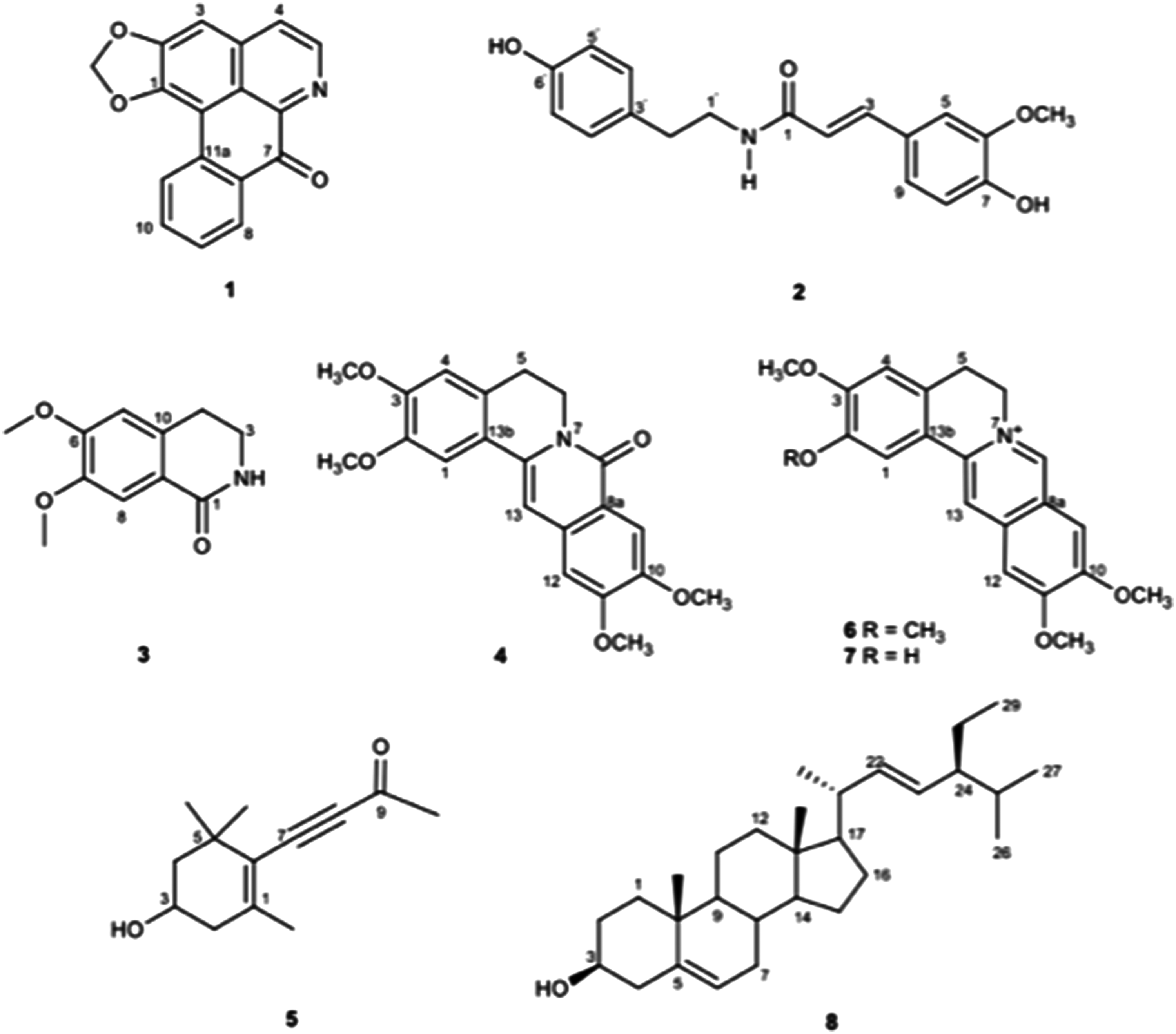
Chemical structures of isolated compounds
Determination of AChE Inhibitory Activities
AChE activity was measured using a modified Ellman method. 12 Briefly, the reaction was carried out in 140 µL (100 µM) of sodium phosphate buffer (pH 8.0), 20 µL of test sample solution (final concentration of 100 µg/mL for the extracts and 100 µM for compounds), and 20 µL of AChE solution, which were mixed in a 96-well microplate and incubated for 15 min at room temperature. Subsequently, 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB) (10 µL) was added and the reaction was then started with the addition of acetylthiocholine (10 µL). The hydrolysis of ACh was determined by monitoring the formation of the yellow 5-thio-2-nitrobenzoate anion at 405 nm for 15 min, which is a result of the reaction of DTNB with thiocholine released by the enzymatic hydrolysis of AChE. All samples and the positive control (galanthamine) were dissolved at concentrations of 100, 20, 4, and 0.8 µg/mL (µM) in MeOH to determine dose–response curves. The plates were read at OD450 using an ELISA plate reader (BioTek). The experiment was repeated 3 times. The inhibition value (in %) was calculated using the following equation: I (%) = (1–S/E) × 100, where S and E are the respective enzyme activities with and without the test sample. The inhibition of AChE activity of each sample was expressed in terms of the IC50 value (µM required to inhibit the hydrolysis of the substrate, AChE by 50%), as calculated from the log-dose inhibition curve.
Antioxidant Activity
The ability of the extracts and isolates to annihilate the DPPH radical (1,1-diphenil-2- picrylhydrazyl) was carried out as described by Blois.
13
Briefly, 200 μL of each sample was added to 200 μL methanolic solution of DPPH (0.135 mM). After mixing and incubation for 30 min at room temperature, the absorbance was measured at 517 nm against the corresponding blank by means of a Shimadzu UV-1800 (Shimadzu USA Manufacturing, Inc.). Quercetin was used as a positive control. The tests were carried out in triplicate. The ability to scavenge the DPPH radical was calculated as inhibition by the following equation:
Results and Discussion
Determination of AChE Inhibitory Activities
In this study, the extracts and compounds isolated from A. tonkinensis were tested for AChE inhibitory activity using a modified Ellman's colorimetric method in a 96-well microplate at various concentrations. Galanthamine was used as a positive control. The acetylcholinesterase inhibitory activity (IC50 values) of extracts and compounds are presented in Table 1. Among the extracts, the MeOH portion of the CH2Cl2 extract showed the strongest inhibition of AChE, followed by the total MeOH extract, the CH2Cl2 extract, the aqueous phase (pH 9), and the aqueous phase, with corresponding IC50 inhibition values of 14.6, 22.7, 32.9, 84.4, and 90.2 µg/mL, respectively. Pseudocolumbamine and pseudopalmatine possessed potent AChE inhibition effects with IC50 values of 18.9 and 8.6 μM, respectively.
The IC50 Value of the Antiacetylcholinesterase Activity of Extracts and Compounds From the A. tonkinensis.

N.T: the compounds were not tested.
According to Hien et al, 14 the methanolic extracts of only three out of 30 medicinal plants in Vietnam (10% of samples) were reported to present AChE inhibition with IC50 values of less than 50 μg/mL. Similar results (ie, the same proportion of plants) were reported by Mathew and Subramanian. 6 In our study, the total MeOH extract with an IC50 value of 22.7 μg/mL against AChE was a good source for further study.
To date, 4 medications have been approved by the FDA for the treatment of AD and other dementia: donepezil (Aricept), rivastigmine (Exelon), galanthamine (Razadyne), and memantine (Namenda). The first 3 (alkaloids) are AChE inhibitors, and more specifically galantamine and rivastigmine are derived from plant alkaloids. Memantine acts by blocking a neurotransmitter called glutamate, which is a nitrogen-containing molecule. Alkaloids have become the most promising drug candidates for the treatment of AD due to their nitrogen-containing structures.15,16 In our study, we investigated the presence of alkaloids in the methanolic extract using a conventional alkaloid extraction method, TLC, and detection with Dragendorff's reagent 17 . Two alkaloids, pseudocolumbamine and pseudopalmatine, were isolated with potent AChE inhibitory activity (IC50 values of 18.9 and 8.6 μM, respectively. This extract and the pure compounds are promising candidates for drug discovery for AD.
Antioxidant Activity
The antioxidant activity was investigated using a DPPH assay with quercetin as a positive control. As shown in Table 2, the best antioxidant efficiency was found for the aqueous phase (pH 9) extract with an IC50 value of 24.5 μg/mL, followed by the MeOH portion extract with an IC50 value of 72.1 μg/mL. Of the isolated compounds, N-trans-feruloyltyramine showed significant antioxidant activity with an IC50 value of 61.2 μM.
The IC50 Values of the Antioxidant Activity of Extracts and Compounds of A. tonkinensis.

N.T: the compounds were not tested.
DPPH is the method of choice for evaluating antioxidant activity in vitro since it is generally considered a valid, accurate, simple, and economic method. 18 In our study, an aqueous phase (pH 9) extract and the MeOH portion of the extract showed significant antioxidant activity in vitro with IC50 values of 24.5 and 72.1 μg/mL, respectively. The results suggest that the active constituents are most likely polar. The aqueous phase (pH 9) extract can be considered a valuable source for the development of novel antioxidant formulations. Interestingly, N-trans-feruloyltyramine exhibited considerable DPPH radical scavenging activity. Our results are in good agreement with the findings reported by Li. 19 This alkamide is assumed to play a major part in the protection of the plant. 20
Conclusions
The total MeOH extract, the MeOH portion of the CH2Cl2 extract, as well as pseudocolumbamine and pseudopalmatine possessed potent AChE inhibitory activities. Besides, the aqueous phase (pH 9) extract showed significant antioxidant activity. They are all considered to be promising candidates for further study. The bioactivities of these extracts are reported for the first time.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211042134 - Supplemental material for In Vitro Acetylcholinesterase Inhibitory and Antioxidant Activity of Alphonsea tonkinensis A.DC
Supplemental material, sj-docx-1-npx-10.1177_1934578X211042134 for In Vitro Acetylcholinesterase Inhibitory and Antioxidant Activity of Alphonsea tonkinensis A.DC by Khan Viet Nguyen, Thu Oanh Thi Nguyen, Duc Viet Ho, Jyrki Heinämäki, Ain Raal and Hoai Thi Nguyen in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hue University (grant no. DHH 2019-04-105).
Supplemental Material
Supplemental material for this article is available online.

